Climate change and the associated disturbances have disrupted the relative stability of tree species composition in hemiboreal forests. The natural ecology of forest communities, including species occurrence and composition, forest structure, and food webs, have been affected. Yet, the hemiboreal forest zone of Lithuania is the least studied in the country for climate change risks and possible management adaption techniques. Moreover, the adverse effects of the phenomenon of global climate change can undermine the resilience of forest ecology in terms of its capacity for natural regeneration to occur successfully on the scale of the expected time. Especially since forests are meta-networks that constantly evolve toward the successional processes and patterns that cannot be reached at an individual species level. Therefore, in the context of hemiboreal forests, assisted natural regeneration lays the groundwork necessary to consider the life-cycle features of trees that affect the organic relationships between individual species and ecological communities indirectly via their effects on growth, reproduction, and survival, such as tree regeneration strategies that correspond to the various trade-offs in the adaptations to competition, stress, and forest disturbances. With the concept of assisted natural regeneration, embedded in the principle of deep ecology, the natural powers of forest ecology can be revitalized and fast-tracked to keep up with the pace of global climate change or even overtake it ahead its disruptions.
- hemiboreal trees
- assisted natural regeneration
- deep ecology
1. Introduction
2. European Hemiboreal Tree Species: The Case of Lithuania
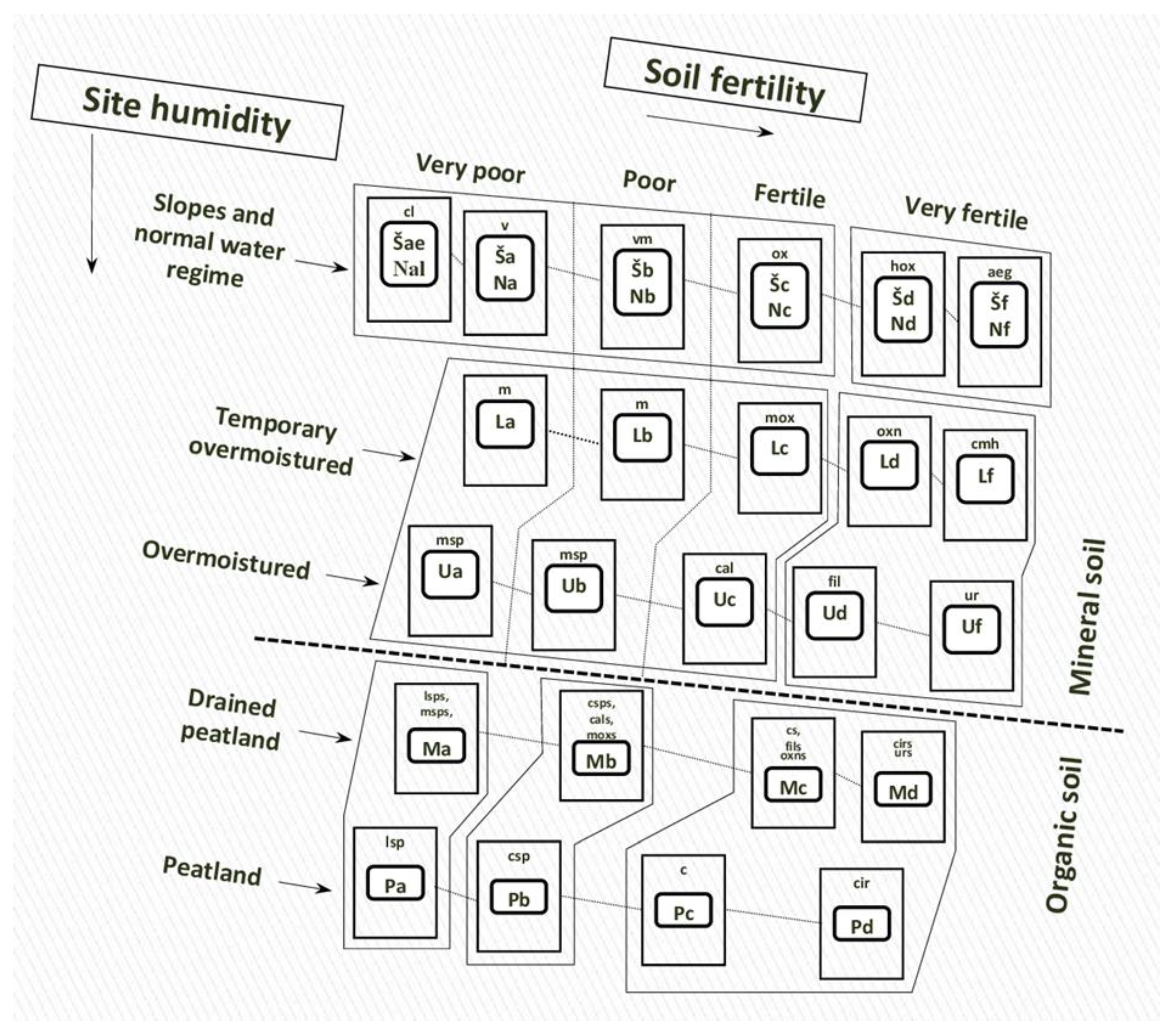
3. Hemiboreal Tree Dynamics
3.1. Tree Regeneration Strategy
| Regeneration Strategy: C—Colonization O—Occupation I—Invasion E—Expansion |
