Meat may contain natural, spoilage, and pathogenic microorganisms based on the origin and characteristics of its dietary matrix. Several decontamination substances are used during or after meat processing, which include chlorine, organic acids, inorganic phosphates, benzoates, propionates, bacteriocins, or oxidizers. Unfortunately, traditional decontamination methods are often problematic because of their adverse impact on the quality of the raw carcass or processed meat. The extended shelf-life of foods is a response to the pandemic trend, whereby consumers are more likely to choose durable products that can be stored for a longer period between visits to food stores. This includes changing purchasing habits from “just in time” products “for now” to “just in case” products, a trend that will not fade away with the end of the pandemic.
- clean label foods
- ozone
- cold plasma
1. Introduction
2. Nonthermal Decontamination Technologies
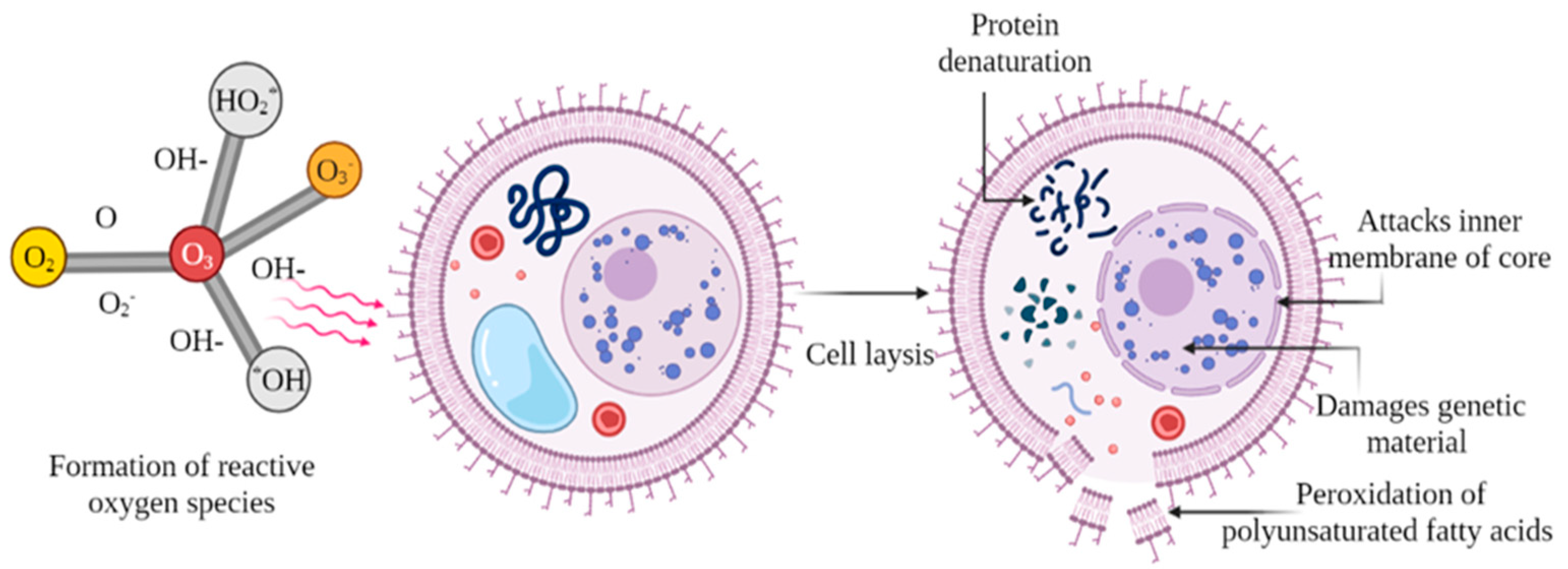
2.1. Ozonation
| Sample | Specification | Microbes | Highlights | Reference |
|---|---|---|---|---|
| Chicken legs | 2–10 mg/L for 1 h combined with vacuum packaging (polyamide/polyethylene bags) stored at 4 °C for 16 days. | TVC, Pseudomonas spp., LAB, Yeast-molds, & Enterobacteriaceae | 6-day shelf-life extension compared to vacuum packaging alone (4-day extension). Positively affected odor, texture, and taste retained an acceptable score for 14–16 days. | [29] |
| Chicken meat (freeze-dried) | 0.6 ppm at 4 °C (90% RH) for 10 min. | TAMB, LAB, E. coli. & Salmonella spp. | 1.1 log CFU/g was observed in TAMB and LAB. E. coli. and Salmonella spp. was not detected. Combination with MAP (20% CO2, 80% N2) improved the texture and sensory proprieties. | [30] |
| Chicken meat (freeze-dried) | 0.4–0.7 ppm at 4 °C (90% RH) for 10–120 min. | LAB & TAMB | Reduced 4.77 and 6.8 log CFU/g, respectively. The combined use of ozone and lyophilization would be useful for extending shelf-life to 8 months. | [22] |
| Chicken breast meat | 10 × 10−6 kg O3/m3/h for 3 days. | Coliform, aerobic, and anaerobic bacteria | Aerobic: 2.96 log CFU/g (untreated = 5.35 log CFU/g) Anaerobic: 2.18 log CFU/g (untreated = 4.63 log CFU/g) Coliform: 1.74 log CFU/g (untreated = 3.35 log CFU/g) |
[31] |
| Duck breast meat | Aerobic: 2.52 log CFU/g (untreated = 4.11 log CFU/g) Anaerobic: 3.46 log CFU/g (untreated = 3.95 log CFU/g) Coliform: 1.39 (untreated = 3.28) |
|||
| Turkey breast meat | 1 × 10−2 kg/m3 at 22 °C (21.6% RH) for 8 h. | TAMB, Enterobacteriaceae & yeast-mold | Reduced 2.9, 2.3 and 1.9 log CFU/g, respectively. | [32] |
| Beef (sliced) | 218–286 mg/m3, 5–20 pulses for 2–40 min with intervals of 30 min. | Heterotrophic microflora & L. monocytogenes | Decreased 1.5 log CFU/g heterotrophic counts. Decreased inoculated L. monocytogenes counts by more than 1 log CFU/g. Exposure times of more than 10 min negatively affected red color and rancidity. | [33] |
2.2. High Hydrostatic Pressure (HHP)
| Meat Type | Treatment Conditions | Storage Conditions | Findings | Reference |
|---|---|---|---|---|
| Chicken fillets | 500 MPa for 10 min. | 4 and 12 °C | HHP resulted in the reduction of the pathogen population below the detection limit of the enumeration method (0.48 log CFU/g), irrespective of the inoculum. HHP extended the shelf life of chicken fillets by 6 and 2 days, at 4 and 12 °C, respectively. | [44] |
