The single cell protein (SCP) refers to the dead, dried microbial cells or total protein extracted from the pure microbial culture of algae, bacteria, filamentous fungi, unicellular algae, and cyanobacteria cultivated on different carbon sources that are used as a protein supplement in human foods or animal feeds. Many studies reported that the wastes from various fruits such as orange, sweet orange, mango, banana, pomegranate, pineapple, grapes, watermelon, papaya, and many others are potential substrates for SCP production. These SCPs can be used as a protein supplement in human foods or animal feeds.
- SCPs
- fruit wastes
- fermentation
- bioconversion
1. Single Cell Protein Production Methods
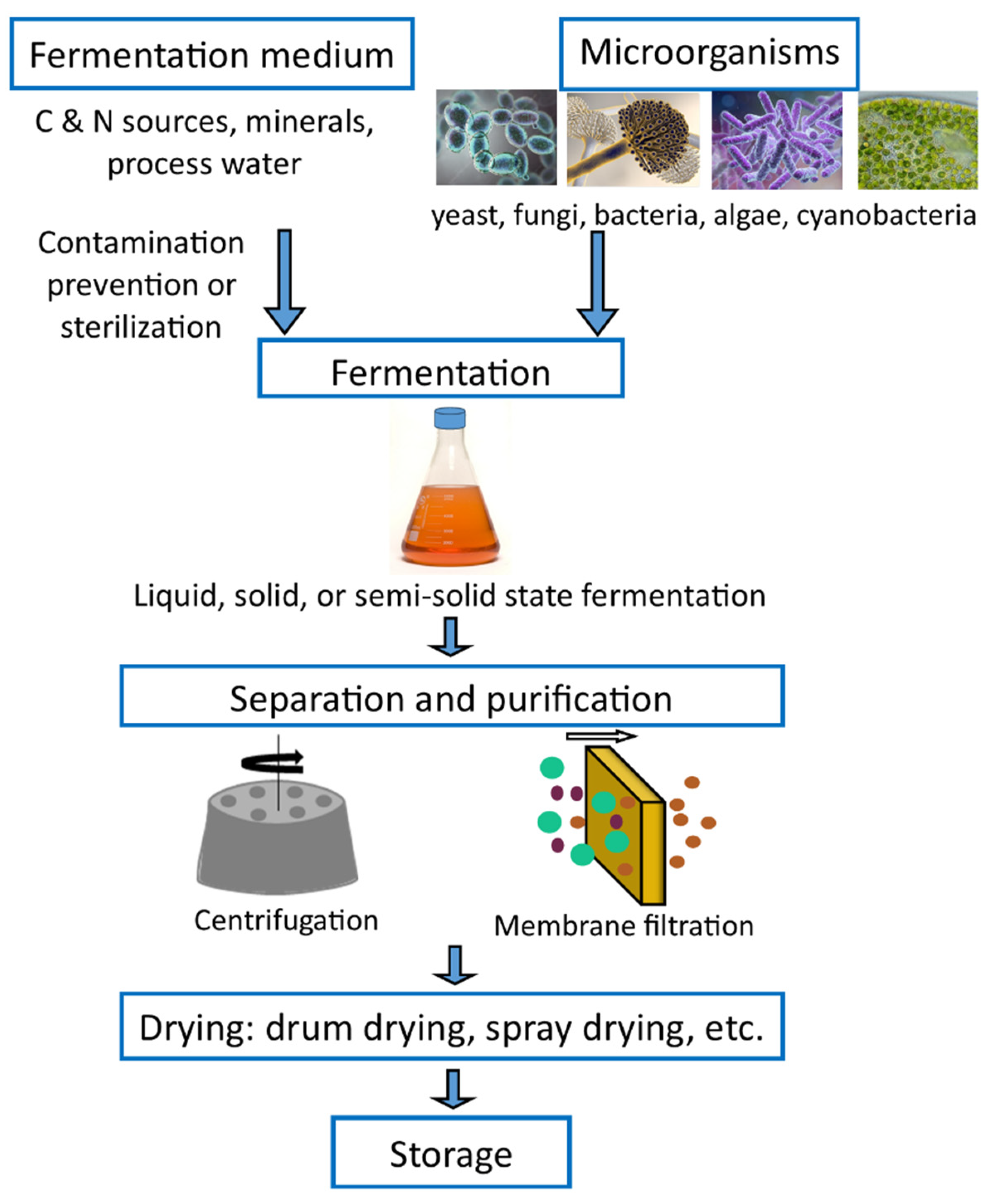
2. Fruit Production and Waste Generation
3. Physico-Chemical Properties of Fruit Waste
4. Fruit Waste as Substrate for Single Cell Protein Production
| Microorganism | Substrate (Fruit Waste) | Type of Fermentation Medium | Reference |
|---|---|---|---|
| Yeast | |||
| Yarrowia lipolytica (formerly Candida lipolytica, or Saccharomyces lipolytica) | Olive fruits wastes | SF/LSF | [50] |
| Candida utilis | Pineapple cannery effluent | SF/LSF | [51] |
| Pineapple waste | SF/LSF | [52] | |
| Mixture of the banana and orange waste | SF/LSF | [53] | |
| Orange peel | SF/LSF | [54] | |
| Mango wastes | SSF | [55] | |
