Some of the most effective anticancer compounds are still derived from plants since the chemical synthesis of chiral molecules is not economically efficient. Rapid discovery of lead compounds with pronounced biological activity is essential for the successful development of novel drug candidates. The genus Astragalus L. is the largest in the family Fabaceae (syn. Leguminosae), with more than 3500 species. Astragalus, excluding Astracantha (formerly Astragalus subgenus Tragacantha), has a world total of ca. 2500 species, of which ca. 500 are in the Americas. Many of the species have conservation status “vulnerable” or “critically endangered”.
- plant anticancer compounds
- in vitro production
- saponins
- flavonoids
- alkaloids
- Astragalus
1. Ethnobotanical Data of Astragalus Species Used against Cancer
2. Secondary Metabolites of Astragalus Species Anticancer Properties
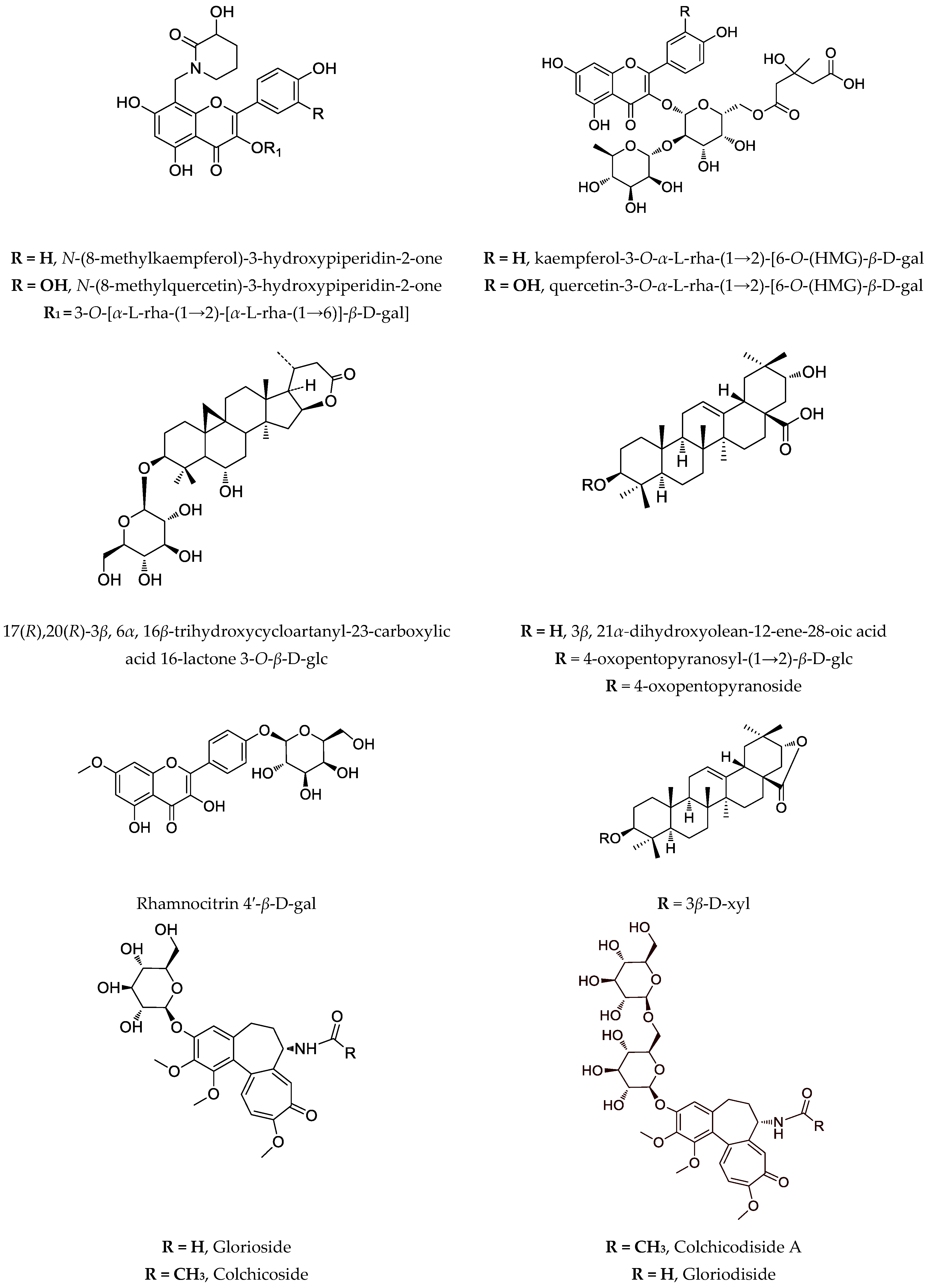
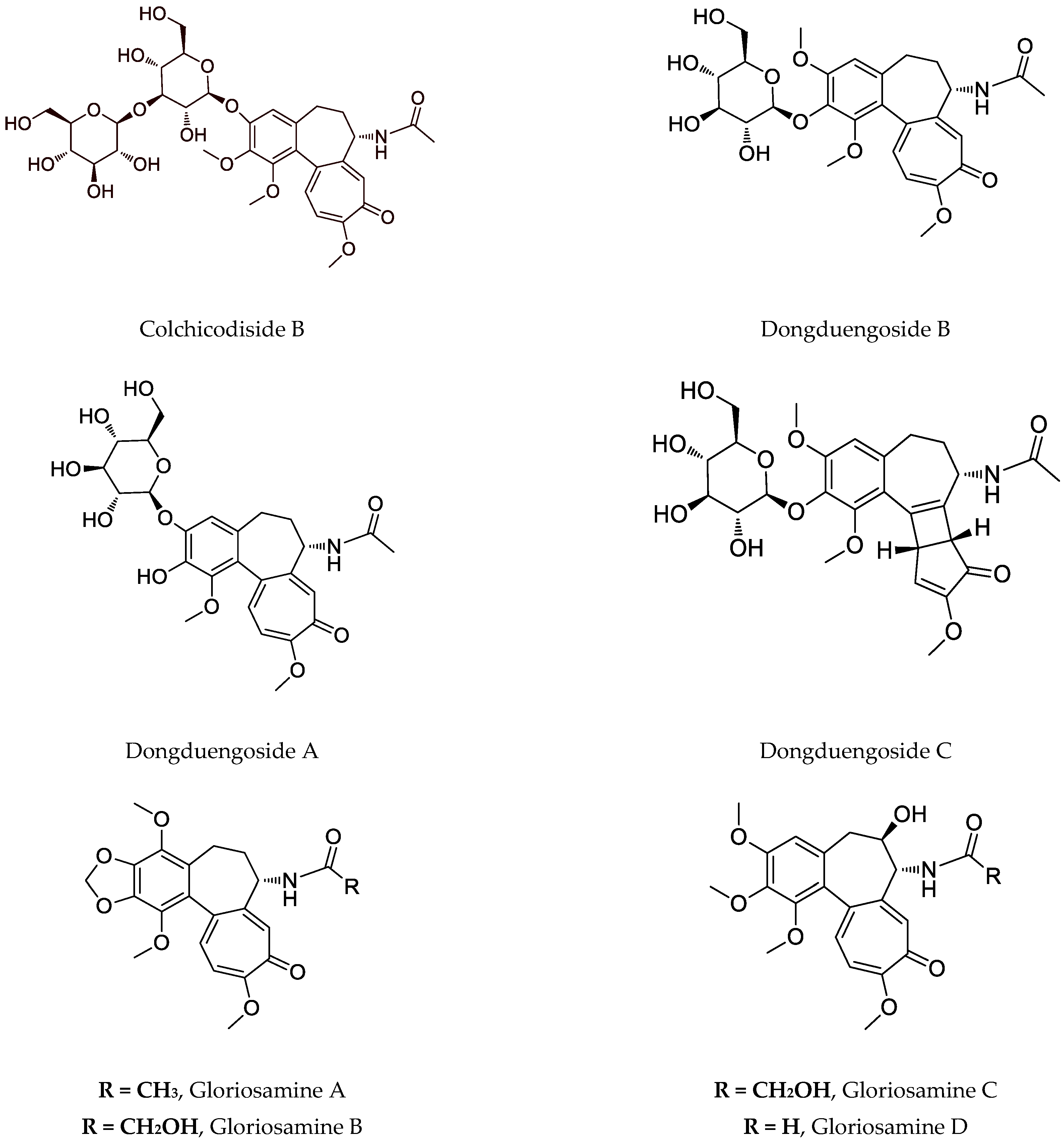
3. Biotechnology of Astragalus Species
3.1. Cell Culture
3.2. Effects of Medium Composition and Growth Regulators
3.3. Effects of End-Product Inhibition
3.4. Genetic Transformation of Astragalus Species by Agrobacterium Rhizogenes
3.5. In Vitro Production of Secondary Metabolite
4. Characteristics of Target Gloriosa Species
4.1. Ethnobotaical Data of Gloriosa Species Used against Cancer
4.2. Secondary Metabolites of Gloriosa Species with Anticancer Properties
| Plant Species | Type | Compounds Isolated | Cytotoxicity on Cell Lines (IC50) | References |
|---|---|---|---|---|
| A. aitosensis | callus, suspension | cycloartane saponins, sterols, flavonoids | n.d. | [9] |
| aerial pars, wild grown * | 5,6-dehydro-6-desoxyastragenol | n.d. | [9] | |
| A. angustifolius | callus, suspension | cycloartane saponins, flavonoids | n.d. | [11] |
| aerial parts, wild grown * | β-sitosterol, cycloastragenol, astragenol, soyasapogenol B, 3-O-[α-L-rha-(1→2)-β-D-xyl-(1→2)-β-D-glc]-3β,22β,24-trihydroxyolean-12-en-29-oic acid |
n. d. HeLa (36 µM); HT-29 (50 µM) |
[22] [11] |
|
| A. asper | aerial parts, wild grown * | saponins, flavonoids | n.d. | [11] |