| Frozen chicken breast | 500 MPa for 1 min and 400 MPa for 5 min. | _ | HHP showed inactivation of Salmonella at 400 MPa for 5 min and 500 MPa for 1 min. | [45] |
| Ground chicken meat | 350 MPa for 10 min + 0.75% carvacrol. | HHP with 0.60% carvacrol treatment resulted in a >5-log pathogen reduction. | [46] | |
| Ground beef | 400 MPa for 15 min at 25, 35, and 45 °C. | 4 and −20 °C for up to 5 days | At 25 °C, 5 log reduction in E. coli O157:H7 was observed further low-temperature storage serves as the hurdle in its survival and recovery after treatment. HHP showed no effect on the chromatic profile of grounded beef. | [47] |
| Vacuum-packed ground beef | 200 and 400 MPa for 5 min at 25 °C. | _ | L. sakei is good pressure-resistant lactic acid bacteria used in combination with HHP at 400 MPa and is efficient in controlling pathogenic E. coli strains. | [48] |
| Uncooked ground beef patties | 300, 400, and 500 MPa for 5 min. | 4 °C for 10 days | HHP combine with Lactobacillus acidophilus showed less total aerobic count (3.35 log CFU/g) than untreated (6.74 log CFU/g) beef patties with 0.80 log CFU/mL yeast and mold count. The combined treatment showed a delayed decrease in pH value, inhibited lipid oxidation with better color retention and the highest sensory score. | [49] |
| Beef patty | 400 and 600 MPa for 5 min. | Refrigerated storage for 18 h | An amount of 2 and 4 log CF/mL reductions after 400 and 600 MPa in Shiga toxin-producing E. coli O157:H7, respectively. Variations in fat concentration of 10 and 20% did not affect. In contrast, 1% NaCl evident more reduction than 2%, indicating bar protective effect of salt. | [50] |
| Vacuum-pack ripened mutton patties | 200 and 400 MPa for 10 min. | 4 °C for 28 days | Significant reduction in total plate count after HHP at both levels, with a significant increase in lightness (L*). Redness (a*), yellowness (b*); hardness, gumminess, and chewiness of patties reduced significantly. | [51] |
| Beef steak | 450 MPa, 600 MPa 1, 3, 6, 10, 15 min. | _ | HHP have the potential to allow the production of a convenient and safe product by achieving 5 log definition of pasteurization of beef steak inoculated with E. coli 0157:H7. | [52] |
| Beef slurry | 600 MPa for 20 min at 75 °C. | _ | Best inactivation of spores of Clostridium perfringens in beef slurry was a 2.2 log reduction. | [53] |
| Beef slurry | 600 MPa for 20 min at 75 °C. | _ | After HHP, a greater reduction (2.2 log) in C. perfringens spores was observed as compared to thermal treatment (no reduction) after 20 min. | [54] |
| Beef slurry | 600 MPa at 70 °C for 20 min. | _ | A 4.9 log reduction in Bacillus cereus spores after treatment at 70 °C but same temperature thermal processing led to 0.5 log reduction in spore. Increasing HHP temperature from 38 to 70 °C increases the spore inactivation for up to 3 logs. | [55] |
| Marinated beef (Longissimus lumborum) | 300, 400, and 600 MPa for 5 min. | Refrigerated storage for 14 days | HHP was proven to provide safe meat along with a sodium reduction in it. Meat marinated with salt and citric acid has no sufficient inactivation of L. innocua and Enterococcus faecium, while when combine with HHP, a 6 log cycle reduction was observed. | [56] |
| Beef burgers | 300 MPa for 10 min at 9.9 °C and 600 MPa 10 min, 10.2 °C. | _ | Mesophilic and psychotropic count remain at the detection limit after HHP at 600 MPa, with no effect on lipid oxidation for at least 6 days. | [57] |
