From 1964 to the early 1970s, Orson Anderson led a research program at the Lamont Geological Observatory in the newly-emerging field of “mineral physics”. In collaboration with colleagues Edward Schreiber and Naohiro Soga, Orson exploited the techniques of physical acoustics to study the behavior of the sound velocities of minerals at elevated pressures and temperatures. This research program also included semi-empirical studies of the relationships between the bulk modulus and the molar volume of solids, the use of lattice dynamics to calculate the elastic moduli of cubic structures as a function of pressure to predict instabilities, and theoretical investigations of the Lagrangian and Eulerian formulations of finite strain equations of state.
2. Background on Mineral Physics
According to Robert Hazen
[1], “mineral physics is the study of mineralogical problems through the application of condensed matter physics”. In reality, mineral physicists use not only physics but also solid-state chemistry. In addition, they study not only minerals but all materials related to natural minerals (e.g., structural analogs, but also glasses, melts, and fluids). The father of mineral physics is widely accepted to be Francis Birch, whose seminal paper in 1952 summarized the state of knowledge of the elasticity and constitution of the Earth’s interior at that time
[2]; however, Birch did not use the name “mineral physics”. Orson often cited the German physicist Eduard Grüneisen as the person who first introduced the term mineral physics
[3]. Over the subsequent two decades, many laboratories began to conduct experiments on the physical properties of minerals at high pressures and temperatures. Among these were Orson Anderson at Lamont (and later at the University of California Los Angeles/UCLA), John Jamieson at the University of Chicago, Alvin Van Valkenburg at the National Bureau of Standards, William Bassett and Taro Takahashi at the University of Rochester, Ho-kwang Mao and Peter Bell at the Carnegie Institution of Washington, Gene Simmons at the Massachusetts Institute of Technology, Mineo Kumazawa at Nagoya University, Thomas Ahrens at the California Institute of Technology, Gerhard Barsch (and later Earl Graham) at the Pennsylvania State University, Hartmut Spetzler at the University of Colorado, Francis Birch (and later Richard O’Connell) at Harvard University, Murli Manghnani at the University of Hawaii, Syun-iti Akimoto at the University of Tokyo, Jean-Paul Poirier at the Institut de Physique du Globe in Paris, and Robert Liebermann [the author] at the Australian National University (and later at Stony Brook University).
3. The Mineral Physics Laboratory at Lamont
The Mineral Physics Laboratory at the Lamont Geological Observatory was established in the early 1960s by Orson Anderson. From 1965 to 1970, I was a graduate student, later postdoc, in that laboratory (Figure 1). The story of how the lab was founded is best told in Orson’s own words.
Figure 1. In 1976, Orson gave one of the invited talks at a workshop on seismic anisotropy in the Castle of Liblice in Czechoslovakia, convened by Vladislav Babuska, Jaroslava Plomerova, and me.
In the following paragraphs, I shall quote from a correspondence written in May 2008
[4] by Anderson to Michael Purdy (Director of Lamont) and to the author in 2008
[5] and 2014
[6], with the permission of Orson (whose words will be highlighted in italics throughout this paper).
I [Anderson] was a physicist at Bell Labs for 10 years. I received my PhD in physics at the University of Utah under the supervision of Henry Eyring and Walter Elsasser in the field of physical acoustics in 1951 and was snapped up by Bell Telephone Laboratories, who needed physicists trained in acoustics but found them sparse in the graduate lists of the country. Transistor physics was blooming at the time, but the telephone industries still ran on electrical mechanical devices, and physical acoustics is the mainstay of the research needed for such devices. I prospered in my science career at Bell Labs under the tutelage of Warren Mason and Hal McSkimin.