| Cyberlindnera spp. | Banana peel hydrolysate | SF/LSF | [56] |
| Geotrichum candidum | Orange peel | SF/LSF | [57] |
| Saccharomyces cerevisiae | Watermelon, mixture of fruit wastes | SF/LSF | [58] |
| Watermelon, pineapple | SF/LSF | [59] | |
| Yam peel | SF/LSF | [60] | |
| Apple, orange peel | SF/LSF | [46] | |
| Cucumber peel, orange peel | SF/LSF | [40] | |
| Pineapple waste | SF/LSF | [32][61][62][63] | |
| Papaya waste | SF/LSF | [64] | |
| Apple, papaya, banana | SF/LSF | [44] | |
| Guava peels and cashew bagasse | SSF | [65] | |
| Rind of pomegranate, mango, banana, apple, sweet orange peel |
SSF | [48] | |
| Orange peels | SSF | [66][67] | |
| Pichia pinus | Mango waste | SF/LSF | [68] |
| Fungi | |||
| Aspergillus niger | Banana peel, orange peel, cucumber peel, pineapple peel, watermelon peel | SF/LSF | [69] |
| Banana peel | SF/LSF | [70] | |
| Banana peel | SF/LSF | [71] | |
| Banana, papaya, orange | SF/LSF | [72] | |
| Lemon peel, orange peel, apple pomace | SSF | [73] | |
| Aspergillus niger Rhizopus oryzae |
Orange peels | SSF | [67] |
| Aspergillus niger Saccharomyces cerevisiae |
Orange peel | SSF | [66] |
| Aspergillus terreus | Banana peel | SSF | [74] |
| Penicillium roqueforti, Penicillium camemberti | Bergamot fruit (citrus fruit) peel | SSF | [75] |
| Phanerochaete chrysosporium, Panus tigrinus |
Banana peel, pineapple peel, papaya peel | SF/LSF | [30] |