| Raw meatballs (beef, veal, beef + veal + pork) | 400 and 600 MPa for 0 and 18 min. | 4 and −12 °C for 18 h | No difference in the extent of inactivation in different species of meat used for meatballs preparation in refrigerated storage (0.9 to 2.9 log CFU/g) as compared to frozen samples (1.0 to 3.0 log CFU/g). A total of 600 MPa requires 1–3 min and 400 MPa requires 9 min for a ≥2.0 log CFU/g reduction. | [58] |
| Emulsified beef sausages | 100–400 MPa for 15 min at 10 °C. | _ | HHP proved to be an effective technique to produce microbial safe beef sausages (reduce total viable count equivalent to the sausages having higher salt concentration) with lower salt concentration. | [59] |
| Dry fermented sausages | 600 MPa for 3 min. | 4 °C for 4 weeks | Inactivation of E. coli O157:H7 in dried fermented sausages was observed to be affected by aw. At aw ≤ 0.90, or moisture protein ratio in the range of 1.9–2.3, led to 6.4 log reduction. Further drying reduced to 2.2 log reduction. Recovery of E. coli O157:H7 was observed for 1 week of storage but in 2-, 3-, and 4-week storage, no further recovery was observed. | [60] |
| Pork cooked sausages | 600 MPa for 3 min. | 4 and 10 °C for 35 days | Cooking of sausages leads to a >6 log reduction in inoculated L. monocytogenes. During storage at 4 °C, no significant growth was observed after HHP. But at 10 °C storage, growth remains below the detection limit up to 21 days after the 4.5 log CFU/mL increase in population was observed. No lactic acid bacterial growth was observed till the end of storage. | [61] |
| Italian salami | 600 MPa for 300 s. | _ | HHP related microbial inactivation depicts an inverse relation with aw. All 20 salami samples showed a 5 log reduction in Salmonella after treatment. | [62] |
| Italian salami | 600 MPa for 300 s. | _ | An amount of 0.34–4.32 log CFU/g reduction during processing in L. innocua was observed which was reduced to 0.48–3.4 log CFU/g after HHP. The efficacy of HHP was associated with aw and higher pH after acidification, drying and seasoning phase. | [63] |
| Nitrite-free emulsion-type sausage | 0.1, 500 MPa for 12 min + 0, 1, 2% vinegar | 4 °C for two weeks followed by at 20 °C for three weeks | HHP (500 MPa; four cycles and each for 3 min) + vinegar (1%) reduced vegetative cells and spores of C. perfringens by 4.8 and 2.8 log CFU/g, respectively. | [64]. |
| Traditional Portuguese ready-to-eat meat sausage (Chouriço de carne) | 300 MPa for 5 min at 10 °C + lactic acid bacteria (Pediococcus acidilactici, HA-6111-2) and its bacteriocin (bacHA-6111-2). | Refregrated storage for 60 days. | The hurdle technology (bacteriocin and pressurization) showed a 0.5 log CFU/g decrease in L. innocua cells compared to non-treated cells. | [65] |
| Dry-cured ham | 450 MPa for 10 min and 600 MPa for 5 min. | 4 °C for 30 days | The efficacy of HHP against L. monocytogenes was reduced by low aw values. The changes in HHP-surviving bacteria gene transcription patterns were strain-dependent. | [66] |
| Cooked ham | 400 MPa for 10 min at 17 °C + alginate films containing enterocins. | 1 or 6 °C for 2 months | Both antimicrobial packaging and pressurization delayed the growth of L. monocytogenes levels below the detection limit (day 90) during 6 °C storage. | [67] |
This entry is adapted from the peer-reviewed paper 10.3390/foods11152173
References
- Cerveny, J.; Meyer, J.D.; Hall, P.A. Compendium of the Microbiological Spoilage of Foods and Beverages. Compend. Microbiol. Spoilage Foods Beverages 2009, 69–86.