Forest Habitat Types (NATURA 2000 Codes)/Codes of the Lithuanian Forest Type Series * and Forest Site Types ** | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mixed Broadleaved Forests | Norway Spruce Mixed Forests | Scots Pine Forests | |||||||||||||||||
| (9020 9080 91F0 91E0) | (9050 9160 9180 9190 9070) | (9010 9060 91D0 91T0) | |||||||||||||||||
| aeg * Nf ** |
cmh Lf |
cal Uc |
Fil Ud |
ur Uf |
cir Pd |
c Pc |
ox Nc |
mox Lc |
hox Nd |
oxn Ld |
cl Nal |
v Na |
vm Nb |
m Lb |
msp Ub |
csp Pb |
lsp Pa |
||
| C | Alnus glutinosa | G | G | G | G | G, S | G, S | G | |||||||||||
| C | Alnus incana | G | G | g | G | s, g | s, g | G | G | ||||||||||
| C | Betula pendula | G | G | g | G | g | S | S | S | S | s | s | S | ||||||
| C | Betula pubescens | G | G | S | S | g | G | S | s | S | |||||||||
| C | Larix decidua | S | s | ||||||||||||||||
| C | Pinus sylvestris | s, m | M | M | M | M | M | M | M | M | M | ||||||||
| O | Fraxinus excelsior | G | G | G | g | G | |||||||||||||
| O | Populus tremula | G | G | G | S | S | s | S | S | ||||||||||
| O | Quercus robur | G | G | G | G | g | |||||||||||||
| O | Ulmus laevis | G | G | g | |||||||||||||||
| I | Acer platanoides | G | g | ||||||||||||||||
| I | Carpinus betulus | G | G | ||||||||||||||||
| I | Picea abies | g | G | S | S | S | S | s | s | s | |||||||||
| I | Ulmus glabra | G | |||||||||||||||||
| E | Fagus sylvatica | G | G | ||||||||||||||||
| E | Tilia cordata | G | g | G | g | ||||||||||||||
3.2. Natural Regeneration of European Hemiboreal Tree Species
This entry is adapted from the peer-reviewed paper 10.3390/d14100892
References
- Olff, H.; Alonso, D.; Berg, M.P.; Eriksson, B.K.; Loreau, M.; Piersma, T.; Rooney, N. Parallel Ecological Networks in Ecosystems. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1755–1779.
- Camazine, S.; Deneubourg, J.-L.; Franks, N.R.; Sneyd, J.; Theraula, G.; Bonabeau, E. Self-Organization in Biological Systems; Princeton University Press: Princeton, NJ, USA, 2020; ISBN 978-0-691-21292-0.
