A myocyte (also known as a muscle cell) is the type of cell found in some types of muscle tissue. Myocytes develop from myoblasts to form muscles in a process known as myogenesis. There are two specialized forms of myocytes with distinct properties: cardiac, and smooth muscle cells. On the other hand, skeletal muscles are formed by morphological units referred to as muscle fibers. Cardiomyocytes are the cells that form the chambers of the heart, and have a single central nucleus. Skeletal muscle fibers help support and move the body and are called syncytia – multinucleated structures formed by fusion of individual myoblasts during embryonic development. Smooth muscle cells control involuntary movements such as the peristalsis contractions in the oesophagus and stomach.
- skeletal muscles
- muscle cells
- myocyte
1. Structure
1.1. Terminology
The unusual microstructure of muscle cells has led cell biologists to create specialized terminology. However, each term specific to muscle cells has a counterpart that is used in the terminology applied to other types of cells:
| Muscle cell | Other organismal cells |
|---|---|
| sarcoplasm | cytoplasm |
| sarcoplasmic reticulum | smooth endoplasmic reticulum (SER) |
| sarcosome | mitochondrion |
| sarcolemma | cell membrane |
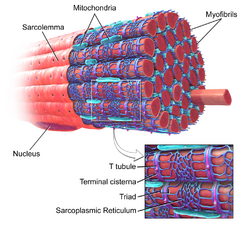
The sarcoplasm is the cytoplasm of a muscle fiber/cell. Most of the sarcoplasm is filled with myofibrils, which are long protein cords composed of myofilaments. The sarcoplasm is also composed of glycogen, a polysaccharide of glucose monomers, which provides energy to the cell with heightened exercise, and myoglobin, the red pigment that stores oxygen until needed for muscular activity.[1]
There are three types of myofilaments:[1]
- Thick filaments, composed of protein molecules called myosin. In striations of muscle bands, these are the dark filaments that make up the A band.
- Thin filaments are composed of protein molecules called actin. In striations of muscle bands, these are the light filaments that make up the I band.
- Elastic filaments are composed of titin, a large springy protein; these filaments anchor the thick filaments to the Z disc.
Together, these myofilaments work to produce a muscle contraction.
The sarcoplasmic reticulum, a specialized type of smooth endoplasmic reticulum, forms a network around each myofibril of the muscle fiber. This network is composed of groupings of two dilated end-sacs called terminal cisternae, and a single transverse tubule, or T tubule, which bores through the cell and emerge on the other side; together these three components form the triads that exist within the network of the sarcoplasmic reticulum, in which each T tubule has two terminal cisternae on each side of it. The sarcoplasmic reticulum serves as reservoir for calcium ions, so when an action potential spreads over the T tubule, it signals the sarcoplasmic reticulum to release calcium ions from the gated membrane channels to stimulate a muscle contraction.[1][2]
The sarcolemma was historically synonymous with the cell membrane of a striated muscle fiber or muscle cell. However, there are two other components which form the sarcolemma - basal lamina and reticular fibers. The cell membrane of the sarcolemma receives and conducts stimuli. In skeletal muscle, at the end of each muscle fiber, the outer layer of the sarcolemma combines with tendon fibers.[3] Within the muscle fiber pressed against the sarcolemma are multiple flattened nuclei; embryologically, this multinuclear condition results from multiple myoblasts fusing to produce each muscle fiber, where each myoblast contributes one nucleus.[1]
1.2. Internal
The cell membrane of a myocyte has several specialized regions, which may include the intercalated disk and the transverse tubular system. The cell membrane is covered by a lamina coat which is approximately 50 nm wide. The laminar coat is separable into two layers; the lamina densa and lamina lucida. In between these two layers can be several different types of ions, including calcium.[4]
The cell membrane is anchored to the cell's cytoskeleton by anchor fibers that are approximately 10 nm wide. These are generally located at the Z lines so that they form grooves and transverse tubules emanate. In cardiac myocytes this forms a scalloped surface.[4]
The cytoskeleton is what the rest of the cell builds off of and has two primary purposes; the first is to stabilize the topography of the intracellular components and the second is to help control the size and shape of the cell. While the first function is important for biochemical processes, the latter is crucial in defining the surface to volume ratio of the cell. This heavily influences the potential electrical properties of excitable cells. Additionally deviation from the standard shape and size of the cell can have negative prognostic impact.[4]
1.3. Myofibrils
Each muscle fiber contains myofibrils, which are very long chains of sarcomeres, the contractile units of the cell. A cell from the biceps brachii muscle may contain 80,000 sarcomeres.[5] The myofibrils of smooth muscle cells are not arranged into sarcomeres. The sarcomeres are composed of thin and thick filaments. Thin filaments are made of actin and attach at Z lines which help them line up correctly with each other.[6] Troponins are found at intervals along the thin filaments. Thick filaments are made of the elongated protein myosin.[7] The sarcomere does not contain organelles or a nucleus. Sarcomeres are marked by Z lines which show the beginning and the end of a sarcomere. Individual myocytes are surrounded by endomysium.