- Alum, E.A.; Urom, S.M.O.C.; Ben, C.M.A. Microbiological Contamination of Food the Mechanisms Impacts and Prevention. Int. J. Sci. Technol. Res. 2015, 4, 65–78.
- Yousefi, M.; Khorshidian, N.; Hosseini, H. Potential Application of Essential Oils for Mitigation of Listeria Monocytogenes in Meat and Poultry Products. Front. Nutr. 2020, 7, 255.
- Stoica, M.; Stoean, S.; Alexe, P. Overview of Biological Hazards Associated with the Consumption of the Meat Products. J. Agroaliment. Process. Technol. 2014, 20, 192–197.
- ur Rahman, U.; Sahar, A.; Ishaq, A.; Aadil, R.M.; Zahoor, T.; Ahmad, M.H. Advanced Meat Preservation Methods: A Mini Review. J. Food Saf. 2018, 38, e12467.
- Khalafalla, F.A.; Ali, H.M.; El-Fouley, A. Microbiological Evaluation of Chicken Meat Products. J. Vet. Med. Res. 2019, 26, 151–163.
- Ribeiro, C.M.; Stefani, L.M.; Lucheis, S.B.; Okano, W.; Cruz, J.C.M.; Souza, G.V.; Casagrande, T.A.C.; Bastos, P.A.S.; Pinheiro, R.R.; Arruda, M.M.; et al. Methicillin-Resistant Staphylococcus Aureus in Poultry and Poultry Meat: A Meta-Analysis. J. Food Prot. 2018, 81, 1055–1062.
- Hu, K.L.; Yu, X.Q.; Chen, J.; Tang, J.N.; Wang, L.Z.; Li, Y.M.; Tang, C. Production of Characteristic Volatile Markers and Their Relation to Staphylococcus Aureus Growth Status in Pork. Meat Sci. 2020, 160, 107956.
- Whyte, P.; McGill, K.; Collins, J.D. An Assessment of Steam Pasteurization and Hot Water Immersion Treatments for the Microbiological Decontamination of Broiler Carcasses. Food Microbiol. 2003, 20, 111–117.
- Gu, J.-G.; Park, J.-M.; Yoon, S.-J.; Ahn, B.-K.; Kang, C.-W.; Song, J.-C.; Kim, J.-M. Assessment of Dipping Treatment with Various Lactic Acid or Sodium Benzoate Concentrations to Extend the Shelf-Life of Spent Hen Breast Meats. Korean J. Food Sci. Anim. Resour. 2011, 31, 428–435.
- Sarjit, A.; Dykes, G.A. Trisodium Phosphate and Sodium Hypochlorite Are More Effective as Antimicrobials against Campylobacter and Salmonella on Duck as Compared to Chicken Meat. Int. J. Food Microbiol. 2015, 203, 63–69.
- Juneja, V.K.; Valenzuela-Melendres, M.; Heperkan, D.; Bautista, D.; Anderson, D.; Hwang, C.A.; Peña-Ramos, A.; Camou, J.P.; Torrentera-Olivera, N. Development of a Predictive Model for Salmonella Spp. Reduction in Meat Jerky Product with Temperature, Potassium Sorbate, PH, and Water Activity as Controlling Factors. Int. J. Food Microbiol. 2016, 236, 1–8.