- Kazansky, A.B. Bootstrapping of Life through Holonomy and Self-Modification. In Proceedings of the 9th International Conference on Computing Anticipatory Systems, Liege, Belgium, 3–8 August 2009; Dubois, D.M., Ed.; American Institute of Physics: Melville, NY, USA, 2010; pp. 297–306.
- Emery, S.M. Succession: A Closer Look. Nat. Educ. 2010, 3, 10–45.
- Schwarz, C.; Gourgue, O.; van Belzen, J.; Zhu, Z.; Bouma, T.J.; van de Koppel, J.; Ruessink, G.; Claude, N.; Temmerman, S. Self-Organization of a Biogeomorphic Landscape Controlled by Plant Life-History Traits. Nat. Geosci. 2018, 11, 672–677.
- Turner, M.G. Landscape Ecology: The Effect of Pattern on Process. Annu. Rev. Ecol. Syst. 1989, 20, 171–197.
- Minore, D.; Laacke, R.J. Natural Regeneration. In Reforestation Practices in Southwest Oregon and Northern California: Chapter 11; Hobbs, S.D., Tesch, S.D., Owston, P.W., Stewart, R.E., Tappeiner, J.C., Wells, G.E., Eds.; Oregon State University Press: Corvallis, OR, USA, 1992; pp. 258–283. ISBN 0-87437-001-9.
- Bajc, M.; Aravanopoulos, F.; Westergren, M.; Fussi, B.; Kavaliauskas, D.; Alizoti, P.; Kiourtsis, F.; Kraigher, H. Manual for Forest Genetic Monitoring; Slovenian Forestry Institute: Ljubljana, Slovenia, 2020.
- Holbrook, M. Adventures in Complexity: An Essay on Dynamic Open Complex Adaptive Systems, Butterfly Effects, Self-Organizing Order, Coevolution, the Ecological Perspective, Fitness Landscapes, Market Spaces, Emergent Beauty at the Edge of Chaos, and All That Jazz. Acad. Mark. Sci. Rev. 2003, 2003, 1–181.
- Borman, M.M.; Pyke, D.A. Successional Theory and the Desired Plant Community Approach. Rangelands 1994, 16, 82–84.
- Chazdon, R.L. Second Growth: The Promise of Tropical Forest Regeneration in an Age of Deforestation; University of Chicago Press: Chicago, IL, USA, 2014; ISBN 978-0-226-11791-1.
- Chazdon, R.L.; Brancalion, P.H.S.; Laestadius, L.; Bennett-Curry, A.; Buckingham, K.; Kumar, C.; Moll-Rocek, J.; Vieira, I.C.G.; Wilson, S.J. When Is a Forest a Forest? Forest Concepts and Definitions in the Era of Forest and Landscape Restoration. Ambio 2016, 45, 538–550.
- Ivanova, N.; Fomin, V.; Kusbach, A. Experience of Forest Ecological Classification in Assessment of Vegetation Dynamics. Sustainability 2022, 14, 3384.
- Jõgiste, K.; Frelich, L.E.; Laarmann, D.; Vodde, F.; Baders, E.; Donis, J.; Jansons, A.; Kangur, A.; Korjus, H.; Köster, K.; et al. Imprints of Management History on Hemiboreal Forest Ecosystems in the Baltic States. Ecosphere 2018, 9, e02503.
- Angelstam, P.; Manton, M.; Pedersen, S.; Elbakidze, M. Disrupted Trophic Interactions Affect Recruitment of Boreal Deciduous and Coniferous Trees in Northern Europe. Ecol. Appl. 2017, 27, 1108–1123.
- European Commission. New EU Forest Strategy for 2030. In Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions; European Union: Brussels, Belgium, 2021; p. 28.
- Ehlers, A.; Worm, B.; Reusch, T.B.H. Importance of Genetic Diversity in Eelgrass Zostera Marina for Its Resilience to Global Warming. Mar. Ecol. Prog. Ser. 2008, 355, 1–7.
- Wright, D.; Bishop, J.M.; Matthee, C.A.; von der Heyden, S. Genetic Isolation by Distance Reveals Restricted Dispersal across a Range of Life Histories: Implications for Biodiversity Conservation Planning across Highly Variable Marine Environments. Divers. Distrib. 2015, 21, 698–710.