| Phanerochaete chrysosporium | Banana peels, pineapple peels, and papaya peels | SF/LSF | [30] |
| Rhizopus oligosporus | Papaya waste, cucumber peelings, pomegranate fruit rind, pineapple fruit skin, and watermelon skin. | SSF | [76] |
| Trichoderma viride, Trichoderma reesei |
Orange peel | SSF | [77] |
| Bacteria | |||
| Rhodococcus opacus | Orange wastes, lemon wastes | SF/LSF | [78] |
| Other natural sources/mixed cultures | |||
| Natural microorganisms in Palmyrah toddy | Papaya, watermelon, and banana peel | SF/LSF | [49] |
| Lactobacillus culture isolated from curd |
Mix fruit wastes such as pineapple peel residue, pomegranate waste, apple waste, and pear waste | SF/LSF | [79] |
5. Types of Fruit Waste
5.1. Fruit Wastes Rich in Simple Sugars
5.2. Fruit Waste Rich in Fibers
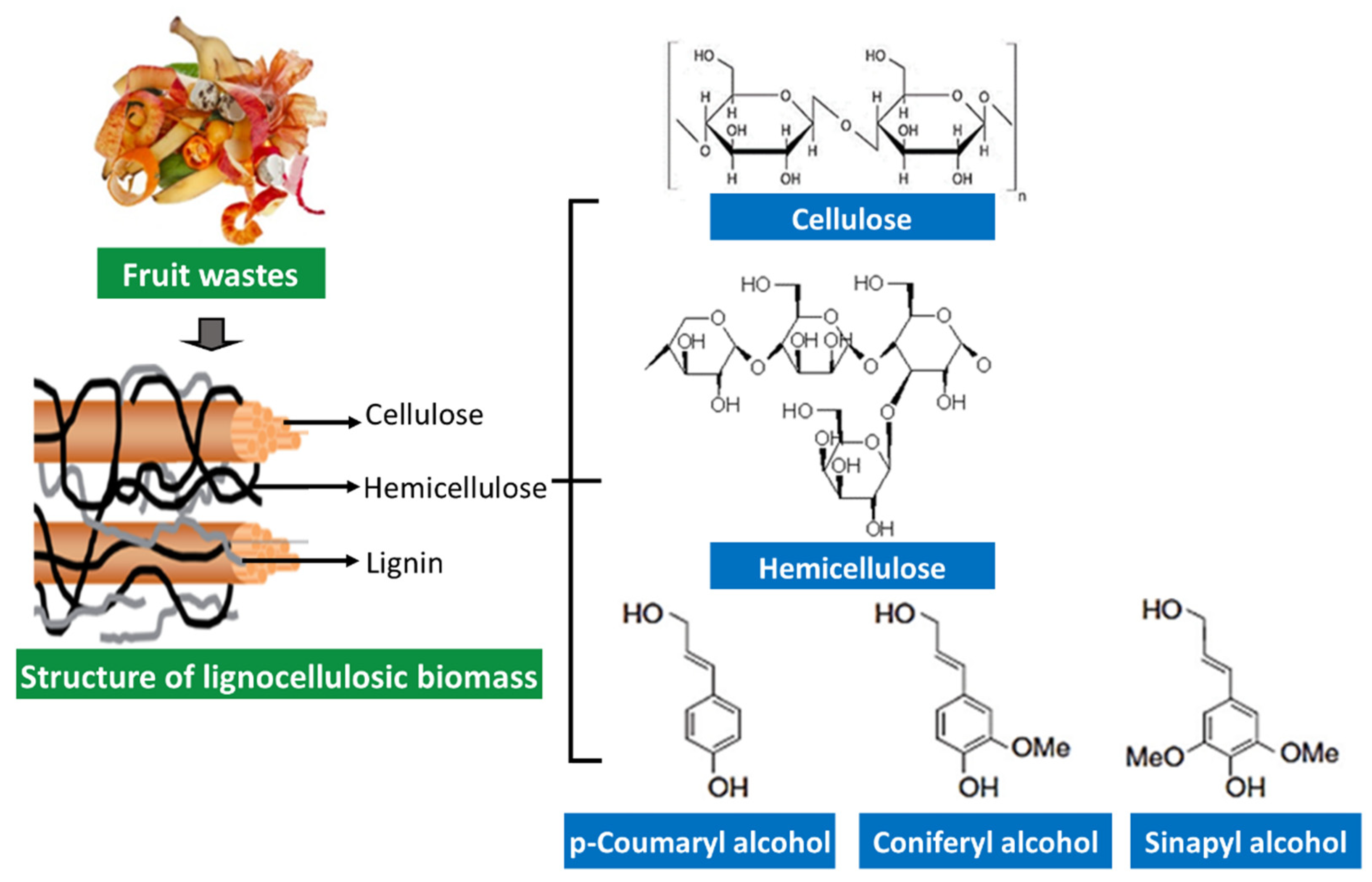
This entry is adapted from the peer-reviewed paper 10.3390/separations9070178
References
- Labuza, T.P.; Santos, D.B.; Roop, R.N. Engineering Factors in Single-Cell Protein Production. I. Fluid Properties and Concentration of Yeast by Evaporation. Biotechnol. Bioeng. 1970, 12, 123–134.
- Bekatorou, A.; Psarianos, C.; Koutinas, A.A. Production of Food Grade Yeasts. Food Technol. Biotechnol. 2006, 44, 407–415.
- Bajpai, P. (Ed.) Single Cell Protein Production from Lignocellulosic Biomass; Springer Briefs in Molecular Science; Springer: Singapore, 2017; ISBN 978-981-10-5873-8.
