Enzymatical processes topic wants to enhance the central role of enzymes, the real workers from whose silent and tireless work depends the well-being of all cells in fact, intra-cellular reactions that take place in the cells are facilitated and therefore accelerated by enzymes.
- enzymatical
- intra-cellular
- enzymes
1. Role of the Enzymes and the First Discoveries on Their Characteristics
Enzymes are biological catalysts that allow to accelerate chemical reactions, or the speed with which they take place. The initial study on enzymes assumed the active site of the enzyme as a rigid structure and the adaptation of a substrate in the active site more or less like a key in the lock. This first idea was suggested for the first time in 1894 by the German biochemist Emil Fischer.[1][2] In a model structured in this way, or according to the "key and lock" version it was possible to guess how the enzymes were specific and coordinated between them. A more useful view of enzyme-substrate interaction derives from the model of induced adaptation.[3] This model assumes that the initial link of the substrate molecule to the active site will distort both the enzyme and the substrate, stabilizing the molecule of the latter in its transition state and thus making the link more susceptible to the catalytic attack.
2. The Importance of Enzymes in Diagnostic
Because the measure of an enzymatic activity is useful for a clinical routine diagnosis the following conditions must be met.
- The enzyme must be present in the blood, in the urine, or in other tissue fluids that can be easily found. Tissue biopsies should not be practiced as routine, but only in cases where the diagnostic value is particularly important.
- The enzyme must be easily dosable and is even better if the method can be automated.
- The quantitative differences between the enzymatic activities of normal and sick subjects must be significant, and there must be a good correlation between the levels of enzymatic activity and the pathological state.
- It is also advisable that the enzyme is sufficiently stable to allow sample conservation at least for limited periods of time.
The serum is the fluid on which most analysis are done. Urine can only be used for few enzymes secreted by the kidneys. The enzymes in the serum can be divided into two categories:
- specific plasma enzymes: for example, enzymes that carry out a plasma activity, such as enzymes involved in blood coagulation, in the activation of the complement, and in the metabolism of lipoproteins;
- non-specific plasma enzymes: this category includes those enzymes that do not carry out physiological functions in the plasma for example enzymes inside the cells: amylase, lipase, phosphatase and other enzymes associated with cellular metabolism, whose presence in a normal serum in high quantity can be attributed to cellular suffering and/or tissue damage.
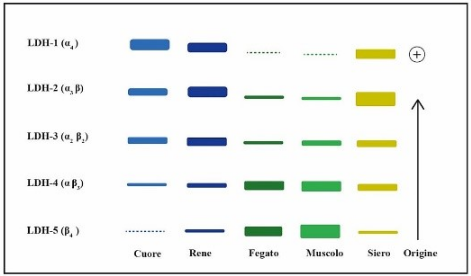
Ideally, for diagnostic purposes, it would be desirable to analyze specific enzymes that would allow to identify the tissue from which they come; but unfortunately it exist isoenzymes that have a different distribution and they are aspecific in various tissues. The most studied case is that of the dehydrogenase lactate. The enzyme consists of four subunits. There are two types of subunits that, combining in various ways, give rise to five different forms of lactate dehydrogenase ɑ1ß, ɑ2ß, ß3, ɑß4, and ß5. These five forms, separable electrophoretically, are differently distributed in tissues (figure 1). Today it is possible, in some cases, to distinguish the isoenzymes, make use of monoclonal antibodies. This method has been applied to recognize the different isoenzymes of human phospho fruttochinase, and to identify what was the absent form in the hereditary deficiencies of phosphofructokinase. The isoenzymatic representations, in addition to give us indications on the origin of the tissue, are also useful in legal medicine. Since numerous enzymes of the serum and of the red blood cells are present in different isoenzimatic forms, the particular distribution in a blood sample can help to identify its origin.
3. Techniques to Study the Enzyme Activities
It's very important to understand enzyme structure and measure their activity because particular changes in their structure establish pathological processes. In fact, enzymes catalyze a variety of biochemical reactions and they control all physiological processes.[4] Several analytical approaches have been developed to meet this need, such as the Protein film electrochemistry, Nuclear Magnetic Resonance, Spettrophotometer UV-VIS and FT-IR, Chromatography such as HPLC (High-Performance Liquid Chromatography) usually coupled to Mass spectrometry, Electron Microscopy and X-ray crystallography.
