Ethylene-propylene-diene rubbers (EPDM) are one of the most important polyolefin materials widely commercialized and used in various industries in recent years. The production of EPDM is based solely on catalytic coordination polymerization processes. The development of new catalysts and processes for the synthesis of EPDM has expanded the range of products and their manufacturing in terms of energy efficiency, processability, and environmental safety.
- single-site catalyst
- ethylene
- propylene
- diene
- EPDM
- rubber
1. Introduction
| EPDM Manufacturer |
Polymer Grade | Ethylene Content, wt% | Diene (ENB) Content, wt% | Mooney Viscosity ML1+4(125 °C) 1 |
Polymer Structures | Ref. |
|---|---|---|---|---|---|---|
| ARLANXEO | Keltan® | 44–71 | 0–11.0 | 22–92 | LCB 2, ND 3, MD 4, BD 5 |
[8] |
| DOW Elastomers | NordelTM | 50–85 | 0–8.5 | 18–85 | ND, MD, BD | [9] |
| ExxonMobil | VistalonTM | 54–77 | 0–10.0 | 16–82 | LCB, ND, MD, BD |
[10] |
| Mitsui Chemical | Mitsui EPTTM | 41–72 | 0–14.0 | 40–78 | LCB, ND, MD, BD |
[11] |
| KumhoPolychem | KEP® | 55–71 | 0–10.0 | 23–95 | No data | [12] |
1 Mooney viscosity is the routine industry standard correlated with molecular weight of EPDM. High Mooney viscosities are preferred. Abbreviation according to ASTM D1646 is ML 1+4 (125 °C), where M—Mooney units, L—large rotor, 1—the sample preheat time (min), 4—the sample test time (min) at 125 °C. 2 Long Chain Branching. 3 Narrow molecular weight distribution.4 Medium molecular weight distribution. 5 Broad molecular weight distribution.
2. Single-Site Catalysts
2.1. Metallocene Catalysts
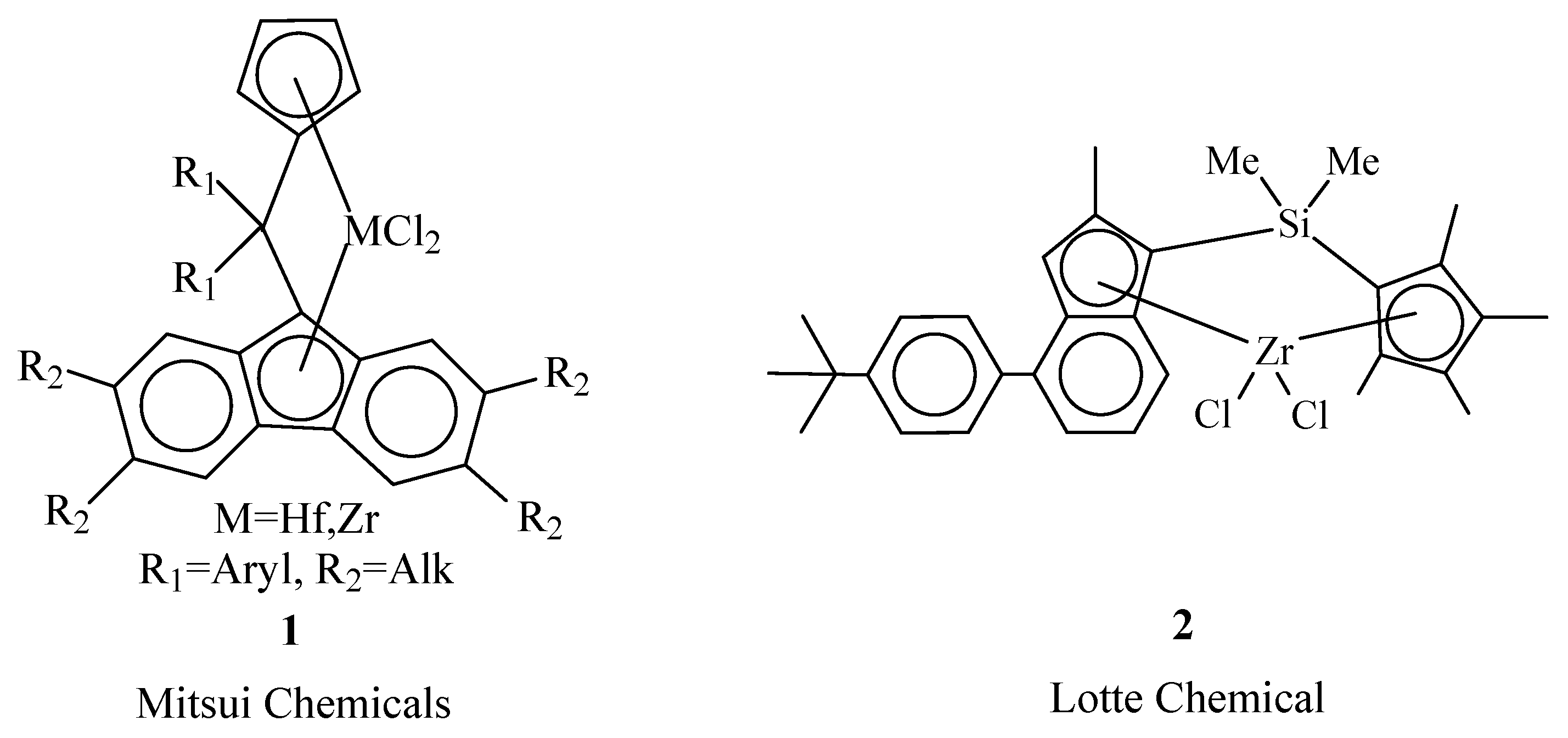
Promising for producing EPDM are two types of complexes: metallocenes of Cs symmetry [4][5][6][7][8] and zirconocenes of C1 symmetry [9]. The capabilities of Cs symmetry catalysts in the synthesis of EPDM can be demonstrated by the patent data of Mitsui Chemicals [16][17][18][19][20]. They claim a new effective class of hafnocene and zirconocene complexes of type 1 (Figure 1) containing different substituents in both the bridging group and the peripheral positions of the Flu ligand. When activated with CPh3B(C6F5)4 (4 and 10 equiv.), the complexes effectively provide ternary copolymerization of E/P/ENB at 80–120 °C. Hafnocenes 1 ensure the incorporation of ENB at a level of 7–11 wt% and form elastomers with high molecular weights (Mw = 1000–2000 kDa) [16][17][18]. The compounds with R1 = 4-MePh, 4-MeOPh, 4-Me2NPh and R2 = Me show the highest activity up to a 10,000–20,000 kg copolymer/(molHf h atm)). Zirconocenes 1 show high efficiency in the synthesis of well-vulcanizing VNB terpolymers with a low degree of branching [19]. Such copolymers cannot be obtained by using conventional vanadium and other metallocene systems. The content of VNB in this EPDM reaches 10 wt%. The molecular weights range from 50 to 600 kDa.