It was a custom at Bell Labs that an important visitor be accompanied for his entire visit by a host who is a young scientist. A Bell Vice President, William Baker, asked me to be the host of Lamont Director Maurice Ewing during his one-day visit in 1962. At the luncheon, I was seated between Baker and Ewing. Ewing said that the most significant lab to him that he visited was the lab of his host. Ewing said that one of Lamont’s perplexing problems was to determine the thickness of sediments over the basement rocks. The analysis of the sonar signal determined the lapsed time, not the thickness. The primary difficulty is that the velocity of sound through the sediment layer is often not known. Ewing thought that they needed the kind of lab that Anderson had and the technical support that went with it. So, he boldly asked my VP if Lamont could borrow me to establish a physical acoustics lab at Lamont. Vice President Baker said yes you could have him two or three days a week. So, it was settled that I was to build a lab at Lamont equipped to handle the sediment problem and to find and hire the scientists necessary to run the lab. I felt a little bit like I was in the slave market, so I spoke up and said I will agree to do that if Lamont will take me on some of their ocean research expeditions.
I hired two postdocs on behalf of Lamont to run the new lab: Edward Schreiber from Alfred University in 1963 and Naohiro Soga from the Research Division of American Standard in 1964 (where he had been working for two years after finishing graduate school at Kyoto University). Both had PhDs in ceramics and industrial chemistry, and I trained them in physical acoustics at my lab at Bell Labs. Schreiber took charge of the high-pressure experiments and Soga the high-temperature experiments. By 1965, Soga and Schreiber, as postdocs, were producing good relevant papers at Lamont, and I had been seduced away from Bell Labs and was now an adjunct professor of geology at Columbia and a full-time employee at Lamont. My research grants paid my salary.
In 1967, Professor Charles Drake of Columbia, the Chairman of the Department of Geology, had me appointed a professor of Geology at Columbia University [an appointment which Orson’s graduate students had been urging for sometime]. I was asked to teach mineralogy. Much later on, Drake told me that Professor Rhodes Fairbridge of Columbia said that, in view of my record in teaching minerals, the biggest mistake that the Department ever made was to appoint me a professor in it. I hasten to add that Drake did not agree with Fairbridges’ opinion. In addition, Orson cited Max Born, Lawrence Bragg, Peter Debye, Peter Paul Ewald, Andre Guinier, and Max von Laue as physicists who had made major contributions to mineralogy.
To learn geophysics, Orson became editor of the Journal of Geophysical Research. It was during that period in which all of the basic plate tectonic papers were published under his editorship.
Even though laboratories in this field of experimental mineralogy arose simultaneously in the two decades after Birch’s 1952 paper, the field did not have a name. The name “mineral physics” first appeared at Lamont. Director Ewing [known affectionately as “Doc” Ewing by all staff] observed to me that my lab was the only one at Lamont without a name; so, I pondered what the name should be and thought it should have physics in it because we were using the tools of physical acoustics, and it ought to be related to geological sciences, so the word mineral seemed an appropriate adjective. Thus, a sign labeled “Mineral Physics” appeared on the door of our lab in an old shed, which was located just north of the new seismology building. As far as I can determine, the words “mineral physics” were not used before the sign was printed and posted. A photograph of the sign, which was made by Ed Schreiber, is shown in Figure 2 [7].
Figure 2. “Mineral Physics” sign from Lamont in 1965. The name proposed by Orson Anderson. The sign and photo were made by Edward Schreiber
[7].
This is why Orson maintains that mineral physics was born at Lamont in the year 1964
[5]. The name “mineral physics” was subsequently adopted by the All-Union Committee of the American Geophysical Union (AGU), for which Orson served at the founding chair. The name has become part of the vocabulary of researchers around the world; by 2007, there were more than 30 mineral physics laboratories in academic and governmental institutions in the U.S.