| A. boeticus | callus, suspension, hairy roots |
saponins, soyasapogenol B, β-sitosterol, flavonoids | n.d. | [42] |
| A. brachycera | hairy roots ** shoots ** |
cycloartane saponins, sterols | n.d. | [9] |
| A. canadensis | hairy roots | cycloartane saponins, cycloastragenol, astragenol, | n.d. | [9] |
| A. centralpinus | aerial parts, wild grown * | flavonoids | n.d. | [11] |
| A. corniculatus | aerial parts, wild grown *** | two oleanane type saponins and a corresponding lactone | Graffi tumour–in vivo, i.p., hamsters (50 mg/kg) ***; in vitro (20 µg/mL) *** | [62] |
| A. edulis | callus | quercetin, kaempferol, isorhamnetin, saponins | n.d. | [42] |
| A. englerianus | hairy roots | cycloartane saponins | n.d. | [9] |
| A. falcatus | hairy roots | cycloartane saponins | n.d. | [9] |
| A. glycypyllos | hairy roots **, callus shoots *** |
cycloastragenol, astragenol, soyasapogenol B epoxycycloartanes |
n.d. T-24 (125 µg/mL); CAL-29 (90 µg/mL); MJ (75 µg/mL); HUT-78 (78 µg/mL) |
[9] [11] [63] |
| aerial parts, wild grown *** | epoxycycloartanes | K-562 (50 µg/mL) ***; HL-60 (40 µg/mL) ***; BV-173 (70 µg/mL) *** | [64] | |
| aerial parts, wild grown *** | epoxycycloartanes | T-24 (168 µg/mL); CAL-29 (105 µg/mL); MJ (126 µg/mL); HUT-78 (87 µg/mL) | [63] | |
| aerial parts, wild grown | 17(R),20(R)-3β,6α,16β-trihydroxycycloartanyl-23-carboxylic acid 16-lactone 3-O-β-D-glucopyranoside | T-24 (66 µg/mL); CAL-29 (52 µg/mL); MJ (52 µg/mL); HUT-78 (18 µg/mL) | [65] | |
| A. hamosus | callus, suspension, hairy roots | saponins, soyasapogenol B, β-sitosterol, astragalin, rutin, isorhamnetin-3-O-glycoside | n.d. | [11][42] |
| aerial parts, wild grown *** | saponins | HL-60 (63 µg/mL); HL-60/Dox (25 µg/mL); SKW-3 (84 µg/mL) | [34] | |
| A. missouriensis | Callus **, suspension, hairy roots | isoquercitrin, quercitrin, rutin, hyperoside, saponins | n.d. | [42][62] |
| A. mongholicus (syn. A. membranaceus) | hairy roots **, shoots ** | astragalosides, β-sitosterol, stigmasterol, campesterol | n.d. | [9][63] |