2.2. “Constrained” Geometry Complexes
Special attention in the synthesis of EPDM is paid to bridged monocyclopentadienyl complexes, the so-called “constrained” geometry complexes (CGCs). CGCs contain h5-Cp-ligand linked by a bridging group with a donor ligand (Don). The bridging group reduces the Cp–M–Don angle by approximately 20–30°as compared to Cp–M–Cp, which ensures high availability of a transition metal in the active site and, thus, a high degree of comonomer incorporation [21][22][23]. The donor is linked to the transition metal by a s-bond and ensures high stability of catalysts of this type at elevated temperatures (up to 160–180 °C).Variations in any part of the CGC ligands as well as the type of transition metal affect catalytic properties of the complexes, such as activity, the ability to insert comonomer(s), and molecular weight characteristics of the resulting polymers.
CGCs 3 (Figure 2) were commercialized by DOW in the mid of the 1990s for homogeneous polymerization processes used for production of linear low-density polyethylene [23][24][25][26]. Complexes of this type are also effective for the synthesis of EPDM [25][26], which allowed DOW to launch a wide range of elastomers (NordelTM IP). CGCs provide production of EPDM with a wide range of Mw (up to 500 kDa), composition (up to 50 wt% ofpropylene and 7.5 wt% ofENB), and the Mooney viscosity (ML1+4(125°C)=18–85).
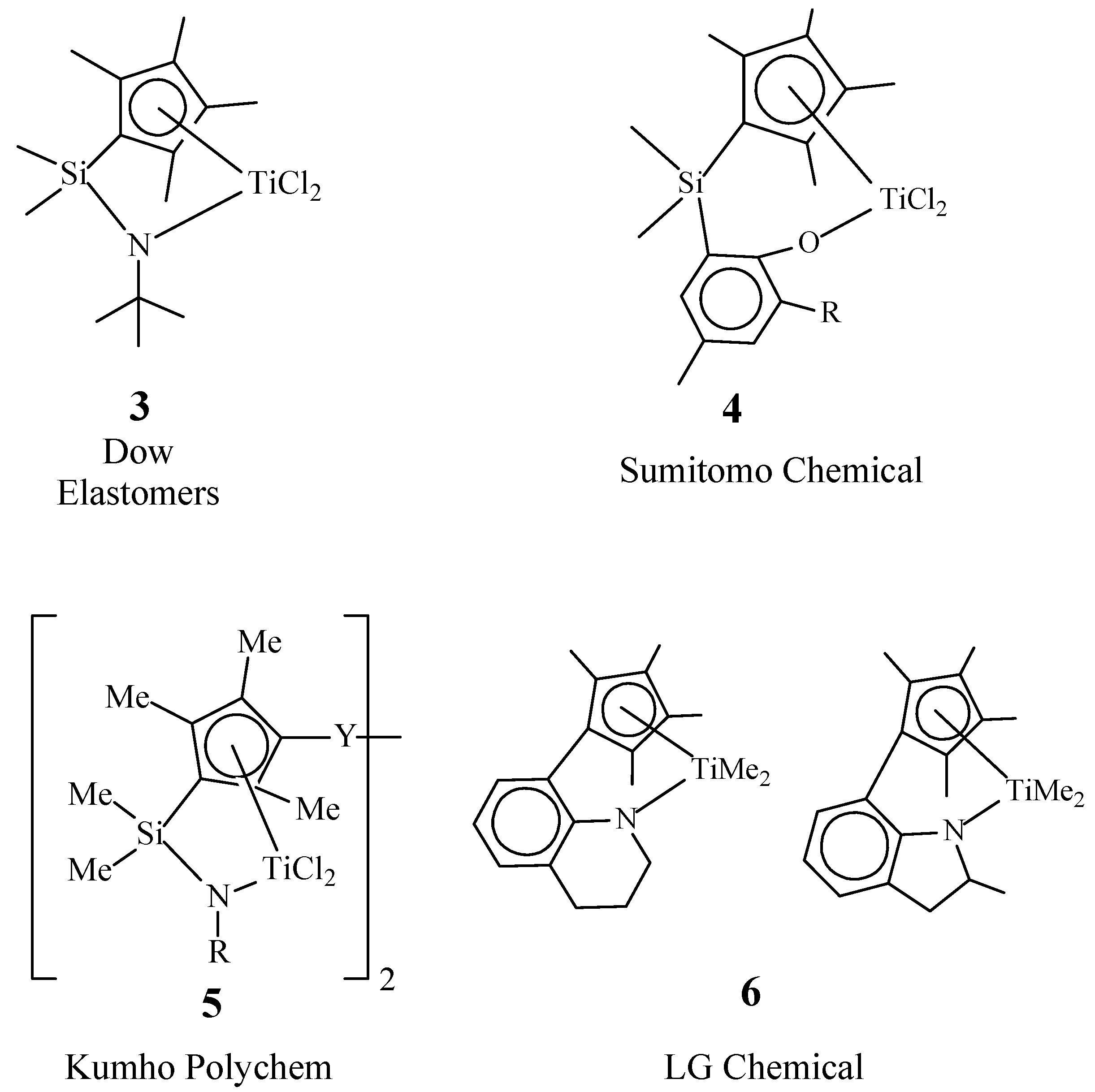
Figure 2. Chemical structures of CGCs 3-6.