The Protein film electrochemistry known as electrochemistry of protein-modified electrodes was used to study the kinetic properties of enzymes. This technique provides real-time information on transient and steady-state catalytic behavior of the enzymes and it allows to observe cooperative two-electron transfer, oxidation state dependent inhibitor binding and anaerobic oxidative inactivation. Despite its advantages, protein film electrochemistry does not provide a fully comprehensive picture of the enzyme–electrode interface but it can be coupled to a large range of complementary techniques.[5]
Nuclear Magnetic Resonance is used to analyze the molecular structures of organic compounds using a superconducting magnet and structural analysis software, the equipment can smoothly analyze complicated molecular structures. While Nuclear Magnetic Resonance is highly versatile, it often fails due to its low sensitivity. Another problem about this technique is the drift in the magnetic field that can lead to spectral peaks broadening and lineshapes being distorted, making interpretation of NMR results very difficult.[6]
Spectrophotometers analyze the molecular structures by absorbing Ultraviolet - Visible or infrared light. This equipment is used for measuring light transmitted or reflected from the sample by irradiating light. When the light is transmitted through (reflected on) the sample, it is absorbed. Based on the absorption wavelength and strength, data on the functional groups contained in the molecules constituting the sample can be obtained.
HPLC/MS-MS is a powerful technique widely used in most chemical and biochemical research settings, both in academia and industry. HPLC is used when it is necessary to separate analytes based on structure or molecular composition. It can be used to quantify, purify and identify analytes in a mixture. The advantages of HPLC as a chromatographic technique are speed and efficiency. Disadvantages are linked to the obtaining of few precise information on the structure of the molecule for the purpose of recognition unequivocal and moreover to the difficulty of analyzing compounds with P.M. <600 Da.[7]
Electron microscopy is a technique recommended for nanomaterial characterization. It is split into scanning electron microscopy (SEM) and Transmission Electron microscopy (TEM). The first, to generate the picture, uses the electrons repelled from the sample area close to the surface, while TEM detects electrons passing through a very thin sample. Despite the electron microscopy is a device with very high resolving power whom allows the observation of samples of infinitesimal sizes, the live systems such as cells cannot be studied by this technique.[8] This means that biological interactions cannot be observed correctly, which limits the applications of electron microscopy in biological research.[9]
X-ray crystallography is a powerful non-destructive technique for determining the molecular structure. It uses the principles of X-ray diffraction to analyze the sample and it's done in many different directions so that the 3D structure can be built up.[10] It is a technique that has helped to deduce the 3D crystal structure of many biological materials. Scientist use this technique, where it's need determining the size of atoms, the lengths and different types of chemical bonds, the atomic arrangement of materials, conformational modifications of the molecules during their chemical reactions.[11] Often it's used to obtain the complete structure of Viruses and Bacteria. Moreover, it's sure that no other technique can compete with X-ray Crystallography because it's able to determine the geometry of the smallest enzymatic molecules, although commercial instruments can reach resolutions of 5 Å or less.[8]
4. Enzymes in Therapy
Thanks to the progresses in the field of the research have provided the bases for the development of new "enzymatic drugs", in fact, it's known the many enzyme substrates can be used in support of traditional medical treatments for different types of pathologies.[12][13] The use of biodynamic components favor cellular metabolic reactivation, obtaining excellent results, because they are able to maintain a cellular stability defined as "allostatic" during the pathological processes and are able to provide the energy necessary for intracellular support.[14] The discoveries and furtherance with which the therapeutic enzymes are being produced have led to new opportunities and prompted new open doors in the field of medication for both therapeutic and analytical purposes.[15] It has been found that enzymatic therapies can open new scenarios on the management of these pathologies, starting from biochemistry (and therefore from study of the cell) also arriving at the resolution of the pathology itself.[16] For example, some researches showed how it's possible to improve the quality of life of patients and nowadays many laboratories are evaluating the current discoveries and advancements in therapeutic enzymes to provide a scope of improvement for the existing enzymes and help overcome the challenges to further develop new ones.[17] It could deduce that these new technologies could help the treatment of cancer patients, obviously not as an antitumor drug, but as useful strategy to improve the quality of life by reducing the adverse symptoms.[18]