| A. monspessulanus | aerial parts, wild grown * | flavoalkaloids, acylated flavonoids, flavonoids | n.d. | [24] |
| A. onobrychis | aerial parts, wild grown * | flavonoids, saponins | n.d. | [66] |
| A. oxyglotis | hairy roots | cycloartane saponins | n.d. | [9] |
| A. sesameus | Shoots ** | - | HL-60/Dox (87 µg/mL); SKW-3 (68 µg/mL) | |
| A. spruneri | aerial parts, wild grown * | flavonoids | n.d. | [67] |
| A. sulcatus | hairy roots | cycloartane saponins, sterols, swensonine | n.d. | [9] |
| A. thracicus | callus, suspension | saponins, flavonoids | n.d. | [11] |
| aerial parts, wild grown * | saponins, flavonoids | HT-29 (52 µg/mL); HL-60 (67 µg/mL); HL-60/Dox (53 µg/mL); SKW-3 (83 µg/mL) | [11] | |
| A. vesicarius ssp. carniolicus | callus | flavonoids | HL-60 (8.8 µg/mL) *; HL-60/Dox (11.8 µg/mL) * | [68] |
| callus | 5-hydroxy-7-methoxy-2′, 5′-dihydroxyisoflavone | HL-60 (38.9 µg/mL); HL-60/Dox (35.2 µg/mL) | [68] | |
| 5, 7-dihydroxy-4′-methoxyisoflavone | HL-60 (41.4 µg/mL); HL-60/Dox (42.4 µg/mL) | [68] | ||
| 7-methoxy-5-hydroxy-4′-methoxy-2′-hydroxyisoflavone | HL-60 (64.1 µg/mL); HL-60/Dox (41.8 µg/mL) | [68] | ||
| 8-pregnyl genistein | HL-60 (36.1 µg/mL); HL-60/Dox (36.1 µg/mL) | [68] | ||
| 5,7-dihydroxy-8-pregnyl-4′-methoxy-2′-hydroxyisoflavone | HL-60 (56.3 µg/mL); HL-60/Dox (56.8 µg/mL) | [68] | ||
| sophorophenolone | HL-60 (78.0 µg/mL); HL-60/Dox (63.0 µg/mL) | [68] | ||
| G. superba | seeds | colchicoside, colchicine, 3-O-demethylcolchicine | PANC-1, PANC02 (GS ++ 0.45–0.59 µg/mL) PANC02 (GS2B + 9.49 µg/mL) |
[49] [69] |
| glorioside, colchicodiside A, gloriodiside, colchicodiside B, colchicodiside C, dongduengoside A-C, colchicine, 2-demethilcolchicine, colchicoside and luteolin 7-O-β-D-glucopyranoside | DLA (29 µg #; 21 µg ##) | [58] [59] [70] |
||
| rhizomes | peptides | SW620 (n.d.) | [50] | |
| roots | colchicine | HT-29 (0.12 μg/mL *) | [71] | |
| G. rothschildiana | aerial parts | gloriosamine A-D, colchicine, colchiciline, colchifoline and N-deacetyl-N-formylcolchicine | - | [57] |
4.3. Biotechnology of Gloriosa Species
This entry is adapted from the peer-reviewed paper 10.3390/cimb44090267
