Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Engineering, Petroleum
The mineral components of the reservoir system have a significant impact on the formation of hydrocarbon deposits. The composition of minerals changes with the migration of hydrocarbons during the further formation and destruction of the deposit.
- heavy oil
- shale oil
- rock minerals
- catalyst
1. Minerals at the Stage of Formation
The mineral components of the reservoir system have a significant impact on the formation of hydrocarbon deposits. The composition of minerals changes with the migration of hydrocarbons during the further formation and destruction of the deposit. Authigenic mineral formation in the zones of hydrocarbon flows and in areas of their concentration is associated with a change in the physicochemical conditions at the contact of mobile hydrocarbons and water and occurs due to the dissolution of gases in water, variations in pH and Eh of the medium, the activity of anaerobic bacteria, etc. [10]. Epigenetic mineral formation in the areas of occurrence of oil and gas deposits has its own unique features and patterns.
Terrestrial heat from the bowels ensures the transformation of organic matter in clayey rocks. On the way to hydrocarbon flow, a “hydrocarbon footprint” is formed. In the zone of this “trace”, glauconite forms in the presence of hydrocarbons, in addition to the appearance of pyrolusite, pyrochlore, often uranium pyrochlore, plumbopirochlore and strontium pyrochlore; meanwhile, ferruginous minerals become converted. In zones of intense heat and hydrocarbon flow, new formations appear in the form of phenocrysts (impregnations), intergrowths of crystals.
At subsequent stages, biotite, ilmenite and calcite are formed in the hydrocarbon flow zone. In the central zone of the “hydrocarbon footprint”, ferruginous minerals hematite, pyrite and magnesioferrite are often found, and, in the far outer zones of the flow, pentahydrocalcite is sometimes formed.
In the zone where the concentration of hydrocarbons coming from the depths and from the oil source strata occurs (as a result of their compaction or dehydration of silty-clay deposits), a hydrocarbon deposit is slowly formed. In areas where hydrocarbons are collected, they are concentrated and converted. The concentration zone is characterized by lower temperatures (50–60 °C). Secondary processes include primarily the precipitation of clay minerals from fluid solution. Hydromicas and kaolinite precipitate. Kaolinite is probably formed as a result of the hydrolysis of CO2-containing silicate rocks with acidic solutions. Dickite, hydromica, illite and saussurite are found here. Kaolinite is the most representative in samples. Kaolinite is often fine-grained, granular. New formations of clay minerals are typical for peripheral zones of hydrocarbon deposits [11].
The formation of kaolinite is associated with periods of intensive growth of structures and mass flows of hydrocarbons up the section [12,13,14], and may be due, in the active tectonic phase, to the appearance of aggressive carbon dioxide solutions coming from the basement and lower horizons of the sedimentary cover. Kaolin formation occurs mainly due to the dissolution of detrital silicates and aluminosilicates of sandy–silty rocks. The formation of kaolinite is favored by high temperatures, pressure and the presence of protons in the solution (low pH values), while a carbonic environment, on the contrary, shifts the process in the opposite direction. The formation of kaolinite is known to be associated with the hydrolysis of silicate rocks:
2K[AlSi3Os] + 7H2O → Al2[Si2O5](OH)4 + 4H2SiO3 + 2КОН.
Clay minerals undergo hydrolysis under conditions of lowering the pH of formation waters, due to the release of CO2 and organic acids and during the decomposition of organic matter. According to previously published works [14,15,16], authigenic kaolinite is formed in a well-permeable medium mainly due to clay cement, consisting of hydromicas, hydromica–montmorillonite formations and chlorite. A more perfect structure, according to Sakhibgareev et al. [17], is possessed by kaolinite from aquifers, which is explained by the preservative effect of oil on authigenic mineral formation. Perfect aggregates of kaolinite are sometimes found in the contours of deposits [12]. Kaolinite formation is less common in oil-bearing reservoirs.
The maximum amount of kaolinite is observed in gas-saturated rocks. Minerals of the kaolinite and chlorite group are the most typical for unproductive sections. Hydromicas and montmorillonite formations are observed within oil deposits, often in the form of basal-pore cement, with predominantly random orientation of clay particles [12]. Organic matter adsorbed on clay material increases the stability of clay minerals [15,18]. When organic matter interacts with clay minerals, the fluid-resistant properties of screens increase significantly [12].
