Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Environmental Sciences
Plastics have been massively produced since the 1940s, when society started to consume large quantities of plastics in every segment of our lives, including industry, agriculture, healthcare, and others. The production of plastics has been expanded in the last decades because of their favored characteristics, such as lightness, durability, flexibility, versatility, and cost-effectiveness.
- degradation
- health risk
- micro(nano)plastics
1. Introduction
Plastics have been massively produced since the 1940s, when society started to consume large quantities of plastics in every segment of our lives, including industry, agriculture, healthcare, and others [1][2]. The production of plastics has been expanded in the last decades because of their favored characteristics, such as lightness, durability, flexibility, versatility, and cost-effectiveness [3]. However, the extensive consumption of plastics has resulted in severe environmental consequences. It is of particular relevance that once plastic litters enter the environment, they can persist for a long time and will be broken down into small debris (alias mesoplastics), microplastics (MPs, <5 mm), and even nanoplastics (NPs, <100 nm) via physical, chemical and biological processes before reaching complete decomposition [4]. The MPs and NPs have attracted public concern due to their small size, worldwide distribution, and potential ecotoxicological effects [5]. As a result, the studies have been increasing in the past few years (Figure 1, retrieval time from 01.2016 to 01.2022). The figure shows that the studies of MPs and NPs have expanded in the past few years.
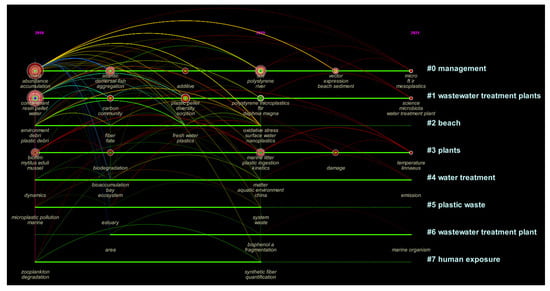
Figure 1. Visual analysis diagram of research studies relating to MPs and NPs since 2016. To date, many previous studies have investigated the global distribution of MPs and recently focused on the NPs [6]. For example, as compared with the studies of MPs, the analysis methodologies of NPs are pretty limited, and only 405 publications (including 71 review papers and 334 research papers) can be retrieved according to the literature retrieval of ‘nanoplastic(s)’with other keywords such as ‘analysis’, ‘extraction’, ‘methodology’, ‘separation’, ‘characterization’, ‘identification’, or ‘quantification’ (Figure 2) (Date up to 1 May 2022). The figure shows that most NPs studies focused on the fate and toxicity in the environment, while studies regarding the measuring methodology are still scarce.
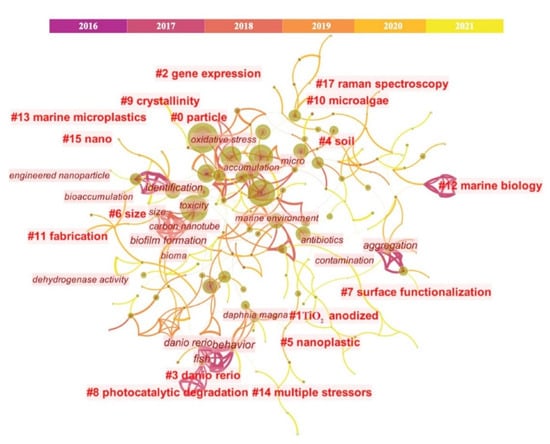
Figure 2. Visual analysis diagram of MPs and NPs measurement since 2016. Note: The retrieval was accomplished with the keywords such as “nanoplastic”, “analysis”, “extraction”, “methodology”, “separation”, “characterization”, “identification”, and “quantification”, via database, including Web of Science, Science Direct.
Recently, Cai et al. [7] summarized the progress and challenges of environmental NPs analysis, in which 95 studies have been reviewed while only 12 of them focused on the field studies (Date up to 1 May 2022). For example, Fang et al. [8] introduced the identification and visualization of MPs and NPs by a newly developed Raman mapping image method; Peiponen et al. [9] also demonstrated the optical identification of MPs and NPs in aquatic environments. However, they all pointed out that improving analytical reliability and accuracy is still challenging, especially for NPs. For instance, how to enhance the weak Raman signal from NPs, and the identification requires a combination of optical detection, etc. In other words, it is a common challenge that the analytic techniques remain to be standardized and explored, especially for improving the detection efficiency and accuracy.
Various physical, biological/microbial, and chemical techniques have been implemented to combat the threat posed by micro(nano)plastic. Currently, some research is being carried out on (micro)plastic degradation. The degradation of (micro)plastics is split into biological and chemical degradation [10]. Sudhakar et al. [11] conducted in situ experiments using polyethylene (PE) and polypropylene (PP) sheets in marine water. They studied Pseudomonas spp., Clostridium spp., unidentified anaerobic, heterotrophic, and iron-reducing bacteria and fungi and found that degradation dependent on study site, plastic type, and season. Another study addressed an in vitro study of Nylon pellets degradation by Bacillus cereus, B. sphericus, Vibrio furnisii, and Brevundimonas vesicularis and found that degradation varied across microbial taxa (highest for B. cereus) [12]. On the other hand, in vitro degradation of PE sheets with B. cereus subgroup A and B. sphericus GC subgroup IV showed degradation rates varied with PE [13]. Bao and coworkers recently studied microplastics released from biodegradable blended plastic and demonstrated bacterial communities in contrasting environments such as water and air, showing variable trends [14]. For instance, plastics having hydrolyzable covalent links (such as polyethylene terephthalate (PET)) can be hydrophilic, which accelerates the hydrolytic degradation of the polymer [14]. Microplastics’ C–C and C–H bonds can be destroyed by ultraviolet (UV) light, causing them to deteriorate [15][16]. However, more research is needed on the chemical breakdown of microplastics, which is currently being carried out via advanced oxidation processes (AOPs) [17].
2. Overall Introduction on Techniques for MPs and NPs Measurement
The studies of MPs and NPs pollution are gaining more and more attention around the globe; however, it should be cautioned that their protocols remain to be standardized profoundly in terms of sampling, preservation, digestion, separation, and identification in particular to some comprehensive, fast and efficient approaches [18]. At present, most of the studies adopted the approaches recommended by NOAA. Figure 3 summarizes the most common methods for the determination of MPs in water [19][20], soil (or sediment, sand) [21][22], organism [23][24], and air [25] in the previous studies
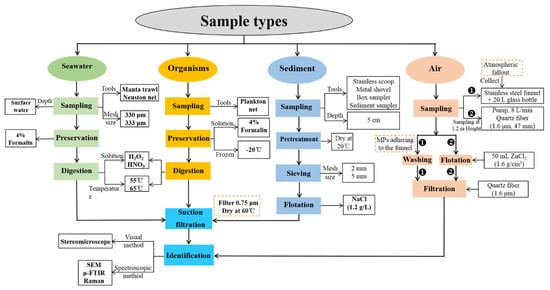
Figure 3. Procedures of MPs investigation in the environment.