- Pandey, A. Recent Process Developments in Solid-State Fermentation. Process Biochem. 1992, 27, 109–117.
- Pandey, A.; Soccol, C.R.; Mitchell, D. New Developments in Solid State Fermentation: I-Bioprocesses and Products. Process Biochem. 2000, 35, 1153–1169.
- Singhania, R.R.; Patel, A.K.; Soccol, C.R.; Pandey, A. Recent Advances in Solid-State Fermentation. Biochem. Eng. J. 2009, 44, 13–18.
- Suman, G.; Nupur, M.; Anuradha, S.; Pradeep, B. Single Cell Protein Production: A Review. Int. J. Curr. Microbiol. App. Sci 2015, 4, 251–262.
- Ravichandran, S.; Vimala, R. Solid State and Submerged Fermentation for the Production of Bioactive Substances: A Comparative Study. Int. J. Sci. Nat. 2012, 3, 480–486.
- Yamuna Rani, K.; Ramachandra Rao, V.S. Control of Fermenters—A review. Bioprocess Eng. 1999, 21, 77–88.
- Linder, T. Making the Case for Edible Microorganisms as an Integral Part of a More Sustainable and Resilient Food Production System. Food Sec. 2019, 11, 265–278.
- FAO. World Food and Agriculture–Statistical Yearbook 2021; FAO Statistical Yearbook–World Food and Agriculture; FAO: Rome, Italy, 2021; ISBN 978-92-5-134332-6.
- WHO. Diet, Nutrition, and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation; World Health Organization: Geneva, Switzerland, 2003; ISBN 978-92-4-120916-8.
- Balali, G.I.; Yar, D.D.; Afua Dela, V.G.; Adjei-Kusi, P. Microbial Contamination, an Increasing Threat to the Consumption of Fresh Fruits and Vegetables in Today’s World. Int. J. Microbiol. 2020, 2020, e3029295.
- Romelle, F.D.; Rani, A.; Manohar, R.S. Chemical Composition of Some Selected Fruit Peels. Eur. J. Food Sci. Technol. 2016, 4, 12–21.
- Ibrahim, U.K.; Kamarrudin, N.; Suzihaque, M.U.H.; Hashib, S.A. Local Fruit Wastes as a Potential Source of Natural Antioxidant: An Overview. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Miri, Malaysia, 1–3 December 2016; Volume 206, p. 012040.
- Sharma, R.; Oberoi, H.S.; Dhillon, G.S. Chapter 2-Fruit and Vegetable Processing Waste: Renewable Feed Stocks for Enzyme Production. In Agro-Industrial Wastes as Feedstock for Enzyme Production; Dhillon, G.S., Kaur, S., Eds.; Academic Press: San Diego, CA, USA, 2016; pp. 23–59. ISBN 978-0-12-802392-1.
- Abdullah; Mat, H.B. The Characteristic of Pineapple Waste from Canning Industry. Adv. Sci. Lett. 2017, 23, 5691–5693.
- Murakonda, S.; Dwivedi, M. Powders from Fruit Waste. In Food Powders Properties and Characterization; Ermiş, E., Ed.; Food Engineering Series; Springer: Cham, Switzerland, 2021; pp. 155–168. ISBN 978-3-030-48908-3.
- Coman, V.; Teleky, B.-E.; Mitrea, L.; Martău, G.A.; Szabo, K.; Călinoiu, L.-F.; Vodnar, D.C. Chapter Five-Bioactive Potential of Fruit and Vegetable Wastes. In Advances in Food and Nutrition Research; Toldrá, F., Ed.; Academic Press: Cambridge, MA, USA, 2020; Volume 91, pp. 157–225.
- Mahato, N.; Sharma, K.; Sinha, M.; Dhyani, A.; Pathak, B.; Jang, H.; Park, S.; Pashikanti, S.; Cho, S. Biotransformation of Citrus Waste-I: Production of Biofuel and Valuable Compounds by Fermentation. Processes 2021, 9, 220.
- Chaouch, M.A.; Benvenuti, S. The Role of Fruit By-Products as Bioactive Compounds for Intestinal Health. Foods 2020, 9, 1716.
- Kumar, H.; Bhardwaj, K.; Sharma, R.; Nepovimova, E.; Kuča, K.; Dhanjal, D.S.; Verma, R.; Bhardwaj, P.; Sharma, S.; Kumar, D. Fruit and Vegetable Peels: Utilization of High Value Horticultural Waste in Novel Industrial Applications. Molecules 2020, 25, 2812.
