Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Dizziness is one of the most common reasons for consultation in adult patients. It is an umbrella term used to describe various sensations, including vertigo, disequilibrium, lightheadedness, or presyncope.
- cervicogenic dizziness
- cervical proprioception
- proprioceptors
1. Introduction
Dizziness is one of the most common reasons for consultation in adult patients [1][2]. It is an umbrella term used to describe various sensations, including vertigo, disequilibrium, lightheadedness, or presyncope (Table 1) [2]. From this perspective, vertigo is just one part of dizziness. However, in the light of the International Bárány Society for NeuroOtology [3], dizziness and vertigo are no longer subordinate but independent allelic symptoms. Dizziness and vertigo may coexist or occur sequentially (Table 2) [3]. In 1955, Ryan and Cope [4] first described dizziness caused by neck disorders as cervical vertigo, also known as cervicogenic vertigo, cervicogenic dizziness or cervical dizziness. Here researchers use cervicogenic dizziness to name this trouble. A recent clinical observation of a large number of cases (1000 cases) found that cervicogenic dizziness accounted for 89% of all dizziness, or vertigo [5]. Cervical spondylosis was one of the common causes of dizziness in the elderly in a community survey [1]. Among patients with cervical vertebral whiplash injuries, the prevalence of dizziness has been variously reported, ranging from 20% to 90% [6]. Nearly half of patients with neck pain have cervicogenic dizziness [1]. However, cervicogenic dizziness is the most controversial among all dizziness because its pathogenesis is unclear, and its diagnosis and treatment are difficult [6][7][8][9].
Table 1. Main categories of dizziness.
| Category | Description |
|---|---|
| Vertigo | A sense of spinning experienced even when someone is perfectly still. |
| Disequilibrium | A loss or lack of equilibrium or stability. |
| Presyncope | Feeling of losing consciousness or blacking out. |
| Lightheadedness | Feeling a little woozy or faint. |
Table 2. Definition of dizziness and vertigo by the International Bárány Society for NeuroOtology.
| Classification | Description |
|---|---|
| Dizziness | The sensation of disturbed or impaired spatial orientation without a hallucinatory or distorted sense of motion. |
| Vertigo | The sensation of self-motion when no self-motion is occurring or the sensation of distorted self-motion during an otherwise normal head movement. |
Cervicogenic dizziness is considered to have four different pathogenesis, but proprioceptive cervicogenic dizziness is the most common and accepted by most scholars [6]. Unlike other forms of dizziness, cervicogenic dizziness is of interest not only to neurologists but also to physiotherapists, pain physicians, and orthopedic surgeons.
2. Pathophysiology
2.1. Cervical Proprioceptors and Proprioception
The sensorimotor system includes all the afferent, efferent, central integrations, and processing parts [10]. Somatosensory is the afferent component of the sensorimotor system and includes the conscious perception of pressure, temperature, vibration, pain, and proprioception [10][11]. Proprioception has often been described as the sixth sensation [12], including kinesthesia, force sensation, and joint position sense [10][11][13].
Proprioceptive information is conducted by specialized nerve endings, called proprioceptors which are situated in the joints, muscles, tendons, and skin [10][12]. Cervical proprioceptive afferents can be primarily divided into three groups: joint receptors, muscle spindle, and Golgi tendon organs (GTO) [12][14][15], which play a significant role in head-eye coordination and posture maintenance [16]. Proprioceptive information in the neck plays a crucial role in monitoring head orientation and offering a reference for the visual and vestibular receptors [2]. In physiological conditions, muscle spindles play an important role in kinesthesia GTOs conduce to the senses of heaviness and force, while cervical joint receptors may act a secondary role in proprioception [15].