The research program in Orson’s laboratory at Lamont included experimental, semi-empirical, and theoretical studies. The principal experimental technique was the determination of sound velocities using ultrasonic interferometry (including both pulse–echo overlap and pulse superposition versions) to a pressure of 2 kbar (0.2 GPa) and a temperature of 200 °C. Minerals investigated included polycrystalline aggregates or single crystals of MgO-periclase
[8], Al
2O
3-corundum
[9], Mg
2SiO
4-forsterite
[10], MgAl
2O
4-spinel
[11], zincite-ZnO
[12], garnet
[13], alpha-SiO
2 [14], CaO
[15], Fe
2O
3-hematite
[16], NiFe
2O
4-ferrite
[17], and (Mg,Fe)
2SiO
4-olivine
[18]. Advances in ceramic technology made available high-quality polycrystalline specimens suitable for high-frequency ultrasonic experiments [i.e., low porosity, small grain size, free of cracks, and densities close to theoretical values derived from X-ray diffraction]. Most of these studies were led by Ed Schreiber and Naohiro Soga, along with visiting scientists (Mineo Kumazawa and Murli Manghnani) and graduate student Robert Liebermann [the author]. For a summary of these ultrasonic studies, see
Figure 3 and the review paper in Reviews of Geophysics
[19]; see also
[20].
In Anderson’s 1995 book “Equations of State of Solids for Geophysics and Ceramic Science” [21], he states: Ed and I showed the community that precision ultrasonic measurements of sound velocity done under pressure [modest indeed by today’s standards] could yield valid equations of state in the shock wave pressure regime. Naohiro and I demonstrated that high-temperature elasticity obeyed certain fundamental laws in solid-state physics first presented by Max Born and Eduard Grüneisen in the 1920s and 1930s. Their paper
[22] was selected as the outstanding contribution to ceramic literature in 1966 and brought them the Ross Coffin Purdy Award of the American Ceramic Society in 1968.
Figure 3. Summary of elastic properties of minerals from Anderson, Schreiber, Liebermann, and Soga
[19].
Another experimental technique adopted and improved by Naohiro Soga and associates involved vibrating specimens of isotropic symmetry to their natural resonances and measuring the frequencies of their normal modes
[23][24]. Initially, these studies used spherical specimens of tektites and lunar beads, but, later, these were extended to rectangular specimens (and the technique was termed rectangular parallelepiped resonance, or RPR) and were particularly useful in measuring the resonances at high temperatures (extension to high pressures was not feasible, as the spherical normal modes were damped by the applied pressure).
When Orson decided to extend this technique to anisotropic specimens, he engaged Columbia undergraduate Harold Demarest to pursue this objective. In the following paragraph, Orson describes the success of this initiative.
A Lamont graduate student, Harry Demarest, decided to see if he could measure the velocity of sound in a small cube of a mineral with cubic symmetry, but he had to do research to find the required equations that yield the velocity of sound. His experimental results showed a series of resonant bands (a spectrum), and Demarest developed the theory for analyzing this spectrum to yield the elastic constants and velocities of sound. His sample was a single crystal, with cubic symmetry, of MgO. This was very exciting because we had opened the door to measuring the elastic constant of a small body having any crystallography symmetry. Furthermore, we did not need glue to bond the crystal to the transducer but used gravity to hold the crystal between the transducers touching opposite corners of the cube. Using glue for the transducers limits temperature measurements to about 700 °C. Without glue, we could hope for measurements for temperatures up to a large fraction of the melting point (in the range 2000–2500 °C). Demarest graduated from Lamont with his PhD, and we replaced him with Dr. Ichiro Ohno as a Lamont postdoc. Ohno was an accomplished theorist from Nagoya who undertook the theoretical problem of analyzing the resonance spectrum of a crystal with any symmetry. At our lab at Lamont, we had invented a new field in physical acoustics, it is now called RUS (Resonant Ultrasound Spectroscopy). For pioneering the field of RUS, the Acoustical Society of America gave me a citation. This citation brought welcome new funding to Lamont and the Mineral Physics Lab from the Office of Naval Research. More importantly, our lab had pierced the barrier to find physical properties at Earth’s mantle and core conditions because of the high temperature capability of RUS. This new work of mineral physics gave the high-temperature answers to help theorists develop the new theory of superplumes [25].
