Cancer represents one of the greatest public health challenges. One of the most cancer-driving events embodies the dysregulation of both the canonical and the non-canonical Wnt/β-catenin pathway. The impact of the Wnt/β-catenin pathway has been widely reviewed in colorectal, breast, and ovarian cancers.
Genetic and epigenetic alterations are commonly detected in colorectal cancers (CRCs). As a matter of fact, 70% of CRCs are connoted by the APC mutations and almost all patients display an overactive Wnt/β-catenin pathway also mediated by oncogenic miRNAs. Therefore, miRNAs have been proposed as anti-cancer and/or diagnostic/prognostic tools. Among cancers, breast cancer (BC) is one of the most expensive health care costs with a high rate of diagnosis and deaths per year. The Wnt/β-catenin cascade and in particular the β-catenin content has been correlated with a dismal prognosis, high tumour grade, and metastasis formation. In addition in triple-negative breast cancer (TNBC) both the canonical and the non-canonical Wnt/β-catenin pathways have been reported as drivers of cancer dissemination, aggressiveness, early age of onset, and poor outcome. To add further complexity, the Wnt5a ligand was found to display both anti-tumour and tumour promoting properties depending on the tumour microenvironment (TME), the activation of specific signalling pathways, and the receptor availability in BC.
Likewise, an abnormal Wnt/β-catenin cascade has been shown to strongly contribute to ovarian cancer (OC) growth, stemness, and drug resistance.
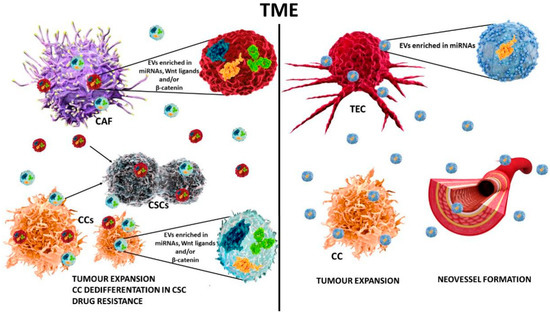
In the last decades, particular attention has been dedicated to investigate the role of extracellular vesicles (EVs) released in the TME in cancer growth and progression. EVs are heterogeneous small membrane-bound carriers with a complex cargo contributing to cell-to-cell communication, tumour growth, invasion, and chemoresistance. Since EVs can be detected in the majority of biological fluids and in the TME, EVs have been proposed as diagnostic and/or prognostic tools, as well as useful therapeutic options. Indeed, EVs engineered with specific anti-tumour molecules or loaded with conventional anti-tumour drugs have been proposed as novel anti-cancer options.
Based on these notions, in the last decades, Wnt/β catenin targeting approaches have been explored to hinder tumour expansion. However so far, the most relevant limitation relies on the crucial role played by the Wnt/β catenin cascade in tissue homeostasis. Therefore, to develop targeting approaches the identification of the specific EV cargo driving tumour progression and the mechanisms accounting for the unbalanced Wnt/β catenin pathway in cancer should be considered as the most challenging issues.
- Wnt/β-catenin dependent pathway
- Wnt/β-catenin independent pathway
- colorectal cancer
- breast cancer
- ovarian cancer
- extracellular vesicles
Cancer is one of the greatest public health challenges. According to the World Health Organization (WHO) 9.6 million cancer deaths have been reported in 2018. The most common cancers include lung, breast, colorectal, prostate, skin cancer (non-melanoma) and stomach. The unbalance of physiological signalling pathways due to the acquisition of mutations in tumour cells is considered the most common cancer driver. The Wingless-related integration site (Wnt)/β-catenin pathway is crucial for tissue development and homeostasis in all animal species and its dysregulation is one of the most relevant events linked to cancer development and dissemination. The canonical and the non-canonical Wnt/β-catenin pathways are known to control both physiological and pathological processes including cancer. Herein the impact of the Wnt/β-catenin cascade in driving cancers from different origin has been examined. Finally, based on the impact of Extracellular Vesicles (EVs) on tumour growth, invasion and chemoresistance, and their role as tumour diagnostic and prognostic tools, an overview of the current knowledge linking EVs to the Wnt/β-catenin pathway is also discussed.