Muscle spindles and GTOs react to the changes in skeletal muscle length and tension, respectively. A high density of muscle spindles has been found in the neck region of humans [16][17][18]. In general, spindle density refers to the number of spindles in wet muscle tissue per gram of muscle sample and is often used to compare the relative abundance of muscle spindles in different muscles of the same species [16][17]. In a highly cited and classic article, Kulkarni et al. [16] found that the suboccipital small muscles in human fetuses had an abundant spindle density and spindle content but lacked GTOs, making them ideal sensors for detecting the joint position and movements of craniovertebral joints. They also found that the inferior oblique muscle had a spindle density of 242/g, while the trapezius muscle had a spindle density of only 2.2/g [16]. Boyd-Clark et al. [17] found that the spindle density of longus Colli (48.6/g) was significantly higher than that of the multifidus muscle (24.3/g) in human autopsies, and the morphology, distribution, and density of spindle did not change with age. Although spindle density has been widely used in comparative studies, muscle mass has never been shown to be an appropriate reference for spindle number [18]. Banks et al. [18] revealed that spindle density itself has a nonlinear relationship with muscle mass, so direct linear comparisons of muscle spindle densities across muscle sizes are misleading. They performed an allometric analysis on the number of spindles in mammalian skeletal muscles and suggested the use of residual value as a simple way to measure the relative abundance of muscle spindle components. There was no difference in relative spindle abundance between large and small muscles as measured by residual values [18]. The longus capitis muscle, semispinalis capitis, and obliquus capitis inferior have the highest relative abundance values (7.5, 4.9, and 3.5, respectively) [18].
It is widely accepted that the joint receptors in proprioception act as joint limit detectors, playing a significant role in the sense of position near the limits of joint motion [12][15][19]. However, joint receptors are critical for the control of feedforward muscle activity and muscle stiffness via the gamma muscle spindle system [20]. Slight flexion of the upper cervical joints can lead to significant changes in the discharge rate of muscle spindle afferents in the perivertebral muscles [21]. Thunberg et al. [22] found reflex connections between receptors in the neck facet joints and fusimotoneurones of dorsal cervical muscles and the transient activation of chemically sensitive nerve endings in facet joints to be capable of triggering positive feedback loops that may produce chronic pain and stiffness in the cervical muscles. Therefore, cervical joint receptors are likely to impact postural control and head-eye movement via their influence on the muscle spindle system [20][21][22].
In 1967, Freeman and Wyke [23] classified four kinds of mechanoreceptors in the knee joints of cats. Except as free nerve endings (type IV), three types of proprioceptors are also found in human cervical facet joints [24][25] and discs [26][27], including the Ruffini corpuscles (type I), Pacinian corpuscles (type II), and GTOs (type III). However, they are much lower than the amount in the muscles. Although there are a small number of mechanoreceptor endings in the facet capsules and discs, the volume of receptors is relatively large. It is likely that receptive fields are large and that one or two mechanoreceptors may be sufficient to monitor the area of each individual facet capsule or disc [24]. Animal studies have also found that the functional proprioceptors in the facet joint capsule can be activated by low-stretching-level activities [25]. In addition, the intervertebral disc is situated on the central axis of cervical motion, and thus, proprioceptors of cervical discs are in a favorable site to monitor subtle changes in the cervical position or direction of motion. [26][28].
2.2. Central and Reflex Connection for Cervical Proprioceptive Signals
The neck proprioceptor can provide information on the movement and position of the head relative to the trunk but not on the movement of the head in space. However, this sensory information can affect vestibular reflexes, which function to stabilize the posture of the head, eyes, and body and to construct a sense of spatial orientation [29]. The vestibular system provides relevant information about the position of the head relative to space, while the visual system identifies the position of the head relative to the external environment [30]. By combining vestibular signals with neck proprioception information, the motion signals can be coordinately transformed into a body-centered frame of reference [31]. In addition, visual-vestibular and proprioceptive-vestibular interactions are critical for postural control and gaze [31].
The cervical proprioceptive system has unique central and reflex connections with the vestibular and visual systems (Figure 1). It is well recognized that the convergence of cervical proprioceptive afferents with vestibular and visual inputs at different levels of neuroaxis includes the vestibular nuclei, thalamus, and cerebral cortex [10][16][29][32][33][34]. Animal studies suggest that cervical proprioceptive afferents are transmitted to the central cervical nucleus through the dorsal root ganglion and project directly to the vestibular nucleus [35]. Neurons in the central cervical nucleus receive afferent information from the vestibular and cervical proprioceptors and provide integrated data about the head position relative to the trunk and space to the cerebellum and reticular formation [36][37]. The spinal cord can also indirectly convey input to the vestibular nuclei via the cerebellum and reticular formation [38]. Moreover, the vestibular nuclei can provide direct inputs to the reticular formation and parabrachial nucleus with projections to sympathetic preganglionic neurons of the thoracic spinal cord, which are involved in producing the rapid adjustment of circulation, digestion, and respiration necessary to maintain homeostasis (through the vestibulo-sympathetic reflex pathway) [39][40]. Reflex activities relating to the cervical, vestibular, and visual systems play a significant role in the coordination of head, neck, and eye movements (Figure 1).