There were two, somewhat amusing episodes during these sound velocity studies:
In 1969, Mineo Kumazawa had arrived at Lamont to study the elasticity of single-crystal diamondz (following earlier work at Bell Labs by colleagues of Anderson). Instead, he was encouraged to study single-crystal olivines. Orson purchased a large single crystal of Burmese peridot from a gem dealer in New York City for a cost of more than $5000; he was so proud of that crystal that he published a photo and news item in the local newspaper in Palisades, NY. After Kumazawa cut the precious crystal into rectangular parallelepipeds for the acoustic studies, someone broke into the mineral physics lab at night and stole the largest pieces (which now had no commercial value). Kumazawa was devastated and embarrassed, and had no recourse but to try to perform his sound velocity measurements on the small pieces remaining, often termed “curf loss”. He succeeded, and he and Orson published a seminal paper in the
Journal of Geophysical Research [18].
The other “amusing” episode occurred in 1970 and involved laboratory measurements of the sound velocities in lunar rocks returned from the Moon by the Apollo missions. In Orson’s own words:
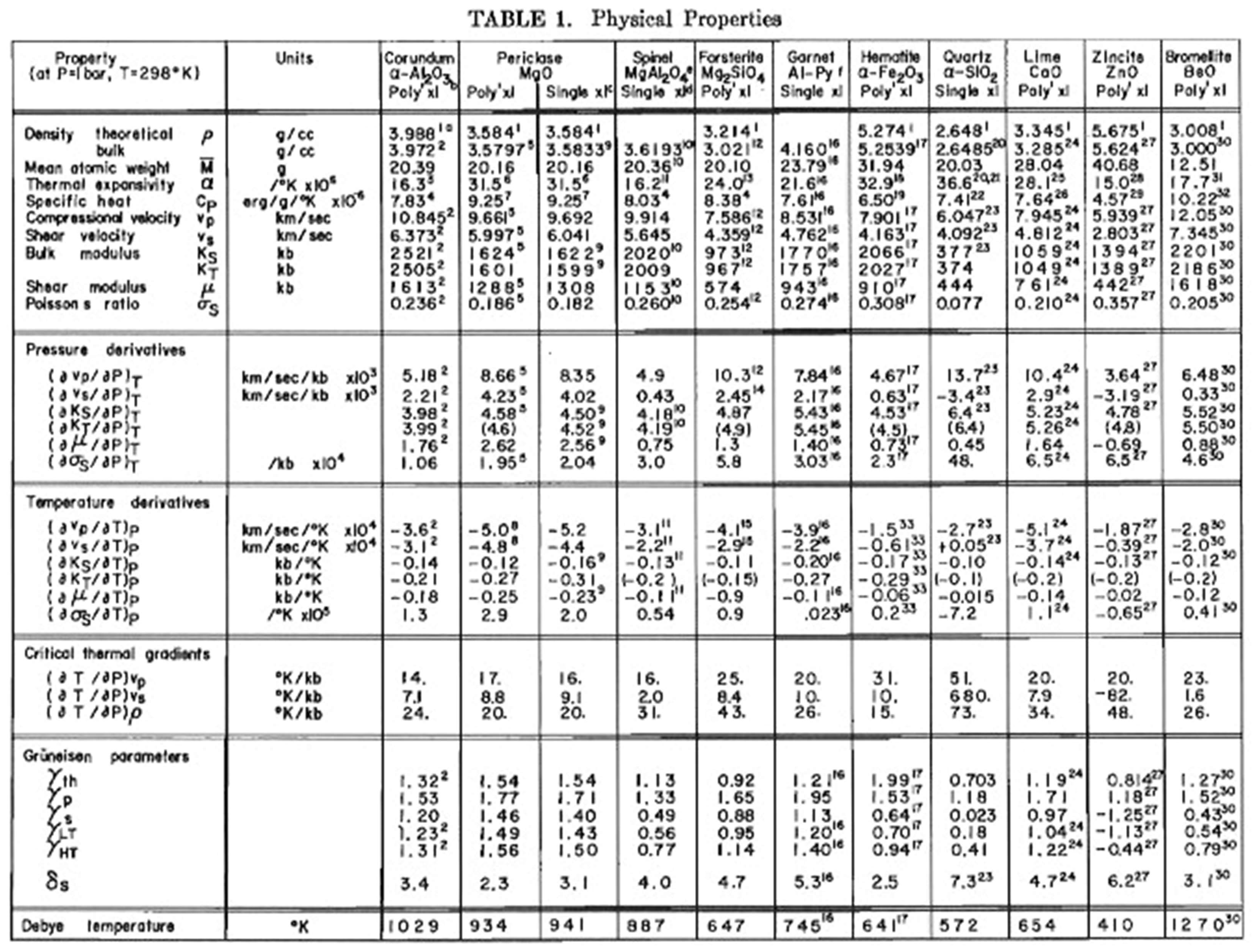
This was the time of the Apollo landings, and we received five rocks from the Moon to measure their physical properties. We were amazed to discover that the surface of the Moon had a velocity of sound of about 1.8–2.1 km per second, approximately that of seawater. Schreiber and I published a paper in Science [26] showing that the average sound velocity of the Moon rocks was close to provolone cheese. This paper brought many request for reprints. Of course, our task was to find why the velocity was so low in the lunar crust. We searched and found small glass spheroids (1 mm or less) in the lunar soils. They were formed by the liquid splash from meteorites hitting the lunar surface. We found the sound velocity of the lunar glass spheroids to be about 6.5 km per second [23], which is what the rocks should have had by their composition. After several years, we found that the velocity of sound of lunar material was low because it had been extensively cracked in a complete vacuum. There was no liquid meniscus between the surface cracks and the sound would not cross over the cracks. More importantly to physical acoustics was our discovery that we could measure the velocity of sound in a tiny sphere. This led to a substantial innovation in physical acoustics because we were resonating a very tiny isotropic sphere and obtaining the velocity of sound. The equations used are found in Rayleigh’s classical book. See discussion of sphere resonant techniques above.
In the introduction to of a memorial volume of PAGEOPH dedicated to Ed Schreiber (
Figure 4)