References
- Xu, L.; Podlech, D. Astragalus Mongholicus Bunge. Flora China 2010, 10, 338–339, 343.
- Tierra, M.; Tierra, L. Chinese Traditional Herbal Medicine; Lotus Light Pub: Twin Lakes, WI, USA, 1998.
- Shahrajabian, M.H. A Review of Astragalus Species as Foodstuffs, Dietary Supplements, a Traditional Chinese Medicine and A Part of Modern Pharmaceutical Science. Appl. Ecol. Env. Res. 2019, 17, 13371–13382.
- Wang, S.F.; Wang, Q.; Jiao, L.J.; Huang, Y.L.; Garfield, D.; Zhang, J.; Xu, L. Astragalus-Containing Traditional Chinese Medicine, with and without Prescription Based on Syndrome Differentiation, Combined with Chemotherapy for Advanced Non-Small-Cell Lung Cancer: A Systemic Review and Meta-Analysis. Curr. Oncol. 2016, 23, 188–195.
- Auyeung, K.K.; Han, Q.-B.; Ko, J.K. Astragalus Membranaceus: A Review of Its Protection Against Inflammation and Gastrointestinal Cancers. Am. J. Chin. Med. 2016, 44, 1–22.
- Lin, S.; An, X.; Guo, Y.; Gu, J.; Xie, T.; Wu, Q.; Sui, X. Meta-Analysis of Astragalus-Containing Traditional Chinese Medicine Combined With Chemotherapy for Colorectal Cancer: Efficacy and Safety to Tumor Response. Front. Oncol. 2019, 9, 749.
- Cho, W.C. Immunomodulatory and Anti-Tumor Activities of Astragalus Ancient Herb—Modern Miracle; LAP LAMBERT Academic Publishing: Saarbrücken, Germany, 2009.
- Li, R.; Chen, W.; Wang, W.; Tian, W.; Zhang, X. Extraction, Characterization of Astragalus Polysaccharides and Its Immune Modulating Activities in Rats with Gastric Cancer. Carbohydr. Polym. 2009, 78, 738–742.
- Ionkova, I.; Shkondrov, A.; Krasteva, I.; Ionkov, T. Recent Progress in Phytochemistry, Pharmacology and Biotechnology of Astragalus Saponins. Phytochem. Rev. 2014, 13, 343–374.
- Li, X.; Qu, L.; Dong, Y.; Han, L.; Liu, E.; Fang, S.; Zhang, Y.; Wang, T. A Review of Recent Research Progress on the Astragalus Genus. Molecules 2014, 19, 18850–18880.
- Krasteva, I.; Shkondrov, A.; Ionkova, I.; Zdraveva, P. Advances in Phytochemistry, Pharmacology and Biotechnology of Bulgarian Astragalus Species. Phytochem. Rev. 2016, 15, 567–590.