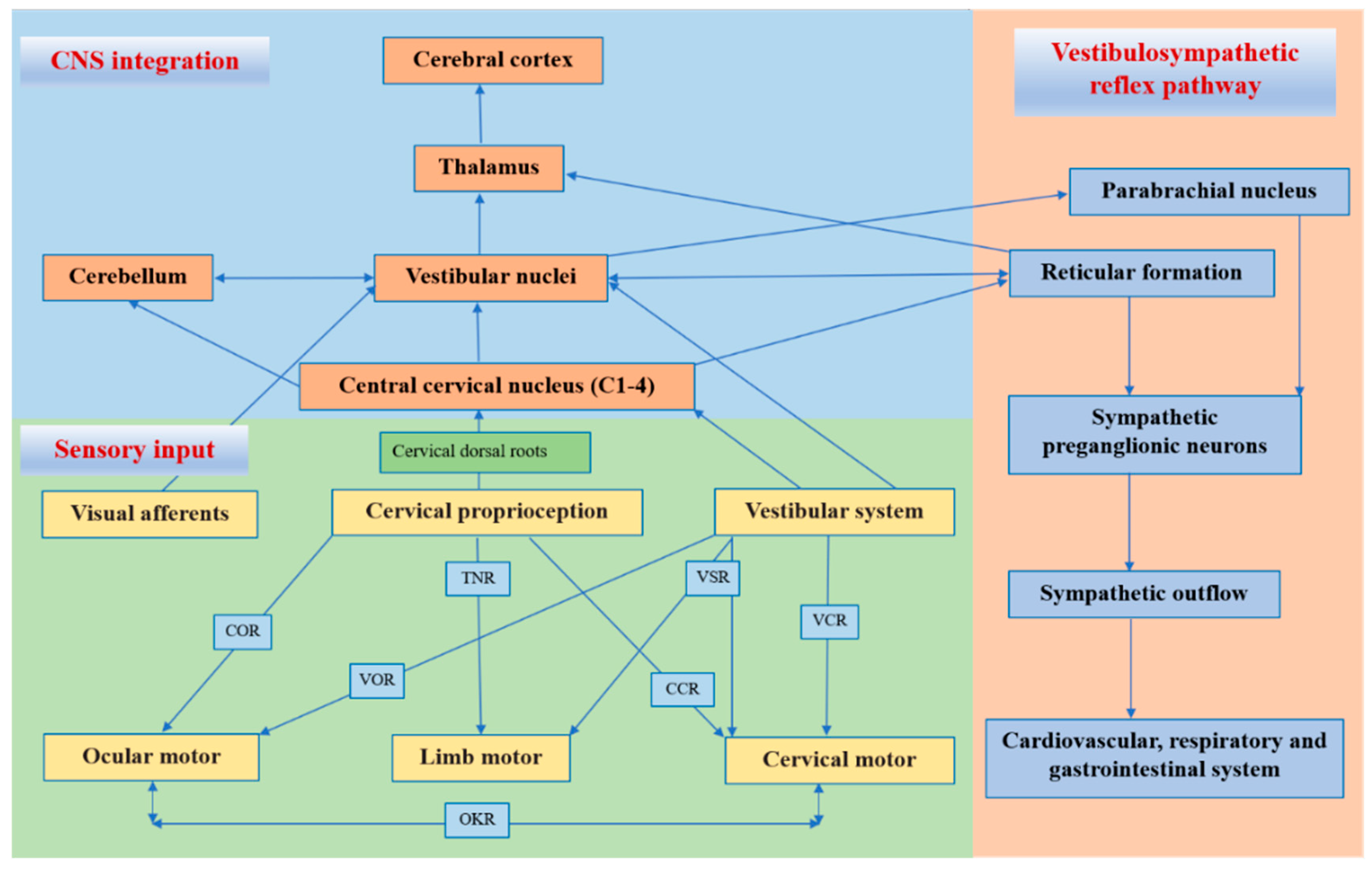
Figure 1. Central and reflex connection for cervical proprioceptive signals. COR = cervico–ocular reflex. VOR = vestibulocular reflex. TNR = tonic neck reflex. VSR = vestibulospinal reflex. CCR = cervico–collic reflex. VCR = vestibulo–collic reflex. OKR = optokinetic reflex.