Myocytes are bound together by perimysium into bundles called fascicles; the bundles are then grouped together to form muscle tissue, which is enclosed in a sheath of epimysium. The perimysium contains blood vessels and nerves which provide for the muscle fibers. Muscle spindles are distributed throughout the muscles and provide sensory feedback information to the central nervous system. Myosin is shaped like a long shaft with a rounded end pointed out towards the surface. This structure forms the cross bridge that connects with the thin filaments.[7]
2. Development
A myoblast is a type of embryonic progenitor cell that differentiates to give rise to muscle cells.[8] Differentiation is regulated by myogenic regulatory factors, including MyoD, Myf5, myogenin, and MRF4.[9] GATA4 and GATA6 also play a role in myocyte differentiation.[10]
Skeletal muscle fibers are made when myoblasts fuse together; muscle fibers therefore are cells with multiple nuclei, known as myonuclei, with each cell nucleus originating from a single myoblast. The fusion of myoblasts is specific to skeletal muscle (e.g., biceps brachii) and not cardiac muscle or smooth muscle.
Myoblasts in skeletal muscle that do not form muscle fibers dedifferentiate back into myosatellite cells. These satellite cells remain adjacent to a skeletal muscle fiber, situated between the sarcolemma and the basement membrane[11] of the endomysium (the connective tissue investment that divides the muscle fascicles into individual fibers). To re-activate myogenesis, the satellite cells must be stimulated to differentiate into new fibers.
Myoblasts and their derivatives, including satellite cells, can now be generated in vitro through directed differentiation of pluripotent stem cells.[12]
Kindlin-2 plays a role in developmental elongation during myogenesis.[13]
2.1. Muscle Fiber Growth
Muscle fibers grow when exercised and shrink when not in use. This is due to the fact that exercise stimulates the increase in myofibrils which increase the overall size of muscle cells. Well exercised muscles can not only add more size but can also develop more mitochondria, myoglobin, glycogen and a higher density of capillaries. However muscle cells cannot divide to produce new cells, and as a result we have fewer muscle cells as an adult than a newborn.[14]
3. Function
3.1. Muscle Contraction
When contracting, thin and thick filaments slide with respect to each other by using adenosine triphosphate. This pulls the Z discs closer together in a process called sliding filament mechanism. The contraction of all the sarcomeres results in the contraction of the whole muscle fiber. This contraction of the myocyte is triggered by the action potential over the cell membrane of the myocyte. The action potential uses transverse tubules to get from the surface to the interior of the myocyte, which is continuous within the cell membrane. Sarcoplasmic reticula are membranous bags that transverse tubules touch but remain separate from. These wrap themselves around each sarcomere and are filled with Ca2+.[7]
Excitation of a myocyte causes depolarization at its synapses, the neuromuscular junctions, which triggers action potential. With a singular neuromuscular junction, each muscle fiber receives input from just one somatic efferent neuron. Action potential in a somatic efferent neuron causes the release of the neurotransmitter acetylcholine.[15]
When the acetylcholine is released it diffuses across the synapse and binds to a receptor on the sarcolemma, a term unique to muscle cells that refers to the cell membrane. This initiates an impulse that travels across the sarcolemma.[14]
When the action potential reaches the sarcoplasmic reticulum it triggers the release of Ca2+ from the Ca2+ channels. The Ca2+ flows from the sarcoplasmic reticulum into the sarcomere with both of its filaments. This causes the filaments to start sliding and the sarcomeres to become shorter. This requires a large amount of ATP, as it is used in both the attachment and release of every myosin head. Very quickly Ca2+ is actively transported back into the sarcoplasmic reticulum, which blocks the interaction between the thin and thick filament. This in turn causes the muscle cell to relax.[14]
3.2. Kinds of Contraction