- Kocharunchitt, C.; Mellefont, L.; Bowman, J.P.; Ross, T. Application of Chlorine Dioxide and Peroxyacetic Acid during Spray Chilling as a Potential Antimicrobial Intervention for Beef Carcasses. Food Microbiol. 2020, 87, 103355.
- Manzoor, A.; Jaspal, M.H.; Yaqub, T.; Haq, A.U.; Nasir, J.; Avais, M.; Asghar, B.; Badar, I.H.; Ahmad, S.; Yar, M.K. Effect of Lactic Acid Spray on Microbial and Quality Parameters of Buffalo Meat. Meat Sci. 2020, 159, 107923.
- Zhao, T.; Zhao, P.; Chen, D.; Jadeja, R.; Hung, Y.C.; Doyle, M.P. Reductions of Shiga Toxin–Producing Escherichia Coli and Salmonella Typhimurium on Beef Trim by Lactic Acid, Levulinic Acid, and Sodium Dodecyl Sulfate Treatments. J. Food Prot. 2014, 77, 528–537.
- Burfoot, D.; Allen, V.; Mulvey, E.; Jewell, K.; Harrison, D.; Morris, V. Reducing Campylobacter Numbers on Chicken Carcasses Using Lactic Acid in Processing Plants. Int. J. Food Sci. Technol. 2015, 50, 2451–2457.
- Hansson, I.; Sandberg, M.; Habib, I.; Lowman, R.; Engvall, E.O. Knowledge Gaps in Control of Campylobacter for Prevention of Campylobacteriosis. Transbound. Emerg. Dis. 2018, 65, 30–48.
- Killinger, K.M.; Kannan, A.; Bary, A.I.; Cogger, C.G. Validation of a 2 Percent Lactic Acid Antimicrobial Rinse for Mobile Poultry Slaughter Operations. J. Food Prot. 2010, 73, 2079–2083.
- Yoon, K.S. Effect of Gamma Irradiation on the Texture and Microstructure of Chicken Breast Meat. Meat Sci. 2003, 63, 273–277.
- Anang, D.M.; Rusul, G.; Bakar, J.; Ling, F.H. Effects of Lactic Acid and Lauricidin on the Survival of Listeria Monocytogenes, Salmonella Enteritidis and Escherichia Coli O157:H7 in Chicken Breast Stored at 4 °C. Food Control 2007, 18, 961–969.
- Ko, J.-K.; Ma, Y.-H.; Song, K.-B. Effect of Chlorine Dioxide Treatment on Microbial Growth and Qualities of Chicken Breast. Prev. Nutr. Food Sci. 2005, 10, 122–129.
- Cantalejo, M.J.; Zouaghi, F.; Pérez-Arnedo, I. Combined Effects of Ozone and Freeze-Drying on the Shelf-Life of Broiler Chicken Meat. LWT 2016, 68, 400–407.
- Nabi, B.G.; Mukhtar, K.; Arshad, R.N.; Radicetti, E.; Tedeschi, P.; Shahbaz, M.U.; Walayat, N.; Nawaz, A.; Inam-Ur-raheem, M.; Aadil, R.M. High-Pressure Processing for Sustainable Food Supply. Sustainability 2021, 13, 13908.
- Roobab, U.; Fidalgo, L.G.; Arshad, R.N.; Khan, A.W.; Zeng, X.A.; Bhat, Z.F.; Bekhit, A.E.D.A.; Batool, Z.; Aadil, R.M. High-Pressure Processing of Fish and Shellfish Products: Safety, Quality, and Research Prospects. Compr. Rev. Food Sci. Food Saf. 2022, 1–29.
- Umair, M.; Jabbar, S.; Ayub, Z.; Muhammad Aadil, R.; Abid, M.; Zhang, J.; Liqing, Z. Recent Advances in Plasma Technology: Influence of Atmospheric Cold Plasma on Spore Inactivation. Food Rev. Int. 2021.
- Gonçalves, A.A.; Lira Santos, T.C. Improving Quality and Shelf-Life of Whole Chilled Pacific White Shrimp (Litopenaeus Vannamei) by Ozone Technology Combined with Modified Atmosphere Packaging. LWT 2019, 99, 568–575.
- Roobab, U.; Afzal, R.; Ranjha, M.M.A.N.; Zeng, X.A.; Ahmed, Z.; Aadil, R.M. High Pressure-Based Hurdle Interventions for Raw and Processed Meat: A Clean-Label Prospective. Int. J. Food Sci. Technol. 2022, 57, 816–826.