- Monspart-Sényi, J. Fruit Processing Waste Management. In Handbook of Fruits and Fruit Processing; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2012; pp. 315–331. ISBN 978-1-118-35253-3.
- Panda, S.K.; Ray, R.C.; Mishra, S.S.; Kayitesi, E. Microbial Processing of Fruit and Vegetable Wastes into Potential Biocommodities: A Review. Crit. Rev. Biotechnol. 2018, 38, 1–16.
- Sadh, P.K.; Kumar, S.; Chawla, P.; Duhan, J.S. Fermentation: A Boon for Production of Bioactive Compounds by Processing of Food Industries Wastes (By-Products). Molecules 2018, 23, 2560.
- Spalvins, K.; Zihare, L.; Blumberga, D. Single Cell Protein Production from Waste Biomass: Comparison of Various Industrial by-Products. Energy Procedia 2018, 147, 409–418.
- De Gregorio, A.; Mandalari, G.; Arena, N.; Nucita, F.; Tripodo, M.M.; Lo Curto, R.B. SCP and Crude Pectinase Production by Slurry-State Fermentation of Lemon Pulps. Bioresour. Technol. 2002, 83, 89–94.
- Sadhu, S.D.; Garg, M.; Kumar, A. 4-Major Environmental Issues and New Materials. In New Polymer Nanocomposites for Environmental Remediation; Hussain, C.M., Mishra, A.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 77–97. ISBN 978-0-12-811033-1.
- Mussatto, S.I.; Ballesteros, L.F.; Martins, S.; Teixeira, J.A. Use of Agro-Industrial Wastes in Solid-State Fermentation Processes; IntechOpen: London, UK, 2012; ISBN 978-953-51-0253-3.
- Saheed, O.K.; Jamal, P.; Karim, M.I.A.; Alam, M.Z.; Muyibi, S.A. Utilization of Fruit Peels as Carbon Source for White Rot Fungi Biomass Production under Submerged State Bioconversion. J. King Saud Univ.-Sci. 2016, 28, 143–151.
- Saheed, O.K.; Jamal, P.; Kari, M.I.A.; Alam, Z.; Muyibi, S.A. Cellulolytic Fruits Wastes: A Potential Support for Enzyme Assisted Protein Production. J. Biol. Sci. 2013, 13, 379–385.
- Umesh, M.; Thazeem, B.; Preethi, K. Valorization of Pineapple Peels through Single Cell Protein Production Using Saccharomyces cerevisiae NCDC 364. Appl. Food Biotechnol. 2019, 6, 255–263.
- Morais, D.R.; Rotta, E.M.; Sargi, S.C.; Bonafe, E.G.; Suzuki, R.M.; Souza, N.E.; Matsushita, M.; Visentainer, J.V. Proximate Composition, Mineral Contents and Fatty Acid Composition of the Different Parts and Dried Peels of Tropical Fruits Cultivated in Brazil. J. Braz. Chem. Soc. 2017, 28, 308–318.
- Ani, P.N.; Abel, H.C. Nutrient, Phytochemical, and Antinutrient Composition of Citrus maxima Fruit Juice and Peel Extract. Food Sci. Nutr. 2018, 6, 653–658.
- Dias, P.G.I.; Sajiwanie, J.W.A.; Rathnayaka, R.M.U.S.K. Chemical Composition, Physicochemical and Technological Properties of Selected Fruit Peels as a Potential Food Source. Int. J. Fruit Sci. 2020, 20, S240–S251.
- Pathak, P.D.; Mandavgane, S.A.; Kulkarni, B.D. Fruit Peel Waste: Characterization and Its Potential Uses. Curr. Sci. 2017, 113, 444–454.
- Rivas, B.; Torrado, A.; Torre, P.; Converti, A.; Domínguez, J.M. Submerged Citric Acid Fermentation on Orange Peel Autohydrolysate. J. Agric. Food Chem. 2008, 56, 2380–2387.