2.3. Altered Cervical Proprioceptive Afferent and Cervicogenic Dizziness
Some studies have found that the “reversible” injury of the neck can cause defects in balance function and vision. [6]. The injection of local anesthetics in the neck is induced by nystagmus and ataxia in animals, and ataxia, a sense of falling or tilting without nystagmus, in humans [41]. In addition, the unilateral disconnection of C1–C3 dorsal roots can produce almost the same effect as a unilateral labyrinthectomy and unilateral transection of the upper cervical afferents, which can result in severe ataxia and nystagmus [42][43]. Furthermore, neck muscle vibrations in humans can induce prolonged eye position changes [44], visual illusory movements [45], and increased body sway [46].
As reviewed above, the cervical spine has very well-developed proprioceptors, and their input information integrates with the vestibular and visual systems in multiple levels of the central nervous system, which act on the neck and eye muscles through a variety of reflex activities to maintain the coordinated movement of the head, eyes, neck, and body. When cervical proprioceptive afferent activities are reduced or increased in the process of multilevel information integration in the central nervous system, cervicogenic dizziness can occur due to sensory mismatch or conflict between the vestibular, visual, and proprioceptive inputs. The conflict or mismatch of central information integration can also explain the pathogenesis of motion sickness and visual vertigo [47]. In general, abnormalities anywhere in neural pathways, from cervical proprioceptors to all levels of the central nervous system, can cause altered proprioceptive inputs and result in dizziness and instability. The function of cervical mechanoreceptors can be altered due to neck trauma, muscular spasms or fatigue, cervical degenerative disease, or neck pain [8][9].
Neck muscle spasms or fatigue can alter proprioception and postural control, and a prolonged contraction of unilateral cervical muscles can significantly increase the sensitivity of the neck proprioceptors [48]. Interstitial inflammatory mediators produced by muscle fatigue can make muscle spindle sensitive [49]. In addition, the spontaneous electromyographic activity of myofascial trigger points is consistent with the hyperactive muscle spindles [50].
Neck pain often causes proprioceptive deficits and muscle tension. Chronic pain seems to be associated with a decrease in painful muscle activity and an increase in the activity of the ipsilateral and contralateral non-painful muscles [51]. Chronic neck pain may also be associated with a reduction in the cross-sectional area of the neck muscles and deficiencies in muscle function in terms of strength, accuracy, acuity, endurance, and range of motion. [52]. However, changes in the structure and function of the neck muscles can change the discharge of proprioception, thus affecting afferent inputs and leading to changes in proprioception [53]. Cervical pain can distort sensorimotor control through side-specific changes. Even when the pain itself is significantly reduced, there can be long-term effects on proprioception [54]. In addition, neck pain can also influence the central modulation of the neck proprioceptive input and subcortical and cortical reorganization at multiple levels of the somatosensory system [55].
Cervical trauma, such as whiplash injuries, can cause cervical proprioception disorders. Neck pain, limitation of movement, and strains on the cervical muscles could modify the proprioceptive input in a whiplash-injured neck [7]. Whiplash injuries can directly damage the proprioceptive receptors in facet joints, discs, and muscles. The release of inflammatory mediators associated with trauma activates chemically sensitive nerve endings in the joints and discs [22]. Muscle morphological changes, such as fatty infiltration [56] and psychological distress when activated in a sympathetic nervous system [57], also influence the function and activity of the cervical muscles.