There are four main different types of muscle contraction: twitch, treppe, tetanus and isometric/isotonic. Twitch contraction is the process in which a single stimulus signals for a single contraction. In twitch contraction the length of the contraction may vary depending on the size of the muscle cell. During treppe (or summation) contraction muscles do not start at maximum efficiency; instead they achieve increased strength of contraction due to repeated stimuli. Tetanus involves a sustained contraction of muscles due to a series of rapid stimuli, which can continue until the muscles fatigue. Isometric contractions are skeletal muscle contractions that do not cause movement of the muscle. However, isotonic contractions are skeletal muscle contractions that do cause movement.[14]
Specialized cardiomyocytes located in the sinoatrial node are responsible for generating the electrical impulses that control the heart rate. These electrical impulses coordinate contraction throughout the remaining heart muscle via the electrical conduction system of the heart. Sinoatrial node activity is modulated, in turn, by nerve fibres of both the sympathetic and parasympathetic nervous systems. These systems act to increase and decrease, respectively, the rate of production of electrical impulses by the sinoatrial node.
4. Fiber Typing
There are numerous methods employed for fiber-typing, and confusion between the methods is common among non-experts. Two commonly confused methods are histochemical staining for myosin ATPase activity and immunohistochemical staining for Myosin heavy chain (MHC) type. Myosin ATPase activity is commonly—and correctly—referred to as simply "fiber type", and results from the direct assaying of ATPase activity under various conditions (e.g. pH).[16] Myosin heavy chain staining is most accurately referred to as "MHC fiber type", e.g. "MHC IIa fibers", and results from determination of different MHC isoforms.[16] These methods are closely related physiologically, as the MHC type is the primary determinant of ATPase activity. Note, however, that neither of these typing methods is directly metabolic in nature; they do not directly address oxidative or glycolytic capacity of the fiber. When "type I" or "type II" fibers are referred to generically, this most accurately refers to the sum of numerical fiber types (I vs. II) as assessed by myosin ATPase activity staining (e.g. "type II" fibers refers to type IIA + type IIAX + type IIXA... etc.).
Below is a table showing the relationship between these two methods, limited to fiber types found in humans. Note the sub-type capitalization used in fiber typing vs. MHC typing, and that some ATPase types actually contain multiple MHC types. Also, a subtype B or b is not expressed in humans by either method.[17] Early researchers believed humans to express a MHC IIb, which led to the ATPase classification of IIB. However, later research showed that the human MHC IIb was in fact IIx,[17] indicating that the IIB is better named IIX. IIb is expressed in other mammals, so is still accurately seen (along with IIB) in the literature. Non human fiber types include true IIb fibers, IIc, IId, etc.
| ATPase type | MHC heavy chain(s) |
|---|---|
| Type I | MHC Iβ |
| Type IC | MHC Iβ > MHC IIa |
| Type IIC | MHC IIa > MHC Iβ |
| Type IIA | MHC IIa |
| Type IIAX | MHC IIa > MHC IIx |
| Type IIXA | MHC IIx > MHC IIa |
| Type IIX | MHC IIx |
Further fiber typing methods are less formally delineated, and exist on more of a spectrum. They tend to be focused more on metabolic and functional capacities (i.e., oxidative vs. glycolytic, fast vs. slow contraction time). As noted above, fiber typing by ATPase or MHC does not directly measure or dictate these parameters. However, many of the various methods are mechanistically linked, while others are correlated in vivo.[20][21] For instance, ATPase fiber type is related to contraction speed, because high ATPase activity allows faster crossbridge cycling.[16] While ATPase activity is only one component of contraction speed, type I fibers are "slow", in part, because they have low speeds of ATPase activity in comparison to type II fibers. However, measuring contraction speed is not the same as ATPase fiber typing.