2.1. Separation Methods for MPs and NPs in Different Matrices
The analytical steps for MPs and NPs in different environmental matrices are generally similar, including sample collection, processing (e.g., centrifugation, digestion, staining), flotation, filtration, and then a series of physical and chemical methods for characterization (Figure 3). In general, trawl or Neuston nets are usually used for MPs sampling from the aqueous environment such as freshwater and seawater. At the same time, stainless steel products (e.g., shovels and spoons) are commonly applied in sampling soil, sediment, and sludge. By comparison, pumps and atmospheric wet and dry deposition (e.g., active and passive sampling) are frequently applied to sampling MPs in the air; and planktonic nets are used to collect aquatic organisms [26][27][28]. After sampling, flotation is essential in separating MPs from the collected samples. At this stage, the large materials will be removed using saturated sodium chloride and sodium iodide [29][30]. Sodium chloride applies to the flotation of low-density plastics, while sodium iodide is for the flotation of high-density plastics. After flotation, the supernatant will be filtered using filter membranes made of cellulose nitrate, nylon, PTFE, sartorius, polycarbonate, and glass membranes [31][32][33]. However, digestion is essential to the flotation of MPs in complex samples (e.g., sludge, wastewater), and a 30% hydrogen peroxide solution is commonly used for digestion [34][35]. In addition, the digestion solution varies with the types of samples and the characteristics of MPs, aiming to achieve maximum recovery efficiency [36]. For example, potassium hydroxide solution is generally used for the digestion of biological samples [37], while proteinase K is for tissues [38] and Fenton’s reagent for sediments [39].
2.2. Identification of MPs and NPs
Various methods, including visual, spectroscopic, and scanning electron microscopy (SEM), are recommended to identify the MPs in environmental samples. Among them, the microscope’s visual method is aimed at observing MPs’ surface morphology (e.g., shape, length, color, etc.) [40], and high-resolution images of surface structure MPs can be achieved using SEM. SEM pictures can be used to compare MPs before and after aging or to see MP biofilm [41]. Further, the spectroscopic method, mainly through Micro Fourier Transform Interferometer (μ-FTIR) or μ-Raman can identify the composition of polymers by analyzing the functional groups on MPs [42], in which the μ-FTIR is applicable to the identification of MPs in size of ca. 20 μm, while μ-Raman to those in size of ca. 1 μm and even smaller [43]. However, since both of these procedures are costly and time-consuming, it is useful to quickly establish techniques for detecting MPs. Next, some new approaches were suggested, such as a support vector machine (SVM) approach is recommended to determine MPs based on hyperspectral imaging and achieving steady detection of various kinds of plastics [18]. Some studies investigated a portable optical sensor to detect transparent and translucent MPs in freshwater. For understanding a variety of MPs and plastic additives, gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-mass spectrometry (LC-MS) are recommended in addition to visual and spectral verification. For instance, LC-MS/GC-MS can be used to examine the degradation products and additives in natural and manufactured fibers [44]. In addition, other integrated methods such as thermal desorption-proton transfer reaction-mass spectrometry (TS-PTR-MS), thermochemolysis coupled to GC-MS, pyrolysis-gas chromatography/mass spectrometry (py-GC-MS) and surface-enhanced Raman spectroscopy (SERS) have been shown to be efficient in the identification of NPs in real-world studies [45]. However, exploring novel methods for rapid quantitative and qualitative identification of MPs in various media, particularly for tiny plastic particles such as NPs, remains challenging [46].
3. Techniques for MPs and NPs Measurement in Various Matrices
3.1. Techniques for MPs Studies in Seawater
As shown in Figure 3 and Table 1, the main steps for MPs studies in seawater include sampling, preservation, digestion, filtration, and identification.
Table 1. Common methods used in the sampling and identification of MPs in seawater.
| Procedures | Common Methods | Refs |
|---|---|---|
| Sampling Tool | Plankton net; Manta trawl; Neuston net; Continue intake; Teflon pump | [47][48][49][50][51] |
| Reservation | Formalin solution (4%, 5%); Refrigerated; Ethanol (70%, 90%) | [52][53][54] |
| Digestion | Hydrogen peroxide (30%); Sodium hydroxide (5M); Nitric acid (65%); Fe (II) solution (0.05 M) | [47][55][56] |
| Separation | Sieving; Density separation, filtration | [57] |
| Pore Size (μm) | 0.45; 0.70; 0.75 | [58] |
| Temperature for drying | 50 ℃; 55 ℃; 60 °C; Air-dried | [59] |
| Identification | Stereomicroscope; SEM; μ-FTIR or Raman | [60] |
3.1.1. Sampling
Seawater samples can be collected by plankton net [61], manta trawl [47], catamaran, neuston net [48] (Figure 4), teflon pump [49] and intake system of the vessel [51], with a common range of mesh size from 120~335 μm [47][54]. Manta trawl and Neuston net with a mesh size of 330 μm or 333 μm [62][63] is the most frequently used in seawater sampling. In general, the vessel is dragged at a speed of 1~5 knots [50][54] for 5~30 min [61][64]. Next, a flow meter is installed at the entrance of the sampling device to measure the water flow [52]. Finally, transfer the sampled surface water to a glass bottle for further analysis.
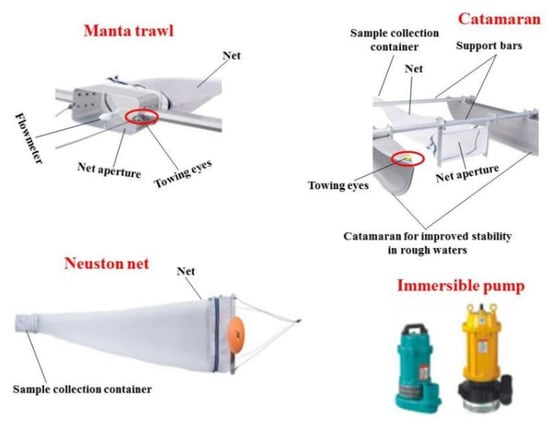
Figure 4. Sampling tools used in the investigation of MPs.
3.1.2. Preservation and Digestion
In general, the aforementioned seawater samples are directly used for detecting MPs [19][33], except for some specific purpose of investigating MPs distribution in the environment [56]. Under such circumstances, it is suggested to preserve fresh seawater with reagents (e.g., 4% or 5% formalin solution) [52], 70 or 95% ethanol [53], or directly refrigerated at 4 °C [65], since that some MPs may transfer from seawaters to phytoplankton, zooplankton and other organisms in higher trophic levels. Subsequently, seawater is digested to degrade the organic matter inside by adding reagents such as 5 M NaOH [66], 65% HNO3 [56], 30–35% H2O2 [20], or wet peroxide oxidation (WPO, a combination of 30% H2O2 and 0.05 M Fe(II) solution (i.e., 7.5 g of FeSO4·7H2O to 500 mL of water and 3 mL of concentrated sulfuric acid)) [53] and others, as discussed later in Section 3.2. Next, the digested solution of the samples on the net will be transferred to a density separator for flotation, which separates the light component MPs (with a density of 0.8–1.4 g/mL) from heavy ones and collects the floating solids using 0.3-mm custom sieve [57]. In contrast, the samples in the volume-reduced water can be directly separated from the mixtures through sieving [53] and filtration with a set of vacuum suction filter devices with membrane pore sizes of 0.45~0.75 μm [54]. Next, the filter paper is subsequently transferred into a petri dish with a cover for drying at 25~60 °C [53].
