A promising approach to overcome current therapies limitations is the use of new therapeutic agents (molecules or nanoparticles) that sensitize cancer cells to radiotherapy (RT) also known as dose enhancers and radiosensitizers. This enhanced radiation local absorption in combination with the accumulation of high Z-elements in the irradiated cancer tissue results in a larger production of harmful diffused photons, photoelectrons, Auger electrons, Compton electrons, and radical species [
19]. AGuIX particles were firstly synthesized in 2011 [
20] to increase the radiobiological effect of high-energy radiation in the tumor. This technology is composed of very small (average size 5 nm diameter) polysyloxane particles with chelated cyclic gadolinium covalently grafted into the inorganic matrix [
21] (
Figure 1). In preclinical experiments, AGuIX NPs showed high radiosensitizing and anti-tumor properties [
22] that combined with a solid and reproducible synthesis process favored their evaluation in clinical settings.
NBTXR3 technology is a novel radio sensitizer comprising crystalized hafnium oxide (HfO2) nanoparticles, locally injected into tumor tissue and activated by RT. HfO2 nanoparticles possess excellent x-ray absorption coefficient because of the high electron-density elements composing the particles and acceptable safety. The particles are 50 nm in size and negatively charged thanks to a phosphate coating applied to maintain colloidal stability [
36]. NBTXR3 followed by RT could improve the treatment of advanced or borderline-resectable cancers compared to RT alone [
36]. Preclinical studies have shown that NBTXR3 working mechanism is mostly physical without targeting specific biological pathways, and its use could be extended to many types of cancer. The system was tested in patients with head and neck squamous cell carcinoma exploring a dose escalation setting [
37]. Within 7 weeks after NBTXR3 injection, nanoparticles in the surrounding tissues disappeared, showing that the system was well tolerated. Additionally, this study [
37] showed that one intratumor administration of NBTXR3 before radiotherapy could yield remarkable local tolerance, homogeneous dispersion of the particles in the tumor tissue, no leakage, and showed promising signs of anticancer activity in terms of pathological responses. In another study [
38] dose optimization and side effects were evaluated for NBTXR3 in association with RT for recurrent and inoperable non-small cell lung cancer patients.
Superparamagnetic iron oxide nanoparticles (SPION) found their application in the biomedical field because of their theranostic properties, since they can allow for MRI and thermo-ablation. SPION working mechanism depends on an external alternate magnetic field determining their action only in the sick tissue. The synthesis of these particles is based on the nanomanipulation of magnetite and maghemite [
47] and their dispersion is obtained through surface modifications (capping) based on organic molecules [
48] and polymeric (i.e., polyethylene glycol) [
49] surface functionalization. Their size ranges from 20 to 150 nm [
47] and the mechanism of tumor accumulation is based on intratumoral injection [
50] or EPR [
51] following IV administration. However, in preclinical studies they were object of intense studies to engineering their surface with targeting molecules [
52] to increase their residence time and internalization in cancer cells. These manipulations focused also on conjugating therapeutic agents on their surface comprising both small molecules [
53] and biologics [
50]. Attempts at exploiting polarized magnetic fields to increase their tumor targeting were proposed as well [
54]. Finally, they were often used to implement the properties of other delivery platforms in hybrid synthetic settings [
55]. Their translational use was deeply investigated mostly for their ability to enhance the MRI resolution, even though their toxicity related to DNA damage and reactive oxygen species formation limited their large application [
56]. FDA-approved formulations of SPION are currently intended as iron replacement and they are secondarily used as contrast agents for kidney imaging [
57,
58]. In this scenario, the only phase 3 and 4 trial focused on this technology aimed at understanding the SPION ability to detect lymphatic metastases in breast [
59] and pancreatic cancer [
60] after IV infusion. The latter trial showed, in comparison with traditional histology, a matching of the 2 methods higher than the 80%. Ongoing clinical trials aim to investigate the efficacy of locally injected SPION magnetic hyperthermia against brain and prostate tumors. In a phase I clinical trial, the authors showed the effect of thermoablation to treat prostate cancer by local injection of SPION and further thermoablation induction under magnetic field application [
61]. Untargeted SPION (Ferumoxytol) are currently evaluated for treatment of primary and metastatic hepatic cancers [
62]. The radiotherapy with SPION supported by magnetic resonance imaging guided linear accelerator allowed to detect and maximize avoidance of residual functionally active hepatic parenchyma from over-the-threshold irradiation thus minimizing SBRT liver damages because of stereotactic body radiation therapy in patients with pre-existing hepatic conditions. The safety, efficacy and tolerability of SPION in combination with spinning magnetic field (SMF) and neoadjuvant chemotherapy in osteosarcoma patients is currently evaluated in a Phase I clinical trial [
63]. The study comprises intra-tumor injection of SPIONs followed by SMF and conventional neoadjuvant chemotherapy from day 1. The authors declare a synergistic effect of SPIONs/SMF with neoadjuvant chemotherapy in increasing cancer cell killing and improving the ratio of limb retention (amputation).
