Stem cells serve as a source of cellular material in embryogenesis and postnatal growth and regeneration. This requires significant proliferative potential ensured by sufficient telomere length. Telomere attrition in the stem cells and their niche cells can result in the exhaustion of the regenerative potential of high-turnover organs, causing or contributing to the onset of age-related diseases. Telomerase activity is present in most types of adult stem cells, though at substantially lower levels. Such lower levels are sufficient for slowing down telomere shortening and expanding the replicative lifespan but cannot prevent replicative senescence. In this case, low telomerase expression may provide protection against the malignant transformation of stem cells.
- stem cells
- cell senescence
- replicative lifespan
- aging
- telomeres
- telomerase
1. Embryonic Stem Cells
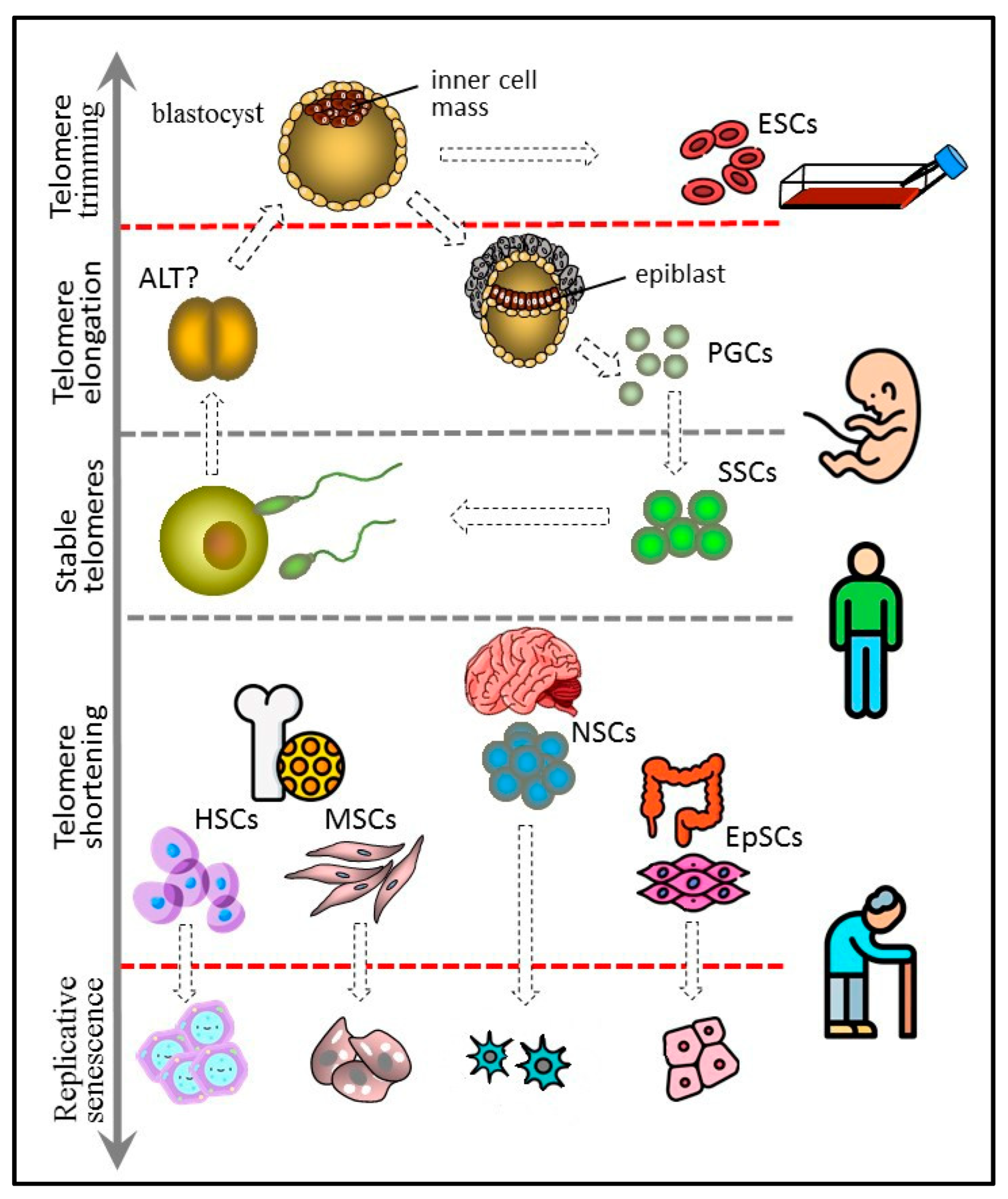
Figure 1. Telomere length dynamics in various types of stem cells. The figure 1 has been designed using icons made by Freepik and Smashicons from www.flaticon.com.