- Zhou, R.; Chen, H.; Chen, J.; Chen, X.; Wen, Y.; Xu, L. Extract from Astragalus Membranaceus Inhibit Breast Cancer Cells Proliferation via PI3K/AKT/MTOR Signaling Pathway. BMC Complement. Altern. Med. 2018, 18, 83.
- Zhang, J.; Liu, L.; Wang, J.; Ren, B.; Zhang, L.; Li, W. Formononetin, an Isoflavone from Astragalus Membranaceus Inhibits Proliferation and Metastasis of Ovarian Cancer Cells. J. Ethnopharmacol. 2018, 221, 91–99.
- Li, W.; Song, K.; Wang, S.; Zhang, C.; Zhuang, M.; Wang, Y.; Liu, T. Anti-Tumor Potential of Astragalus Polysaccharides on Breast Cancer Cell Line Mediated by Macrophage Activation. Mater. Sci. Eng. C 2019, 98, 685–695.
- Li, S.; Sun, Y.; Huang, J.; Wang, B.; Gong, Y.; Fang, Y.; Liu, Y.; Wang, S.; Guo, Y.; Wang, H.; et al. Anti-Tumor Effects and Mechanisms of Astragalus Membranaceus (AM) and Its Specific Immunopotentiation: Status and Prospect. J. Ethnopharmacol. 2020, 258, 112797.
- Shen, L.; Gwak, S.R.; Cui, Z.Y.; Joo, J.C.; Park, S.J. Astragalus-Containing Chinese Herbal Medicine Combined With Chemotherapy for Cervical Cancer: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2021, 12, 587021.
- Le Marchand, L. Cancer Preventive Effects of Flavonoids—A Review. Biomed. Pharmacother. 2002, 56, 296–301.
- Thomasset, S.C.; Berry, D.P.; Garcea, G.; Marczylo, T.; Steward, W.P.; Gescher, A.J. Dietary Polyphenolic Phytochemicals—Promising Cancer Chemopreventive Agents in Humans? A Review of Their Clinical Properties. Int. J. Cancer 2007, 120, 451–458.
- Council of Europe; European Pharmacopoeia Commission; European Directorate for the Quality of Medicines & Healthcare. European Pharmacopoeia; Council of Europe: Strasbourg, France, 2019.
- Zong, A.; Cao, H.; Wang, F. Anticancer Polysaccharides from Natural Resources: A Review of Recent Research. Carbohydr. Polym. 2012, 90, 1395–1410.
- Jia, L.; Lv, D.; Zhang, S.; Wang, Z.; Zhou, B. Astragaloside IV Inhibits the Progression of Non-Small Cell Lung Cancer Through the Akt/GSK-3β/β-Catenin Pathway. Ooncol. Res. 2019, 27, 503–508.
- Gülcemal, D.; Masullo, M.; Bedir, E.; Festa, M.; Karayıldırım, T.; Alankus-Caliskan, O.; Piacente, S. Triterpene Glycosides from Astragalus Angustifolius. Planta Med. 2012, 78, 720–729.
- Bourezzane, S.; Haba, H.; Long, C.; Benkhaled, M. Chemical Composition and Antioxidant Activity of Astragalus Monspessulanus L. Growing in Semiarid Areas of Algeria. J. Serb. Chem. Soc. 2018, 83, 31–38.
- Krasteva, I.; Bratkov, V.; Bucar, F.; Kunert, O.; Kollroser, M.; Kondeva-Burdina, M.; Ionkova, I. Flavoalkaloids and Flavonoids from Astragalus Monspessulanus. J. Nat. Prod. 2015, 78, 2565–2571.
- Elenga, P.; Nikolov, S.; Panova, D. Triterpene Glycosides and Sterols from Astragalus Glycyphyllos L. Pharmazie 1986, 41, 300.
- Elenga, P.; Nikolov, S.; Panova, D. Triterpene Glycosides from Astragalus Glycyphyllos L.—A New Natural Compound of the Overground Parts. Pharmazie 1987, 42, 422–423.
- Linnek, J.; Mitaine-Offer, A.; Miyamoto, T.; Lacaille-Dubois, M. Two Cycloartane-Type Glycosides from the Roots of Astragalus Glycyphyllos. Planta Med. 2008, 74, PB141.
- Shkondrov, A.; Krasteva, I.; Bucar, F.; Kunert, O.; Kondeva-Burdina, M.; Ionkova, I. A New Tetracyclic Saponin from Astragalus Glycyphyllos L. and Its Neuroprotective and HMAO-B Inhibiting Activity. Nat. Prod. Res. 2020, 34, 511–517.