3.1.3. Observation, Enumeration, and Identification
The items similar to MPs (based on morphology and color) will be selected from the above-dried samples for further analysis. In general, MPs can be analyzed by qualitative analysis and quantitative analysis, in which the qualitative analysis applies to the analysis of the physical morphologies and the identification of the chemical components of MPs [67], while the quantitative analysis is mainly to calculate the quantity and weight of MPs by inspection [68]. First, the MPs’ shape, color, and size were observed by a stereoscopic microscope [55] and characterized by SEM [69]. Next, the suspected MPs particles or fibers can be selected using tweezers. The quantity of MPs is counted by manual counting (prone to the miscarriage of justice) [68]. At the same time, the qualitative analysis is achieved by μ-FTIR [61], μ-Raman [60] are commonly used in previous studies, other approaches such as SEM-EDS [53], ESEM-EDS [67], Pyr-GG-MS [70], or other methods such as tagging method [71].
3.2. Techniques for MPs Studies in Sediment and Soil
Among the types of sediments, such as sea (core) sand, beach sand, and municipal soil [34][38], beach sand is the most studied type. Beach sands are collected by stainless steel spoon, shovel, or frame with a quadrat from the surface to a depth of 1~5 cm [59][72] (As shown in Table 2). Sands are usually preserved in aluminum foil or stainless containers and then dried through freeze-dried [50], air-dried [73], or oven at 40–60 °C [74][75] [76]. Once dried, the samples can be sieved with a mesh size of 1~5 mm [75][77].
Table 2. Typical sampling and identification of MPs and NPs in sediment or soil.
| Parameters | Range/Chemicals/Instruments | Refs |
|---|---|---|
| Sampling depth (cm) | 1; 2; 3; 5; 10 | [59][72] |
| Mesh size (mm) | 1; 2; 5 | [59][75] |
| Dry temperature | Freeze-dried; Air-dried; 40 ℃; 50 ℃; 60 ℃ | [74][75] |
| Separation solution | NaCl; NaI; KI; ZnCl2 (1.37~1.7 g/mL); ZnBr2; Canola oil | [77][78] |
| Filter paper mesh size (μm) | 0.7; 0.8; 1.0; 1.2 | [50][79] |
| Identification | Stereomicroscope; SEM; μ-FTIR or μ-Raman; Py-GC-MS | [60][61][80] |
Next, the dried and sieved samples will be separated by density flotation [79], which separates the light component MPs (with a density of 0.8–1.4 g/mL) from the reconstituted sediment (with a density of 2.65 g/mL) [68]. In detail, a certain volume of sediment samples can be mixed evenly by stirring and shaking, letting it settle down until reaching solid-liquid separation, and collecting the MPs suspended or floating on the supernatant using filter paper.
As summarized in Table 3, the common saturated salt solution includes 1.2 g/mL NaCl [75], 1.71 g/mL ZnBr2 [81], 1.6 g/mL ZnCl2 [82], and 1.57 g/mL NaI [77], and others. NaCl is the most widely used because of its low cost and no harm to the environment [79]. However, some high-density plastic particles cannot float in the solution. Flotation reagents such as NaI, KI [83], ZnCl2, NaBr [84], and ZnBr2 [81] are also used to float denser polymers in some studies, but they are costly and may cause chemical hazards to filter paper and therefore disturb the identification of samples, and be a hazard to the environment.
Table 3. Advantages and disadvantages of density separation solutions.
| Solution (Density) | Advantages | Disadvantages | Refs |
|---|---|---|---|
| NaCl (1.2 g/mL) | Low cost, environmental sound | High-density plastic particles are not applicable | [75] |
| NaI (1.57 g/mL) | Cheaper, less environmental dangerous | Oxidizes filter paper, makes filter black, and makes it difficult to distinguish MPs | [81] |
| KI (1.254 g/mL) | Applicable to high-density polymer | Expensive, oxidize filter paper | [83] |
| ZnCl2 (1.6 g/mL) | The high recovery rate of plastic particles (even high-density ones) | High cost, hazardous to the environment | [82] |
| ZnBr22 (1.71 g/mL) | Good recoveries for larger MPs | Expensive, severely hazardous to the environment | [81] |
| Canola oil | High recovery rate | Interfere the chemical analysis of MPs | [85] |
| NaBr | Low cost, Environmental friendly | Not applicable to some types of plastics | [84] |
After floatation, the supernatant is filtered with a mesh size of 0.47~2 μm through vacuum suction filter equipment [72][86]. More MPs would be collected by the use of a 0.7 μm or smaller filter [50] as compared with larger ones (e.g., 2 μm or larger mesh size) [76]. At last, the suspected items similar to MPs can be identified according to the same procedures as the seawater sample [87].
3.3. Techniques for MPs Studies in Organisms
The existence of MPs in marine organisms have been studied in birds [88], fish [55], mussel [89], terrestrial invertebrate [90], earthworms [91], bivalves [92], and invertebrates [93], in terms of the whole animal [93], digestive tract [88], all soft tissue [92], and gastrointestinal tract [94]. Two sampling devices are commonly used to collect organism samples in water [95]. One is the direct use of plankton nets such as Bongo nets and shrimp nets, the residual organisms on the net can be collected after being washed with 10% formalin solution. The other is that collect a certain volume of the water column with a clean container and then extract the organism from the water column for further analysis.
Preservation methods used in biological organisms are stricter than seawater or sediment samples. Immediately, frozen the organism samples at −20 °C [96] or preserved them in formalin solution [97] or ethanol [98] (Table 4). The pretreatment of organism samples is a combination of digestion and filtration. According to the size of the biological sample, digestion can be divided into two types. One is the complete digestion (i.e., digestion of the whole biological sample [93], which applies to small organisms such as mussels [89] and oysters [99]. The other is partial digestion dissolves part of the soft tissue or digestive organs obtained by dissection and tissue section [100], which applies to larger biological samples such as fish [101], waterfowl [102], and others (e.g., turtles) [103].
Table 4. Common methods used in the sampling and identification of MPs in organisms.
| Items | Parameters | Refs |
|---|---|---|
| Biological species | Birds; Fish; Mussel; Bivalves; Invertebrates | [55][89][93] |
| Tissue type | Digestive tract; All soft tissue; Whole animal; Gastrointestinal tract | [93][94] |
| Preservation | Frozen at −20 °C; Ethanol; Formalin | [60][89] |
| Digestion | Acid (HNO3); Enzymatic; H2O2; NaClO; Alkaline | [56][66] |
| Digestion temperature | 25 ℃; 65 °C; 100 ℃ | [76][104][105] |
| Separation | Flotation; Filtration; Sieving | [71][104] |
| Identification | Stereomicroscope; SEM; μ-FTIR or μ-Raman | [52][60][61] |
Digestion is classified as acid, alkaline, and enzyme digestion [67] (Table 5). Based on the previous studies, acid digestion solutions mainly include H2O2 (30% or 35%) [47], HNO3 (65% or 69%) [56], NaClO [66] and other combined acids (i.e., 5% or 37% HCl, 65% or 69% HNO3, 40% HF) [66][104] at room temperature (usually 25 °C) [104], 65 °C [106], and 100 °C [76], in which 65 °C is the most frequently used. They show different advantages and disadvantages. For example, H2O2 has no effects on plastic particles, but it cannot remove all organic materials; NaClO can completely eliminate organisms, but it fails to reach an optimum digestion efficiency in a short time [66], and the concentrated acid, especially corrosive acid may adversely impair the structural and chemical integrity of MPs and may result in strong chemical hazards to the environment as well [56][66][76]. By comparison, enzymatic digestion has high efficiency, but it may destroy nylon fiber plastics and cannot completely digest biogenic materials [92][96]. Once the biological tissues are completely digested, a vacuum suction filter device is further used to filter the digested mixtures, and the identification of MPs follows the procedures of seawater samples.