The post-sedimentary formation of chlorite minerals is facilitated by the development of anaerobic bacterial activity, accompanied by iron reduction and an increase in the pH of pore solutions, the increased content of which is often accompanied by a deterioration in the reservoir properties of rocks. Minerals of the hydromica group, including illite, are rare in oil-saturated reservoirs. In pelitomorphic rocks, the process of illitization is more likely. Illite formation is assumed to be more alkaline than kaolinite formation [19]. Among the epigenetic carbonates, calcite is the most common; siderite, dolomite and ankerite are common [20].
In the sandy–silty reservoirs of Western Siberia, the most common cements are clayey and carbonate. In zones of secondary carbonatization, often associated with the migration of hydrocarbons, several generations of calcite stand out. It is believed that the bulk of calcite is formed at the stage of early epigenesis, before oil enters the trap. Calcite of late generations is formed in connection with the processes of sulfate reduction near hydrocarbon deposits [21]. The structures of diagenetic carbonates are most often pelitomorphic and granular with nodules. Among carbonates, there are calcite, manganocalcite, rhodochrosite, dolomite, siderite, ankerite, often manganosiderite, manganese calcite, magnesium-containing siderite, ferruginous magnesite and the Mg content in these carbonates in oil-saturated sandstones increases. In the oil-bearing contours, predominantly calcium-iron magnesite with a high content of Mg crystallizes [14].
For some deposits of the Ural-Volga region and Western Siberia, a multi-stage formation of deposits has been proven [22,23].
In a productive oil-saturated reservoir, where the preservative effect of hydrocarbons has a significant impact on the course of secondary processes, the leading process is silicification, as a result of which productive sandstones often contain up to 5–10% regeneration quartz cement.
As a result of the interaction of hydrocarbons with host rocks, subvertical zonal-ring-shaped geochemical, geophysical and biogeochemical fields are established above oil and gas deposits. Subvertical annular zones of secondary alteration of rocks arise as a result of the processes of chemical and biochemical destruction of hydrocarbons dissipating from deposits and the interaction of hydrocarbons with mineral components of rocks [19]. The migration of hydrocarbons causes a change in the parameters of the mineral environment in various geochemical zones.
In the hydrodynamic systems of the oil and gas section, in addition to temperature and pressure, the processes of secondary mineral formation are significantly affected by the mineralization of solutions and their pH and Eh. First of all, this affects the pH and Eh values of water-mineral media [24].
Carbon dioxide and hydrogen sulfide are of great importance in the successive occurrence and spatial variability of the concentration fields of various elements in the diffusion flow of hydrocarbons. They have a significant effect on the Eh and pH values of rocks and are involved in the processes of leaching and authigenesis. Carbon dioxide is formed during the oxidation of hydrocarbons, is contained in a dissolved state in waters, is present in the mineral environment, enhances the removal of such cations as Fe2+, Mn2+, Cu2+, Co2+ and alkaline earth metals: Mg2+, Ca2+, Sr2+ and Ba2+. Newly formed minerals are deposited in sediments located in the contour of hydrocarbon anomalies. Ions that have a greater affinity for sulfur: Fe2+, Cu2+, Co2+ and Mi2+ form sulfides, and those that have a greater affinity for oxygen: Mg2+, Ca2+, Sr2+, Ba2+ form carbonates.
Numerous studies have noted that, in overproductive deposits in the form of halos that outline hydrocarbon deposits, elevated concentrations of elements are established: Mi, Fe, Ti, Cu, Pb, Sn, V, Ni, Co, Si, Al, Ca, Ba, Sr, etc. Paragenetic associations of chemical elements, the nature of their relationship with neoformations, the sequence of deposition and selective confinement to different parts of productive sections are revealed. The multivalent and chalcophile elements Fe, Mi, Pb and Zn tend to precipitate in reducing media together with siderite and sulfides. Complexes of compounds with organic components form Cu, V, Ni and Co. In the presence of CO2, Ca, Ba and Sr, carbonates are intensively dissolved, turning into soluble hydrocarbons.
In other words, rocks containing hydrocarbons include a wide variety of compounds that actively influence the transformation of hydrocarbons at the stage of reservoir formation. During the development of deposits by thermal methods, many rock components can actively participate in chemical processes that provide an increase in the mobility of heavy oil or the destruction of kerogen in shale deposits.