1. Introduction
The human wingless-related integration site (Wnt) genes encode 19 evolutionarily conserved glycoproteins with 22-24 Cys residues. In the endoplasmic reticulum (ER), the Wnt ligands are post-translationally acetylated by porcupine, a membrane associated O-acyl transferase. Acetylation leads to palmitoylation, which is required for the release and binding of Wnt to the frizzled (FZD) receptors. This, in turn, drives the biological response [1].
The Wnt signalling pathway regulates crucial cellular processes including cell fate determination, organogenesis during embryonic development, normal adult homeostasis, motility, polarity and stem cell renewal [2]. Moreover, its contribution in cancer has been extensively investigated [3].
The Wnt pathway has been widely studied and reviewed, and a general understanding of the transduction cascade has been clarified. The Wnt cascade has been subdivided into different branches due to its complexity [4,5]. They include the canonical Wnt/β-catenin (Wnt/β-catenin dependent pathway) and the non-canonical Wnt/β-catenin pathway (β-catenin-independent pathway). The latter was further allocated into two additional branches, the planar cell polarity (PCP) and the Wnt/calcium pathways [2]. Both of them contribute to cancer development and dissemination.
2. Wnt Canonical Pathway: β-Catenin Dependent
The canonical pathway turns around the β-catenin intracellular level (Figure 1). In the absence of Wnt proteins, the β-catenin “destruction complex” keeps low β-catenin in the cell. The “destruction complex” mainly consists of two kinases: casein kinase 1α (CK1α), glycogen synthase kinase 3 β (GSK-3β) and two scaffolds: axis inhibition (Axin), and adenomatous polyposis coli (APC). Firstly, β-catenin undergoes phosphorylation by CK1α at serine 45 (Ser45), Ser33, Ser37 and threonine 41 (Thr41) by GSK-3β. Then, the E3 ubiquitin ligase, denoted as β-transducin repeat-containing protein (βTrCP), marks β-catenin ubiquitination and degradation [1]. This prevents β-catenin nuclear translocation while allowing histone deacetylation and chromatin compaction by the Groucho repressor, translating into the inhibition of gene transcription [6] (Figure 1a).
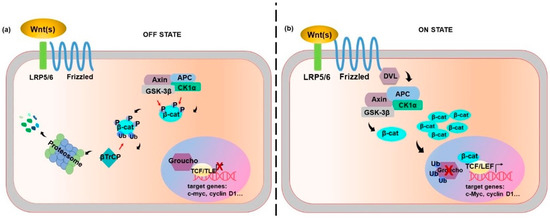
The activation of the canonical Wnt signal requires both the FZD family receptors and the low-density-lipoprotein-related protein 5/6 (LRP5/LRP6) co-receptors, phosphorylation of which is essential for receptor activation. Wnt binding to its receptor results in dishevelled (DVL) phosphorylation, leading to Axin de-phosphorylation and decline of its cytoplasmic content [7]. Thereby, β-catenin can be released from the “destruction complex”, and its degradation prevented while stabilization is allowed. Accumulation of β-catenin turns into its nuclear translocation [7].
Although several nuclear β-catenin binding partners have been involved in the control of gene transcription, the most relevant β-catenin partners are the members of the T-cell factor/lymphoid enhancer factor (TCF/LEF) family of transcription factors [7]. This complex binds to the promoter region of target genes and regulates their transcription.
Once in the nucleus, the engagement of β-catenin transiently converts the TCF/LEF into transcriptional activators, which displace Groucho and induce chromatin remodelling and transcriptional activity (Figure 1b).
A number of genes are targeted by Wnt-β-catenin. Among them, genes involved in positive- and negative-feedback regulation, cell-cycle progression, and stem cell homeostasis are the most commonly included genes.