Table 5. Advantages and disadvantages of different digestion methods in MPs study.
| Digestion Method | Digestion Solution | Advantage | Disadvantage | Refs |
|---|---|---|---|---|
| Acid digestion | HCl (5% or 37%) | Fully digested fish tissues | May degrade some plastic polymers | [66] |
| HNO3 (65% or 69%) | Full digestion and inexpensive | Corrosive acid, may degrade plastics | [92] | |
| HClO4 (65% or 68%) | Common reagent in labs, easy to obtain | Corrosive acid, may degrade plastics | [105] | |
| HF (40%) | Full digestion | may impact the structural or chemical integrity of plastics | [56] | |
| H2O2 (30% or 35%) | No effect on any plastic particles | Incomplete digestion | [96] | |
| Alkaline digestion | KOH (10%) | The most suitable solution to digest fish tissue | May hinder FTIR if not cleaned | [66] |
| NaOH (5M) | Low chemical harm and low cost | Unknown effects on polymer types | [55] | |
| Enzymatic digestion | Enzyme (proteinase and collagenase) | High digestion efficiency | Incomplete digestion and may be worse for nylon fibres | [60] |
3.4. Research Methods of MPs in Air
Air is a mixture of gases and suspended particles [107]. Recent literature suggests the presence of MPs in the air [28]. There are quite limited studies on the occurrence of MPs in the air [108][109][110]. In general, atmospheric fallout with dry and wet deposition can be collected through a stainless-steel funnel, which connects with a 20 L glass bottle at the bottom [111][112]. Once the fallout was collected, rinse the funnel with reverse osmosed water to recover all particles (including MPs) adhering to the funnel. After being rinsed, all samples in a 20 L glass bottle were immediately filtered on quartz fiber GF/A (with a size of 1.6 μm). At last, observe the MPs on the filter using a stereomicroscope and measure the length of the fibers during the enumeration (e.g., the software ‘Histolab (Microvision instruments—Evry—France)’ coupled with the stereomicroscope) [111][112]. The other method for indoor air sampling is as follows, a pump (Stand-alone sampling pump GH300, Deltanova, France) at 8 L/min allowed to sample indoor air on quartz fiber GF/A (1.6 μm, 47 mm), and the height and time of samplings depends on the location selected. Next, 50 mL zinc chloride solution (1.6 g/cm3) is used as a flotation solution to separate the 5.5 mg sample in a separation funnel. Next, a subsample of 1 mL was taken from the homogenized floating fraction and filtered on quartz fiber GF/A (1.6 μm, 47 mm). At last, all samples were observed by stereomicroscope and qualitatively analyzed by using μ-FTIR or μ-Raman [111].
3.5. Measurement of MPs in Other System
In addition to the natural environmental media, there are some techniques for detecting the MPs in other specific media, such as wastewater [65], drinking water or tap water [113][114], and sludge [115]. Among them, grabbing samples (i.e., single samples collected at one time) is the most typical method for collecting wastewater samples from wastewater treatment plants [43]. In detail, a telescopic sampling rod (Telescoop, Waterra Ltd., Solihull, UK) was used to collect wastewater samples, which can be further transferred into a 50 mL centrifuge tube for centrifugation at 2038× g for 2 min, and then the resulting pellet was treated with 30 mL of Fenton’s reagent [61]. By comparison, the sampling and pretreatment of tap water and drinking water is much easier because their components are simpler than wastewater. The samples are mainly taken from bottled water and water plants [116][117]. Next, the samples will be processed in terms of staining [118], filtration [119], or digestion [120] for subsequent observation. Since the sludge in wastewater treatment plants commonly contains complex components, they are usually to be air-dried, sieved, and kept as soil samples [34] and then analyzed using the same methods as that of MPs analysis in soil.
This entry is adapted from the peer-reviewed paper 10.3390/su142114338
References
- North, E.J.; Halden, R.U. Plastics and environmental health: The road ahead. Rev. Environ. Health 2013, 28, 1–8.
- Bao, R.; Wang, Z.; Qi, H.; Mehmood, T.; Cai, M.; Zhang, Y.; Yang, R.; Peng, L.; Liu, F. Occurrence and distribution of microplastics in wastewater treatment plant in a tropical region of China. J. Clean. Prod. 2022, 349, 131454.
- Ivleva, N.P.; Wiesheu, A.C.; Niessner, R. Microplastic in aquatic ecosystems. Angew. Chem. Int. Ed. 2017, 56, 1720–1739.
- Thompson, R.C.; Olsen, Y.; Mitchell, R.P.; Davis, A.; Rowland, S.J.; John, A.W.G.; McGonigle, D.; Russell, A.E. Lost at sea: Where is all the plastic? Science 2004, 304, 838.
- Sridharan, S.; Kumar, M.; Bolan, N.S.; Singh, L.; Kumar, S.; Kumar, R.; You, S. Are microplastics destabilizing the global network of terrestrial and aquatic ecosystem services? Environ. Res. 2021, 198, 111243.
- Wang, L.; Wu, W.-M.; Bolan, N.S.; Tsang, D.C.; Li, Y.; Qin, M.; Hou, D. Environmental fate, toxicity and risk management strategies of nanoplastics in the environment: Current status and future perspectives. J. Hazard. Mater. 2021, 401, 123415.
- Cai, H.; Xu, E.G.; Du, F.; Li, R.; Liu, J.; Shi, H. Analysis of environmental nanoplastics: Progress and challenges. Chem. Eng. J. 2021, 410, 128208.
- Fang, C.; Sobhani, Z.; Zhang, X.; McCourt, L.; Routley, B.; Gibson, C.T.; Naidu, R. Identification and visualisation of microplastics/nanoplastics by Raman imaging (iii): Algorithm to cross-check multi-images. Water Res. 2021, 194, 116913.
- Peiponen, K.-E.; Räty, J.; Ishaq, U.; Pélisset, S.; Ali, R. Outlook on optical identification of micro- and nanoplastics in aquatic environments. Chemosphere 2019, 214, 424–429.
- Lambert, S.; Sinclair, C.; Boxall, A. Occurrence, degradation, and effect of polymer-based materials in the environment. In Reviews of Environmental Contamination and Toxicology; Whitacre, D.M., Ed.; Springer International Publishing: Cham, Switzerland, 2014; Volume 227, pp. 1–53.