[27], William Bassett and I quoted from their 1970 Science paper “Properties and Composition of Lunar Materials: Earth Analogies”.
Figure 4. The Schreiber Volume edited by Liebermann and Sondergeld
[27].
To account for these very low velocities, we decided to consider materials other than those listed initially by Birch or the later more detailed compilations of Anderson and Liebermann [28]. [The search was aided by considerations of much earlier speculations concerning the nature of the Moon by Erasmus in 1542 [29]; “With this merry toy…he made his friends to believe the Moon to be made of green cheese”], and a significant group of materials was found which have velocities that cluster around those actually observed for lunar rocks (see Figure 5).
Figure 5. From Schreiber and Anderson
[26] showing the comparison between sound velocities for the lunar rocks and for various earth materials.
The materials [cheeses] were chosen so as to represent a broad geographic distribution in order to preclude any bias that might be introduced by regional sampling. It is seen that these materials exhibit compressional velocities that are in consonance with those measured for lunar rocks—which leads us to suspect that perhaps old hypotheses are best after all and should not be lightly discarded.
As seen in Figure 5, the Vp = a + bp curve of Birch [30] for the rock types diabase, gabbros, and eclogites fit the cheeses surprisingly well. This apparent inconsistency, in that the cheeses do obey these relationships by having a velocity appropriate to their density, in contrast to the lunar rocks with which they compare so well, may readily accounted for when one considers how much better aged the lunar materials are.”
In 1970, Anderson and Schreiber used these velocity measurements in a Christmas card (Figure 6) for which they wrote, “It brightens the spirits in times like these to know the Moon is made of cheese”.
Figure 6. From Christmas card sent by Schreiber and Anderson to colleagues in 1970. Based on Schreiber and Anderson
[26] showing velocities of lunar rocks compared with terrestrial cheeses.
From 1964 to the early 1970s, Anderson and his colleagues published many papers in peer-reviewed scientific journals; after leaving Lamont for the University of California in 1971, Orson put these Collected Reprints into a special volume to share with his friends and colleagues (see
Figure 7). In addition to his 1995 book (
Figure 8), Orson also was a co-author, with Ed Schreiber and Naohiro Soga, of a book entitled “Elastic Constants and Their Measurement”
[31] (see
Figure 9), in which they discussed the theory of elasticity and various experimental techniques to measure elastic properties, indirect methods to estimate elastic constants, and thermodynamic approaches to estimate the pressure and temperature derivatives of elastic constants.