- Gök, V.; Aktop, S.; Özkan, M.; Tomar, O. The Effects of Atmospheric Cold Plasma on Inactivation of Listeria Monocytogenes and Staphylococcus Aureus and Some Quality Characteristics of Pastırma—A Dry-Cured Beef Product. Innov. Food Sci. Emerg. Technol. 2019, 56, 102188.
- Gertzou, I.N.; Karabagias, I.K.; Drosos, P.E.; Riganakos, K.A. Effect of Combination of Ozonation and Vacuum Packaging on Shelf Life Extension of Fresh Chicken Legs during Storage under Refrigeration. J. Food Eng. 2017, 213, 18–26.
- Zouaghi, F.; Cantalejo, M.J. Study of Modified Atmosphere Packaging on the Quality of Ozonated Freeze-Dried Chicken Meat. Meat Sci. 2016, 119, 123–131.
- Muhlisin, M.; Utama, D.T.; Lee, J.H.; Choi, J.H.; Lee, S.K. Effects of Gaseous Ozone Exposure on Bacterial Counts and Oxidative Properties in Chicken and Duck Breast Meat. Korean J. Food Sci. Anim. Resour. 2016, 36, 405–411.
- Ayranci, U.G.; Ozunlu, O.; Ergezer, H.; Karaca, H. Effects of Ozone Treatment on Microbiological Quality and Physicochemical Properties of Turkey Breast Meat. Ozone Sci. Eng. 2020, 42, 95–103.
- Giménez, B.; Graiver, N.; Giannuzzi, L.; Zaritzky, N. Treatment of Beef with Gaseous Ozone: Physicochemical Aspects and Antimicrobial Effects on Heterotrophic Microflora and Listeria Monocytogenes. Food Control 2021, 121, 1–9.
- Cooke, E.M. Principles and Practice of Disinfection, Preservation and Sterilization. J. Clin. Pathol. 1983, 36, 364.
- Kaavya, R.; Pandiselvam, R.; Abdullah, S.; Sruthi, N.U.; Jayanath, Y.; Ashokkumar, C.; Chandra Khanashyam, A.; Kothakota, A.; Ramesh, S.V. Emerging Non-Thermal Technologies for Decontamination of Salmonella in Food. Trends Food Sci. Technol. 2021, 112, 400–418.
- USDA-FSIS. Safe and Suitable Ingredients Used in the Production of Meat and Poultry, and Egg Products; USDA-FSIS: Washington, DC, USA, 2016. Available online: https://www.google.com/search?q=USDA-FSIS.+Safe+and+Suitable+Ingredients+Used+in+the+Production+of+Meat+and+Poultry%2C+and+Egg+Products%3B+USDA-FSIS%3A+Washington%2C+DC%2C+USA%2C+2016&oq=USDA-FSIS.+Safe+and+Suitable+Ingredients+Used+in+the+Production+of+M (accessed on 16 May 2022).
- USDA-FSIS USDA FSIS, 2001, Letter from Robert C. Post (FSIS, Washington, DC) to Mark D. Dopp (Am. Meat Institute, Arlington, VA) Dated 21 December 2001. Available online: https://www.fsis.usda.gov/ (accessed on 16 May 2022).
- Pandiselvam, R.; Subhashini, S.; Banuu Priya, E.P.; Kothakota, A.; Ramesh, S.V.; Shahir, S. Ozone Based Food Preservation: A Promising Green Technology for Enhanced Food Safety. Ozone Sci. Eng. 2019, 41, 17–34.
- Shynkaryk, M.V.; Pyatkovskyy, T.; Mohamed, H.M.; Yousef, A.E.; Sastry, S.K. Physics of Fresh Produce Safety: Role of Diffusion and Tissue Reaction in Sanitization of Leafy Green Vegetables with Liquid and Gaseous Ozone-Based Sanitizers. J. Food Prot. 2015, 78, 2108–2116.
- Pandiselvam, R.; Kaavya, R.; Jayanath, Y.; Veenuttranon, K.; Lueprasitsakul, P.; Divya, V.; Kothakota, A.; Ramesh, S.V. Ozone as a Novel Emerging Technology for the Dissipation of Pesticide Residues in Foods—A Review. Trends Food Sci. Technol. 2020, 97, 38–54.
- Roobab, U.; Khan, A.W.; Lorenzo, J.M.; Arshad, R.N.; Chen, B.R.; Zeng, X.A.; Bekhit, A.E.D.; Suleman, R.; Aadil, R.M. A Systematic Review of Clean-Label Alternatives to Synthetic Additives in Raw and Processed Meat with a Special Emphasis on High-Pressure Processing (2018–2021). Food Res. Int. 2021, 150, 110792.