- Sudhakar, M.; Trishul, A.; Doble, M.; Suresh Kumar, K.; Syed Jahan, S.; Inbakandan, D.; Viduthalai, R.R.; Umadevi, V.R.; Sriyutha Murthy, P.; Venkatesan, R. Biofouling and biodegradation of polyolefins in ocean waters. Polym. Degrad. Stab. 2007, 92, 1743–1752.
- Sudhakar, M.; Priyadarshini, C.; Doble, M.; Sriyutha Murthy, P.; Venkatesan, R. Marine bacteria mediated degradation of nylon 66 and 6. Int. Biodeterior. Biodegrad. 2007, 60, 144–151.
- Sudhakar, M.; Doble, M.; Murthy, P.S.; Venkatesan, R. Marine microbe-mediated biodegradation of low- and high-density polyethylenes. Int. Biodeterior. Biodegrad. 2008, 61, 203–213.
- Krzan, A.; Hemjinda, S.; Miertus, S.; Corti, A.; Chiellini, E. Standardization and certification in the area of environmentally degradable plastics. Polym. Degrad. Stab. 2006, 91, 2819–2833.
- Born, M.P.; Brüll, C. From model to nature—A review on the transferability of marine (micro-) plastic fragmentation studies. Sci. Total Environ. 2022, 811, 151389.
- Rutkowska, M.; Heimowska, A.; Krasowska, K.; Janik, H. Biodegradability of polyethylene starch blends in sea water. Pol. J. Environ. Stud. 2002, 11, 267–271.
- Du, H.; Xie, Y.; Wang, J. Microplastic degradation methods and corresponding degradation mechanism: Research status and future perspectives. J. Hazard. Mater. 2021, 418, 126377.
- Shan, J.; Zhao, J.; Zhang, Y.; Liu, L.; Wu, F.; Wang, X. Simple and rapid detection of microplastics in seawater using hyperspectral imaging technology. Anal. Chim. Acta 2019, 1050, 161–168.
- Cincinelli, A.; Scopetani, C.; Chelazzi, D.; Lombardini, E.; Martellini, T.; Katsoyiannis, A.; Fossi, M.C.; Corsolini, S. Microplastic in the surface waters of the Ross Sea (Antarctica): Occurrence, distribution and characterization by FTIR. Chemosphere 2017, 175, 391–400.
- Zheng, Y.; Li, J.; Sun, C.; Cao, W.; Wang, M.; Jiang, F.; Ju, P. Comparative study of three sampling methods for microplastics analysis in seawater. Sci. Total Environ. 2021, 765, 144495.
- Liu, H.; Yang, X.; Liu, G.; Liang, C.; Xue, S.; Chen, H.; Ritsema, C.J.; Geissen, V. Response of soil dissolved organic matter to microplastic addition in Chinese loess soil. Chemosphere 2017, 185, 907–917.
- Ziajahromi, S.; Drapper, D.; Hornbuckle, A.; Rintoul, L.; Leusch, F.D.L. Microplastic pollution in a stormwater floating treatment wetland: Detection of tyre particles in sediment. Sci. Total Environ. 2020, 713, 136356.
- Pitt, J.A.; Kozal, J.S.; Jayasundara, N.; Massarsky, A.; Trevisan, R.; Geitner, N.; Wiesner, M.; Levin, E.D.; Di Giulio, R.T. Uptake, tissue distribution, and toxicity of polystyrene nanoparticles in developing zebrafish (Danio rerio). Aquat. Toxicol. 2018, 194, 185–194.
- von Friesen, L.W.; Granberg, M.E.; Hassellöv, M.; Gabrielsen, G.W.; Magnusson, K. An efficient and gentle enzymatic digestion protocol for the extraction of microplastics from bivalve tissue. Mar. Pollut. Bull. 2019, 142, 129–134.
- Cai, L.; Wang, J.; Peng, J.; Tan, Z.; Zhan, Z.; Tan, X.; Chen, Q. Characteristic of microplastics in the atmospheric fallout from Dongguan city, China: Preliminary research and first evidence. Environ. Sci. Pollut. Res. 2017, 24, 24928–24935.
- Chen, G.; Fu, Z.; Yang, H.; Wang, J. An overview of analytical methods for detecting microplastics in the atmosphere. TrAC Trends Anal. Chem. 2020, 130, 115981.
- Fu, Z.; Chen, G.; Wang, W.; Wang, J. Microplastic pollution research methodologies, abundance, characteristics and risk assessments for aquatic biota in China. Environ. Pollut. 2020, 266, 115098.
- Mehmood, T.; Peng, L. Polyethylene scaffold net and synthetic grass fragmentation: A source of microplastics in the atmosphere? J. Hazard. Mater. 2022, 429, 128391.
- Lin, J.; Xu, X.-P.; Yue, B.-Y.; Li, Y.; Zhou, Q.-Z.; Xu, X.-M.; Liu, J.-Z.; Wang, Q.-Q.; Wang, J.-H. A novel thermoanalytical method for quantifying microplastics in marine sediments. Sci. Total Environ. 2021, 760, 144316.
- Mai, L.; Bao, L.-J.; Shi, L.; Wong, C.S.; Zeng, E.Y. A review of methods for measuring microplastics in aquatic environments. Environ. Sci. Pollut. Res. 2018, 25, 11319–11332.
- Ceccarini, A.; Corti, A.; Erba, F.; Modugno, F.; La Nasa, J.; Bianchi, S.; Castelvetro, V. The hidden microplastics: New insights and figures from the thorough separation and characterization of microplastics and of their degradation byproducts in coastal sediments. Environ. Sci. Technol. 2018, 52, 5634–5643.
- Coppock, R.L.; Cole, M.; Lindeque, P.K.; Queirós, A.M.; Galloway, T.S. A small-scale, portable method for extracting microplastics from marine sediments. Environ. Pollut. 2017, 230, 829–837.
- Song, Y.K.; Hong, S.H.; Eo, S.; Jang, M.; Han, G.M.; Isobe, A.; Shim, W.J. Horizontal and vertical distribution of microplastics in Korean coastal waters. Environ. Sci. Technol. 2018, 52, 12188–12197.
- Li, Q.; Wu, J.; Zhao, X.; Gu, X.; Ji, R. Separation and identification of microplastics from soil and sewage sludge. Environ. Pollut. 2019, 254, 113076.
- Nguyen, B.; Claveau-Mallet, D.; Hernandez, L.M.; Xu, E.G.; Farner, J.M.; Tufenkji, N. Separation and analysis of microplastics and nanoplastics in complex environmental samples. Acc. Chem. Res. 2019, 52, 858–866.
- Liu, M.; Lu, S.; Chen, Y.; Cao, C.; Bigalke, M.; He, D. Analytical methods for microplastics in environments: Current advances and challenges. In Microplastics in Terrestrial Environments: Emerging Contaminants and Major Challenges; He, D., Luo, Y., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 3–24.