In recent years, one can note increasing interest to catalysts of this type from several leading EPDM manufacturers. From the late 1990s to the present, active developments in the field of CGC molecular design have been carried out at Sumitomo Chemical, KumhoPolychem, and LG Chem, which have made it possible to significantly expand the range of promising catalysts for the synthesis of EPDM [27][28][29][30][31][32][33][34].
2.3. Half-Sandwich Titanium Complexes
Another promising class of catalysts for the synthesis of EPDM are half-sandwich titanocenes 7–11(Figure 3) containing N-donor ligands [35][36][37][38][39][40][41][42][43][44][45][46][47][48][49][50][51][52]. Catalysts are included in the new technology was called Keltan Advanced Catalyst Elastomer (Keltan ACE™)
Keltan ACETM catalysts are characterized by high catalytic activity (up to 1200 tons copolymer/(molTi h)) in ternary copolymerization of olefins and dienes and make it possible to obtain copolymers with a high content of dienes (up to 15 wt% of ENB, 5 wt% ofVNB and 20 wt% ofDCPD). Most of these catalysts are stable in copolymerization processes at temperatures up to 120°C and form copolymers with high Mw values up to 2000 kDa. A variety of developed complexes of this type makes it possible to obtain EPDM with a wide range of properties [40][41][42][43][44][45][46][47][48][49][50][51][52].
On the basis of Keltan ACE™ technology, large-scale production of EPDM has been organized at the plants in Geleen, Netherlands, and Changzhou, China, each with listed annual capacity of 160000 tons, along with 40000 tons per year in Triunfo, Brazil [39][54][55].
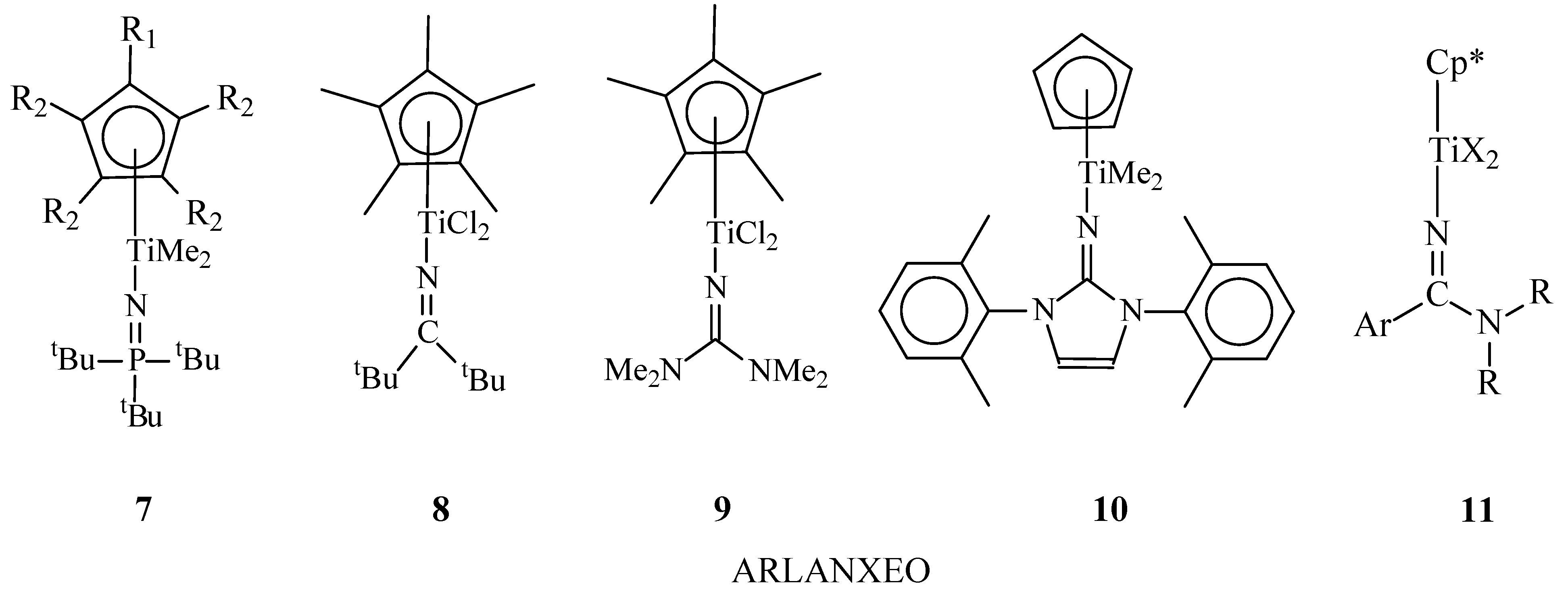
Figure 3. Chemical structures of half-sandwich titanium complexes 7-11.