- Roobab, U.; Inam-Ur-Raheem, M.; Khan, A.W.; Arshad, R.N.; Zeng, X.; Aadil, R.M. Innovations in High-Pressure Technologies for the Development of Clean Label Dairy Products: A Review. Food Rev. Int. 2021, 1–22.
- Global High Pressure Processing (HPP) Food Market—Analysis by Product Type, Distribution Channel, by Region, by Country (2020 Edition): Market Insights, Outlook Post COVID-19 Pandemic (2020–2025); Research and Markets: Dublin, Ireland, 2020.
- Argyri, A.A.; Papadopoulou, O.S.; Nisiotou, A.; Tassou, C.C.; Chorianopoulos, N. Effect of High Pressure Processing on the Survival of Salmonella Enteritidis and Shelf-Life of Chicken Fillets. Food Microbiol. 2018, 70, 55–64.
- Cap, M.; Paredes, P.F.; Fernández, D.; Mozgovoj, M.; Vaudagna, S.R.; Rodriguez, A. Effect of High Hydrostatic Pressure on Salmonella Spp Inactivation and Meat-Quality of Frozen Chicken Breast. LWT 2020, 118, 108873.
- Chuang, S.; Sheen, S.; Sommers, C.H.; Zhou, S.; Sheen, L.Y. Survival Evaluation of Salmonella and Listeria Monocytogenes on Selective and Nonselective Media in Ground Chicken Meat Subjected to High Hydrostatic Pressure and Carvacrol. J. Food Prot. 2020, 83, 37–44.
- Zhou, Y.; Karwe, M.V.; Matthews, K.R. Differences in Inactivation of Escherichia Coli O157: H7 Strains in Ground Beef Following Repeated High Pressure Processing Treatments and Cold Storage. Food Microbiol. 2016, 58, 7–12.
- Wang, L.; Kong, X.; Jiang, Y. Recovery of High Pressure Processing (HPP) Induced Injured Escherichia Coli O157:H7 Inhibited by Lactobacillus Sakei on Vacuum-Packed Ground Beef. Food Biosci. 2021, 41, 100928.
- Lee, H.; Shahbaz, H.M.; Yang, J.; Jo, M.H.; Kim, J.U.; Yoo, S.; Kim, S.H.; Lee, D.U.; Park, J. Effect of High Pressure Processing Combined with Lactic Acid Bacteria on the Microbial Counts and Physicochemical Properties of Uncooked Beef Patties during Refrigerated Storage. J. Food Process. Preserv. 2021, 45, e15345.
- Eccoña Sota, A.; Cap, M.; Rodriguez, A.; Szerman, N.; Speroni, F.; Vaudagna, S.R. Effects of High Hydrostatic Pressure and Beef Patty Formulations on the Inactivation of Native Strains of Shiga Toxin-Producing Escherichia Coli O157:H7. Food Bioprocess Technol. 2021, 14, 1194–1198.
- Banerjee, R.; Jayathilakan, K.; Chauhan, O.P.; Naveena, B.M.; Devatkal, S.; Kulkarni, V.V. Vacuum Packaged Mutton Patties: Comparative Effects of High Pressure Processing and Irradiation. J. Food Process. Preserv. 2017, 41, e12880.
- Sun, S.; Sullivan, G.; Stratton, J.; Bower, C.; Cavender, G. Effect of HPP Treatment on the Safety and Quality of Beef Steak Intended for Sous Vide Cooking. LWT-Food Sci. Technol. 2017, 86, 185–192.
- Evelyn; Milani, E.; Silva, F.V.M. Comparing High Pressure Thermal Processing and Thermosonication with Thermal Processing for the Inactivation of Bacteria, Moulds, and Yeasts Spores in Foods. J. Food Eng. 2017, 214, 90–96.
- Evelyn; Silva, F.V.M. High Pressure Thermal Processing for the Inactivation of Clostridium Perfringens Spores in Beef Slurry. Innov. Food Sci. Emerg. Technol. 2016, 33, 26–31.
- Evelyn; Silva, F.V.M. Modeling the Inactivation of Psychrotrophic Bacillus Cereus Spores in Beef Slurry by 600 MPa HPP Combined with 38–70 °C: Comparing with Thermal Processing and Estimating the Energy Requirements. Food Bioprod. Process. 2016, 99, 179–187.