- Isah, T.; Umar, S.; Mujib, A.; Sharma, M.P.; Rajasekharan, P.E.; Zafar, N.; Frukh, A. Secondary Metabolism of Pharmaceuticals in the Plant in Vitro Cultures: Strategies, Approaches, and Limitations to Achieving Higher Yield. Plant Cell Tissue Organ Cult. 2018, 132, 239–265.
- Krasteva, I.; Nikolov, S.; Kaloga, M.; Mayer, G. Triterpenoid Saponins from Astragalus Corniculatus. Z. Für. Nat. B 2006, 61, 1166–1169.
- Krasteva, I.; Nikolov, S.; Kaloga, M.; Mayer, G. A New Saponin Lactone from Astragalus Corniculatus. Nat. Prod. Res. 2007, 21, 941–945.
- Krasteva, I.N.; Toshkova, R.A.; Nikolov, S.D. Protective Effect of Astragalus Corniculatus Saponins against Myeloid Graffi Tumour in Hamsters. Phytother. Res. 2004, 18, 255–257.
- Toshkova, R.A.; Krasteva, I.N.; Wesselinova, D.W.; Nikolov, S.D. Influence of Purified Saponin Mixture from Astragalus Corniculatus Bieb. on Phagocytic Cells in Graffi-Tumor Bearing Hamsters. J. Ethnopharmacol. 2007, 109, 394–399.
- Krasteva, I.; Momekov, G.; Zdraveva, P.; Konstantinov, S.; Nikolov, S. Antiproliferative Effects of a Flavonoid and Saponins from Astragalus Hamosus against Human Tumor Cell Lines. Pharmacogn. Mag. 2008, 4, 269–272.
- Dineva, I.; Krasteva, I.; Berger, M.; Konstantinov, S. In Vitro Antineoplastic Activity of Some Cytoreductive Drugs versus New Compounds of Plant Origin. Intern. J. Curr. Chem. 2010, 1, 281–290.
- Du, M.; Wu, X.J.; Ding, J.; Hu, Z.B.; White, K.N.; Branford-White, C.J. Astragaloside IV and Polysaccharide Production by Hairy Roots of Astragalus Membranaceus in Bioreactors. Biotechnol. Lett. 2003, 25, 1853–1856.
- Wilson, S.A.; Roberts, S.C. Recent Advances towards Development and Commercialization of Plant Cell Culture Processes for the Synthesis of Biomolecules: Development and Commercialization of Plant Cell Culture. Plant Biotechnol. J. 2012, 10, 249–268.
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and Resupply of Pharmacologically Active Plant-Derived Natural Products: A Review. Biotechnol. Adv. 2015, 33, 1582–1614.
- Ionkova, I. Genetic Transformation in Astragalus spp. In Transgenic Medicinal Plants; Biotechnology in Agriculture and Forestry; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1999; pp. 55–72.
- Ionkova, I.; Kartnig, T.; Alfermann, W. Cycloartane Saponin Production in Hairy Root Cultures of Astragalus Mongholicus. Phytochemistry 1997, 45, 1597–1600.
- Giri, A.; Narasu, M.L. Transgenic Hairy Roots. Biotechnol. Adv. 2000, 18, 1–22.
- Ionkova, I.; Alfermann, A. Transformation of Astragalus Species by Agrobacterium Rhizogenes and Their Saponin Production. Planta Med. 1990, 56, 634–635.
- Ionkova, I. Optimization of Flavonoid Production in Cell Cultures of Astragalus Missouriensis Nutt. (Fabaceae). Pharmacogn. Mag. 2009, 5, 92–97.
- Maroyi, A. The Genus Gloriosa (Colchicaceae): Ethnobotany, Phylogeny and Taxonomy; Wageningen University: Wageningen, The Netherlands, 2012.
- Samy, R.P.; Thwin, M.M.; Gopalakrishnakone, P.; Ignacimuthu, S. Ethnobotanical Survey of Folk Plants for the Treatment of Snakebites in Southern Part of Tamilnadu, India. J. Ethnopharmacol. 2008, 115, 302–312.
- Shah, A.; Bharati, K.A.; Ahmad, J.; Sharma, M.P. New Ethnomedicinal Claims from Gujjar and Bakerwals Tribes of Rajouri and Poonch Districts of Jammu and Kashmir, India. J. Ethnopharmacol. 2015, 166, 119–128.
- Maroyi, A. Gloriosa Superba L. (Family Colchicaceae): Remedy or Poison? J. Med. Plants Res. 2011, 5, 6112–6121.
- Ade, R.; Rai, M.K. Review: Current Advances in Gloriosa Superba L. Biodiversitas 2009, 10, 210–214.
- Capistrano, R.; Vangestel, C.; Wouters, A.; Dockx, Y.; Pauwels, P.; Stroobants, S.; Apers, S.; Lardon, F.; Pieters, L.; Staelens, S. Efficacy Screening of Gloriosa Superba Extracts in a Murine Pancreatic Cancer Model Using 18F-FDG PET/CT for Monitoring Treatment Response. Cancer Biother. Radiopharm. 2016, 31, 99–109.