Figure 7. Collected Reprints from the mineral physics laboratory of Orson Anderson at Lamont from 1964 to 1970.
Figure 8. 1995 book by Orson Anderson based on notes for his solid-state geophysics course at Lamont Geological Observatory in the 1960s.
Figure 9. Book by Schreiber, Anderson, and Soga
[31].
During his eight years on the geology faculty at Columbia University, Orson supervised four graduate students: Bob Liebermann, whom he recruited from the Seismology Group at Lamont; Leon Thomsen, whom he recruited from the Goddard Institute for Space Science in New York; Harry Demarest, whom he recruited when he was an undergraduate at Columbia; and Nicholas Warren, who followed Orson to UCLA. In the acknowledgements for his 1995 book
[21], Orson thanked us:
Leon for teaching me Lagrangian theory, Bob for teaching me persistence, and Harry for laying the computational foundation of resonant ultrasound spectroscopy.Orson initially co-opted me to do library research for a review paper he had promised to write for the Air Force Office of Scientific Research (AFOSR). In those days before electronic journals, it was necessary to access hard copies and take notes or, if lucky, make photocopies. Fortunately, Columbia has an excellent library containing journals going back to the 19th century, and I also visited the Library of Congress during an AGU meeting in Washington. This 1966 report for the AFOSR was later published in Volume IVB of Physical Acoustics, edited by Warren Mason
[28].
Both Thomsen and I graduated in 1969 with the theses “On the Fourth-Order Anharmonic Equation of State of Solids”
[32] and “Effect of Iron Content upon the Elastic Properties of Oxides and Some Applications to Geophysics”
[33], respectively. In collaboration with Subir Banerjee (then at the Ampex Corporation and now at the University of Minnesota), we discovered anomalies in the compressional and shear properties of hematite in the region of the Morin transition, which we ascribed to magnetoelastic interactions
[34][35].
The semi-empirical research in Orson’s laboratory included investigations of the relationship of the bulk modulus K to the volume V of oxides
[36], which later evolved to a proposed “law of corresponding states for oxides”
[37]; for restrictions of this “law”, see
[38]. The latter showed that Anderson’s law is valid only for oxides having their mean atomic weight to be near the value of 20 or 21, while other oxides follow the more common law of chemical physics prescribed by lattice dynamics.
During this same time period, Don Anderson, at Caltech, had formulated a “seismic equation of state”
[39]. The two Andersons (Don and Orson) spent a few years dueling over which equations of state was the more appropriate; see
[40]. As the two protagonists had almost identical initials, D.L. and O.L., many authors began to spell out their first names when referencing their papers.
In another semi-empirical research initiative, Anderson and Liebermann
[41] used lattice dynamics and a Born repulsive potential between atoms to calculate the elastic constants of cubic lattices NaCl, CsCl, and ZnS as functions of pressure and to predict the vanishing of a shear constant (either c
44 or c’ = 1/2(c
11 − c
12)); see also
[42].
The primary theoretical research in the mineral physics lab in the 1960s was conducted by graduate student Leon Thomsen, to whom I am indebted for the following history
[43].
As described in detail above, Orson’s group got the opportunity to measure acoustic velocities on lunar rocks, which had been recovered on the Apollo mission. Comparing with terrestrial analogs, they found that the lunar rock velocities were close to that of sapsago cheese, a hard cheese from Switzerland. As such, they actually published a scholarly paper likening the Moon to green cheese
[26].
Perhaps because of this, I took Orson’s class in mineral physics. During that semester, a paper by Anderson appeared in the
Journal of Geophysical Research, of which he was the editor. I detected a logical error in that paper, and I brought it to Orson’s attention. Orson said, “You are correct; write a short paper discussing my error. I will see that it gets accepted by the
Journal of Geophysical Research under your name”. This was a great lesson in academic ethics
[44].