4.1. Effects on Enzymatic Processes Coordination in Pathology
Thanks to the highly effective vaccines who have been introduced, the spread of SARS-CoV-2 are slowing. However, many sick people need of support and treatments against COVID-19. The main supportive cares are NSAIDs, glucocorticoid, dexamethasone and oxygen therapy. All these therapies help the patient to relieve symptoms, to support breathing and to reduce the cytokine storm. Fortunately, the research is looking new efficient drugs able to improve the health condition of COVID patients. For example, fluvoxamine is a selective serotonin reuptake inhibitor (SSRI) used for the treatment of obsessive-compulsive disorder such as depression. Despite there is insufficient evidence for the COVID-19 treatment because need results from well-conducted clinical trials, several studies showed it is possible used it for the treatment of high-risk outpatients with early diagnosed COVID-19 because it was found fluvoxamine binds the sigma-1 receptor of the immune cells, resulting in reduced production of inflammatory cytokines.[19][20] New rising researches are highlighting the important role of nutrition and of the use of enzymatical components to modulate biochemical reactions of the infected cells and improve general physical condition of COVID-19 patients, to face the disease in the best possible way.[21][22] In the end, monoclonal therapies are catching up but in the United States they are used for early stages and in the high-risk cases.[23] The authorization about their use is based on the results of a large clinical trial that found the antibodies prevented symptoms people who were tested positive. Indeed, monoclonal antibody therapies would be able to prevent hospitalizations, reduce viral loads and lessen symptom severity because they recognize spike proteins and don't allow to the virus to enter into human cells, giving to the immune system a leg up until it can mount its own response.[24][25]
4.2. Coordination of Enzymatic Processes in Neurodegenerative Diseases
Multiple sclerosis (MS) is the most frequent neurological disease in young adults with a complex and still uncertain pathogenesis. It's an immune-mediated disease of the central nervous system in which macrophages and T cells infiltrate the brain and induce myelin damage, neuroinflammation and neurodegeneration. Despite being of unknown etiology, we now know that the oligodendrocyte loss, activation of brain resident immune cells such as microglia and astrocytes and expression and release of oxidative-stress related molecules are critically involved in the disease process.[26] In several researches are emerging evidences that mitochondrial dysfunction and associated oxidative stress play an important role in driving neurodegeneration, in fact it's known that demyelinated axons must consume more energy to maintain conduction. As a result, axons in MS lesions contain more mitochondria but, surprisingly, less oxidative phosphorylation.[27] Reactive oxygen species (ROS) have been implicated as mediators of demyelination and axonal damage because they cause damage to lipids and proteins resulting in cell death by necrosis or apoptosis.[28] Thus, in medical practice is employed treatment with antioxidants to prevent propagation of tissue damage and improve both survival and neurological outcome. By nowadays, several studies have been performed to see whether dietary intake of several antioxidants prevents or reduces the progression of MS. Moreover, it was discovered that enzymatic cascades, such as protease activity, cytokine and chemokine activation and complement activation increasing the development and progression of Multiple Sclerosis.[29] Scientists are studying a mechanism to modulate the enzyme activities to improve the quality of life of patients with MS. From first results, some researches show the use of enzymatical substrates play a key role in ROS reduction and in enzyme cellular reset.[30] These two factors indicate the improvement of antioxidant action, the activation of the mitochondrial activity and the modulation of main enzymes involved in oxidative balance such as Catalase, Superoxide Dismutase, Glutathione S-transferases and NADPH Oxidase.[16]
4.3. Role of Enzymatic Inhibitors in HCV Infections