- Zhang, J.; Gao, D.; Li, Q.; Zhao, Y.; Li, L.; Lin, H.; Bi, Q.; Zhao, Y. Biodegradation of polyethylene microplastic particles by the fungus Aspergillus flavus from the guts of wax moth Galleria mellonella. Sci. Total Environ. 2020, 704, 135931.
- Schwaferts, C.; Niessner, R.; Elsner, M.; Ivleva, N.P. Methods for the analysis of submicrometer- and nanoplastic particles in the environment. TrAC Trends Anal. Chem. 2019, 112, 52–65.
- Vermeiren, P.; Muñoz, C.; Ikejima, K. Microplastic identification and quantification from organic rich sediments: A validated laboratory protocol. Environ. Pollut. 2020, 262, 114298.
- Karlsson, T.M.; Kärrman, A.; Rotander, A.; Hassellöv, M. Comparison between manta trawl and in situ pump filtration methods, and guidance for visual identification of microplastics in surface waters. Environ. Sci. Pollut. Res. 2020, 27, 5559–5571.
- Fu, D.; Zhang, Q.; Fan, Z.; Qi, H.; Wang, Z.; Peng, L. Aged microplastics polyvinyl chloride interact with copper and cause oxidative stress towards microalgae Chlorella vulgaris. Aquat. Toxicol. 2019, 216, 105319.
- Hahn, A.; Gerdts, G.; Völker, C.; Niebühr, V. Using FTIRS as pre-screening method for detection of microplastic in bulk sediment samples. Sci. Total Environ. 2019, 689, 341–346.
- Elkhatib, D.; Oyanedel-Craver, V. A critical review of extraction and identification methods of microplastics in wastewater and drinking water. Environ. Sci. Technol. 2020, 54, 7037–7049.
- Sorensen, L.; Groven, A.S.; Hovsbakken, I.A.; Del Puerto, O.; Krause, D.F.; Sarno, A.; Booth, A.M. UV degradation of natural and synthetic microfibers causes fragmentation and release of polymer degradation products and chemical additives. Sci. Total Environ. 2021, 755, 143170.
- Reichel, J.; Graßmann, J.; Letzel, T.; Drewes, J.E. Systematic development of a simultaneous determination of plastic particle identity and adsorbed organic compounds by thermodesorption–pyrolysis GC/MS (TD-Pyr-GC/MS). Molecules 2020, 25, 4985.
- Li, P.; Li, Q.; Hao, Z.; Yu, S.; Liu, J. Analytical methods and environmental processes of nanoplastics. J. Environ. Sci. 2020, 94, 88–99.
- Gewert, B.; Ogonowski, M.; Barth, A.; MacLeod, M. Abundance and composition of near surface microplastics and plastic debris in the Stockholm Archipelago, Baltic Sea. Mar. Pollut. Bull. 2017, 120, 292–302.
- Di Mauro, R.; Kupchik, M.J.; Benfield, M.C. Abundant plankton-sized microplastic particles in shelf waters of the northern Gulf of Mexico. Environ. Pollut. 2017, 230, 798–809.
- Zhao, S.; Zhu, L.; Li, D. Microplastic in three urban estuaries, China. Environ. Pollut. 2015, 206, 597–604.
- Tsang, Y.Y.; Mak, C.W.; Liebich, C.; Lam, S.W.; Sze, E.T.P.; Chan, K.M. Microplastic pollution in the marine waters and sediments of Hong Kong. Mar. Pollut. Bull. 2017, 115, 20–28.
- Desforges, J.-P.W.; Galbraith, M.; Dangerfield, N.; Ross, P.S. Widespread distribution of microplastics in subsurface seawater in the NE Pacific Ocean. Mar. Pollut. Bull. 2014, 79, 94–99.
- van der Hal, N.; Ariel, A.; Angel, D.L. Exceptionally high abundances of microplastics in the oligotrophic Israeli Mediterranean coastal waters. Mar. Pollut. Bull. 2017, 116, 151–155.
- Anderson, P.J.; Warrack, S.; Langen, V.; Challis, J.K.; Hanson, M.L.; Rennie, M.D. Microplastic contamination in Lake Winnipeg, Canada. Environ. Pollut. 2017, 225, 223–231.
- Castillo, A.B.; Al-Maslamani, I.; Obbard, J.P. Prevalence of microplastics in the marine waters of Qatar. Mar. Pollut. Bull. 2016, 111, 260–267.
- Güven, O.; Gökdağ, K.; Jovanović, B.; Kıdeyş, A.E. Microplastic litter composition of the Turkish territorial waters of the Mediterranean Sea, and its occurrence in the gastrointestinal tract of fish. Environ. Pollut. 2017, 223, 286–294.
- Miller, M.E.; Kroon, F.J.; Motti, C.A. Recovering microplastics from marine samples: A review of current practices. Mar. Pollut. Bull. 2017, 123, 6–18.
- Masura, J.; Baker, J.; Foster, G.; Arthur, C. Laboratory methods for the analysis of microplastics in the marine environment: Recommendations for quantifying synthetic particles in waters and sediments. NOAA Tech. Memo. NOS-ORR-48 2015.
- Zhang, W.; Zhang, S.; Wang, J.; Wang, Y.; Mu, J.; Wang, P.; Lin, X.; Ma, D. Microplastic pollution in the surface waters of the Bohai Sea, China. Environ. Pollut. 2017, 231, 541–548.
- Song, Y.K.; Hong, S.H.; Jang, M.; Han, G.M.; Rani, M.; Lee, J.; Shim, W.J. A comparison of microscopic and spectroscopic identification methods for analysis of microplastics in environmental samples. Mar. Pollut. Bull. 2015, 93, 202–209.
- Collard, F.; Gilbert, B.; Eppe, G.; Parmentier, E.; Das, K. Detection of anthropogenic particles in fish stomachs: An isolation method adapted to identification by Raman spectroscopy. Arch. Environ. Contam. Toxicol. 2015, 69, 331–339.
- Tagg, A.S.; Sapp, M.; Harrison, J.P.; Ojeda, J.J. Identification and quantification of microplastics in wastewater using focal plane array-based reflectance micro-FT-IR imaging. Anal. Chem. 2015, 87, 6032–6040.
- Green, D.S.; Kregting, L.; Boots, B.; Blockley, D.J.; Brickle, P.; da Costa, M.; Crowley, Q. A comparison of sampling methods for seawater microplastics and a first report of the microplastic litter in coastal waters of Ascension and Falkland Islands. Mar. Pollut. Bull. 2018, 137, 695–701.
- Kedzierski, M.; Falcou-Préfol, M.; Kerros, M.E.; Henry, M.; Pedrotti, M.L.; Bruzaud, S. A machine learning algorithm for high throughput identification of FTIR spectra: Application on microplastics collected in the Mediterranean Sea. Chemosphere 2019, 234, 242–251.
- Aytan, Ü.; Valente, A.; Senturk, Y.; Usta, R.; Sahin, F.B.E.; Mazlum, R.E.; Agirbas, E. First evaluation of neustonic microplastics in Black Sea waters. Mar. Environ. Res. 2016, 119, 22–30.
- Hidayaturrahman, H.; Lee, T.-G. A study on characteristics of microplastic in wastewater of South Korea: Identification, quantification, and fate of microplastics during treatment process. Mar. Pollut. Bull. 2019, 146, 696–702.