- Rodrigues, I.; Trindade, M.A.; Caramit, F.R.; Candoğan, K.; Pokhrel, P.R.; Barbosa-Cánovas, G.V. Effect of High Pressure Processing on Physicochemical and Microbiological Properties of Marinated Beef with Reduced Sodium Content. Innov. Food Sci. Emerg. Technol. 2016, 38, 328–333.
- Mizi, L.; Cofrades, S.; Bou, R.; Pintado, T.; López-Caballero, M.E.; Zaidi, F.; Jiménez-Colmenero, F. Antimicrobial and Antioxidant Effects of Combined High Pressure Processing and Sage in Beef Burgers during Prolonged Chilled Storage. Innov. Food Sci. Emerg. Technol. 2019, 51, 32–40.
- Porto-fett, A.C.S.; Jackson-davis, A.; Kassama, L.S.; Daniel, M.; Oliver, M.; Jung, Y.; Luchansky, J.B. Inactivation of Shiga Toxin-Producing Escherichia Coli in Refrigerated and Frozen Meatballs Using High Pressure Processing. Microorganisms 2020, 8, 360.
- Zhu, Y.; Yan, Y.; Yu, Z.; Wu, T.; Bennett, L.E. Effects of High Pressure Processing on Microbial, Textural and Sensory Properties of Low-Salt Emulsified Beef Sausage. Food Control 2022, 133, 108596.
- Balamurugan, S.; Gemmell, C.; Lau, A.T.Y.; Arvaj, L.; Strange, P.; Gao, A.; Barbut, S. High Pressure Processing during Drying of Fermented Sausages Can Enhance Safety and Reduce Time Required to Produce a Dry Fermented Product. Food Control 2020, 113, 107224.
- Balamurugan, S.; Inmanee, P.; De Souza, J.E.; Strange, P.; Pirak, T.; Barbut, S. Effects of High Pressure Processing and Hot Water Pasteurization of Cooked Sausages on Inactivation of Inoculated Listeria monocytogenes, Natural Populations of Lactic Acid Bacteria, Pseudomonas spp., and Coliforms and Their Recovery during Storage at 4 A. J. Food Prot. 2018, 81, 1245–1251.
- Bonilauri, P.; Grisenti, M.S.; Daminelli, P.; Merialdi, G.; Ramini, M.; Bardasi, L.; Taddei, R.; Cosciani-Cunico, E.; Dalzini, E.; Frustoli, M.A.; et al. Reduction of Salmonella Spp. Populations in Italian Salami during Production Process and High Pressure Processing Treatment: Validation of Processes to Export to the U.S. Meat Sci. 2019, 157, 107869.
- Bonilauri, P.; Merialdi, G.; Ramini, M.; Bardasi, L.; Taddei, R.; Grisenti, M.S.; Daminelli, P.; Cosciani-Cunico, E.; Dalzini, E.; Frustoli, M.A.; et al. Modeling the Behavior of Listeria Innocua in Italian Salami during the Production and High-Pressure Validation of Processes for Exportation to the U.S. Meat Sci. 2021, 172, 108315.
- Lee, S.H.; Choe, J.; Shin, D.J.; Yong, H.I.; Choi, Y.; Yoon, Y.; Jo, C. Combined Effect of High Pressure and Vinegar Addition on the Control of Clostridium Perfringens and Quality in Nitrite-Free Emulsion-Type Sausage. Innov. Food Sci. Emerg. Technol. 2019, 52, 429–437.
- Castro, S.M.; Kolomeytseva, M.; Casquete, R.; Silva, J.; Teixeira, P.; Castro, S.M.; Queirós, R.; Saraiva, J.A. Biopreservation Strategies in Combination with Mild High Pressure Treatments in Traditional Portuguese Ready-to-Eat Meat Sausage. Food Biosci. 2017, 19, 65–72.
- Pérez-Baltar, A.; Alía, A.; Rodríguez, A.; Córdoba, J.J.; Medina, M.; Montiel, R. Impact of Water Activity on the Inactivation and Gene Expression of Listeria Monocytogenes during Refrigerated Storage of Pressurized Dry-Cured Ham. Foods 2020, 9, 1092.
- Marcos, B.; Aymerich, T.; Monfort, J.M.; Garriga, M. High-Pressure Processing and Antimicrobial Biodegradable Packaging to Control Listeria Monocytogenes during Storage of Cooked Ham. Food Microbiol. 2008, 25, 177–182.