Hepatitis C virus (HCV) is a (+)ssRNA enveloped virus of the family Flaviviridae. HCV infection affects approximately 3% of the world's population and causes chronic liver diseases, including liver fibrosis, cirrhosis, and hepatocellular carcinoma.[31] HCV life-cycle is linked to several endocellular enzymes because they contribute to RNA replication and to nucleocapsid assembly and its maturation. These enzymes play an important role in the formation of multiple copies of HCV genome. Particularly, this process is catalyzed by an enzymes called NS5B RNA-dependent RNA polymerase. The so-formed positive strand RNA genomes are next translated to create new viral enzymes and proteins.[32] Although current antiviral therapy comprising direct-acting antivirals can achieve a quite satisfying sustained virological response rate, it is still limited by viral resistance, long treatment duration, combined adverse reactions, and high costs. Moreover, the currently marketed antivirals fail to prevent the HCV entry, because it's a highly orchestrated process involving protein interactions and enzyme activities of host cells. In fact, Most drugs act on inhibiting adhesion factors among host cell and virus, eliminating viral infection from the very beginning of the viral life cycle. This way allowes to optimize and widen the prevention of HCV infection, but it would be suitable use other factors able to contrast viral replication inside the cells. Nowday, the introduction of new multi-genotypic direct acting antivirals in clinical practice has revolutionized HCV treatment, permitting the achievement of >95% rates of sustained virological response in many patients.[33] However, virological failures can occur associated with development of resistance. Therefore, interferons alpha-2 are used in the treatment of HCV in combination with Ribavirin because the interferon, after binding with a specific receptor, induces an enzyme multidirectional response by, e.g., increasing the concentrations of 2’,5’-oligoadenylate synthase and β2-microglobulin in serum causing an antiviral and antiproliferative activity, while Ribavirin inhibits specifically viral polymerase (replication enzyme) by disturbing the synthesis of mRNA and nucleic acids.[32] Other studies show the use of enzymatical inhibitors can help the treatment of the patient with HCV infection, in fact, since 2011, DNA and RNA polymerase inhibitors, RNA protease inhibitors, serine protease inhibitors have been approved for clinical use because they demonstrate high efficacy against HCV replication.[31]
5. Enzymes in Agriculture
With the high use of pesticides and herbicides, both the soil and the plants are deprived of enzymes capable of working organic matter. This leads to the depletion of the soil, nutrients and an increase in infections. Following these observation European Union (EU) has issued a new legislation and the Italian Senate, on 20 May 2021, approved a draft law "Ddl 988" about Provisions for the protection, development and competitiveness of agricultural, agri-food and aquaculture production using biological methods. The latter would allow to work with enzymes in agriculture, enriching the soil with vital nutrients for plants and obtain all the substances necessary to overcome adversity. Thanks to enzymological studies in agricolture it's possible to renew the soil by means of natural processes that characterize all living organisms and ecological systems. From the researches are well known the advantage of the crop rotation because it allows to the enzymes to settle in the soil and periodically enrich it guaranteeing resistance to parasites for cultivated plants.[34]
The content is sourced from: https://handwiki.org/wiki/Biology:Enzymatical_processes
References
- "How Emil Fischer was led to the lock and key concept for enzyme specificity". Advances in Carbohydrate Chemistry and Biochemistry 50: 1–20. 1994. doi:10.1016/S0065-2318(08)60149-3. PMID 7942253. https://dx.doi.org/10.1016%2FS0065-2318%2808%2960149-3
- The lock and key model for Molecular Recognition. Is it time for a paradigm shift?. 2018.
- "The phenomenon of enzymatic adaptation and its bearings on problems of genetics and cellular differentiation". Selected Papers in Molecular Biology by Jacques Monod. Elsevier. 2 December 2012. pp. 68–134. ISBN 978-0-323-14263-2. https://books.google.com/books?id=TchQ6gPzO_4C&pg=PA68.
- "Methods of Measuring Enzyme Activity Ex Vivo and In Vivo". Annual Review of Analytical Chemistry 11 (1): 509–533. June 2018. doi:10.1146/annurev-anchem-061417-125619. PMID 29505726. Bibcode: 2018ARAC...11..509O. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=6147230
- "Advancing Techniques for Investigating the Enzyme-Electrode Interface". Accounts of Chemical Research 52 (5): 1439–1448. May 2019. doi:10.1021/acs.accounts.9b00087. PMID 31042353. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=6533600
- Applications of NMR spectroscopy. Volume 2. Atta-ur- Rahman, M. Iqbal Choudhary. Sharjah, United Arab Emirates. 2015. ISBN 978-1-60805-965-2. OCLC 906183598. https://www.sciencedirect.com/topics/chemistry/nuclear-magnetic-resonance-instrumentation.