3. Extracellular Vesicles and the Wnt Pathway
| EV Cargo | EV Source | Target Cells | Related Cancers | Expression Level | Pathway Interaction | Impact on Tumour Cells | Ref. |
|---|---|---|---|---|---|---|---|
| Mutant β-catenin in EVs | LIM1215 | RKO | CRC | Upregulated | β-catenin | migration, metastasis tumour growth |
[129] |
| 14-3-3ζ in EVs | HEK293T | COS-7, SW480 |
CRC | Upregulated | β-catenin GSK-3β DVL2 |
survival migration |
[130] |
| Wnt ligands in EVs | CAFs | CRC | CRC | Upregulated | β-catenin | dedifferentiation drug resistance colony formation |
[131,132] |
| β-catenin in EVs | milk | HCC | HCC | Silenced | β-catenin | proliferation tumour growth |
[133,134] |
| DKK-1 in EVs | MM | MM | MM | Upregulated | β-catenin | osteoclast activity osteoblast differentiation |
[135] |
| EVs | OSCC | OSCC | OSCC | Upregulated | β-catenin | metastasis stemness chemoresistance |
[136] |
| Wnt5b in EVs | Caco-2 and PANC-1 |
A549 | Lung cancer |
Upregulated | β-catenin dependent and independent pathways | proliferation migration |
[137] |
| EVs | CAFs | BC | BC | Upregulated | Wnt-PCP | cell growth and motility | [138] |
4. Conclusions
Cell-to cell communication is part of the evolutional processes. Wnt ligands are essential for homeostasis and, in the last 30 years, genetic, biochemical, and molecular investigations have uncovered several Wnt signalling components [2,3]. Driving interest on this topic mainly relies on dysregulation of the Wnt/β-catenin signalling and cancer development/progression [3]. Moreover, Wnt/β-catenin cascade seems to contribute to the TME shape, which plays a crucial role in the control of tumour progression and immune regulation. Many different Wnt proteins have been described, and, among them, Wnt5a plays a critical role, taking part in both the canonical and the non-canonical Wnt/β-catenin pathway [104,105].
The identification of specific tools able to interfere with the Wnt/β-catenin cascade has been a hotspot for many years. This is particularly true for CRC, in which almost 70% of CRC patients display APC mutations [15]. Apart from CRC, the Wnt/β-catenin pathway is gaining attention in several malignancies, such as breast, ovarian, melanoma, prostate and paediatric osteosarcoma [53,124,125]. In this regard, BC and in particular TNBC are featured by the abnormal activation of both the canonical and non-canonical Wnt/β-catenin pathway [113,114]. Likewise, a hyper-active Wnt/β-catenin cascade has been shown to play a crucial role in the progression, stemness, and drug resistance in OC [70,119]. Several miRNAs have been identified to modulate this cascade and thereby widely studied as screening markers or targets in different tumour settings [142].
In the TME, intercellular communication has been recently reported as mediated by the transfer of EV molecular cargo and revised in [143]. Their cargo also includes a number of Wnt components. Of note, wild-type and mutant β-catenin, able to promote survival and proliferation of recipient cells and, in several instances, dedifferentiation towards a CSC phenotype, have been detected in EVs (Figure 3a). Moreover, their role in mediating drug resistance has been reported. Furthermore, since EVs are released within the TME, their contribution in cancer growth and progression has been extensively investigated [144]. EV shedding, blockade, or engineering have been proposed as innovative anti-tumour instruments for fine-tuning the Wnt/β catenin pathway [142,145].
In recent decades, several efforts have been directed to the development of Wnt/β catenin targeting approaches in order to interfere with tumour progression. However, these efforts have been limited by the crucial role of the Wnt/β catenin pathway in preserving tissue homeostasis. Therefore, future energies should be directed to clearly dissect the mechanisms driving the unbalanced Wnt/β catenin pathway in cancer, and the EV mechanism of action should be considered amid them. Should they be identified, targeting approaches would become a suitable anti-cancer option
This entry is adapted from the peer-reviewed paper 10.3390/ijms21207697