The folate receptor alpha (FRα) represents a promising target in oncology because of its over-expression in tumors (i.e., ovarian, breast and lung cancers), low and restricted distribution in normal tissues [
68], emerging insights about its tumor promoting functions, and association with patient prognosis. ELU001 is a new molecular C’Dot Drug Conjugate (CDC). ELU001 comprises a very small silica core (6 nm) functionalized with ~12 folic acid targeting moieties and ~22 exatecan topoisomerase-1 inhibitor payloads linked to via Cathepsin-B cleavable linkers covalently bound to the surface of the nanoparticles. Because of their small size, ELU001 are characterized by tumor penetration ability via receptor-mediated endocytosis and are rapidly eliminated by the kidneys. ELU001 high avidity is believed to promote internalization into FRα over-expressing cancer cells, selectively delivering its therapeutic payload. The first Phase I/II clinical trial [
69] dedicated to ELU-FRα-1 is under recruiting phase for advanced, recurrent or refractory FRα over-expressing tumors, considered being topoisomerase 1 inhibitor-sensitive [
70] and with no other therapeutic options available. The study will focus on dose escalation and safety to determine the recommended Phase 2 dose and on the expansion of the patient cohort, where specific cancer types will be evaluated for efficacy and safety of this technology.
3. Polymeric Particles
3.1. CALAA-01
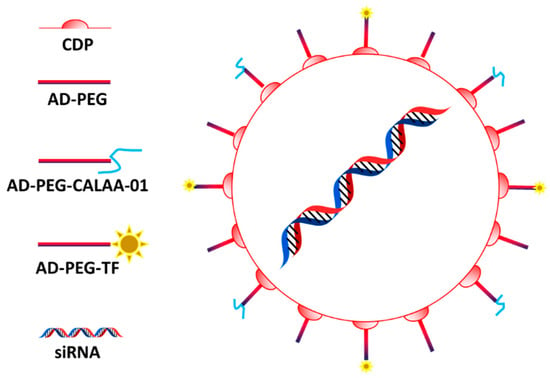
CALAA-01 is considered the first targeted polymeric carrier designed for delivering siRNA tested in human [
71]. A positively charged cyclodextrin core allows the loading the negative nucleic acid payload. Polydispersity, stability, circulation, and targeting properties depend on surface PEGylation and transferrin modification (
Figure 2) [
72].
Figure 2. CALAA-01 structure: The system is composed by a core of cyclodestrin encapsulating the siRNA and functionalized with PEG bearing adamantine (AD-PEG) and transferrin (AD-PEG-TF). Figure from Kurreck et al. [
72].
The use of transferrin as targeting agent is very common in nanomedicine applied to cancer disease, since many tumors over-express the receptor for this molecule [
73]. When fully assembled, the carriers have an average size of 50–70 nm and can be trafficked in the endosomal compartment of the cells after internalization favored by the interaction with transferrin receptor, even though EPR is still fundamental for the particles to reach cancer cells [
71]. After sequestration in the endosomal compartment, the system can disassemble and eventually escape from these vesicles, probably thanks to the positive charge of the cyclodextrin structure. The system was enriched with imidazole groups to favor the buffering properties of the system and induce particle endosomal escape via proton sponge effect [
71]. In a Phase 1 clinical trial [
74], the system was tested for its efficacy and safety against solid tumors (i.e., melanoma) by delivering siRNA against the M2 subunit of ribonucleotide reductase. The trial was terminated before being completed, perhaps because of toxicity issues probably related to the carrier [
75], and the results were not reported. However, a work on this trial was published showing the ability of this technology to induce tumor regression and decreasing the expression of functional ribonucleotide reductase [
76].