2. Germline Stem Cells
3. Hematopoietic Stem Cells
4. Mesenchymal Stem Cells
5. Neural Stem Cells
6. Tissue-Specific Epithelial Stem Cells
This entry is adapted from the peer-reviewed paper 10.3390/biomedicines10102335
References
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147.
- Nolte, J. Lrrc34 Interacts with Oct4 and Has an Impact on Telomere Length in Mouse Embryonic Stem Cells. Stem Cells Dev. 2021, 30, 1093–1102.
- Meerdo, L.N.; Reed, W.A.; White, K.L. Telomere-to-centromere ratio of bovine clones, embryos, gametes, fetal cells, and adult cells. Cloning Stem Cells 2005, 7, 62–73.
- Marión, R.M.; López de Silanes, I.; Mosteiro, L.; Gamache, B.; Abad, M.; Guerra, C.; Megías, D.; Serrano, M.; Blasco, M.A. Common telomere changes during in vivo reprogramming and early stages of tumorigenesis. Stem Cell Rep. 2017, 8, 460–475.
- Agarwal, S.; Loh, Y.H.; McLoughlin, E.M.; Huang, J.; Park, I.H.; Miller, J.D.; Huo, H.; Okuka, M.; dos Reis, R.M.; Loewer, S.; et al. Telomere elongation in induced pluripotent stem cells from dyskeratosis congenita patients. Nature 2010, 464, 292–296.
- Kamada, M.; Mitsui, Y.; Matsuo, T.; Takahashi, T. Reversible transformation and de-differentiation of human cells derived from induced pluripotent stem cell teratomas. Hum. Cell 2016, 29, 1–9.
- Liu, L.; Bailey, S.M.; Okuka, M.; Munoz, P.; Li, C.; Zhou, L.; Wu, C.; Czerwiec, E.; Sandler, L.; Seyfang, A.; et al. Telomere lengthening early in development. Nat. Cell Biol. 2007, 9, 1436–1441.
- Zeng, S.; Liu, L.; Sun, Y.; Xie, P.; Hu, L.; Yuan, D.; Chen, D.; Ouyang, Q.; Lin, G.; Lu, G. Telomerase-mediated telomere elongation from human blastocysts to embryonic stem cells. J. Cell Sci. 2014, 127, 752–762.
- Zhao, S.; Wang, F.; Liu, L. Alternative lengthening of telomeres (ALT) in tumors and pluripotent stem cells. Genes 2019, 10, 1030.
- Zalzman, M.; Falco, G.; Sharova, L.V.; Nishiyama, A.; Thomas, M.; Lee, S.L.; Stagg, C.A.; Hoang, H.G.; Yang, H.T.; Indig, F.E.; et al. Zscan4 regulates telomere elongation and genomic stability in ES cells. Nature 2010, 464, 858–863.
- Dan, J.; Zhou, Z.; Wang, F.; Wang, H.; Guo, R.; Keefe, D.L.; Liu, L. Zscan4 Contributes to Telomere Maintenance in Telomerase-Deficient Late Generation Mouse ESCs and Human ALT Cancer Cells. Cells 2022, 11, 456.
- Dan, J.; Liu, Y.; Liu, N.; Chiourea, M.; Okuka, M.; Wu, T.; Ye, X.; Mou, C.; Wang, L.; Wang, L.; et al. Rif1 maintains telomere length homeostasis of ESCs by mediating heterochromatin silencing. Dev. Cell 2014, 29, 7–19.