Because of these types of relationships, Type I and Type II fibers have relatively distinct metabolic, contractile, and motor-unit properties. The table below differentiates these types of properties. These types of properties—while they are partly dependent on the properties of individual fibers—tend to be relevant and measured at the level of the motor unit, rather than individual fiber.[16]
| Properties | Type I fibers | Type IIA fibers | Type IIX fibers |
|---|---|---|---|
| Motor Unit Type | Slow Oxidative (SO) | Fast Oxidative/Glycolytic (FOG) | Fast Glycolytic (FG) |
| Twitch speed | Slow | Fast | Fast |
| Twitch force | Small | Medium | Large |
| Resistance to fatigue | High | High | Low |
| Glycogen content | Low | High | High |
| Capillary supply | Rich | Rich | Poor |
| Myoglobin | High | High | Low |
| Red color | Dark | Dark | Pale |
| Mitochondrial density | High | High | Low |
| Capillary density | High | Intermediate | Low |
| Oxidative enzyme capacity | High | Intermediate-high | Low |
| Z-line width | Intermediate | Wide | Narrow |
| Alkaline ATPase activity | Low | High | High |
| Acidic ATPase activity | High | Medium-high | Low |
- Fiber color
Traditionally, fibers were categorized depending on their varying color, which is a reflection of myoglobin content. Type I fibers appear red due to the high levels of myoglobin. Red muscle fibers tend to have more mitochondria and greater local capillary density. These fibers are more suited for endurance and are slow to fatigue because they use oxidative metabolism to generate ATP (adenosine triphosphate). Less oxidative type II fibers are white due to relatively low myoglobin and a reliance on glycolytic enzymes.
- Twitch speed
Fibers can also be classified on their twitch capabilities, into fast and slow twitch. These traits largely, but not completely, overlap the classifications based on color, ATPase, or MHC.
Some authors define a fast twitch fiber as one in which the myosin can split ATP very quickly. These mainly include the ATPase type II and MHC type II fibers. However, fast twitch fibers also demonstrate a higher capability for electrochemical transmission of action potentials and a rapid level of calcium release and uptake by the sarcoplasmic reticulum. The fast twitch fibers rely on a well-developed, anaerobic, short term, glycolytic system for energy transfer and can contract and develop tension at 2–3 times the rate of slow twitch fibers. Fast twitch muscles are much better at generating short bursts of strength or speed than slow muscles, and so fatigue more quickly.[22]
The slow twitch fibers generate energy for ATP re-synthesis by means of a long term system of aerobic energy transfer. These mainly include the ATPase type I and MHC type I fibers. They tend to have a low activity level of ATPase, a slower speed of contraction with a less well developed glycolytic capacity. They contain high mitochondrial volumes, and the high levels of myoglobin that give them a red pigmentation. They have been demonstrated to have high concentrations of mitochondrial enzymes, thus they are fatigue resistant. Slow twitch muscles fire more slowly than fast twitch fibers, but are able to contract for a longer time before fatiguing.[22]
- Type distribution
Individual muscles tend to be a mixture of various fiber types, but their proportions vary depending on the actions of that muscle and the species. For instance, in humans, the quadriceps muscles contain ~52% type I fibers, while the soleus is ~80% type I.[23] The orbicularis oculi muscle of the eye is only ~15% type I.[23] Motor units within the muscle, however, have minimal variation between the fibers of that unit. It is this fact that makes the size principal of motor unit recruitment viable.