3.2. Micelles
Polymeric micelles are nanocarriers composed of a core–shell structure that can be generated via self-assembly of amphiphilic block copolymers [
77]. Because of their self-assembly and amphiphilic nature, micelles are relatively easy to synthesize compared to other technologies and for this reason they are often studied as drug delivery vehicles for poor water-soluble compounds [
78]. Hydrophilic polymers including (but not limited to) PEG, polyoxazolines, chitosan, dextran, and hyaluronic acids can wrap their hydrophobic core, while the therapeutic payloads can be also chemically conjugated to these structures [
79]. Micelle surface can be easily modified in function of the number of monomers used in their fabrication and conjugation of tumor-specific ligands is easy and reproducible [
79,
80]. Regarding their clinical translation, one of the major limitations of micelles is represented by their low mechanical properties and re-assembly when their amount in aqueous solution is below the so-called critical micellar concentration [
77]. Currently, there are several micellar-based nanoformulations approved for improving cancer treatment, and others are in advanced clinical trials. The chemotherapeutic drug Paclitaxel (PTX) that has a very low solubility in water (less than 0.1 µg/mL) is often used in micellar-based systems to avoid Cremophor-EL and ethanol formulations, resulting in adverse reactions like dyspnea, hypotension, angioedema, and generalized hives (2–4% of patients). Genexol-PM, a monomethoxy-poly (ethylene glycol)-block-poly(D,L-lactide) with a mean size of 20–50 nm was approved in clinics in several Asian countries (South Korea, Philippines, India, and Vietnam) for breast cancer, lung cancer, and ovarian cancer [
78]. In Genexol-PM, PTX is physically incorporated into the inner core of the micelles that target the tumors via EPR effect. Currently, Genexol-PM in combination with carboplatin is tested for its safety as an adjuvant treatment in patients with newly diagnosed ovarian cancer that underwent cytoreductive surgery [
81]. Other polymeric micelles represent a promising vehicle for PTX delivery, and they show similarity with Genexol-PM including a core–shell structure with physical entrapment of PTX, PEG coating, small size, and passive targeting through the EPR mechanism. Despite there are limited evidence of superior efficacy of polymeric-PTX compared to Cremophor-PTX, micelles allow administration of an increased PTX dose and offer improved patient safety. Similar platforms worth to mention are Apalea/Paclical (mean size 20–30 nm) and pm-Pac (mean size 20 nm) that target the tumor via EPR [
82,
83]. Apalea/Paclical contains retinoic acid to solubilize PTX and is approved in different countries (Russian Federation, Kazakhstan, and European Union) against platinum-sensitive ovarian, peritoneal, and fallopian tube cancer [
84,
85]. Finally, pm-Pac (mean size 20 nm) successfully passed a Phase III study as first-line treatment in combination with cisplatin for advanced non-small cell lung cancer (NSCLC) [
86,
87]. Trials (Phase 1–3) dedicated to investigate the benefits of micellar-based technologies are currently ongoing in China [
88] and Japan [
89] where they showed similar therapeutic benefits compared to PTX, but less toxicity in treating metastatic or recurrent breast cancer.