- "Svantaggi e vantaggi di un HPLC". http://it.scienceaq.com/Chemistry/100216383.html.
- "Enzimi in "Enciclopedia del Novecento"" (in it-IT). https://www.treccani.it/enciclopedia/enzimi_(Enciclopedia-del-Novecento).
- Nanomaterials for food packaging : materials, processing technologies, and safety issues. Miguel Ângelo Parente Ribeiro Cerqueira, Jose Maria Lagaron, Lorenzo Miguel Pastrana Castro, António Augusto Martins de Oliveira Soares Vicente. Cambridge, MA. 2018. ISBN 978-0-323-51272-5. OCLC 1035556365. https://www.sciencedirect.com/topics/materials-science/electron-microscopy.
- "x ray crystallography". Molecular Pathology 53 (1): 8–14. February 2000. doi:10.1136/mp.53.1.8. PMID 10884915. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1186895
- Molecular virology of human pathogenic viruses. London, UK. 2017. ISBN 9780128008386. OCLC 949276536. https://www.sciencedirect.com/topics/medicine-and-dentistry/x-ray-crystallography.
- "Enzymes as therapeutic agents". Enzymes in Human and Animal Nutrition. 2018. pp. 301–312. doi:10.1016/B978-0-12-805419-2.00015-0. ISBN 978-0-12-805419-2. https://dx.doi.org/10.1016%2FB978-0-12-805419-2.00015-0
- "Enzymes approved for human therapy: indications, mechanisms and adverse effects". BioDrugs 29 (1): 31–55. February 2015. doi:10.1007/s40259-015-0116-7. PMID 25648140. https://dx.doi.org/10.1007%2Fs40259-015-0116-7
- "Oral nutritional supplement prevents weight loss and reduces side effects in patients in advanced lung cancer chemotherapy". Amino Acids 52 (3): 445–451. March 2020. doi:10.1007/s00726-020-02822-7. PMID 32034492. https://dx.doi.org/10.1007%2Fs00726-020-02822-7
- "Therapeutic enzymes: Discoveries, production and applications". Journal of Drug Delivery Science and Technology 63: 102455. June 2021. doi:10.1016/j.jddst.2021.102455. https://dx.doi.org/10.1016%2Fj.jddst.2021.102455
- "Reduction in Fatigue Symptoms Following the Administration of Nutritional Supplements in Patients with Multiple Sclerosis". Medical Sciences 9 (3): 52. July 2021. doi:10.3390/medsci9030052. PMID 34287336. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=8293375
- "Enzymes in therapy of biofilm-related oral diseases". Biotechnology and Applied Biochemistry 64 (3): 337–346. May 2017. doi:10.1002/bab.1490. PMID 26969579. https://dx.doi.org/10.1002%2Fbab.1490
- "Cysteine cathepsins: multifunctional enzymes in cancer". Nature Reviews. Cancer 6 (10): 764–775. October 2006. doi:10.1038/nrc1949. PMID 16990854. https://dx.doi.org/10.1038%2Fnrc1949
- "Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19: the TOGETHER randomised, platform clinical trial" (in English). The Lancet. Global Health 10 (1): e42–e51. October 2021. doi:10.1016/S2214-109X(21)00448-4. PMID 34717820. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=8550952
- "Fluvoxamine" (in en). https://www.covid19treatmentguidelines.nih.gov/therapies/immunomodulators/fluvoxamine/.
- "The Role of Nutrition in COVID-19 Susceptibility and Severity of Disease: A Systematic Review". The Journal of Nutrition 151 (7): 1854–1878. July 2021. doi:10.1093/jn/nxab059. PMID 33982105. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=8194602
- "How Nutrition can help to fight against COVID-19 Pandemic". Pakistan Journal of Medical Sciences 36 (COVID19–S4): S121–S123. May 2020. doi:10.12669/pjms.36.COVID19-S4.2776. PMID 32582329. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7306972
- "Hospitalized Adults: Therapeutic Management" (in en). https://www.covid19treatmentguidelines.nih.gov/management/clinical-management/hospitalized-adults--therapeutic-management/.