- Le, R.; Huang, Y.; Zhang, Y.; Wang, H.; Lin, J.; Dong, Y.; Li, Z.; Guo, M.; Kou, X.; Zhao, Y.; et al. Dcaf11 activates Zscan4-mediated alternative telomere lengthening in early embryos and embryonic stem cells. Cell Stem Cell 2021, 28, 732–747.e9.
- Markiewicz-Potoczny, M.; Lobanova, A.; Loeb, A.M.; Kirak, O.; Olbrich, T.; Ruiz, S.; Lazzerini Denchi, E. TRF2-mediated telomere protection is dispensable in pluripotent stem cells. Nature 2021, 589, 110–115.
- Rivera, T.; Haggblom, C.; Cosconati, S.; Karlseder, J. A balance between elongation and trimming regulates telomere stability in stem cells. Nat. Struct. Mol. Biol. 2017, 24, 30–39.
- Varela, E.; Munoz-Lorente, M.A.; Tejera, A.M.; Ortega, S.; Blasco, M.A. Generation of mice with longer and better preserved telomeres in the absence of genetic manipulations. Nat. Commun. 2016, 7, 11739.
- Li, J.S.; Miralles Fuste, J.; Simavorian, T.; Bartocci, C.; Tsai, J.; Karlseder, J.; Lazzerini Denchi, E. TZAP: A telomere-associated protein involved in telomere length control. Science 2017, 355, 638–641.
- Zhao, Y.; Zhang, G.; He, C.; Mei, Y.; Shi, Y.; Li, F. The 11th C2H2 zinc finger and an adjacent C-terminal arm are responsible for TZAP recognition of telomeric DNA. Cell Res. 2018, 28, 130–134.
- Nikolic, A.; Volarevic, V.; Armstrong, L.; Lako, M.; Stojkovic, M. Primordial Germ Cells: Current Knowledge and Perspectives. Stem Cells Int. 2016, 2016, 1741072.
- Guo, J.; Grow, E.J.; Yi, C.; Mlcochova, H.; Maher, G.J.; Lindskog, C.; Murphy, P.J.; Wike, C.L.; Carrel, D.T.; Goriely, A.; et al. Chromatin and single-cell RNA-seq profiling reveal dynamic signaling and metabolic transitions during human spermatogonial stem cell development. Cell Stem Cell 2017, 21, 533–546.e6.
- Kerr, C.L.; Shamblott, M.J.; Gearhart, J.D. Pluripotent stem cells from germ cells. Methods Enzymol. 2006, 419, 400–426.
- Tang, W.W.C.; Kobayash, T.; Irie, N.; Dietmann, S.; Surani, M.A. Specification and epigenetic programming of the human germ line. Nat. Rev. Genet. 2016, 17, 585–600.
- Sharma, S.; Wistuba, J.; Pock, T.; Schlatt, S.; Neuhaus, N. Spermatogonial stem cells: Updates from specification to clinical relevance. Hum. Reprod. Updat. 2019, 25, 275–297.
- Pech, M.F.; Garbuzov, A.; Hasegawa, K.; Sukhwani, M.; Zhang, R.J.; Benayoun, B.A.; Brockman, S.A.; Lin, S.; Brunet, A.; Orwig, K.E.; et al. High telomerase is a hallmark of undifferentiated spermatogonia and is required for maintenance of male germline stem cells. Genes Dev. 2015, 29, 2420–2434.
- Kanatsu-Shinohara, M.; Ogonuki, N.; Iwano, T.; Lee, J.; Kazuki, Y.; Inoue, K.; Miki, H.; Takehashi, M.; Toyokuni, S.; Shinkai, Y.; et al. Genetic and epigenetic properties of mouse male germline stem cells during long-term culture. Development 2005, 132, 4155–4163.
- Ferlin, A.; Rampazzo, E.; Rocca, M.S.; Keppel, S.; Frigo, A.C.; De Rossi, A.; Foresta, C. In young men sperm telomere length is related to sperm number and parental age. Hum. Reprod. 2017, 28, 3370–3376.
- Dunlop, C.E.; Telfer, E.E.; Anderson, R.A. Ovarian germline stem cells. Stem Cell Res. Ther. 2014, 5, 98.