- Orozco, R.S.; Hernández, P.B.; Morales, G.R.; Núñez, F.U.; Villafuerte, J.O.; Lugo, V.L.; Ramírez, N.F.; Díaz, C.E.B.; Vázquez, P.C. Characterization of Lignocellulosic Fruit Waste as an Alternative Feedstock for Bioethanol Production. BioResources 2014, 9, 1873–1885.
- Ververis, C.; Georghiou, K.; Danielidis, D.; Hatzinikolaou, D.G.; Santas, P.; Santas, R.; Corleti, V. Cellulose, Hemicelluloses, Lignin and Ash Content of Some Organic Materials and Their Suitability for Use as Paper Pulp Supplements. Bioresour. Technol. 2007, 98, 296–301.
- Mondal, A.K.; Sengupta, S.; Bhowal, J.; Bhattacharya, D.K. Utilization of Fruit Wastes in Producing Single Cell Protein. Int. J. Sci. Environ. 2012, 1, 430–438.
- Nasseri, A.T.; Rasoul-Amini, S.; Morowvat, M.H.; Ghasemi, Y. Single Cell Protein: Production and Process. Am. J. Food Technol. 2011, 6, 103–116.
- Adoki, A. Factors Affecting Yeast Growth and Protein Yield Production from Orange, Plantain and Banana Wastes Processing Residues Using Candida Sp. Afr. J. Biotechnol. 2008, 7, 290–295.
- Malav, A.; Dube, P. Single Cell Protein Production Using Various Microbial Mass: A Review. IJAR 2017, 5, 2190–2194.
- Kandari, V.; Gupta, S. Bioconversion of Vegetable and Fruit Peel Wastes in Viable Product. J. Microbiol. Biotechnol. Res. 2012, 2, 308–312.
- Adedayo; Ajiboye, E.A.; Akintunde, J.K.; Odaibo, A. Single Cell Proteins: As Nutritional Enhancer. Adv. Appl. Sci. Res. 2011, 2, 396–409.
- Bacha, U.; Nasir, M.; Khalique, A.; Anjum, A.; Jabbar, M. Comparative Assessment of Various Agro-Industrial Wastes for Saccharomyces cerevisiae Biomass Production and Its Quality Evaluation as Single Cell Protein. J. Anim. Plant Sci. 2011, 21, 844–849.
- Akanni, G.; Ntuli, V.; Preez, D. Cactus Pear Biomass, a Potential Lignocellulose Raw Material for Single Cell Protein Production (SCP): A Review. Int. J. Curr. Microbiol. App. Sci 2014, 3, 171–197.
- Khan, M.; Khan, S.; Zafar, A.; Tanveer, A. Production of Single Cell Protein from Saccharomyces cerevisiae by Utilizing Fruit Wastes. Nanobiotechnica Univers. 2010, 1, 127–132.
- Thiviya, P.; Kapilan, R.; Madhujith, T. Bioconversion of Fruit Wastes of Papaya, Watermelon, and Banana into Single Cell Protein Production. Trop. Agric. Res. 2021, 32, 503–514.
- Rages, A.A.; Haider, M.M. Alkaline Hydrolysis of Olive Fruits Wastes for the Production of Single Cell Protein by Candida lipolytica. Biocatal. Agric. Biotechnol. 2021, 33, 101999.
- Nigam, J.N. Single Cell Protein from Pineapple Cannery Effluent. World J. Microbiol. Biotechnol. 1998, 14, 693–696.
- Rosma, A.; Ooi, K.I. Production of Candida utilis Biomass and Intracellular Protein Content: Effect of Agitation Speed and Aeration Rate. MJM 2006, 2, 15–18.
- Munawar, R.; Irfan, M.; Nadeem, M.; Syed, Q.; Siddique, Z. Biosynthesis of Single Cell Biomass of Candida Utuilis by Submerged Fermentation. Pak. J. Sci. 2010, 62, 1–5.
- Carranza-Méndez, R.C.; Chávez-González, M.L.; Sepúlveda-Torre, L.; Aguilar, C.N.; Govea-Salas, M.; Ramos-González, R. Production of Single Cell Protein from Orange Peel Residues by Candida utilis. Biocatal. Agric. Biotechnol. 2022, 40, 102298.
- Somda, M.K.; Nikiema, M.; Keita, I.; Mogmenga, I.; Kouhounde, S.H.S.; Dabire, Y.; Coulibaly, W.H.; Taale, E.; Traore, A.S. Production of Single Cell Protein (SCP) and Essentials Amino Acids from Candida utilis FMJ12 by Solid State Fermentation Using Mango Waste Supplemented with Nitrogen Sources. AJB 2018, 17, 716–723.