- "Anti-SARS-CoV-2 Monoclonal Antibodies" (in en). https://www.covid19treatmentguidelines.nih.gov/therapies/anti-sars-cov-2-antibody-products/anti-sars-cov-2-monoclonal-antibodies/.
- "What Is Monoclonal Antibody Therapy & Who Is Eligible to Receive It?" (in en). https://www.houstonmethodist.org/blog/articles/2021/jan/what-is-monoclonal-antibody-therapy-and-who-is-eligible-to-receive-it/.
- Ohl, Kim; Tenbrock, Klaus; Kipp, Markus (March 2016). "Oxidative stress in multiple sclerosis: Central and peripheral mode of action" (in en). Experimental Neurology 277: 58–67. doi:10.1016/j.expneurol.2015.11.010. PMID 26626971. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7094520
- Nijland, Philip G.; Molenaar, Remco J.; van der Pol, Susanne M. A.; van der Valk, Paul; van Noorden, Cornelis J. F.; de Vries, Helga E.; van Horssen, Jack (December 2015). "Differential expression of glucose-metabolizing enzymes in multiple sclerosis lesions" (in en). Acta Neuropathologica Communications 3 (1): 79. doi:10.1186/s40478-015-0261-8. ISSN 2051-5960. PMID 26637184. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=4670517
- Offen, Daniel; Gilgun-Sherki, Yossi; Melamed, Eldad (2004-03-01). "The role of oxidative stress in the pathogenesis of multiple sclerosis: The need for effective antioxidant therapy". Journal of Neurology 251 (3): 261–268. doi:10.1007/s00415-004-0348-9. ISSN 0340-5354. PMID 15015004. http://link.springer.com/10.1007/s00415-004-0348-9.
- Scarisbrick, I.A. (2008). "The Multiple Sclerosis Degradome: Enzymatic Cascades in Development and Progression of Central Nervous System Inflammatory Disease". Current Topics in Microbiology and Immunology 318: 133–175. doi:10.1007/978-3-540-73677-6_6. ISBN 978-3-540-73676-9. ISSN 0070-217X. PMID 18219817. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=4097322
- van Horssen, Jack; Witte, Maarten E.; Schreibelt, Gerty; de Vries, Helga E. (2011-02-01). "Radical changes in multiple sclerosis pathogenesis" (in en). Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease. Molecular Basis of Multiple Sclerosis 1812 (2): 141–150. doi:10.1016/j.bbadis.2010.06.011. ISSN 0925-4439. PMID 20600869. https://www.sciencedirect.com/science/article/pii/S0925443910001213.
- Qian, Xi-Jing; Zhu, Yong-Zhe; Zhao, Ping; Qi, Zhong-Tian (2016-01-06). "Entry inhibitors: New advances in HCV treatment". Emerging Microbes & Infections 5: e3. doi:10.1038/emi.2016.3. ISSN 2222-1751. PMID 26733381. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=4735057
- Zając, Marianna; Muszalska, Izabela; Sobczak, Agnieszka; Dadej, Adrianna; Tomczak, Szymon; Jelińska, Anna (2019-03-01). "Hepatitis C – New drugs and treatment prospects" (in en). European Journal of Medicinal Chemistry 165: 225–249. doi:10.1016/j.ejmech.2019.01.025. ISSN 0223-5234. PMID 30685524. https://www.sciencedirect.com/science/article/pii/S0223523419300352.
- Ceccherini-Silberstein, Francesca; Cento, Valeria; Di Maio, Velia Chiara; Perno, Carlo Federico; Craxì, Antonio (2018-10-01). "Viral resistance in HCV infection" (in en). Current Opinion in Virology 32: 115–127. doi:10.1016/j.coviro.2018.10.005. ISSN 1879-6257. PMID 30439589. https://www.sciencedirect.com/science/article/pii/S1879625718300208.
- "Crop rotation and residue management effects on soil enzyme activities, glomalin and aggregate stability under zero tillage in the Indo-Gangetic Plains". Soil and Tillage Research 184: 291–300. 1 December 2018. doi:10.1016/j.still.2018.08.006. https://dx.doi.org/10.1016%2Fj.still.2018.08.006