2.4. Post-MetalloceneChelate Catalysts
The most promising catalysts of this class for the synthesis of EPDM are aryloxyether12 complexes of hafnium and zirconium (Figure 4). The compounds were discovered using the approaches of Combinatorial Chemistry and High-Throughput Screening [56][57][58][59][60] and have been actively developed by Dow Elastomers for the last 15 years [25][61][62][63][64][65][66][67][68][69][70][71][72][73][74][75].
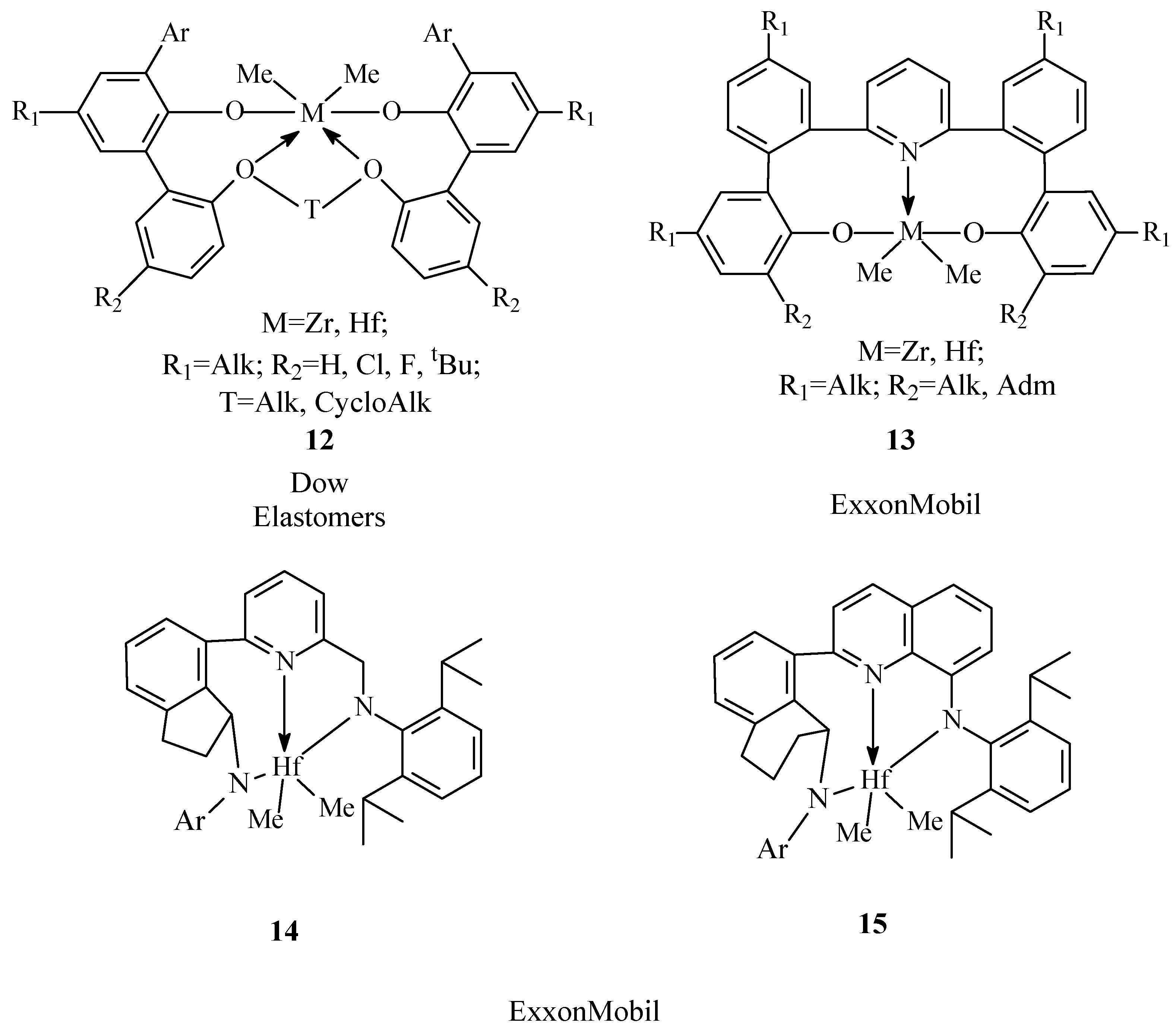
Figure 4. Chemical structures of aryloxyether12 complexes.
Aryloxyethercomplexes 12 have the most unique catalytic characteristics and by sum of properties surpass many known classes of catalysts for the production of EPDM [25][61][62][63][64][65][66][67][68]. The advantages of the catalysts are the ability to be effectively activated by low molar amounts of the activator (AlMAO/M=10-200, Bborate/M up to 5 mol/mol), and high activity in copolymerization processes in high-temperature solution polymerization conditions (120-180°C). The advantage of a high-temperature process carried out in the solution polymerization (the polymer is soluble in the reaction medium) is the formation of amorphous copolymers with broad molecular weight distribution. A feature of these systems is also their ability to form copolymers with high molecular weights (Mw=100-1000 kDa) and low content of gel fraction [25]. Such catalysts were used to obtainrubbers with a high content of propylene (up to 50 wt%) and dienecomonomers of various types (ENB, VNB, DCPD, etc., up to 16 wt%). Aryloxyether complexes are the basis of highly efficient Advanced Molecular Catalyst technology (AMC) developed in 2015 for obtaining new NordelTM rubber grades with improved properties [25]. Based on this technology, a plant with annual capacity of 200 thousand tons of EPDM was launched in Plakimin (USA) in 2018 [69].