- Jiru, T.M.; Melku, B. Single Cell Protein Production from Torula Yeast (Cyberlindnera Sp.) Using Banana Peel Hydrolysate. J. Adv. Microbiol. 2018, 13, 1–7.
- Ziino, M.; Lo Curto, R.B.; Salvo, F.; Signorino, D.; Chiofalo, B.; Giuffrida, D. Lipid Composition of Geotrichum candidum Single Cell Protein Grown in Continuous Submerged Culture. Bioresour. Technol. 1999, 67, 7–11.
- Stabnikova, O.; Wang, J.-Y.; Bo Ding, H. Joo-HwaTay Biotransformation of Vegetable and Fruit Processing Wastes into Yeast Biomass Enriched with Selenium. Bioresour. Technol. 2005, 96, 747–751.
- Abarshi, M.M.; Mada, S.B.; Amin, M.I.; Salihu, A.; Garba, A.; Mohammad, H.A. Effect of Nutrient Supplementation on Single Cell Protein Production from Watermelon and Pineapple Peels. Niger. J. Basic Appl. Sci. 2017, 25, 130–136.
- Aruna, T.E.; Aworh, O.C.; Raji, A.O.; Olagunju, A.I. Protein Enrichment of Yam Peels by Fermentation with Saccharomyces cerevisiae (BY4743). Ann. Agric. Sci. 2017, 62, 33–37.
- Mensah, J.K.M.; Twumasi, P. Use of Pineapple Waste for Single Cell Protein (SCP) Production and the Effect of Substrate Concentration on the Yield. J. Food Process Eng. 2017, 40, e12478.
- Mujdalipah, S.; Putri, M.L. Utilization of Pineapple Peel and Rice Washing Water to Produce Single Cell Proteins Using Saccharomyces cerevisiae. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Bogor, Indonesia, 9–10 October 2019; Volume 472, p. 012029.
- Nurmalasari, A.; Maharani, S. Addition of Carbon Sources to Pineapple Waste Media in the Production of Single Cell Protein Biomass Saccharomyces Cerevisiae. J. Ris. Biol. Dan Apl. 2020, 2, 70–76.
- Umesh, M.; Priyanka, K.; Thazeem, B.; Preethi, K. Production of Single Cell Protein and Polyhydroxyalkanoate from Carica papaya Waste. Arab. J. Sci. Eng. 2017, 42, 2361–2369.
- Muniz, C.E.S.; Santiago, Â.M.; Gusmão, T.A.S.; Oliveira, H.M.L.; de Sousa Conrado, L.; de Gusmão, R.P. Solid-State Fermentation for Single-Cell Protein Enrichment of Guava and Cashew by-Products and Inclusion on Cereal Bars. Biocatal. Agric. Biotechnol. 2020, 25, 101576.
- Azam, S.; Khan, Z.; Bashir, A.; Khan, I.; Ali, J. Production of Single Cell Protein from Orange Peels Using Aspergillus niger and Saccharomyces cerevisiae. Glob. J. Biotechnol. Biochem. 2014, 9, 14–18.
- Hamdy, H.S. Production of Mini-Food by Aspergillus niger, Rhizopus oryzae and Saccharomyces cerevisiae Using Orange Peels. Rom. Biotechnol. Lett. 2013, 18, 7929–7946.
- Rashad, M.M.; Moharib, S.A.; Jwanny, E.W. Yeast Conversion of Mango Waste or Methanol to Single Cell Protein and Other Metabolites. Biol. Wastes 1990, 32, 277–284.
- Oshoma, C.E.; Eguakun-Owie, S.O. Conversion of Food Waste to Single Cell Protein Using Aspergillus niger. J. Appl. Sci. Environ. Manag. 2018, 22, 350–355.
- Yabaya, A.; Ado, S.A. Mycelial Protein Production by Aspergillus niger Using Banana Peels. Sci. World J. 2008, 3, 9–12.
- Kamal, M.; Ali, M.; Shishir, M.R.I.; Saifullah, M.; Haque, M.; Mondal, S.C. Optimization of Process Parameters for Improved Production of Biomass Protein from Aspergillus Niger Using Banana Peel as a Substrate. Food Sci. Biotechnol. 2019, 28, 1693–1702.