Cervical degenerative diseases are the most common cervical disorders in humans. Patients with cervical spondylosis are often accompanied by neck pain, neck muscle fatigue, neck stiffness, or dizziness. Most chronic idiopathic neck pain is caused by the degeneration of the cervical disc or facet joints [58]. Recent studies found that a multitude of mechanoreceptors, including Ruffini corpuscles and nociceptive receptors growing into the degenerative cervical intervertebral disc, is related to dizziness and neck pain [26][27][59]. The degenerative changes of the cervical disc are always related to inflammation and abnormal mechanical stimulation [26]. In the inflammatory setting, combined with a marked increase in the number of mechanoreceptors, it is likely that the firing characteristics of the mechanoreceptors may become overactive, which in turn induces erroneous proprioceptive afferents [26][27]. In addition, the mechanoreceptors in the cervical intervertebral discs and facet joints can control and monitor the activity of muscle spindles and paraspinal muscles [20]. The electrical stimulation of the facet joint capsule and the disc can cause the contraction of the paravertebral muscles [60]. In pathological situations, such as cervical facet osteoarthritis or intervertebral disc degeneration, the erroneous proprioceptive afferents are directly generated by the mechanoreceptors into the cervical facet joint capsules and the discs or are indirectly generated by neck pain, which becomes mismatched with the normal vestibular and visual information in multiple levels of the central nervous systems and results in the symptoms of dizziness, disorientation, and balance disturbances [26]. Cervical spinal cord or nerve root compression due to cervical myelopathy or radiculopathy can also affect proprioceptive transmission and cause proprioceptive deficits [61][62]. In addition, surgical decompression can reduce dizziness symptoms in patients with cervical root or spinal cord compression [27][63][64][65][66]. Information from the cervical muscle spindles and GTOs match each other in the appropriate cervical alignment [14][67]. Therefore, an abnormal cervical curvature in cervical degenerative diseases may cause abnormal activities in the muscle spindles and GTOs, leading to an aberrant proprioceptive input.
Cervicogenic dizziness is often accompanied by autonomic nervous disorder symptoms, such as palpitations, nausea, and vomiting. As mentioned above, neuroanatomic studies have shown that there are neural projections between the vestibular nucleus, central cervical nucleus, reticular formation, and parabrachial nucleus. Therefore, it is reasonable to believe that the sensory mismatch of vestibular, visual, and neck proprioceptive systems in the vestibular nucleus integration will affect the function of the reticular formation and parabrachial nucleus, leading to an abnormal sympathetic outflow, subsequently causing cardiovascular and gastrointestinal symptoms. In addition, sympathetic innervation is directly related to the intrafusal fibers [68], and a sympathetic outflow can intensely inhibit the proprioceptive input of cat cervical muscle spindles [21].
This entry is adapted from the peer-reviewed paper 10.3390/jcm11216293
References
- Vural, M.; Karan, A.; Albayrak Gezer, İ.; Çalışkan, A.; Atar, S.; Yıldız Aydın, F.; Coşkun Benlidayı, İ.; Gökşen, A.; Koldaş Doğan, Ş.; Karacan, G.; et al. Prevalence, etiology, and biopsychosocial risk factors of cervicogenic dizziness in patients with neck pain: A multi-center, cross-sectional study. Turk. J. Phys. Med. Rehabil. 2021, 67, 399–408.
- Barin, K.; Dodson, E.E. Dizziness in the elderly. Otolaryngol. Clin. North Am. 2011, 44, 437–454.
- Bisdorff, A.; Von Brevern, M.; Lempert, T.; Newman-Toker, D.E. Classification of vestibular symptoms: Towards an international classification of vestibular disorders. J. Vestib. Res. 2009, 19, 1–13.
- Ryan, G.M.; Cope, S. Cervical vertigo. Lancet 1955, 269, 1355–1358.
- Takahashi, S. Importance of cervicogenic general dizziness. J. Rural. Med. 2018, 13, 48–56.
- Li, Y.; Peng, B. Pathogenesis, diagnosis, and treatment of cervical vertigo. Pain Physician 2015, 18, E583–E595.
- Yacovino, D.A.; Hain, T.C. Clinical characteristics of cervicogenic-related dizziness and vertigo. Semin Neurol. 2013, 33, 244–255.
- Wrisley, D.M.; Sparto, P.J.; Whitney, S.L.; Furman, J.M. Cervicogenic dizziness: A review of diagnosis and treatment. J. Orthop. Sports Phys. Ther. 2000, 30, 755–766.
- Brandt, T.; Bronstein, A.M. Cervical vertigo. J. Neurol. Neurosurg. Psychiatry 2001, 71, 8–12.