- Zhang, H.; Panula, S.; Petropoulos, S.; Edsgärd, D.; Busayavalasa, K.; Liu, L.; Li, X.; Risal, S.; Shen, Y.; Shao, J.; et al. Adult human and mouse ovaries lack DDX4-expressing functional oogonial stem cells. Nat. Med. 2015, 21, 1116–1118.
- Turner, S.; Hartshorne, G.M. Telomere lengths in human pronuclei, oocytes and spermatozoa. Mol. Hum. Reprod. 2013, 19, 510–518.
- Wright, W.E.; Piatyszek, M.A.; Rainey, W.E.; Byrd, W.; Shay, J.W. Telomerase activity in human germline and embryonic tissues and cells. Dev. Genet. 1996, 18, 173–179.
- Brummendorf, T.H.; Balabanov, S. Telomere length dynamics in normal hematopoiesis and in disease states characterized by increased stem cell turnover. Leukemia 2006, 29, 273–275.
- Brazvan, B.; Ebrahimi-Kalan, A.; Velaei, K.; Mehdipour, A.; Aliyari Serej, Z.; Ebrahimi, A.; Ghorbani, M.; Cheraghi, O.; Nozad Charoudeh, H. Telomerase activity and telomere on stem progeny senescence. Biomed. Pharmacother. 2018, 102, 9–17.
- Patrick, M.; Weng, N.P. Expression and regulation of telomerase in human T cell differentiation, activation, aging and diseases. Cell Immunol. 2019, 345, 103989.
- Yang, D.; de Haan, G. Inflammation and aging of hematopoietic stem cells in their niche. Cells 2021, 10, 1849.
- De Haan, G.; Lazare, S.S. Aging of hematopoietic stem cells. Blood 2018, 131, 479–487.
- Brzeźniakiewicz-Janus, K.; Rupa-Matysek, J.; Gil, L. Acquired aplastic anemia as a clonal disorder of hematopoietic stem cells. Stem Cell Rev. Rep. 2020, 16, 472–481.
- Fiorini, E.; Santoni, A.; Colla, S. Dysfunctional telomeres and hematological disorders. Differentiation 2018, 100, 1–11.
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999, 284, 143–147.
- Yarygin, K.N.; Lupatov, A.Y.; Sukhikh, G.T. Modulation of immune responses by mesenchymal stromal cells. Bull. Exp. Biol. Med. 2016, 161, 561–565.
- Choumerianou, D.M.; Martimianaki, G.; Stiakaki, E.; Kalmanti, L.; Kalmanti, M.; Dimitriou, H. Comparative study of stemness characteristics of mesenchymal cells from bone marrow of children and adults. Cytotherapy 2010, 12, 881–887.
- Yang, Y.K.; Ogando, C.R.; Wang See, C.; Chang, T.Y.; Barabino, G.A. Changes in phenotype and differentiation potential of human mesenchymal stem cells aging in vitro. Stem Cell Res. Ther. 2018, 9, 131.
- Guerrero, E.N.; Vega, S.; Fu, C.; De León, R.; Beltran, D.; Solis, M.A. Increased proliferation and differentiation capacity of placenta-derived mesenchymal stem cells from women of median maternal age correlates with telomere shortening. Aging 2021, 13, 24542–24559.
- Hiyama, E.; Hiyama, K. Telomere and telomerase in stem cells. Br. J. Cancer 2007, 96, 1020–1024.
- Ryu, E.; Hong, S.; Kang, J.; Woo, J.; Park, J.; Lee, J.; Seo, J.S. Identification of senescence-associated genes in human bone marrow mesenchymal stem cells. Biochem Biophys Res Commun. 2008, 371, 431–436.
- Fehrer, C.; Lepperdinger, G. Mesenchymal stem cell aging. Exp. Gerontol. 2005, 40, 926–930.
- Wang, Q.; Li, J.; Wang, S.; Deng, Q.; Wang, K.; Dai, X.; An, Y.; Dong, G.; Ke, W.; Chen, F.; et al. Single-cell transcriptome profiling reveals molecular heterogeneity in human umbilical cord tissue and culture-expanded mesenchymal stem cells. FEBS J. 2021, 288, 5311–5330.