3. Conclusions
In conclusion, it should be noted that despite the fact that EPDM have being produced by the industry for more than 50 years, there is a high potentialin both improving the technologies for the synthesis of EPDM and expanding the range of their grades. This potential is ensured by the development of new-generation catalytic systems based on Group IVB complexes, which are alternatives to low-activity, unstable, and toxic vanadium catalysts. In particular, this is evidenced by high research activity over the past 10–15 years of the world’s leading manufacturers of EPDM in the development of new systems and their active commercialization. The main advantage of modern systems compared to vanadium ones is their environmental safety, higher activity, and thermal stability, with the possibility of producing EPDM with different contents of comonomers and microstructures, including highly branched and bimodal terpolymers. The use of these catalytic systems has already made it possible to bring to market new grades of EPDM with a wide range of properties (for example, Keltan® (ARLANXEO), NordelTM IP (Dow Elastomers), VistalonTM (Exxon), Mitsui EPTTM (Mitsui), etc.).
The huge potential for tuning the structures of catalytic systems allows researchers to count on the further creation of new efficient systems for the synthesis of EPDM and, accordingly, the production of elastomers with a high content of diene(s) and the creation of new polymer microstructureswith an even wider range of properties.
Funding:This research was funded by the Ministry of Science and Higher Education of the Russian Federation (State Task, No. registration AAAA-A19-119041090087-4).
This entry is adapted from the peer-reviewed paper 10.3390/catal12070704
References
- Noordermeer, J.W. Ethylene-Propylene Elastomers. In Encyclopedia of Polymer Science and Technology, John Wiley & Sons: New York, NY, USA, 2002; pp. 178–196.
- Ravishankar, P.S. Treatise on EPDM. Rubber Chem. Technol.2012, 85, 327–349.
- Van Duin, M.; van Doremaele, G.; van der Aar, N. Defining EPDM for the past and the next 50 years. KGK2017, 11–12, 14–23.
- Bravaya, N.M.; Faingol’d, E.E.; Badamshina, E.R.; Sanginov, E.A. Advances in synthesis of ethylene–propylene–diene elas-tomers by ion-coordination polymerization on single-site catalytic systems of new generation. Polym. Sci. Ser. C2020, 62, 1–16.
- Transition Metal Catalyzed Polymerizations: Ziegler-Natta and Metathesis Polymerizations; Quirk, R.P., Hoff, R.E., Eds.; Cambridge University Press: Cambridge, UK, 1988.
- Karpeles, R.; Grossi, A.V. EPDM rubber technology. In Handbook of Elastomers, 2nd ed.; Bhowmick, A.K., Stephens, H.L., Eds; Marcel Dekker: New York, NY, USA, 2001; pp. 845–876.
- Ma, Y.; Reardon, D.; Gambarotta, S.; Yap, G.; Zahalka, H.; Lemay, C. Vanadium-catalyzed ethylene-propylene copolymeri-zation: the question of the metal oxidation state in Ziegler-Natta polymerization promoted by (β-diketonate)3V. Organometallics 1999, 18, 2773–2781.
- ARLANXEOWebsite/Find Products/Ethylene Propylene Diene Rubber. Available online: https://www.arlanxeo.com/en/products/finder?q=%3aname-asc%3abrandCategory%3akeltan (accessed on 21 June 2022).
- NORDEL™ EPDM Product Selection Guide—265-11001-01-nordel-epdm-product-selection-guide.pdf. Available online: https://www.dow.com/content/dam/dcc/documents/en-us/catalog-selguide/265/265-11001-01-nordel-epdm-product-selection-guide.pdf (accessed on 21 June 2022).
- ExxonMobil/Home/Resources/Technical Data Sheets/EPDM Rubber. Available online: https://www.exxonmobilchemical.com/en/resources/product-data-sheets/epdm-rubber (accessed on 21 June 2022).
- MITSUI EPT™/EPDM Rubber. Available online: https://us.mitsuichemicals.com/service/product/mitsui-ept.htm (accessed on 21 June 2022).
- KUMHO POLYCHEM Website/EPDM/Overview. Available online: https://www.polychem.co.kr/eng/product/epdm?seq=1 (accessed on 21 June 2022).
- Resconi, L.; Cavallo, L.; Fait, A.; Piemontesi, F. Selectivity in propene polymerization with metallocene catalysts. Chem. Rev. 2000, 100, 1253–1346.
- Altaf, A.A.; Badshah, A.; Khan, N.; Marwat, S.; Ali, S. Zirconium complexes in homogeneous ethylene polymerization. J. Coord. Chem. 2011, 64, 1815–1836.
- Gillis, D.J.; Karpeles, R. Process for Producing Polyolefin Elastomer Employing a Metallocene Catalyst. Patent US 6225426 B1, 1 May 2001.
- Endo, K.; Hiwara, M.; Matsuura, S.; Mizobuchi, Y.; Yamamura, Y.; Noguchi, Y.; Ishii, Yu.; Sakai, T.; Shishido, K.; Ichino, K.; Inomata, K.; Takeuchi, F.; Ide, K. Ethylene Alpha-Olefin Non-Conjugated Polyene Copolymer, Use Thereof, and Manufac-turing Method Thereof. Patent US 2016/0347894 A1, 1 December 2016.