Docetaxel (DTX) is another taxane with solubility issues. An analog of Genexol-PM, called Nanoxel-PM micelles loaded with DTX [
90] is currently under clinical trial for efficacy evaluation against different cancers [
91]. Additionally, it is tested as neoadjuvant in patients with breast cancer in combination with DOX and cyclophosphamide [
92] and against salivary duct carcinoma in combination with anti-HER2 monoclonal antibody [
93]. Other versions of this therapeutic formulation are tested in trials as well [
93]. Micelle-based technologies are under clinical trial also for evaluating their ability to deliver cisplatin. NC-6004 micelles have an average size of 30 nm and are composed by PEG and poly-glutamic acid copolymers (PGlu). NC-6004 combination with gemcitabine (GEM) has been studied in NSCLC patients, biliary tract, and bladder cancer patients [
94] resulting in long-lasting antitumor activity and favorable safety profile. Similar data were registered in combination with Pembrolizumab in the treatment of head and neck cancer [
95] and in combination with GEM against advanced solid tumors [
96]. Similar formulations are widely investigated [
97] including the NK012 where the payload SN-38 is covalently attached to the PGlu structure. Here, the efficacy of NK012 was tested in patients with not-resectable colon cancer, but more data are necessary to evaluate its benefits in comparison with the common treatment irinotecan [
98,
99]. A novel epirubicin drug conjugated polymeric micelle (NC-6300; 40–80 nm in diameter) was developed by conjugating the payload to PEG polyaspartate block copolymer through a pH-sensitive linker which enables the selective epirubicin release in tumor. This technology exploits tumor pH as targeting, representing a perfect example of smart technology in clinics in the treatment of cutaneous and not cutaneous angiosarcoma [
100] and advanced, metastatic, or unresectable solid tumors, including soft-tissue sarcomas [
101].
3.3. EP0057
EP0057 (formerly known as CRLX101) is a formulation of camptothecin (CPT) conjugated with a cyclodextrin polymer backbone and is currently being evaluated clinically in multiple refractory solid tumors [
102,
103,
104,
105,
106]. The micelles have a size of approximately 30–40 nm and significantly increase CPT (topoisomerase I inhibitor) solubility while preserving its active lactone form [
107]. EP0057 also exhibits better patient tolerance than other CPT analogs. The nanoparticles-drug conjugate is administered via intravenous infusions, and nanoparticles preferentially accumulate in the tumors through EPR [
107]. EP0057 has been shown to inhibit significantly also hypoxia-inducible factor-1 alpha (HIF-1α) and therefore serving as a radiosensitizer with the potential to improve the efficacy of chemoradiation therapy [
108,
109,
110].
Prior studies showed EP0057 efficacy in recurrent or persistent, epithelial ovarian, fallopian tube or colorectal, peritoneal, and gastroesophageal cancer [
111,
112], where it showed promising results [
112]. Ongoing clinical trials are designed to evaluate the efficacy and safety (Phase 1/2) of this therapeutic in lung [
102], gastric [
113] and ovarian [
103] cancer in combination with Olaparib, as well as to evaluate its pharmacokinetics properties (PK) [
102,
114] using a population model. From the data obtained from 27 patients enrolled on two-Phase II clinical trials, the release of CPT was characterized by an initial rapid clearance, which decreased via first-order decay to the steady-state value by 4 h after the infusion. A second Phase I/IIa clinical study involved 22 efficacy-evaluable patients with metastatic renal cell carcinoma, who received increasing doses of EP0057 combined with bevacizumab [
106]. Partial response or stable disease was observed in 86% of the patients, with a median progression free survival (mPFS) of 9.9 months. Most patients achieved a reduction of tumor and increased the progression-free survival compared to their previous therapy [
115]. In addition, a Phase Ib/II study of EP0057 combined with PTX in women with recurrent epithelial ovarian cancer reported a 31.6% overall response rate, including one complete response with a 5.4 month median progression-free survival [
105]. However, the analysis of trials including data highlights the need of more investigation to evaluate the clinical benefits of this therapeutic also because of the onset of considerable side effects [
116,
117,
118,
119].
3.4. NanoPac
Other attempts to formulate PTX in nanostructure to avoid the use of Cremophore-EL were performed. NanoPac (also known as, Nanotax) are pure PTX nanoparticles generated in supercritical carbon oxide environment in the presence of organic solvents. These particles have a size of 600–800 nm inhibiting their clearance and making their use helpful for topical and local administration (i.e., inhalation) [
120], with no targeting mechanism associated. A Phase 2 trial focused on investigating the effects of different concentration of NanoPac against prostate cancer directly injected into the prostate. Interestingly, the lower dose of drug showed higher benefits in terms of tumor reduction compared to higher doses. The drug showed reasonable side effects, also at the highest dose used [
121]. In similar experimental settings, other trials measured the ability of intra-cystic injected Nanopac to contrast the progress of pancreatic cancer [
122] and of intraperitoneal administration against ovarian cancer [
123]. Additionally, in these cases, lower doses of NanoPac showed higher clinical benefits even though the occurrence of side effect was significantly more pronounced.