- Karami, A.; Golieskardi, A.; Choo, C.K.; Romano, N.; Ho, Y.B.; Salamatinia, B. A high-performance protocol for extraction of microplastics in fish. Sci. Total Environ. 2017, 578, 485–494.
- Kun, W.; Kunde, L.; Dongxing, Y. Research progress on the analysis of microplastics in the environment. Environ. Chem. 2017, 36, 27–36.
- Hidalgo-Ruz, V.; Gutow, L.; Thompson, R.C.; Thiel, M. Microplastics in the marine environment: A review of the methods used for identification and quantification. Environ. Sci. Technol. 2012, 46, 3060–3075.
- Vianello, A.; Boldrin, A.; Guerriero, P.; Moschino, V.; Rella, R.; Sturaro, A.; Da Ros, L. Microplastic particles in sediments of Lagoon of Venice, Italy: First observations on occurrence, spatial patterns and identification. Estuar. Coast. Shelf Sci. 2013, 130, 54–61.
- Fries, E.; Dekiff, J.H.; Willmeyer, J.; Nuelle, M.-T.; Ebert, M.; Remy, D. Identification of polymer types and additives in marine microplastic particles using pyrolysis-GC/MS and scanning electron microscopy. Environ. Sci. Process. Impacts 2013, 15, 1949–1956.
- Li, J.; Liu, H.; Chen, J.P. Microplastics in freshwater systems: A review on occurrence, environmental effects, and methods for microplastics detection. Water Res. 2018, 137, 362–374.
- Yu, X.; Peng, J.; Wang, J.; Wang, K.; Bao, S. Occurrence of microplastics in the beach sand of the Chinese inner sea: The Bohai Sea. Environ. Pollut. 2016, 214, 722–730.
- Retama, I.; Jonathan, M.P.; Shruti, V.C.; Velumani, S.; Sarkar, S.K.; Roy, P.D.; Rodríguez-Espinosa, P.F. Microplastics in tourist beaches of Huatulco Bay, Pacific coast of southern Mexico. Mar. Pollut. Bull. 2016, 113, 530–535.
- Näkki, P.; Setälä, O.; Lehtiniemi, M. Bioturbation transports secondary microplastics to deeper layers in soft marine sediments of the northern Baltic Sea. Mar. Pollut. Bull. 2017, 119, 255–261.
- Fischer, E.K.; Paglialonga, L.; Czech, E.; Tamminga, M. Microplastic pollution in lakes and lake shoreline sediments–a case study on Lake Bolsena and Lake Chiusi (central Italy). Environ. Pollut. 2016, 213, 648–657.
- Nel, H.A.; Dalu, T.; Wasserman, R.J. Sinks and sources: Assessing microplastic abundance in river sediment and deposit feeders in an Austral temperate urban river system. Sci. Total Environ. 2018, 612, 950–956.
- Akhbarizadeh, R.; Moore, F.; Keshavarzi, B.; Moeinpour, A. Microplastics and potentially toxic elements in coastal sediments of Iran’s main oil terminal (Khark Island). Environ. Pollut. 2017, 220, 720–731.
- Maes, T.; Jessop, R.; Wellner, N.; Haupt, K.; Mayes, A.G. A rapid-screening approach to detect and quantify microplastics based on fluorescent tagging with Nile Red. Sci. Rep. 2017, 7, 44501.
- Yu, X.; Ladewig, S.; Bao, S.; Toline, C.A.; Whitmire, S.; Chow, A.T. Occurrence and distribution of microplastics at selected coastal sites along the southeastern United States. Sci. Total Environ. 2018, 613–614, 298–305.
- Wahl, A.; Le Juge, C.; Davranche, M.; El Hadri, H.; Grassl, B.; Reynaud, S.; Gigault, J. Nanoplastic occurrence in a soil amended with plastic debris. Chemosphere 2021, 262, 127784.
- Quinn, B.; Murphy, F.; Ewins, C. Validation of density separation for the rapid recovery of microplastics from sediment. Anal. Methods 2017, 9, 1491–1498.
- Zobkov, M.; Esiukova, E. Microplastics in Baltic bottom sediments: Quantification procedures and first results. Mar. Pollut. Bull. 2017, 114, 724–732.
- Abayomi, O.A.; Range, P.; Al-Ghouti, M.A.; Obbard, J.P.; Almeer, S.H.; Ben-Hamadou, R. Microplastics in coastal environments of the Arabian Gulf. Mar. Pollut. Bull. 2017, 124, 181–188.
- Li, C.; Cui, Q.; Zhang, M.; Vogt, R.D.; Lu, X. A commonly available and easily assembled device for extraction of bio/non-degradable microplastics from soil by flotation in NaBr solution. Sci. Total Environ. 2021, 759, 143482.
- Crichton, E.M.; Noël, M.; Gies, E.A.; Ross, P.S. A novel, density-independent and FTIR-compatible approach for the rapid extraction of microplastics from aquatic sediments. Anal. Methods 2017, 9, 1419–1428.
- Blumenröder, J.; Sechet, P.; Kakkonen, J.E.; Hartl, M.G.J. Microplastic contamination of intertidal sediments of Scapa Flow, Orkney: A first assessment. Mar. Pollut. Bull. 2017, 124, 112–120.
- Sruthy, S.; Ramasamy, E.V. Microplastic pollution in Vembanad Lake, Kerala, India: The first report of microplastics in lake and estuarine sediments in India. Environ. Pollut. 2017, 222, 315–322.
- Terepocki, A.K.; Brush, A.T.; Kleine, L.U.; Shugart, G.W.; Hodum, P. Size and dynamics of microplastic in gastrointestinal tracts of Northern Fulmars (Fulmarus glacialis) and Sooty Shearwaters (Ardenna grisea). Mar. Pollut. Bull. 2017, 116, 143–150.
- Kolandhasamy, P.; Su, L.; Li, J.; Qu, X.; Jabeen, K.; Shi, H. Adherence of microplastics to soft tissue of mussels: A novel way to uptake microplastics beyond ingestion. Sci. Total Environ. 2018, 610–611, 635–640.
- Maxwell, S.H.; Melinda, K.F.; Matthew, G. Counterstaining to separate nile red-stained microplastic particles from terrestrial invertebrate biomass. Environ. Sci. Technol. 2020, 54, 5580–5588.
- Wang, J.; Coffin, S.; Sun, C.; Schlenk, D.; Gan, J. Negligible effects of microplastics on animal fitness and HOC bioaccumulation in earthworm Eisenia fetida in soil. Environ. Pollut. 2019, 249, 776–784.
- Courtene-Jones, W.; Quinn, B.; Murphy, F.; Gary, S.F.; Narayanaswamy, B.E. Optimisation of enzymatic digestion and validation of specimen preservation methods for the analysis of ingested microplastics. Anal. Methods 2017, 9, 1437–1445.
- Taylor, M.L.; Gwinnett, C.; Robinson, L.F.; Woodall, L.C. Plastic microfibre ingestion by deep-sea organisms. Sci. Rep. 2016, 6, 33997.