2. At the Production Stage as Catalysts
The course of catalytic processes during thermal action on oil deposits has long been known. The term aquathermolysis itself provides for the important role of the minerals of the reservoir rock [25]. Aquathermolysis is considered as one of the methods of in situ upgrading of high-viscosity crude oil, along with in situ combustion, low-temperature oxidation and other methods, which are common for formation heating and injection of reagents and/or catalysts [25,26,27,28,29].
The catalytic participation of rock-forming minerals was studied on an oil sample from the Ashalcha field [30]. Physical modeling of the catalytic aquathermolysis of heavy oil with different chemical compositions has been performed. The processes were carried out at a temperature of 300 °C in the presence of kaolin (montmorillonite content 44%) and catalysts based on transition metal carboxylates (Fe, Co, Cu). The process medium was a mixture of carbon dioxide and water vapor. Distinctive features of hydrothermal–catalytic conversion of various types of oil were evaluated by fractional, structural-group, microelement composition and change in the H:C ratio. These variations are due to the initial properties of crude oil and the degree of activation of C-C, C-N, C-O and C-S bond destruction reactions, leading to different levels of increase in the content of saturated fractions and a decrease in the content of resins and asphaltenes. The thermal analysis method was used to evaluate the oil content on the rock before and after the experiments. The high molecular weight components of naphthenic aromatic oil type B2 showed a greater adsorption capacity for rocks compared to oil type A1. Therefore, the adsorption of catalyst components on rocks is also greater [31], as shown by the effect of montmorillonite, kaolinite, quartz, calcite, etc., on the change in the molecular weight of asphaltenes in heavy oils from various fields during aquathermolysis [32].
The process of aquathermolysis has been studied for the kerogen-containing Domanik rock. Fragments of kerogen fall into the fraction of aromatic hydrocarbons. Thermal action changes the ratio of ortho-, para- and meta-isomers of alkyltoluenes. The content of heavy aromatic hydrocarbons increases with increasing temperature, and the ratio of kerogen macroelements changes under the influence of thermal exposure, which is an indicator of the process of maturation of organic matter [33].
In situ combustion is generating a considerable interest in terms of enhancing heavy oil recovery. It is commonly known that promoting the combustion front is based on the formation and oxidation of coke. In their analysis of heavy oil combustion and pyrolysis from China in the presence of montmorillonite, a major type of clay, by means of thermogravimetric analyzer (TGA) and a fixed-bed reactor [34], the authors found that the characteristic temperatures of thermal conversion decreased with montmorillonite in the oxidizing atmosphere while remaining unaffected in the pyrolysis atmosphere. Moreover, the authors reported that the fuel deposition increased in both atmospheres because of montmorillonite’s strong adsorption. It has been found as well that the presence of montmorillonite obviously promoted the progress of coke formation and increased coke yield in the oxidizing atmosphere. In addition, the content of O was found more contrarily to the contents of C and H, which were found decreased in coke. The obtained results pointed towards the idea that the oxidation activity of coke was improved, while trailing occurred due to the blocking in the pore structures of the montmorillonite skeleton. However, in the pyrolysis atmosphere, the presence of montmorillonite did not influence the coke formation temperature but increased coke yield. What is more, the content of C was found increased, and the contents of H and O were found decreased in coke. On the other hand, the coke oxidation activity was reduced with a more serious trailing phenomenon. In fact, montmorillonite significantly affected coke formation through its strong adsorption to polar components in heavy oil and obvious catalysis on dehydrogenation as an acid catalyst in the pyrolysis atmosphere. It has enhanced the oxygenation due to its large surface area and catalyzed polycondensation in the air atmosphere.
In other works [35], authors have studied the influence of the rock matrix on oxidation kinetics by means of kinetic experiments for a Colombian heavy crude using various porous media, such as synthetic sand, drilling cuttings, reservoir core and outcrop. Authors have mainly aimed to evaluate the kinetic behavior of packing with porous media other than the original rock matrix to determine the feasibility of using them as a replacement for the original reservoir rock while maintaining the representativeness of the results. It was supposed that the importance resides in the fact that the observed kinetics can be used to assess the technical feasibility for the implementation of in situ combustion projects. The experimental design included kinetic cell tests varying the porous medium and keeping the same fluid and the same operating conditions. The obtained results showed the possibility of creating reliable laboratory studies for the air injection process using packing other than the reservoir core as a more convenient alternative [35].