- Hwang, E.S.; Ok, J.S.; Song, S. Chemical and physical approaches to extend the replicative and differentiation potential of stem cells. Stem Cell Rev. Rep. 2016, 12, 315–326.
- Wolbank, S.; Stadler, G.; Peterbauer, A.; Gillich, A.; Karbiener, M.; Streubel, B.; Wieser, M.; Katinger, H.; van Griensven, M.; Redl, H.; et al. Telomerase immortalized human amnion- and adipose-derived mesenchymal stem cells: Maintenance of differentiation and immunomodulatory characteristics. Tissue Eng. Part A 2009, 15, 1843–1854.
- Trachana, V.; Petrakis, S.; Fotiadis, Z.; Siska, E.K.; Balis, V.; Gonos, E.S.; Kaloyianni, M.; Koliakos, G. Human mesenchymal stem cells with enhanced telomerase activity acquire resistance against oxidative stress-induced genomic damage. Cytotherapy 2017, 19, 808–820.
- Poltavtseva, R.A.; Silachev, D.N.; Pavlovich, S.V.; Kesova, M.I.; Yarygin, K.N.; Lupatov, A.Y.; Van’ko, L.V.; Shuvalova, M.P.; Sukhikh, G.T. Neuroprotective effect of mesenchymal and neural stem and progenitor cells on sensorimotor recovery after brain injury. Bull Exp. Biol. Med. 2012, 153, 586–590.
- Ferrón, S.R.; Marqués-Torrejón, M.A.; Mira, H.; Flores, I.; Taylor, K.; Blasco, M.A.; Fariñas, I. Telomere shortening in neural stem cells disrupts neuronal differentiation and neuritogenesis. J. Neurosci. 2009, 29, 14394–14407.
- Grammatikakis, I.; Zhang, P.; Mattson, M.P.; Gorospe, M. The long and the short of TRF2 in neurogenesis. Cell Cycle 2016, 15, 3026–3032.
- Lobanova, A.; She, R.; Pieraut, S.; Clapp, C.; Maximov, A.; Denchi, E.L. Different requirements of functional telomeres in neural stem cells and terminally differentiated neurons. Genes Dev. 2017, 31, 639–647.
- Martín-Rivera, L.; Herrera, E.; Albar, J.P.; Blasco, M.A. Expression of mouse telomerase catalytic subunit in embryos and adult tissues. Proc. Natl. Acad. Sci. USA 1998, 95, 10471–10476.
- Schwob, A.E.; Nguyen, L.J.; Meiri, K.F. Immortalization of neural precursors when telomerase is overexpressed in embryonal carcinomas and stem cells. Mol. Biol. Cell 2008, 19, 1548–1560.
- Zhou, Q.G.; Liu, M.Y.; Lee, H.W.; Ishikawa, F.; Devkota, S.; Shen, X.R.; Jin, X.; Wu, H.Y.; Liu, Z.; Liu, X.; et al. Hippocampal TERT regulates spatial memory formation through modulation of neural development. Stem Cell Rep. 2017, 9, 543–556.
- Richardson, R.M.; Nguyen, B.; Holt, S.E.; Broaddus, W.C.; Fillmore, H.L. Ectopic telomerase expression inhibits neuronal differentiation of NT2 neural progenitor cells. Neurosci. Lett. 2007, 421, 168–172.
- Spilsbury, A.; Miwa, S.; Attems, J.; Saretzki, G. The role of telomerase protein TERT in Alzheimer’s disease and in tau-related pathology in vitro. J. Neurosci. 2015, 35, 1659–1674.
- Liu, M.Y.; Nemes, A.; Zhou, Q.G. The emerging roles for telomerase in the central nervous system. Front. Mol. Neurosci. 2018, 11, 160.
- Caporaso, G.L.; Lim, D.A.; Alvarez-Buylla, A.; Chao, M.V. Telomerase activity in the subventricular zone of adult mice. Mol. Cell Neurosci. 2003, 23, 693–702.
- Ming, G.L.; Song, H. Adult neurogenesis in the mammalian brain: Significant answers and significant questions. Neuron 2011, 70, 687–702.