- Budchart, P.; Khamwut, A.; Sinthuvanich, C.; Ratanapo, S.; Poovorawan, Y.; T-Thienprasert, N.P. Partially Purified Gloriosa Superba Peptides Inhibit Colon Cancer Cell Viability by Inducing Apoptosis Through P53 Upregulation. Am. J. Med. Sci. 2017, 354, 423–429.
- Murugesan, A.K.; Pannerselvam, B.; Javee, A.; Rajenderan, M.; Thiyagarajan, D. Facile Green Synthesis and Characterization of Gloriosa Superba L. Tuber Extract-Capped Silver Nanoparticles (GST-AgNPs) and Its Potential Antibacterial and Anticancer Activities against A549 Human Cancer Cells. Environ. Nanotechnol. Monit. Manag. 2021, 15, 100460.
- Gelmi, M.L.; Mottadelli, S.; Pocar, D.; Riva, A.; Bombardelli, E.; De Vincenzo, R.; Scambia, G. N-Deacetyl-N-Aminoacylthiocolchicine Derivatives: Synthesis and Biological Evaluation on MDR-Positive and MDR-Negative Human Cancer Cell Lines. J. Med. Chem. 1999, 42, 5272–5276.
- Reuter, S.; Prasad, S.; Phromnoi, K.; Ravindran, J.; Sung, B.; Yadav, V.R.; Kannappan, R.; Chaturvedi, M.M.; Aggarwal, B.B. Thiocolchicoside Exhibits Anticancer Effects through Downregulation of NF-ΚB Pathway and Its Regulated Gene Products Linked to Inflammation and Cancer. Cancer Prev. Res. 2010, 3, 1462–1472.
- Shi, Q.; Chen, K.; Chen, X.; Brossi, A.; Verdier-Pinard, P.; Hamel, E.; McPhail, A.T.; Tropsha, A.; Lee, K.-H. Antitumor Agents. 183. Syntheses, Conformational Analyses, and Antitubulin Activity of Allothiocolchicinoids. J. Org. Chem. 1998, 63, 4018–4025.
- Balkrishna, A.; Das, S.K.; Pokhrel, S.; Joshi, A.; Verma, S.; Sharma, V.K.; Sharma, V.; Sharma, N.; Joshi, C.S. Colchicine: Isolation, LC–MS QTof Screening, and Anticancer Activity Study of Gloriosa Superba Seeds. Molecules 2019, 24, 2772.
- Lin, Z.-Y.; Kuo, C.-H.; Wu, D.-C.; Chuang, W.-L. Anticancer Effects of Clinically Acceptable Colchicine Concentrations on Human Gastric Cancer Cell Lines. Kaohsiung J. Med. Sci. 2016, 32, 68–73.
- Kitajima, M.; Tanaka, A.; Kogure, N.; Takayama, H. Four New Colchicinoids, Gloriosamines A–D, from Gloriosa Rothschildiana. Tetrahedron Lett. 2008, 49, 257–260.
- Zarev, Y.; Foubert, K.; Ionkova, I.; Apers, S.; Pieters, L. Isolation and Structure Elucidation of Glucosylated Colchicinoids from the Seeds of Gloriosa Superba by LC-DAD-SPE-NMR. J. Nat. Prod. 2017, 80, 1187–1191.
- Sahakitpichan, P.; Chimnoi, N.; Namsa-aid, A.; Panyadee, A.; Ruchirawat, S.; Kanchanapoom, T. Colchicinoid Glucosides from Seedless Pods of Thai Origin Gloriosa Superba. Phytochem. Lett. 2016, 16, 299–302.
- Riva, S.; Sennino, B.; Zambianchi, F.; Danieli, B.; Panza, L. Effect of Organic Cosolvents on the Stability and Activity of the β-1,4-Galactosyltransferase from Bovine Colostrum. Carbohydr. Res. 1997, 305, 525–531.
- Pišvejcová, A.; Rossi, C.; Hušáková, L.; Křen, V.; Riva, S.; Monti, D. β-1,4-Galactosyltransferase-Catalyzed Glycosylation of Sugar Derivatives: Modulation of the Enzyme Activity by α-Lactalbumin, Immobilization and Solvent Tolerance. J. Mol. Catal. B: Enzym. 2006, 39, 98–104.
- Georgieva, A.; Popov, G.; Shkondrov, A.; Toshkova, R.; Krasteva, I.; Kondeva-Burdina, M.; Manov, V. Antiproliferative and Antitumour Activity of Saponins from Astragalus Glycyphyllos on Myeloid Graffi Tumour. J. Ethnopharmacol. 2021, 267, 113519.