- Wagner, J.; Wang, Z.-M.; Ghosal, S.; Rochman, C.; Gassel, M.; Wall, S. Novel method for the extraction and identification of microplastics in ocean trawl and fish gut matrices. Anal. Methods 2017, 9, 1479–1490.
- Desforges, J.-P.W.; Galbraith, M.; Ross, P.S. Ingestion of microplastics by zooplankton in the northeast Pacific Ocean. Arch. Environ. Contam. Toxicol. 2015, 69, 320–330.
- Hermsen, E.; Pompe, R.; Besseling, E.; Koelmans, A.A. Detection of low numbers of microplastics in North Sea fish using strict quality assurance criteria. Mar. Pollut. Bull. 2017, 122, 253–258.
- Vendel, A.L.; Bessa, F.; Alves, V.E.N.; Amorim, A.L.A.; Patrício, J.; Palma, A.R.T. Widespread microplastic ingestion by fish assemblages in tropical estuaries subjected to anthropogenic pressures. Mar. Pollut. Bull. 2017, 117, 448–455.
- Alomar, C.; Estarellas, F.; Deudero, S. Microplastics in the Mediterranean Sea: Deposition in coastal shallow sediments, spatial variation and preferential grain size. Mar. Environ. Res. 2016, 115, 1–10.
- Sussarellu, R.; Suquet, M.; Thomas, Y.; Lambert, C.; Fabioux, C.; Pernet Marie Eve, J.; Le Goïc, N.; Quillien, V.; Mingant, C.; Epelboin, Y.; et al. Oyster reproduction is affected by exposure to polystyrene microplastics. Proc. Natl. Acad. Sci. USA 2016, 113, 2430–2435.
- Cole, M.; Lindeque, P.; Halsband, C.; Galloway, T.S. Microplastics as contaminants in the marine environment: A review. Mar. Pollut. Bull. 2011, 62, 2588–2597.
- Romeo, T.; Pietro, B.; Pedà, C.; Consoli, P.; Andaloro, F.; Fossi, M.C. First evidence of presence of plastic debris in stomach of large pelagic fish in the Mediterranean Sea. Mar. Pollut. Bull. 2015, 95, 358–361.
- English, M.D.; Robertson, G.J.; Avery-Gomm, S.; Pirie-Hay, D.; Roul, S.; Ryan, P.C.; Wilhelm, S.I.; Mallory, M.L. Plastic and metal ingestion in three species of coastal waterfowl wintering in Atlantic Canada. Mar. Pollut. Bull. 2015, 98, 349–353.
- Mascarenhas, R.; Santos, R.; Zeppelini, D. Plastic debris ingestion by sea turtle in Paraíba, Brazil. Mar. Pollut. Bull. 2004, 49, 354–355.
- Santana, M.F.M.; Ascer, L.G.; Custódio, M.R.; Moreira, F.T.; Turra, A. Microplastic contamination in natural mussel beds from a Brazilian urbanized coastal region: Rapid evaluation through bioassessment. Mar. Pollut. Bull. 2016, 106, 183–189.
- Li, J.; Qu, X.; Su, L.; Zhang, W.; Yang, D.; Kolandhasamy, P.; Li, D.; Shi, H. Microplastics in mussels along the coastal waters of China. Environ. Pollut. 2016, 214, 177–184.
- Dehaut, A.; Cassone, A.-L.; Frère, L.; Hermabessiere, L.; Himber, C.; Rinnert, E.; Rivière, G.; Lambert, C.; Soudant, P.; Huvet, A.; et al. Microplastics in seafood: Benchmark protocol for their extraction and characterization. Environ. Pollut. 2016, 215, 223–233.
- Mehmood, T.; Ahmad, I.; Bibi, S.; Mustafa, B.; Ali, I. Insight into monsoon for shaping the air quality of Islamabad, Pakistan: Comparing the magnitude of health risk associated with PM10 and PM2. 5 exposure. J. Air Waste Manag. Assoc. 2020, 70, 1340–1355.
- Gasperi, J.; Wright, S.L.; Dris, R.; Collard, F.; Mandin, C.; Guerrouache, M.; Langlois, V.; Kelly, F.J.; Tassin, B. Microplastics in air: Are we breathing it in? Curr. Opin. Environ. Sci. Health 2018, 1, 1–5.
- Gaston, E.; Woo, M.; Steele, C.; Sukumaran, S.; Anderson, S. Microplastics differ between indoor and outdoor air masses: Insights from multiple microscopy methodologies. Appl. Spectrosc. 2020, 74, 1079–1098.
- Wright, S.L.; Ulke, J.; Font, A.; Chan, K.L.A.; Kelly, F.J. Atmospheric microplastic deposition in an urban environment and an evaluation of transport. Environ. Int. 2020, 136, 105411.
- Dris, R.; Gasperi, J.; Mirande, C.; Mandin, C.; Guerrouache, M.; Langlois, V.; Tassin, B. A first overview of textile fibers, including microplastics, in indoor and outdoor environments. Environ. Pollut. 2017, 221, 453–458.
- Dris, R.; Gasperi, J.; Saad, M.; Mirande, C.; Tassin, B. Synthetic fibers in atmospheric fallout: A source of microplastics in the environment? Mar. Pollut. Bull. 2016, 104, 290–293.
- Sarkar, D.J.; Das Sarkar, S.; Das, B.K.; Praharaj, J.K.; Mahajan, D.K.; Purokait, B.; Mohanty, T.R.; Mohanty, D.; Gogoi, P.; Kumar, V.S.; et al. Microplastics removal efficiency of drinking water treatment plant with pulse clarifier. J. Hazard. Mater. 2021, 413, 125347.
- Tong, H.; Jiang, Q.; Hu, X.; Zhong, X. Occurrence and identification of microplastics in tap water from China. Chemosphere 2020, 252, 126493.
- Bretas Alvim, C.; Bes-Piá, M.A.; Mendoza-Roca, J.A. Separation and identification of microplastics from primary and secondary effluents and activated sludge from wastewater treatment plants. Chem. Eng. J. 2020, 402, 126293.
- Pivokonsky, M.; Cermakova, L.; Novotna, K.; Peer, P.; Cajthaml, T.; Janda, V. Occurrence of microplastics in raw and treated drinking water. Sci. Total Environ. 2018, 643, 1644–1651.
- Wiesheu, A.C.; Anger, P.M.; Baumann, T.; Niessner, R.; Ivleva, N.P. Raman microspectroscopic analysis of fibers in beverages. Anal. Methods 2016, 8, 5722–5725.
- Mason, S.A.; Welch, V.G.; Neratko, J. Synthetic polymer contamination in bottled water. Front. Chem. 2018, 6, 407.
- Schymanski, D.; Goldbeck, C.; Humpf, H.-U.; Fürst, P. Analysis of microplastics in water by micro-Raman spectroscopy: Release of plastic particles from different packaging into mineral water. Water Res. 2018, 129, 154–162.
- Mintenig, S.M.; Löder, M.G.J.; Primpke, S.; Gerdts, G. Low numbers of microplastics detected in drinking water from ground water sources. Sci. Total Environ. 2019, 648, 631–635.
This entry is offline, you can click here to edit this entry!