- Riemann, B.L.; Lephart, S.M. The sensorimotor system, part I: The physiologic basis of functional joint stability. J. Athl. Train. 2002, 37, 71–79.
- Hillier, S.; Immink, M.; Thewlis, D. Assessing proprioception: A systematic review of possibilities. Neurorehabil. Neural. Repair 2015, 29, 933–949.
- Tuthill, J.C.; Azim, E. Proprioception. Curr. Biol. 2018, 28, R194–R203.
- Jerosch, J.; Prymka, M. Proprioception and joint stability. Knee Surg. Sports Traumatol. Arthrosc. 1996, 4, 171–179.
- Kröger, S. Proprioception 2.0: Novel functions for muscle spindles. Curr. Opin. Neurol. 2018, 31, 592–598.
- Proske, U.; Gandevia, S.C. The proprioceptive senses: Their roles in signaling body shape, body position and movement, and muscle force. Physiol. Rev. 2012, 92, 1651–1697.
- Kulkarni, V.; Chandy, M.J.; Babu, K.S. Quantitative study of muscle spindles in suboccipital muscles of human foetuses. Neurol. India 2001, 49, 355–359.
- Boyd-Clark, L.C.; Briggs, C.A.; Galea, M.P. Muscle spindle distribution, morphology, and density in longus colli and multifidus muscles of the cervical spine. Spine 2002, 27, 694–701.
- Banks, R.W. An allometric analysis of the number of muscle spindles in mammalian skeletal muscles. J. Anat. 2006, 208, 753–768.
- Proske, U.; Gandevia, S.C. The kinaesthetic senses. J. Physiol. 2009, 587, 4139–4146.
- Sjölander, P.; Johansson, H.; Djupsjöbacka, M. Spinal and supraspinal effects of activity in ligament afferents. J. Electromyogr. Kinesiol. 2002, 12, 167–176.
- Richmond, F.J.; Abrahams, V.C. Physiological properties of muscle spindles in dorsal neck muscles of the cat. J. Neurophysiol. 1979, 42, 604–617.
- Thunberg, J.; Hellström, F.; Sjölander, P.; Bergenheim, M.; Wenngren, B.; Johansson, H. Influences on the fusimotor-muscle spindle system from chemosensitive nerve endings in cervical facet joints in the cat: Possible implications for whiplash induced disorders. Pain 2001, 91, 15–22.
- Freeman, M.A.; Wyke, B. The innervation of the knee joint. An anatomical and histological study in the cat. J. Anat. 1967, 101, 505–532.
- McLain, R.F. Mechanoreceptor endings in human cervical facet joints. Spine 1994, 19, 495–501.
- Cavanaugh, J.M.; Lu, Y.; Chen, C.; Kallakuri, S. Pain generation in lumbar and cervical facet joints. J. Bone Joint Surg. Am. 2006, 88, 63–67.
- Yang, L.; Yang, C.; Pang, X.; Li, D.; Yang, H.; Zhang, X.; Yang, Y.; Peng, B. Mechanoreceptors in diseased cervical intervertebral disc and vertigo. Spine 2017, 42, 540–546.
- Yang, L.; Chen, J.; Yang, C.; Pang, X.; Li, D.; Wu, B.; Wu, Y.; Lu, X.; Xu, J.; Chen, X.; et al. Cervical intervertebral disc degeneration contributes to dizziness: A clinical and immunohistochemical study. World Neurosurg. 2018, 119, e686–e693.
- Peng, B. Cervical vertigo: Historical reviews and advances. World Neurosurg. 2018, 109, 347–350.
- Gdowski, G.T.; McCrea, R.A. Neck proprioceptive inputs to primate vestibular nucleus neurons. Exp. Brain Res. 2000, 135, 511–526.
- Humphreys, B.K. Cervical outcome measures: Testing for postural stability and balance. J. Manipulative Physiol. Ther. 2008, 31, 540–546.
- Angelaki, D.E.; Cullen, K.E. Vestibular system: The many facets of a multimodal sense. Annu. Rev. Neurosci. 2008, 31, 125–150.
- Rubin, A.M.; Young, J.H.; Milne, A.C.; Schwarz, D.W.; Fredrickson, J.M. Vestibular-neck integration in the vestibular nuclei. Brain Res. 1975, 96, 99–102.