- Orr, H.A. Fitness and Its Role in Evolutionary Genetics. Nat. Rev. Genet. 2009, 10, 531–539.
- Yang, Y.; Wang, L.; Yang, Z.; Xu, C.; Xie, J.; Chen, G.; Lin, C.; Guo, J.; Liu, X.; Xiong, D.; et al. Large Ecosystem Service Benefits of Assisted Natural Regeneration. J. Geophys. Res. Biogeosciences 2018, 123, 676–687.
- Manton, M.; Ruffner, C.; Kibirkštis, G.; Brazaitis, G.; Marozas, V.; Pukienė, R.; Makrickiene, E.; Angelstam, P. Fire Occurrence in Hemi-Boreal Forests: Exploring Natural and Cultural Scots Pine Fire Regimes Using Dendrochronology in Lithuania. Land 2022, 11, 260.
- Karazija, S. Forest Types of Lithuania; Mokslas: Vilnius, Lietuva, 1988; ISBN 978-5-420-00421-0.
- Brazaitis, G.; Marozas, V.; Augutis, D.; Preikša, Ž.; Šaudytė-Manton, S. Lithuanian Forest Habitat Management Recommendations–“Guidelines for the Management of Natural Forest Habitat Types of EC Importance”; Naturalit: Vilnius, Lietuva, 2021.
- Vaičys, M.; Mažvila, J. The Influence of Soil Characteristics on Plant Productivity and Ecological Stability. Ekologija 2009, 55, 99–106.
- Buivydaitė, V. Classification of Soils of Lithuania Based on FAO-UNESCO Soil Classification System and WRB. In Proceedings of the 17th World Congress of Soil Science, Bangkok, Thailand, 14–20 August 2002; The Soil and Fertilizer Society of Thailand: Bangkok, Thailand, 2002; pp. 814–826.
- Kulbokas, G. Experiences from Lithuania-UNECE Technical Workshop on European Forest Types. In Presented at the UNECE Technical Workshop on European Forest Types: Reporting Using the New European Forest Types, Bordeaux, France, 19–21 May 2010; Lithuanian State Forest Service: Kaunas, Lithuania, 2010; pp. 1–16.
- Angelstam, P. Landscape Analysis as a Tool for the Scientific Management of Biodiversity. Ecol. Bull. 1997, 46, 140–170.
- Fomin, V.; Mikhailovich, A.; Zalesov, S.; Popov, A.; Terekhov, G. Development of Ideas within the Framework of the Genetic Approach to the Classification of Forest Types. Balt. For. 2020, 27, 26–39.
- Christensen, N.L.; Peet, R.K. Secondary Forest Succession on the North Carolina Piedmont. In Forest Succession: Concepts and Application; West, D.C., Shugart, H.H., Botkin, D.B., Eds.; Springer Advanced Texts in Life Sciences; Springer: New York, NY, USA, 1981; pp. 230–245. ISBN 978-1-4612-5950-3.
- Angelstam, P.; Kuuluvainen, T. Boreal Forest Disturbance Regimes, Successional Dynamics and Landscape Structures: A European Perspective. Ecol. Bull. 2004, 51, 117–136.
- Angelstam, P.; Dönz-Breuss, M. Measuring Forest Biodiversity at the Stand Scale: An Evaluation of Indicators in European Forest History Gradients. Ecol. Bull. 2004, 51, 305–332. Available online: https://www.jstor.org/stable/20113319 (accessed on 19 September 2022).
- Jandl, R.; Spathelf, P.; Bolte, A.; Prescott, C.E. Forest Adaptation to Climate Change—Is Non-Management an Option? Ann. For. Sci. 2019, 76, 48.
- Russell, G.J. Turnover Dynamics Across Ecological and Geological Scales. In Turnover Dynamics Across Ecological and Geological Scales; Columbia University Press: New York, NY, USA, 2001; pp. 377–404. ISBN 978-0-231-50580-2.
- Neophytou, C.; Heer, K.; Milesi, P.; Peter, M.; Pyhäjärvi, T.; Westergren, M.; Rellstab, C.; Gugerli, F. Genomics and Adaptation in Forest Ecosystems. Tree Genet. Genomes 2022, 18, 12.
