|
Aspect
|
Function
|
Localization
|
Mediators
|
References
|
|
Epithelial barrier
|
Intestinal barrier
formation and support
Enhancing epithelial healing
Neuropods formation
|
Mucosa
|
proEGF
TGF-β
S-nitrosoglutathione
15d-PGJ2
NGF-β *
Artemin *
|
[38,39,40,41,42,43,44,45]
|
|
Intestinal motility
|
Control of GI motility #
|
Myenteric plexus
|
ATP
|
[46,47,48]
|
|
Enteric neurotransmission
|
Neuronal communication
|
ENS
|
ATP
NFG
GSH
|
[49]
|
|
Immune response
|
Activation of EGCs
|
ENS
|
MHC II class
IL-1β
IL-6
TGF-β
proEGF
GSH
PGE2
|
[50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70]
|
|
Visceral sensitivity
|
Sensitizing/activating nociceptors
|
ENS
|
ATP
GABA
IL-1β
neurotrophins
|
[8,71,72]
|
* Mediators released by enteroendocrine cells; # EGC loss results in impaired GI motility. Abbreviations: 15d-PGJ2, 15-deoxy-Δ12,14-prostaglandin J2; ATP, adenosine triphosphate; EGC, enteric glial cell; EGF, Epidermal growth factor; ENS, enteric nervous system; GABA, gamma amino butyric acid; GI, gastrointestinal; GSH, glutathione; IL, interleukin; MHC, major histocompatibility complex; NGF, nerve growth factor; PGE2, prostaglandin E2; proEGF, proepidermal growth factor; TGF, Transforming growth factor.
3. The Endocannabinoid System
The term cannabinoid comprises a group of at least 66 biologically active terpenophenols which are found in cannabis (
Cannabis sativa) and their synthetic analogs [
127]. Cannabinoids are molecules that act on the endogenous cannabinoid system (ECS), also known as the endocannabinoid system, and are usually divided into three main groups: phytocannabinoids (cannabinoids found in plants), endocannabinoids (endogenous compounds found in animals that modulate cannabinoid receptors); and synthetic cannabinoids (synthetic compounds that may or may not be structurally related that also produce agonistic effects in cannabinoid receptors) [
128].
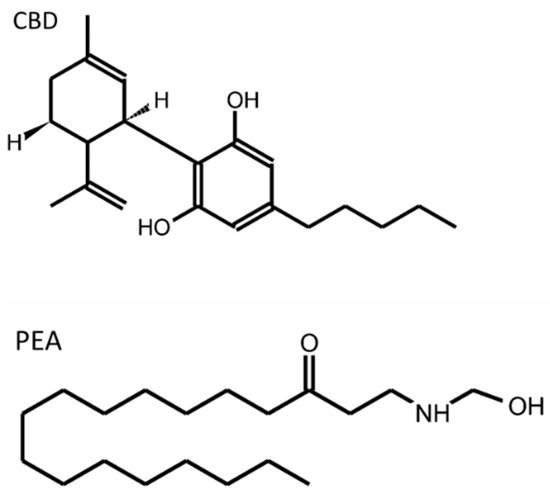
Figure 2 shows the molecular structure of the two cannabinoid compounds that have been more deeply studied in relation to EGCs.
The ECS is composed of cannabinoid receptors (CB1, CB2), their endogenous ligands (endocannabinoids, ECBs), and the enzymes involved in the biosynthesis and degradation of cannabinoids.
Cannabinoid receptors belong to the G-protein coupled receptors (GPCR) family. Their activation results in the inhibition of adenyl cyclase activity and suppression of voltage gated Ca
2+ channels [
129]. Noteworthy, CB receptors possess more than one endogenous agonist: anandamide (
N-arachidonoyl ethanolamine, AEA) and 2-arachidonoyl glycerol (2-AG). ECBs are derivatives of the arachidonic acid, synthesized on demand from the membrane phospholipids in the post-synaptic cells in response to increased levels of intracellular Ca
2+, released immediately after synthesis, and then diffuse throughout the cellular membrane without being stored in vesicles. The action of ECBs is mediated through CB1 or CB2 receptors. Noteworthy, ECBs exhibit different selectivity and affinity: AEA is a partial agonist of CB1 with very weak activity at CB2 receptors, while 2-AG is characterized as a potent agonist of both receptors. Besides these compounds, there are other ECBs that remain less known: 2-arachidonyl glyceryl ether (2-AGE, a CB1 selective agonist) [
130], N-arachidonoyl dopamine (NADA, a CB1 agonist) [
131], and O-arachidonoyl ethanolamine (O-AEA or virodhamine, a partial CB1 agonist and full CB2 agonist) [
132].
Interestingly, it was demonstrated that ECBs may interact with other receptors. For example, AEA binds to TRPV1 [
133,
134]. The effects of TRPV1 activation depend on the site of action: when AEA interacts with pre-synaptic TRPV1 it promotes glutamate release, while the activation of post-synaptic TRPV1 by AEA leads to the reduction of glutamate signaling, inhibition of 2-AG biosynthesis and the blockage of the retrograde action at CB1 receptors. The multi-target action of ECBs may be related to the co-expression of CB receptors and TRPV1 channels in neuronal and non-neuronal cells. It was assessed that TRPV1 are co-localized with CB1 or CB2 receptors in the primary sensory neurons of the DRG in rats [
135,
136,
137], perivascular neurons [
138], vagus nerve [
139], and in the axons of neurons in the CNS [
140,
141,
142]. Moreover, CB receptors are co-expressed with TRPV1 in the endothelial cells of the brain microvessels (both CB1, CB2) [
143], in the endothelial cells from the rodent mesenteric arteries with cirrhosis (CB1) [
144], dendritic cells [
145], muscle cells (in both rodents and humans), [
146], osteoclasts [
147], keratinocytes [
148], and melanocytes [
149]. G protein-coupled receptor 55 (GPR55) is an orphan receptor that constitutes another, non-classical target, for ECBs. Lysophosphatidylinositol (LPI) was identified as the endogenous ligand for GPR55 [
150]. However, AEA and O-AEA can activate these receptors [
151,
152]. Finally, besides TRPV1 and GPR55, ECBs exhibit binding affinity at peroxisome proliferator-activated receptors (PPAR): AEA, O-AEA, and 2-AGE bind PPARα, 2-AG binds to PPARβ/δ, while AEA, 2-AG and 2-AGE bind to PPARγ in vitro [
152].
After the activation of CB receptors, the remaining ECBs are degraded in the process of hydrolysis or oxidization. The first enzyme discovered to be involved in ECBs degradation was named fatty acid amide hydrolase (FAAH). Its most preferred substrate was found to be AEA. A few years later, other enzymes were discovered, and their properties were characterized: monoacylglycerol lipase (MAGL), α, β-hydrolase-6 (ABHD6), and α, β-hydrolase-12 (ABHD12) [
153,
154,
155]. The process of oxidation involves cyclooxygenase-2 (COX-2) and several lipooxygenases [
155].