- Mergner, T.; Hlavacka, F.; Schweigart, G. Interaction of vestibular and proprioceptive inputs. J. Vestib. Res. 1993, 3, 41–57.
- Armstrong, B.; McNair, P.; Taylor, D. Head and neck position sense. Sports Med. 2008, 38, 101–117.
- Matsushita, M.; Gao, X.; Yaginuma, H. Spinovestibular projections in the rat, with particular reference to projections from the central cervical nucleus to the lateral vestibular nucleus. J. Comp. Neurol. 1995, 361, 334.
- Dutia, M.B. The muscles and joints of the neck: Their specialisation and role in head movement. Prog. Neurobiol. 1991, 37, 165–178.
- Hongo, T.; Kitama, T.; Yoshida, K. Integration of vestibular and neck afferent signals in the central cervical nucleus. Prog. Brain Res. 1988, 76, 155–162.
- Pompeiano, O. Spinovestibular relations: Anatomical and physiological aspects. Prog. Brain Res. 1972, 37, 263–296.
- McCall, A.A.; Miller, D.M.; Yates, B.J. Descending influences on vestibulospinal and vestibulosympathetic reflexes. Front. Neurol. 2017, 8, 112.
- Balaban, C.D. Vestibular nucleus projections to the parabrachial nucleus in rabbits: Implications for vestibular influences on the autonomic nervous system. Exp. Brain Res. 1996, 108, 367–381.
- de Jong, P.T.; de Jong, J.M.; Cohen, B.; Jongkees, L.B. Ataxia and nystagmus induced by injection of local anesthetics in the Neck. Ann. Neurol. 1977, 1, 240–246.
- Biemond, A.; De Jong, J.M. On cervical nystagmus and related disorders. Brain 1969, 92, 437–458.
- Ishikawa, K.; Matsuzaki, Z.; Yokomizo, M.; Terada, N.; Miyazaki, S.; Togawa, K. Effect of unilateral section of cervical afferent nerve upon optokinetic response and vestibular nystagmus induced by sinusoidal rotation in guinea pigs. Acta Otolaryngol. Suppl. 1998, 537, 6–10.
- Lennerstrand, G.; Han, Y.; Velay, J.L. Properties of eye movements induced by activation of neck muscle proprioceptors. Graefes Arch. Clin. Exp. Ophthalmol. 1996, 234, 703–709.
- Taylor, J.L.; McCloskey, D.I. Illusions of head and visual target displacement induced by vibration of neck muscles. Brain 1991, 114, 755–759.
- Kavounoudias, A.; Gilhodes, J.C.; Roll, R.; Roll, J.P. From balance regulation to body orientation: Two goals for muscle proprioceptive information processing? Exp. Brain Res. 1999, 124, 80–88.
- Bronstein, A.M.; Golding, J.F.; Gresty, M.A. Visual vertigo, motion sickness, and disorientation in vehicles. Semin. Neurol. 2020, 40, 116–129.
- Malmström, E.M.; Karlberg, M.; Holmström, E.; Fransson, P.A.; Hansson, G.A.; Magnusson, M. Influence of prolonged unilateral cervical muscle contraction on head repositioning--decreased overshoot after a 5-min static muscle contraction task. Man. Ther. 2010, 15, 229–234.
- Johansson, H.; Sojka, P. Pathophysiological mechanisms involved in genesis and spread of muscular tension in occupational muscle pain and in chronic musculoskeletal pain syndromes: A hypothesis. Med. Hypotheses 1991, 35, 196–203.
- Hubbard, D.R.; Berkoff, G.M. Myofascial trigger points show spontaneous needle EMG activity. Spine 1993, 18, 1803–1807.
- Lund, J.P.; Donga, R.; Widmer, C.G.; Stohler, C.S. The pain-adaptation model: A discussion of the relationship between chronic musculoskeletal pain and motor activity. Can. J. Physiol. Pharmacol. 1991, 69, 683–694.
- De Pauw, R.; Coppieters, I.; Kregel, J.; De Meulemeester, K.; Danneels, L.; Cagnie, B. Does muscle morphology change in chronic neck pain patients?—A systematic review. Man. Ther. 2016, 22, 42–49.