- Shkondrov, A.; Krasteva, I.; Ionkova, I.; Popova, P.; Zarev, Y.; Mihaylova, R.; Konstantinov, S. Production of Saponins from in Vitro Cultures of Astragalus Glycyphyllos and Their Antineoplastic Activity. Biotechnol. Biotechnol. Equip. 2019, 33, 1413–1418.
- Shkondrov, A. Phytochemical Investigation of Species from Genus Astragalus L. (Fabaceae); Medical University of Sofia: Sofia, Bulgaria, 2017.
- Mihaylova, R.; Shkondrov, A.; Aluani, D.; Ionkova, I.; Tzankova, V.; Krasteva, I. In Vitro Antitumour and Immunomodulating Activity of Saponins from Astragalus Glycyphyllos. Biotechnol. Biotechnol. Equip. 2021, 35, 1948–1955.
- Benbassat, N.; Nikolov, S. Flavonoids from Astragalus Onobrychis. Planta Med. 1995, 61, 100.
- Kondeva-Burdina, M.; Shkondrov, A.; Simeonova, R.; Vitcheva, V.; Krasteva, I.; Ionkova, I. In Vitro/in Vivo Antioxidant and Hepatoprotective Potential of Defatted Extract and Flavonoids Isolated from Astragalus Spruneri Boiss. (Fabaceae). Food Chem. Toxicol. 2018, 111, 631–640.
- Popova, P.; Zarev, Y.; Mihaylova, R.; Momekov, G.; Ionkova, I. Antiproliferative Activity of Extract from in Vitro Callus Cultures of Astragalus Vesicarius Ssp. Carniolicus (A. Kern.) Chater. Pharmacia 2021, 68, 217–221.
- Capistrano, I.R.; Vangestel, C.; Vanpachtenbeke, H.; Fransen, E.; Staelens, S.; Apers, S.; Pieters, L. Coadministration of a Gloriosa Superba Extract Improves the in Vivo Antitumoural Activity of Gemcitabine in a Murine Pancreatic Tumour Model. Phytomedicine 2016, 23, 1434–1440.
- Saradhadevi, M.; Gnanadesigan, M.; Kapildev, G.; Vasanth, D. Dataset on Antitumor Properties of Silver Nanoparticles from Gloriosa Superba (L.) Seed on Dalton Lymphoma Ascites (DLA) Tumor: Facile and Biocompatible Approach. Data Brief 2017, 14, 524–530.
- Manosroi, A.; Akazawa, H.; Akihisa, T.; Jantrawut, P.; Kitdamrongtham, W.; Manosroi, W.; Manosroi, J. In Vitro Anti-Proliferative Activity on Colon Cancer Cell Line (HT-29) of Thai Medicinal Plants Selected from Thai/Lanna Medicinal Plant Recipe Database “MANOSROI III. ” J. Ethnopharmacol. 2015, 161, 11–17.
- Mamatha, H.; Farooqi, A.A.; Joshi, S.S.; Prasad, T.G. Pollen studies in gloriosa superba linn. Acta Hortic. 1993, 331, 371–376.
- Samarajeewa, P.K. Clonal Propagation of Gloriosa Superba L. Indian J. Exp. Biol. 1993, 31, 719–720.
- Sivakumar, G.; Krishnamurthy, K.; Rajendran, T.D. Embryoidogenesis and Plant Regeneration from Leaf Tissue of Gloriosa Superba. Planta Med 2003, 69, 479–481.
- Jana, S.; Shekhawat, G.S. Critical Review on Medicinally Potent Plant Species: Gloriosa Superba. Fitoterapia 2011, 82, 293–301.
- Arumugam, A.; Gopinath, K. In Vitro Micropropagation Using Corm Bud Explants: An Endangered Medicinal Plant of Gloriosa Superba L. Asian J. Biotechnol. 2012, 4, 120–128.
- Sivakumar, S.; Siva, G.; Sathish, S.; Prem Kumar, G.; Vigneswaran, M.; Vinoth, S.; Kumar, T.S.; Sathishkumar, R.; Jayabalan, N. Influence of Exogenous Polyamines and Plant Growth Regulators on High Frequency in Vitro Mass Propagation of Gloriosa Superba L. and Its Colchicine Content. Biocatal. Agric. Biotechnol. 2019, 18, 101030.
- Mahendran, D.; Kavi Kishor, P.B.; Sreeramanan, S.; Venkatachalam, P. Enhanced Biosynthesis of Colchicine and Thiocolchicoside Contents in Cell Suspension Cultures of Gloriosa Superba L. Exposed to Ethylene Inhibitor and Elicitors. Ind. Crops Prod. 2018, 120, 123–130.