- Cameron, H.A.; McKay, R.D. Adult neurogenesis produces a large pool of new granule cells in the dentate gyrus. J. Comp. Neurol. 2001, 435, 406–417.
- Augusto-Oliveira, M.; Arrifano, G.P.F.; Malva, J.O.; Crespo-Lopez, M.E. Adult hippocampal neurogenesis in different taxonomic groups: Possible functional similarities and striking controversies. Cells 2019, 8, 125.
- Sorrells, S.F.; Paredes, M.F.; Cebrian-Silla, A.; Sandoval, K.; Qi, D.; Kelley, K.W.; James, D.; Mayer, S.; Chang, J.; Auguste, K.I.; et al. Human hippocampal neurogenesis drops sharply in children to undetectable levels in adults. Nature 2018, 555, 377–381.
- Takubo, K.; Izumiyama-Shimomura, N.; Honma, N.; Sawabe, M.; Arai, T.; Kato, M.; Oshimura, M.; Nakamura, K. Telomere lengths are characteristic in each human individual. Exp. Gerontol. 2002, 37, 523–531.
- Anitha, A.; Thanseem, I.; Vasu, M.M.; Viswambharan, V.; Poovathinal, S.A. Telomeres in neurological disorders. Adv. Clin. Chem. 2019, 90, 81–132.
- Flores, I.; Canela, A.; Vera, E.; Tejera, A.; Cotsarelis, G.; Blasco, M.A. The longest telomeres: A general signature of adult stem cell compartments. Genes Dev. 2008, 22, 654–667.
- Buckingham, E.M.; Klingelhutz, A.J. The role of telomeres in the ageing of human skin. Exp. Dermatol. 2011, 20, 297–302.
- Orioli, D. Dellambra, E. Epigenetic regulation of skin cells in natural aging and premature aging diseases. Cells 2018, 7, 268.
- Enzo, E.; Secone Seconetti, A.; Forcato, M.; Tenedini, E.; Polito, M.P.; Sala, I.; Carulli, S.; Contin, R.; Peano, C.; Tagliafico, E.; et al. Single-keratinocyte transcriptomic analyses identify different clonal types and proliferative potential mediated by FOXM1 in human epidermal stem cells. Nat. Commun. 2021, 12, 2505.
- Sarin, K.Y.; Cheung, P.; Gilison, D.; Lee, E.; Tennen, R.I.; Wang, E.; Artandi, M.K.; Oro, A.E.; Artandi, S.E. Conditional telomerase induction causes proliferation of hair follicle stem cells. Nature 2005, 436, 1048–1052.
- Vulliamy, T.; Marrone, A.; Szydlo, R.; Walne, A.; Mason, P.J.; Dokal, I. Disease anticipation is associated with progressive telomere shortening in families with dyskeratosis congenita due to mutations in TERC. Nat. Genet. 2004, 36, 447–449.
- Gourronc, F.A.; Robertson, M.M.; Herrig, A.K.; Lansdorp, P.M.; Goldman, F.D.; Klingelhutz, A.J. Proliferative defects in dyskeratosis congenita skin keratinocytes are corrected by expression of the telomerase reverse transcriptase, TERT, or by activation of endogenous telomerase through expression of papillomavirus E6/E7 or the telomerase RNA component, TERC. Exp. Dermatol. 2010, 19, 279–288.
- Ibrahim, B.; Sheerin, A.N.; Jennert-Burston, K.; Bird, J.L.; Massala, M.V.; Illsley, M.; James, S.E.; Faragher, R.G. Absence of premature senescence in Werner’s syndrome keratinocytes. Exp. Gerontol. 2016, 83, 139–147.
- Buckingham, E.M.; Goldman, F.D.; Klingelhutz, A.J. Dyskeratosis congenita dermal fibroblasts are defective in supporting the clonogenic growth of epidermal keratinocytes. Aging Dis. 2012, 3, 427–437.
- Liu, N.; Yin, Y.; Wang, H.; Zhou, Z.; Sheng, X.; Fu, H.; Guo, R.; Wang, H.; Yang, J.; Gong, P.; et al. Telomere dysfunction impairs epidermal stem cell specification and differentiation by disrupting BMP/pSmad/P63 signaling. PLoS Genet. 2019, 15, e1008368.