The total number of skeletal muscle fibers has traditionally been thought not to change. It is believed there are no sex or age differences in fiber distribution; however, proportions of fiber types vary considerably from muscle to muscle and person to person. Sedentary men and women (as well as young children) have 45% type II and 55% type I fibers. People at the higher end of any sport tend to demonstrate patterns of fiber distribution e.g. endurance athletes show a higher level of type I fibers. Sprint athletes, on the other hand, require large numbers of type IIX fibers. Middle distance event athletes show approximately equal distribution of the two types. This is also often the case for power athletes such as throwers and jumpers. It has been suggested that various types of exercise can induce changes in the fibers of a skeletal muscle.[24] It is thought that if you perform endurance type events for a sustained period of time, some of the type IIX fibers transform into type IIA fibers. However, there is no consensus on the subject. It may well be that the type IIX fibers show enhancements of the oxidative capacity after high intensity endurance training which brings them to a level at which they are able to perform oxidative metabolism as effectively as slow twitch fibers of untrained subjects. This would be brought about by an increase in mitochondrial size and number and the associated related changes, not a change in fiber type.
The content is sourced from: https://handwiki.org/wiki/Biology:Myocyte
References
- Saladin, K (2012). Anatomy & Physiology: The Unity of Form and Function (6th ed.). New York: McGraw-Hill. pp. 403–405. ISBN 978-0-07-337825-1.
- Sugi, Haruo; Abe, T; Kobayashi, T; Chaen, S; Ohnuki, Y; Saeki, Y; Sugiura, S; Guerrero-Hernandez, Agustin (2013). "Enhancement of force generated by individual myosin heads in skinned rabbit psoas muscle fibers at low ionic strength". PLoS ONE 8 (5): e63658. doi:10.1371/journal.pone.0063658. PMID 23691080. Bibcode: 2013PLoSO...863658S. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=3655179
- Bentzinger, CF; Wang, YX; Rudnicki, MA (1 February 2012). "Building muscle: molecular regulation of myogenesis.". Cold Spring Harbor Perspectives in Biology 4 (2): a008342. doi:10.1101/cshperspect.a008342. PMID 22300977. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=3281568
- Ferrari, Roberto. "Healthy versus sick myocytes: metabolism, structure and function". Oxford University Press. Archived from the original on 19 February 2015. https://web.archive.org/web/20150219100248/http://eurheartjsupp.oxfordjournals.org/content/ehjsupp/4/suppl_G/G1.full.pdf. Retrieved 12 February 2015.
- Takizawa, Peter. "Muscle and Neuromuscular Junction". Yale University. http://medcell.med.yale.edu/lectures/files/muscle.pdf.
- Tamarkin, Dawn. "Myofibril Composition". STCC Foundation Press. Archived from the original on 8 September 2006. https://web.archive.org/web/20060908131930/http://faculty.stcc.edu/AandP/AP/AP1pages/Units5to9/Unit7/myofibri.htm. Retrieved 12 February 2015.
- "Structure and Function of Skeletal Muscles". Archived from the original on 15 February 2015. https://web.archive.org/web/20150215092857/http://courses.washington.edu/conj/motor/musclereview.htm. Retrieved 13 February 2015.
- page 395, Biology, Fifth Edition, Campbell, 1999
- "Molecular mechanisms regulating myogenic determination and differentiation". Front Biosci 5: D750–67. 2000. doi:10.2741/Perry. PMID 10966875. https://dx.doi.org/10.2741%2FPerry
- "Loss of both GATA4 and GATA6 blocks cardiac myocyte differentiation and results in acardia in mice". Dev. Biol. 317 (2): 614–9. May 2008. doi:10.1016/j.ydbio.2008.03.013. PMID 18400219. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2423416
- Zammit, PS; Partridge, TA; Yablonka-Reuveni, Z (November 2006). "The skeletal muscle satellite cell: the stem cell that came in from the cold.". Journal of Histochemistry and Cytochemistry 54 (11): 1177–91. doi:10.1369/jhc.6r6995.2006. PMID 16899758. https://dx.doi.org/10.1369%2Fjhc.6r6995.2006
- "Differentiation of pluripotent stem cells to muscle fiber to model Duchenne muscular dystrophy". Nature Biotechnology 33 (9): 962–9. August 2015. doi:10.1038/nbt.3297. PMID 26237517. http://www.hal.inserm.fr/inserm-01484878.