- Ichino, K.; Kikuchi, Yo.; Tohi, Ya.; Matsugi, T.; Yanagimoto, Ya.; Arino, M.; Shishido, K.; Hosoya, M. Eth-ylene/α-Olefin/Non-Conjugated Polyene Copolymer, and Production Process and Use Thereof. Patent US 10435494 B2, 08 November 2018.
- Ichino, K.; Yamaguchi, T.; Aita, Yu.; Noguchi, Yu. Thermoplastic Elastomer Composition, Use Thereof, Method for Producing Same, Ethylene/α-Olefin/Unconjugated Polyene Copolymer and Use Thereof. Patent US 2018/0072877 A1, 15 March 2018.
- Ichino, K.; Kikuchi, Yo.; Shishido, K.; Tanaka, J.; Arino, M. Ethylene/Alpha-Olefin/Non-Conjugated Polyene Copolymer, Method for Producing the Same, and Use Thereof. Patent US 2021/0009730 A1, 14 January 2021.
- Hong, Y.J.; Shin, E.H.; Jung, S.W.; Woo, H.Y.; Lee, R.H.; Min, J.K.; Chae, B.H. Process for Ethylene/Propylene/Dieneterpolymer Using New Transition Metal Compound. Patent KR 101792956 B1, 11 March 2017.
- Chen, E.Y.-X.; Marks, T.J. Cocatalysts for metal-catalyzed olefin polymerization: Activators, activation processes, and struc-ture-activity relationships. Chem. Rev. 2000, 100, 1391–1434.
- Zijlstra, H.S.; Harder, S. Methylalumoxane—History, production, properties, and applications. Eur. J. Inorg. Chem. 2015, 2015, 19– 43.
- Bochmann, M. The chemistry of catalyst activation: The case of group 4 polymerization catalysts. Organometallics 2010, 29, 4711–4740.
- Ali, A.; Tufail, M.K.; Jamil, M.I.; Yaseen, W.; Iqbal, N.; Hussain, M.; Ali, A.; Aziz, T.; Fan, Z.; Guo, L. Comparative analysis of ethylene/diene copolymerization and ethylene/propylene/diene terpolymerization using ansa-zirconocene catalyst with alkyl-aluminum/borate activator: The effect of conjugated and nonconjugated dienes on catalytic behavior and polymer micro-structure. Molecules 2021, 26, 2037.
- Ali, A.; Akram, M.A.; Guo, Y.; Wu, H.-L.; Liu, W.; Khan, A.; Liu, H.; Fu, Z.; Fan, Z. Ethylene–propylene copolymerization and their terpolymerization with dienes using ansa-zirconocene catalysts activated by borate/alkylaluminum. J. Macromol. Sci. Part A 2020, 57, 156–164.
- Ali, A.; Nadeem, M.; Lu, J.; Moradian, J.M.; Rasheed, T.; Aziz, T.; Maouche, C.; Guo, Y.; Awais, M.; Zhiqiang, F.; et al. Rapid kinetic evaluation of homogeneous single-site metallocene catalysts and cyclic diene: How do the catalytic activity, molecular weight, and diene incorporation rate of olefins affect each other? RSC Adv. 2021, 11, 31817–31826.
- Bravaya, N.M.; Panin, A.N. Method of Producing Copolymers of Olefin Monomers with Cyclic or Linear Dienes. Patent RU 2477289 C1, 10 March 2013.
- Bravaya, N.M.; Panin, A.N.; Faingol’d, E.E.; Saratovskikh, S.L.; Babkina, O.N.; Zharkov, I.V.; Perepelitsina, E.O. Isobutyl-alumoxanes as high-performance activators of rac-Et(2-MeInd)2ZrMe2 in copolymerization of ethylene with propylene and ternary copolymerization of ethylene, propylene, and 5-ethylidene-2-norbornene.Polym. Bull. 2016, 73, 473–491.
- Faingol’d, E.E.; Bravaya, N.M.; Panin, A.N.; Saratovskikh, S.L.; Babkina, O.N. Use of Aryl Oxides of Isobutyl Aluminium as Activators Dialkylmetallocene Catalysts for Homopolymerisation Ethylene, Propylene, Copolymerisation of Ethylene with Propylene and Triple Copolymerisation of Ethylene, Propylene and Diene, Homogeneous Metallocene Catalyst Systems for Synthesis of Homo-and Copolymers of Olefins and Dienes, Method of Producing Homo- and Copolymers of Olefins and Dienes, Method for Stabilisation of Homo- and Copolymers of Olefins and Dienes. Patent RU 2588496 C2, 27 June 2016.
- Faingol’d, E.E.; Bravaya, N.M.; Panin, A.N.; Babkina, O.N.; Saratovskikh, S.L.; Privalov, V.I. Isobutylaluminum aryloxides as metallocene activators in homo- and copolymerization of olefins. J. Appl. Polym. Sci. 2016, 133, 43276.
- Faingol’d, E.E.; Zharkov, I.V.; Bravaya, N.M.; Panin, A.N.; Saratovskikh, S.L.; Babkina, O.N.; Shilov, G.V. Sterically crowded dimericdiisobutylaluminumaryloxides: Synthesis, characteristics, and application as activators in homo- and copolymerization of olefins. J. Organomet. Chem. 2018, 871, 86–95.
- Faingol’d, E.E.; Saratovskikh, S.L.; Panin, A.N.; Babkina, O.N.; Zharkov, I.V.; Garifullin, N.O.; Shilov, G.V.; Bravaya, N.M. Ethylene/propylene and ethylene/propylene/5-ethylidene-2-norbornene copolymerizations on metallo-cene/(2,6-tBu2PhO-)AliBu2 catalyst systems. Polymer 2021, 220, 123559.