- Petrokas, R.; Baliuckas, V.; Manton, M. Successional Categorization of European Hemi-Boreal Forest Tree Species. Plants 2020, 9, 1381.
- Yamamoto, S. Gap Regeneration of Major Tree Species in Different Forest Types of Japan. Vegetatio 1996, 127, 203–213.
- Ulft, L. Regeneration in Natural and Logged Tropical Rain Forest-Modelling Seed Dispersal and Regeneration of Tropical Trees in Guyana; Tropenbos-Guyana Series 12; Tropenbos International: Georgetown, Guyana, 2004; ISBN 90-5113-076-7.
- Franklin, J. Regeneration and Growth of Pioneer and Shade-tolerant Rain Forest Trees in Tonga. New Zealand J. Bot. 2003, 41, 669–684.
- Ning, Z.; Hong, J.; Yong-Yan, J. A Phenology Study on the Common Tree Species of Natural Secondary Forests in Northeast China. Chin. J. Plant Ecol. 1990, 14, 336.
- Clark, D.A.; Clark, D.B. Life History Diversity of Canopy and Emergent Trees in a Neotropical Rain Forest. Ecol. Monogr. 1992, 62, 315–344.
- Whitmore, T.C. Canopy Gaps and the Two Major Groups of Forest Trees. Ecology 1989, 70, 536–538.
- Grime, J.P. Evidence for the Existence of Three Primary Strategies in Plants and Its Relevance to Ecological and Evolutionary Theory. Am. Nat. 1977, 111, 1169–1194.
- Hill, M.O.; Roy, D.B.; Thompson, K. Hemeroby, Urbanity and Ruderality: Bioindicators of Disturbance and Human Impact. J. Appl. Ecol. 2002, 39, 708–720.
- Petrokas, R.; Kavaliauskas, D. Concept for Genetic Monitoring of Hemiboreal Tree Dynamics in Lithuania. Land 2022, 11, 1249.
- Navasaitis, M.; Ozolinčius, R.; Smaliukas, D.; Balevičienė, J.M. Lietuvos Dendroflora: Monografija; Lutute: Kaunas, Lietuva, 2003; ISBN 978-9955-575-35-1.
- Shorohova, E.; Kuuluvainen, T.; Kangur, A.; Jõgiste, K. Natural Stand Structures, Disturbance Regimes and Successional Dynamics in the Eurasian Boreal Forests: A Review with Special Reference to Russian Studies. Ann. For. Sci. 2009, 66, 201.
- Angelstam, P.K. Maintaining and Restoring Biodiversity in European Boreal Forests by Developing Natural Disturbance Regimes. J. Veg. Sci. 1998, 9, 593–602.
- Rull, V. Quaternary Palaeoecology and Ecological Theory. Orsis 1990, 5, 91–111.
- Hunter, M.L.; Schmiegelow, F.K.A. Wildlife, Forests, and Forestry: Principles of Managing Forests for Biological Diversity, 2nd ed.; Prentice Hall: Boston, MA, USA, 2011; ISBN 978-0-13-501432-5.
- Birks, H. Late-Quaternary Biotic Changes in Terrestrial and Lacustrine Environments, with Particular Reference to North-West Europe. In Handbook of Holocene Palaeoecology and Palaeohydrology; Berglund, B.E., Ed.; Wiley-Interscience; John Wiley & Sons Ltd.: Chichester, UK, 1986.
- Birks, H. Contributions of Quaternary Botany to Modern Ecology and Biogeography. Plant Ecol. Divers. 2019, 12, 189–385.
- Godvod, K.; Brazaitis, G.; Bačkaitis, J.; Kulbokas, G. The Development and Growth of Larch Stands in Lithuania. J. For. Sci. 2018, 64, 199–206.
- Odum, E.P.; Barrett, G.W. Fundamentals of Ecology, 5th ed.; Thomson Brooks/Cole: Belmont, CA, USA, 2005; ISBN 978-0-534-42066-6.