I switched my focus to Geophysics and moved my office from the Goddard Institute for Space Science to Lamont. Orson guided my research into a PhD thesis
[32][45]. This was a challenge to the conventional (Eulerian) formulation of finite strain theory promoted by the de facto dean of mineral physics, Francis Birch of Harvard; see details in
Section 1 of my 1970 paper
[45]. My thesis turned out to be a theoretical dead end, but it provided me with the basic mathematical foundation and insights which eventually led me to make important contributions in the analysis of seismic anisotropy” (end of quote from Thomsen).
At Columbia, Orson taught a graduate course on “solid-state geophysics”, which started with elementary lattice dynamics and continued to equations-of-state of solids. I adapted these notes and used them at the Australian National University from 1973 to 1976 and at Stony Brook from 1977 to 2013. These lecture notes later formed the basis for his 1995 book “Equations of State of Solids for Geophysics and Ceramic Science” (see
Figure 8)
[21].
As a native of Utah, Orson had a fascination with the Four Corners area of the western U.S. As he was now a professor of geology at Columbia and using ultrasonic techniques to measure sound velocities of rocks and minerals, Anderson organized field trips in 1966 and 1967 to the diatremes of northeastern Arizona, including those at Mule Ear, Garnet Ridge, Cane Valley, and Buell Park. To reach Mule Ear, it was necessary to hire a rubber raft and a guide to transport us from Bluff, Utah, to the field site (
Figure 10a). The Columbia team was a strange one for geological field work, as it included a solid-state physicist (Anderson), a ceramist (Schreiber), an industrial chemist (Soga), and two seismologists (Peter Molnar and the author), so-called “strangers in paradise;” fortunately, we had an excellent field guide, Eugene Shoemaker from the US Geological Survey in Flagstaff, Arizona (
Figure 10b). Though we did not find any diamond-bearing rocks, we did obtain many interesting specimens and photos (see
Figure 10c). In 1968, we drove to the Garnet Ridge diatreme, where Orson and Mineo Kumazawa posed for special photos (
Figure 10d,e). Kumazawa, Helmstaedt, and Masaki
[46] later published a paper on the elastic properties of eclogite xenoliths from the diatremes of the Eastern Colorado Plateau, including a lherzolite from the Mule Ear diatreme (see photos of field trips to diatremes at end of text
Figure 10a–e).
Figure 10. (a) Orson Anderson and Naohiro Soga being transported to the Mule Ear diatreme (in the background) via a rubber raft piloted by guide Kenny Ross; (b) field geologist Eugene Shoemaker showing outcrop from the Mule Ear diatreme to Edward Schreiber (ceramist), Orson Anderson (solid-state physicist with back turned), Naohiro Soga (industrial chemist), and Peter Molnar (seismologist); (c) Ed Schreiber viewing Alhambra Rock; (d) Orson Anderson on the Garnet Ridge diatreme; (e) Mineo Kumazawa at the Garnet Ridge diatreme.
In the late 1960s, the Lamont mineral physics team dispersed, and the Mineral Physics Laboratory closed. The sign was inherited by Ed Schreiber and later given to Orson, who still has it in Utah (see
Figure 2 from
[7]).
Naohiro Soga moved to Rice University and subsequently to Kyoto University, where he continued to apply lattice dynamics to inorganic network-forming materials such as silicates; he deduced their interatomic bond strengths, as well as other types of force constants arising from the network structural vibrations in order to clarify their physical properties.
Orson Anderson moved to the University of California Los Angeles (UCLA) in 1971 and served on the faculty until 2002.
Edward Schreiber moved to Queens College of the City University of New York. He died tragically in a car accident in 1991.
In 1998, a Topical Session of the American Geophysical Union was convened in honor of Orson Anderson, who gave the last talk. A collection papers was later published in
American Mineralogist (Volume 85, 2000), which represented topics of interest to Orson, who was a co-author on four of the papers
[47].