- Baier, M.C.; Zuideveld, M.A.; Mecking, S. Post-metallocenes in the industrial production of polyolefins. Angew. Chem. Int. Edit. 2014, 53, 9722–9744.
- Braunschweig, H.; Breitling, F.M. Constrained geometry complexes—synthesis and applications. Coord. Chem. Rev. 2006, 250, 2691–2720.
- Klosin, J.; Fontaine, P.P.; Figueroa, R. Development of group IV molecular catalysts for high temperature ethylene-α-olefin copolymerization reactions. Account Chem. Res. 2015, 48, 2004–2016.
- Canich, J.A.M. Olefin Polymerization Catalysts. Patent EP 0420436 A1, 3 April 1991.
- Thakur, V.; Shan, C.L.P.; Li, G.; Han, T.; Doelder, J.D. Sponge EPDM by design. Plast. Rubber Compos. 2019, 48, 32–41.
- Stevens, J.C.; Timmers, F.J.; Wilson, D.R.; Schmidt, G.F.; Nickias, P.N.; Rosen, R.K.; Knight, G.W.; Lai, S.-Y. Constrained Geometry Addition Polymerization Catalysts, Processes for Their Preparation, Precursors Therefor, Methods of Use, and Novel Polymers Formed Therewith. Patent EP 0416815 A2, 13 March 1991.
- Katayama, H.; Nabika, M.; Imai, A.; Miyashita, A.; Watanabe, T.; Johohji, H.; Oda, Y.; Hanaoka, H. Transition Metal Complex, Process for Producing the Same, Olefin Polymerization Catalyst Containing the Transition Metal Complex and Process for Producing Olefin Polymers. Patent US 6329478 B1, 11 December 2001.
- Kim, S.-B.; Goo, H.-R.; Hong, Y.-J.; Lee, M.-H.; Do, Y.-G.; Park, M.-H.; Park, J.-H. Dinuclear Metallocene Catalysts and Process for the Manufacture of EP and EPDM Polymers Using Them. Patent KR 101216691 B1, 28 December 2012.
- Kim, H.-J.; Kim, H.-S.; Kim, S.K.; Yoon, S.; Koo, H.R.; Son, A. Catalyst composition for preparing elastic copolymer, and method for preparing elastic copolymer, which comprises ethylene and alpha-olefin or ethylene, alpha-olefin and unconjugated diene, by using same. Patent US 11220566 B2, 11 January 2022.
- Park, S.H.; Yoon, S.C.; Kim, S.K.; Ko, J.S.; Park, S.E.; Choi, S.Y. Elastic Diene Terpolymer and Preparation Method Thereof. Patent US 9428600 B2, 30 June 2016.
- Kim, S.K.; Park, S.H.; Yoon, S.C.; Ko, J.S.; Choi, S.Y. Elastic Diene Terpolymer and Preparation Method Thereof. Patent US 9493593 B2, 15 November 2016.
- Yoon, S.-C.; Park, S.-H.; Ko, J.-S.; Choi, S.-Y. Elastic Terpolymer Including Diene Group and Preparation Method Thereof. Patent US 9410008 B2, 9 August 2016.
- Park, S.H.; Yoon, S.C.; Kim, S.K.;Ko, J.S.; Park, S.E.; Choi, S.Y. Elastic Diene Terpolymer and Preparation Method Thereof. Patent US 9650460 B2, 16 May 2017.
- Wang, Q.; Brown, S.J. Process to Prepare Ethylene Propylene Elastomer. Patent EP 1162214 A1, 12 December 2001.
- Windmuller, P.J.H.; van Doremaele, G.H.J. Process for the Production of a Polymer Comprising Monomeric Units of Ethylene, an α-Olefin and a Vinyl Norbornene. Patent WO 2005/005496 A2, 20 January 2005.
- Ijpeij, E.G.; Arts, H.J.; van Doremaele, G.H.J.; Beijer, F.H.; van Der Burgt, F.; Zuideveld, M.A. Process for the Preparation of a Polyolefin. Patent WO 2005/014601 A2, 17 February 2005.
- Windmuller, P.; Doremaele, G. Process for the Production of a Polymer Comprising Monomeric Units of Ethylene, an α-Oplefin and a Vinyl Norbornene. Patent US 2006/0205900 A1, 14 September 2006.
- Windmuller, P.; Doremaele, G. Process for the Production of a Polymer Comprising Monomeric Units of Ethylene, an α-Oplefin and a Vinyl Norbornene. Patent US 7829645 B2, 9 November 2010.
- Van Doremaele, G.; van Duin, M.; Valla, M.; Berthoud, A. On the development of titanium κ1-amidinate complexes, com-mercialized as Keltan ACE™ technology, enabling the production of an unprecedented large variety of EPDM polymer structures. J. Polym. Sci. Pol. Chem. 2017, 55, 2877–2891.
- Van Doremaele, G.H.J.; Zuideveld, M.A.; Leblanc, A.; Norambuena, V.F.Q. Catalyst Component for the Polymerization of Olefins Having a Guanidinate Ligand. Patent US 8987393 B2, 24 March 2015.
- Ijpeij, E.; Arts, H.; van Doremaele, G.; Windmuller, P.; van der Burgt, F.; Zuideveld, M.A. Polymerization Catalyst Comprising Amidine Ligand. Patent US 7956140 B2, 7 June 2011.