- Reddy, R.S.; Tedla, J.S.; Dixit, S.; Abohashrh, M. Cervical proprioception and its relationship with neck pain intensity in subjects with cervical spondylosis. BMC Musculoskelet. Disord. 2019, 20, 447.
- Malmström, E.M.; Westergren, H.; Fransson, P.A.; Karlberg, M.; Magnusson, M. Experimentally induced deep cervical muscle pain distorts head on trunk orientation. Eur. J. Appl. Physiol. 2013, 113, 2487–2499.
- Tinazzi, M.; Fiaschi, A.; Rosso, T.; Faccioli, F.; Grosslercher, J.; Aglioti, S.M. Neuroplastic changes related to pain occur at multiple levels of the human somatosensory system: A somatosensory-evoked potentials study in patients with cervical radicular pain. J. Neurosci. 2000, 20, 9277–9283.
- Elliott, J.; Jull, G.; Noteboom, J.T.; Darnell, R.; Galloway, G.; Gibbon, W.W. Fatty infiltration in the cervical extensor muscles in persistent whiplash-associated disorders: A magnetic resonance imaging analysis. Spine 2006, 31, E847–E855.
- Passatore, M.; Roatta, S. Influence of sympathetic nervous system on sensorimotor function: Whiplash associated disorders (WAD) as a model. Eur. J. Appl. Physiol. 2006, 98, 423–449.
- Cohen, S.P.; Hooten, W.M. Advances in the diagnosis and management of neck pain. BMJ 2017, 358, j3221.
- Wu, B.; Yang, L.; Peng, B. Ingrowth of nociceptive receptors into diseased cervical intervertebral disc is associated with discogenic neck pain. Pain Med. 2019, 20, 1072–1077.
- Indahl, A.; Kaigle, A.; Reikerås, O.; Holm, S. Electromyographic response of the porcine multifidus musculature after nerve stimulation. Spine 1995, 20, 2652–2658.
- Persson, L.; Karlberg, M.; Magnusson, M. Effects of different treatments on postural performance in patients with cervical root compression. A randomized prospective study assessing the importance of the neck in postural control. J. Vestib. Res. 1996, 6, 439–453.
- Takayama, H.; Muratsu, H.; Doita, M.; Harada, T.; Yoshiya, S.; Kurosaka, M. Impaired joint proprioception in patients with cervical myelopathy. Spine 2005, 30, 83–86.
- Yi, Y.Y.; Xu, H.W.; Zhang, S.B.; Hu, T.; Wang, S.J.; Wu, D.S. Does the C3/4 disc play a role in cervical spondylosis with dizziness? A retrospective study. Int. Orthop. 2020, 44, 1159–1168.
- Li, C.; Qi, Y.; Liu, G.; Yin, X.; Jin, Y.; Jiang, Z.; Li, P.; Kang, X.; Ye, C. Long-term clinical outcomes of percutaneous cervical nucleoplasty for cervical degenerative diseases with neck pain and cervical vertigo. World Neurosurg. 2020, 133, e205–e210.
- Peng, B.; Yang, L.; Yang, C.; Pang, X.; Chen, X.; Wu, Y. The effectiveness of anterior cervical decompression and fusion for the relief of dizziness in patients with cervical spondylosis: A multicentre prospective cohort study. Bone Joint J. 2018, 100, 81–87.
- Li, J.; Jiang, D.J.; Wang, X.W.; Yuan, W.; Liang, L.; Wang, Z.C. Mid-term outcomes of anterior cervical fusion for cervical spondylosis with sympathetic symptoms. Clin. Spine Surg. 2016, 29, 255–260.
- Blecher, R.; Krief, S.; Galili, T.; Biton, I.E.; Stern, T.; Assaraf, E.; Levanon, D.; Appel, E.; Anekstein, Y.; Agar, G.; et al. The proprioceptive system masterminds spinal alignment: Insight into the mechanism of scoliosis. Dev. Cell. 2017, 42, 388–399.e3.
- Radovanovic, D.; Peikert, K.; Lindström, M.; Domellöf, F.P. Sympathetic innervation of human muscle spindles. J. Anat. 2015, 226, 542–548.
This entry is offline, you can click here to edit this entry!