Heavy oil extraction resolves economic and environmental challenges as a result of low prices and costly energy usage for their exploitation and transport. However, enhancing heavy oil recovery requires novel and well-engineered technologies. In situ combustion is a technique that increases heat generation by means of air injection assisted with nano catalysts. The application of such technology requires performance assessment and a kinetic database for each application and each type of reservoir with specific rock–oil in place characteristics. In their work [36], Durman et al. have studied the hydrocracking kinetics of dolomite core pack at a temperature range of 335 °C to 365 °C in a period of 12 h–72 h under constant pressure of 10 MPa, which are the typical ISUT’s conditions for carbonate reservoirs. The obtained results showed similar results with the previous kinetic model. The scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDX) techniques have showed that nanocatalysts of 13 nm to 1220 nm with two types, spherical and agglomerated-shaped, have deposited on the rock. The X-ray fluorescence spectroscopy (XRF) and the ISUT’s kinetic modeling studies have proposed the convenience of using the same model for the reservoir’s studies. This includes the effects of different conditions, such as temperature, pressure or residence time, used during ISUT’s process application.
The roles of various clay and non-clay minerals present in the steam distillation process in reservoir formation have been studied by Tavakkoli et al. [37]. Two heavy oil samples mixed with water and crushed rock mixed with different clay minerals were kept under steam pressure at 150, 200 and 250 °C in a batch autoclave reactor for 40 h. The obtained results were compared with respect to the changes in the density, viscosity and chemical composition of the remaining heavy oil.
Nowadays, improving the efficiency of enhanced oil recovery methods presents a difficult task. Mukhammatdinov et al. have attempted to improve oil quality and reduce resins and asphaltenes content in heavy oil by applying hydrothermal treatment to a sample of Aschalcha oil. The authors simulated the process of heavy oil hydrothermal processing in the presence of iron tallates under reservoir conditions (200 °C, 30 bar). The composition and physicochemical characteristics of the obtained substances were characterized by elemental and SARA analysis, MALDI, GC–MS and FT-IR. The authors have extracted rocks and analyzed them by means of XRD and DSA (drop shape analyzer). The obtained data from this study showed that the introduction of a catalyst in combination with a hydrogen donor reduces the content of resins by 22 wt.% and increases saturated hydrocarbons by 27 wt.%. What is more, the destructive hydrogenation has been found to result in a decrease in the sulfur content in the upgrading products. The authors believe that this is crucial for the oil reservoirs of Tatarstan Republic because of their high sulfur content. In addition, DSA data showed that the hydrophilicity of the rock surface increases due to inhibition of the coke formation after the introduction of the catalytic complex, which means that the oil recovery factor can be increased as a result of reservoir rocks’ oil-wetting properties alteration [38].
In another work, Sitnov et al. studied the process of heavy oil aquathermolysis in the presence of dolomite and calcite. It has been shown that calcite reduces the amount of asphaltenes and resins in addition to their condensation and aromaticity as a result of C-S-C bonds cleavage, as shown by SARA analysis and IR spectroscopy. Moreover, the gas chromatography analysis in this study pointed towards the increment of hydrocarbon content with homologous series of С19-С30 as a result of heavy oil thermal conversion. However, the presence of dolomite leads to the isomerization of new formed alkanes (Figure 1). The authors have shown as well the high maturity of organic matters in thermally conversed heavy oil from a model system with dolomite by geochemical coefficients calculation [39].
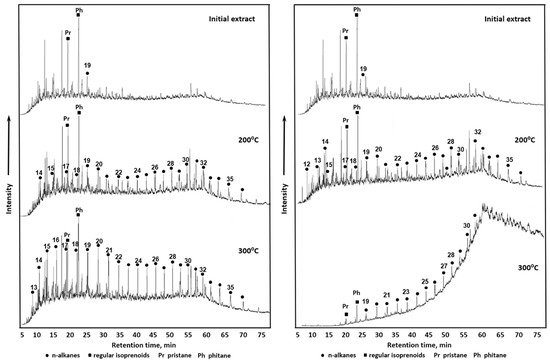
Figure 1. GC–MS chromatograms (TIC = total ion chromatography) of saturated hydrocarbon fraction of initial oil and after thermal treatment in the presence of calcite (CaCO3)—left and in the presence of calcite (CaCO3)—right.
This entry is adapted from the peer-reviewed paper 10.3390/en15166047
This entry is offline, you can click here to edit this entry!