- Van Doremaele, G.H.J.; Berthoud, A.; Norambuena, V.Q.; Rupnicki, L.; Karbaum, P. Metal Complex with a Cyclic Amidine Ligand. Patent WO 2015113957 A1, 6 August 2015.
- Collins, R.A.; Russell, A.F.; Scott, R.T.W.; Bernardo, R.; van Doremaele, G.H.J.; Berthoud, A.; Mountford, P. Monometallic and bimetallic titanium κ1-amidinate complexes as olefin polymerization catalysts. Organometallics 2017, 36, 2167–2181.
- Berthoud, A.; van Doremaele, G.; Norambuena, V.Q.; Scott, R.; Zuideveld, M.A.; Arts, H.J. Bimetallic Complex Comprising Cyclopentadienyl and Amidine Ligands. Patent US 9862736 B2, 9 January 2018.
- Van Doremaele, G.H.J.; Zuideveld, M.A.; Mountford, P.; Heath, A.; Scott, R.T.W. Ti Catalyst System Comprising Substituted Cyclopentadienyl, Amidine and Diene Ligand. Patent WO 2011076775 A1, 30 June 2011.
- Van Doremaele, G.H.J.; Zuideveld, M.A. Borane Activated ti Catalyst System Comprising Amidine and Diene Ligand. Patent WO 2011076772 A1, 30 June 2011.
- Karbaum, P.; Scott, R.T.W.; van De Moosdijk, J. Metal Complex Comprising Amidine and Substituted Cyclopentadienyl Lig-ands. Patent US 10472431 B2, 12 November 2019.
- Berthoud, A.; Van Doremaele, G.H.J.; Scott, R.T.W.; Perez, F.; Bernardo, R. Catalyst System. Patent WO 2017/029141 A1, 23 February 2017.
- Scott, R.T.W.; Zuideveld, M.A.; Mountford, P. Metal Complex with a Lewis Base Ligand. Patent WO 2014180913 A1, 13 November 2014.
- Scott, R.T.W.; Bernardo, R.; van de Moosdijk, J.; Berthoud, A. Metall Complex Comprising Amidine and Substituted Cyclo-pentadienyl Ligands. Patent EP 3272761 A1, 24 January 2018.
- Goryunov, G.P.;Samsonov, O.V.; Uborsky, D.V.; Voskoboynikov, A.Z.; Berthoud, A.; Valla, M. Metal Complex Comprising Amidine and Indole Fused Cyclopentadienyl Ligands. Patent US 20210269465 A1, 2 September 2021.
- Bernardo, R.; van Doremaele, G.; van Meerendonk, W.; Windmuller, P. Catalyst Mixture. Patent US 20220056162 A1, 24 February 2022.
- DSM continues to invest in innovative growth in performance materials cluster. Available online: https://www.pressreleasefinder.com/pr/DSMELPR005/en/ (accessed on 19 May 2022).
- History | ARLANXEO Website/Customer Portal. Available online: https://www.arlanxeo.com/en/company/history (accessed on 19 May 2022).
- Arlanxeoto close Texas Keltan EPDM plant | Rubber News. Available online: https://www.rubbernews.com/suppliers/arlanxeo-close-texas-keltan-epdm-plant (accessed on 19 May 2022).
- Boussie, T.R.; Bruemmer, O.; Diamond, G.; Goh, C.; Lapointe, A.M.; Leclerc, M.K.; Shoemaker, J.A. Bridged bi-Aromatic Lig-ands, Complexes, Catalysts and Processes for Polymerizing and Polymers Therefrom. Patent WO 03/091262A1, 6 November 2003.
- Boussie, T.; Bruemmer, O.; Diamond, G.; LaPointe, A.; Leclerc, M.; Micklatcher, C.; Sun, P.; Bei, X. Bridged bi-Aromatic Cat-alysts, Complexes, and Methods of Using the Same. Patent US 20060025548 A1, 2 February 2006.
- Boussie, T.; Diamond, G.; LaPointe, A.; Leclerc, M.; Micklatcher, C.; Sun, P.; Bei, X. Bridged bi-Aromatic Catalysts, Complexes, and Methods of Using the Same. Patent US 20060052554 A1, 9 March 2006.
- Boussie, T.R.; Diamond, G.M.; Goh, C.; Hall, K.A.; LaPointe, A.M.; Leclerc, M.; Lund, C.; Murphy, V.; Shoemaker, J.A.W.; Tracht, U.; et al. A fully integrated high-throughput screening methodology for the discovery of new polyolefin catalysts: discovery of a new class of high temperature single-site group (IV) copolymerization catalysts. J. Am. Chem. Soc. 2003, 125, 4306–4317.
- Boussie, T.R.; Diamond, G.M.; Goh, C.; Hall, K.A.; LaPointe, A.M.; Leclerc, M.; Murphy, V.; Shoemaker, J.A.W.; Turner, H.; Rosen, R.K.; et al. Nonconventional catalysts for isotactic propene polymerization in solution developed by using high-throughput-screening technologies. Angew. Chem. Int. Ed. 2006, 45, 3278–3283.
- Konze, W.V.; Vanderlende, D.D. Polyolefin Solution Polymerization Process and Polymer. Patent US 8349984 B2, 8 January 2013.
- Boone, H.W.; Iverson, C.N.; Konze, W.V.; Vanderlende, D.D. Ethylene/Alpha-Olefin/Diene Solution Polymerization Process and Polymer. Patent US 8299189 B2, 30 October 2012.
- Konze, W.V.; Stevens, J.C.; Vanderlende, D.D. High Efficiency Solution Polymerization Process. Patent WO 2007/136495 A3, 17 January 2008.