- "Kindlin-2 is required for myocyte elongation and is essential for myogenesis". BMC Cell Biol. 9: 36. 2008. doi:10.1186/1471-2121-9-36. PMID 18611274. PMC 2478659. Archived from the original on 2008-10-11. https://web.archive.org/web/20081011120308/http://www.biomedcentral.com/1471-2121/9/36.
- Ziser, Stephen. "Muscle Cell Anatomy & Function". Archived from the original on 23 September 2015. https://web.archive.org/web/20150923181106/http://www.austincc.edu/sziser/Biol%202404/2404LecNotes/2404LNExII/Muscle%20Physiology.pdf. Retrieved 12 February 2015.
- "Muscle Fiber Excitation". University of Washington. Archived from the original on 27 February 2015. https://web.archive.org/web/20150227010858/http://courses.washington.edu/conj/bess/muscle/musclexcitation.html. Retrieved 11 February 2015.
- MacIntosh, Brian R.; Gardiner, Phillip F.; McComas, Alan J. (2006). Skeletal Muscle: Form and Function. Human Kinetics. ISBN 978-0-7360-4517-9. https://books.google.com/books?id=-EfxEhSFaMYC&pg=PA151.
- Smerdu, V; Karsch-Mizrachi, I; Campione, M; Leinwand, L; Schiaffino, S (December 1994). "Type IIx myosin heavy chain transcripts are expressed in type IIb fibers of human skeletal muscle.". The American Journal of Physiology 267 (6 Pt 1): C1723–8. doi:10.1152/ajpcell.1994.267.6.C1723. PMID 7545970. https://dx.doi.org/10.1152%2Fajpcell.1994.267.6.C1723
- 3.0.CO;2-7. PMID 10998639. https://dx.doi.org/10.1002%2F1097-0029%2820000915%2950%3A6%3C500%3A%3AAID-JEMT7%3E3.0.CO%3B2-7" id="ref_18">Pette, D; Staron, RS (Sep 15, 2000). "Myosin isoforms, muscle fiber types, and transitions". Microscopy Research and Technique 50 (6): 500–9. doi:10.1002/1097-0029(20000915)50:6<500::AID-JEMT7>3.0.CO;2-7. PMID 10998639. https://dx.doi.org/10.1002%2F1097-0029%2820000915%2950%3A6%3C500%3A%3AAID-JEMT7%3E3.0.CO%3B2-7
- Staron, Robert S.; Johnson, Peter (November 1993). "Myosin polymorphism and differential expression in adult human skeletal muscle". Comparative Biochemistry and Physiology B 106 (3): 463–475. doi:10.1016/0305-0491(93)90120-T. https://dx.doi.org/10.1016%2F0305-0491%2893%2990120-T
- Buchthal, F; Schmalbruch, H (Aug 1970). "Contraction times and fibre types in intact human muscle.". Acta Physiologica Scandinavica 79 (4): 435–52. doi:10.1111/j.1748-1716.1970.tb04744.x. PMID 5472111. https://dx.doi.org/10.1111%2Fj.1748-1716.1970.tb04744.x
- Garnett, RA; O'Donovan, MJ; Stephens, JA; Taylor, A (Feb 1979). "Motor unit organization of human medial gastrocnemius.". The Journal of Physiology 287 (1): 33–43. doi:10.1113/jphysiol.1979.sp012643. PMID 430414. PMC 1281479. http://jp.physoc.org/content/287/1/33.
- "Archived copy". Archived from the original on 2007-11-21. https://web.archive.org/web/20071121100903/http://sportsmedicine.about.com/od/anatomyandphysiology/a/MuscleFiberType.htm. Retrieved 2007-11-27. Sports Medicine About.com
- Johnson, M. A.; Polgar, J; Weightman, D; Appleton, D (1973). "Data on the distribution of fibre types in thirty-six human muscles. An autopsy study". Journal of the Neurological Sciences 18 (1): 111–29. doi:10.1016/0022-510x(73)90023-3. PMID 4120482. https://dx.doi.org/10.1016%2F0022-510x%2873%2990023-3
- Michael Yessis (2006). Build A Better Athlete. Ultimate Athlete Concepts. ISBN 978-1-930546-78-3.