- Montgomery, R.K.; Carlone, D.L.; Richmond, C.A.; Farilla, L.; Kranendonk, M.E.; Henderson, D.E.; Baffour-Awuah, N.Y.; Ambruzs, D.M.; Fogli, L.K.; Algra, S.; et al. Mouse telomerase reverse transcriptase (mTert) expression marks slowly cycling intestinal stem cells. Proc. Natl. Acad. Sci. USA 2011, 108, 179–184.
- Carlone, D.L.; Breault, D.T. Slowly cycling versus rapidly cycling intestinal stem cells: Distinct roles or redundancy. Cell Cycle 2011, 10, 723–724.
- Allaire, J.M.; Crowley, S.M.; Law, H.T.; Chang, S.Y.; Ko, H.J.; Vallance, B.A. The intestinal epithelium: Central coordinator of mucosal immunity. Trends Immunol. 2018, 39, 677–696.
- Clevers, H. The intestinal crypt, a prototype stem cell compartment. Cell 2013, 154, 274–284.
- Hiyama, E.; Tatsumoto, N.; Kodama, T.; Hiyama, K.; Shay, J.; Yokoyama, T. Telomerase activity in human intestine. Int. J. Oncol. 1996, 9, 453–458.
- Schepers, A.G.; Vries, R.; van den Born, M.; van de Wetering, M.; Clevers, H. Lgr5 intestinal stem cells have high telomerase activity and randomly segregate their chromosomes. EMBO J. 2011, 30, 1104–1109.
- Pasztoi, M.; Ohnmacht, C. Tissue niches formed by intestinal mesenchymal stromal cells in mucosal homeostasis and immunity. Int. J. Mol. Sci. 2022, 23, 5181.
- Nath, A.; Chakrabarti, P.; Sen, S.; Barui, A. Reactive oxygen species in modulating intestinal stem cell dynamics and function. Stem Cell Rev. Rep. 2022.
- Ruiz, A.; Flores-Gonzalez, J.; Buendia-Roldan, I.; Chavez-Galan, L. Telomere shortening and its association with cell dysfunction in lung diseases. Int. J. Mol. Sci. 2021, 23, 425.
- Kathiriya, J.J.; Brumwell, A.N.; Jackson, J.R.; Tang, X.; Chapman, H.A. Distinct airway epithelial stem cells hide among club cells but mobilize to promote alveolar regeneration. Cell Stem Cell 2020, 26, 346–358.e4.
- Rao, W.; Wang, S.; Duleba, M.; Niroula, S.; Goller, K.; Xie, J.; Mahalingam, R.; Neupane, R.; Liew, A.A.; Vincent, M.; et al. Regenerative metaplastic clones in COPD lung drive inflammation and fibrosis. Cell 2020, 181, 848–864.e18.
- Hong, X.; Wang, L.; Zhang, K.; Liu, J.; Liu, J.P. Molecular mechanisms of alveolar epithelial stem cell senescence and senescence-associated differentiation disorders in pulmonary fibrosis. Cells 2022, 11, 877.
- Wang, L.; Chen, R.; Li, G.; Wang, Z.; Liu, J.; Liang, Y.; Liu, J.P. FBW7 mediates senescence and pulmonary fibrosis through telomere uncapping. Cell Metab. 2020, 32, 860–877.e9.
- Lee, S.; Yu, Y.; Trimpert, J.; Benthani, F.; Mairhofer, M.; Richter-Pechanska, P.; Wyler, E.; Belenki, D.; Kaltenbrunner, S.; Pammer, M.; et al. Virus-induced senescence is driver and therapeutic target in COVID-19. Nature 2021, 599, 283–289.
- Saito, S.; Ku, C.C.; Wuputra, K.; Pan, J.B.; Lin, C.S.; Lin, Y.C.; Wu, D.C.; Yokoyama, K.K. Biomarkers of Cancer Stem Cells for Experimental Research and Clinical Application. J. Pers. Med. 2022, 12, 715.