- Bind, A.; Kumar, M.; Singh, D. Optimization of SCP Production of Aspergillus niger Using Different Fruit Peels-Indian Journals. Int. J. Bioinform. Biol. Sci. 2013, 1, 1–8.
- Orzua, M.C.; Mussatto, S.I.; Contreras-Esquivel, J.C.; Rodriguez, R.; de la Garza, H.; Teixeira, J.A.; Aguilar, C.N. Exploitation of Agro Industrial Wastes as Immobilization Carrier for Solid-State Fermentation. Ind. Crops Prod. 2009, 30, 24–27.
- Jaganmohan, P.; Daas, B.P.; Prasad, S.V. Production of Single Cell Protein (SCP) with Aspergillus terreus Using Solid State Fermentation. Eur. J. Biol. Sci. 2013, 5, 38–43.
- Scerra, V.; Caridi, A.; Foti, F.; Sinatra, M.C. Influence of Dairy Penicillium Spp. on Nutrient Content of Citrus Fruit Peel1Contribution from the Ministry of Scientific Research and Technology–Research Fund 60%: M. C. Sinatra.1. Anim. Feed. Sci. Technol. 1999, 78, 169–176.
- Khan, M.; Khan, S.S.; Ahmed, Z.; Tanveer, A. Production of Fungal Single Cell Protein Using Rhizopus Oligosporus Grown on Fruit Wastes. Biol. Forum 2009, 1, 26–28.
- Ahmadi, F.; Zamiri, M.J.; Khorvash, M.; Banihashemi, Z.; Bayat, A.R. Chemical Composition and Protein Enrichment of Orange Peels and Sugar Beet Pulp after Fermentation by Two Trichoderma Species. Iran. J. Vet. Res. 2015, 16, 25–30.
- Mahan, K.M.; Le, R.K.; Wells, T., Jr.; Anderson, S.; Yuan, J.S.; Stoklosa, R.J.; Bhalla, A.; Hodge, D.B.; Ragauskas, A.J. Production of Single Cell Protein from Agro-Waste Using Rhodococcus Opacus. J. Ind. Microbiol. Biotechnol. 2018, 45, 795–801.
- Patel, N.; Patel, A.; Patel, H.; Patel, M.; Patel, U. Production of Single Cell Protein from Mix Fruits Waste Using Lactobacillus. Int. J. Pharm. Biol. Sci. 2019, 9, 164–168.
- Sadh, P.K.; Duhan, S.; Duhan, J.S. Agro-Industrial Wastes and Their Utilization Using Solid State Fermentation: A Review. Bioresour. Bioprocess. 2018, 5, 1.
- Domingues, R.; Bondar, M.; Palolo, I.; Queirós, O.; de Almeida, C.D.; Cesário, M.T. Xylose Metabolism in Bacteria—Opportunities and Challenges towards Efficient Lignocellulosic Biomass-Based Biorefineries. Appl. Sci. 2021, 11, 8112.
- Sandle, T. 22-Microbiological Challenges to the Pharmaceuticals and Healthcare. In Pharmaceutical Microbiology; Sandle, T., Ed.; Woodhead Publishing: Oxford, UK, 2016; pp. 281–294. ISBN 978-0-08-100022-9.
- Abu Yazid, N.; Barrena, R.; Komilis, D.; Sánchez, A. Solid-State Fermentation as a Novel Paradigm for Organic Waste Valorization: A Review. Sustainability 2017, 9, 224.
- Anwar, Z.; Gulfraz, M.; Irshad, M. Agro-Industrial Lignocellulosic Biomass a Key to Unlock the Future Bio-Energy: A Brief Review. J. Radiat. Res. Appl. Sci. 2014, 7, 163–173.
- Kalaichelvan, P.T.; Arulpandi, I. Bioprocess Technology; MJP Publishers: Chennai, India, 2019; ISBN 978-81-8094-032-3.
- Chen, H. Chemical Composition and Structure of Natural Lignocellulose. In Biotechnology of Lignocellulose: Theory and Practice; Chen, H., Ed.; Springer: Dordrecht, The Netherlands, 2014; pp. 25–71. ISBN 978-94-007-6898-7.
