Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Cell Biology
Meiosis is an essential cell-division process for ensuring genetic diversity across generations. Meiotic recombination ensures the accuracy of genetic interchange between homolous chromosomes and segregation of parental alleles. Programmed DNA double-strand breaks (DSBs), catalyzed by the evolutionarily conserved topoisomerase VIA (a subunit of the archaeal type II DNA topoisomerase)-like enzyme Spo11 and several other factors, is a distinctive feature of meiotic recombination initiation.
- meiosis
- double-strand break (DSB)
- homologous recombination
- chromosome
- hotspots
1. Introduction
In flowering plants, reproductive cells develop in the ‘sporophytic generation’ and then differentiate into the gamete-forming ‘gametophytic generation’ [1,2]. During the reproductive process, the production of gametes requires that the genetic complement be reduced by one-half [3,4,5]. This specialized nuclear division, called meiosis, includes one round of DNA replication followed by two successive rounds of cell division, thereby ensuring the ploidy of the zygotic genome [6,7]. In the first meiotic division (meiosis I), homologous chromosomes are brought in close to pair and undergo synapsis, promoting the reciprocal exchange of parental chromosome fragments and thereby increasing the genetic diversity of the progeny [7,8,9]. Meiotic recombination is initiated by the formation of programmed DNA double-strand breaks (DSBs) catalyzed by the evolutionarily conserved type II topoisomerase–like enzyme SPO11 and several accessary proteins [9,10,11,12,13]. After resection, two SPO11 molecules remain covalently bound to each 5′ end of the nicked DNA, which is then processed by the MRX complex (Mre11-Rad50-Xrs2) with the cooperation of Sae2 to release the SPO11-bound nicked DNA oligonucleotide [14,15,16,17,18,19]. Following the action of the 5′ to 3′ exonuclease Exo1, the DNA ends are further degraded to produce 3′ single-stranded DNA tails [9,20,21,22].
2. Defining Meiotic DSB Hotspots in Different Species
Meiotic DSBs are not randomly distributed along eukaryotic chromosomes; rather, they are concentrated within discrete regions described as DSB hotspots [92]. To generate a high-resolution physical map of the meiotic DSB landscape, several methodologies have been established over the past three decades. SPO11-oligo mapping is achieved by immunoprecipitating tagged-Spo11 bound with oligonucleotides, which subsequently go through end-labeling, purification and sequencing [98]. This method has been effectively applied in several yeast species, mouse and Arabidopsis (Table 2) [92,99,100,101,102]. SSDS takes advantage of an antibody specifically recognizing either one of two DNA recombinases, RAD51 or DMC1, and utilizes chromatin immunoprecipitation to enrich single-stranded DNA that has undergone single-end invasion. With the high-throughput sequencing of enriched single-stranded DNA, the genome-wide distribution of DSB hotpots was successfully obtained in maize, mouse and human [103,104,105] (Table 2).
Table 2. Meiotic DSB hotspots identified in different species by SPO11-oligo mapping or single-stranded DNA sequencing (SSDS).
| Species | Genome Size | Chromosome No. | Number of DSBs | DSB Hotspot No. | Most Common DSB Location |
Average Width (kb) | Predominantly DSB Formation Among Transposon |
Method | Hotspot Detection | References |
|---|---|---|---|---|---|---|---|---|---|---|
| S. cerevisiae (SK1) | 12.1 Mb | 16 | ~175 | 3604–4099 | Gene promoters | 0.248–0.264 | Ty retrotransposons | SPO11-oligos | Enrichment threshold | [92,101,110,111] |
| S. cerevisiae (YPS128) | 12.1 Mb | 16 | ~175 | 4177 | Gene promoters | 0.265 | n/a | SPO11-oligos | Enrichment threshold | [112] |
| S. cerevisiae (UWOPS03-461.4) | 12.1 Mb | 16 | ~175 | 3881 | Gene promoters | 0.256 | n/a | SPO11-oligos | Enrichment threshold | [112] |
| S. pombe | 13.8 Mb | 3 | ~60 | 603 | All chromosome regions | 1.4 | n/a | Rec12-oligos | Enrichment threshold | [113] |
| M. musculus (9R×13R) | 2.8 Gb | 20 | ~250 | 9874– 15,677 |
Intergenic | ~2.000–3.400 | LTR retrotransposons SINE |
SSDS | Peak calling | [103,104] |
| M. musculus (9R) | 2.8 Gb | 20 | ~250 | 14,869 | Intergenic | ~2.000 | n/a | SSDS | Peak calling | [104] |
| M. musculus (13R) | 2.8 Gb | 20 | ~250 | 15,481 | Intergenic | ~2.000 | n/a | SSDS | Peak calling | [104] |
| M. musculus (B6) |
2.8 Gb | 20 | ~250 | 18,313 | Intergenic | ~2.000 | n/a | SSDS | Peak calling | [104] |
| M. musculus (B6) |
2.8 Gb | 20 | ~250 | 13,960 | Intergenic | ~0.281 | n/a | SPO11-oligos | Enrichment threshold | [100] |
| Arabidopsis thaliana | 135 Mb | 5 | ~250–300 | 5914 | Gene promoters and terminators | 0.823 | Helitron /Pogo/Tc1/Mariner DNA transposons | SPO11-1-oligos | Peak calling | [99] |
| Zea mays | 2.4 Gb | 10 | ~500 | 3126 | All chromosome regions | 1.2 | Gypsy retrotransposons | SSDS | Peak calling | [105] |
DSB hotspot designation is governed by a hierarchy of interrelated factors, including cis-regulatory elements, chromatin accessibility and high-order chromosome architecture (Table 2) [106]. Of particular prominence is the striking presence of DSB hotspots in repetitive DNA, such as transposon elements, although this aspect varies among species [10]. In maize, only ~25% of DSB hotspots have been discovered near genes, and the remaining hotspots are distributed in repetitive DNA—predominantly in Gypsy retrotransposons [105]. Although the Arabidopsis genome has fewer transposons compared with maize, a proportion of DSB hotspots were identified in Helitron/Pogo/Tc1/Mariner/MuDR DNA transposons [99]. Similarly, although most DSB hotspots tend to occur in genic regions, a significant overlap have been observed within the MULE-MuDR, TcMar-Mariner, hAT-Charlie and PiggyBac transposon families [107]. Moreover, DSB hotspots are evident in Ty retrotransposons in budding yeast. What is well established is that the meiotic recombination within or adjacent to repetitive elements is rare because this could lead to homologous recombination between non-allelic repeats, likely resulting in harmful chromosomal rearrangements and compromising genome stability [108,109]. Therefore, the paradox between the appearance of DSB hotspots and the suppression of recombination in these repetitive regions suggests that a mechanism exists to safeguard the repair of DSB in repetitive regions and prevent inadvertent crossing over.
3. Control of Meiotic DSB Formation by Protein Phosphorylation
3.1. Cyclin-Dependent Kinases (CDKs)
CDKs in conjunction with their cyclin partners represent an ancient molecular switch that promotes and regulates cell-cycle progression [4,114,115]. The fundamental theme of how CDKs mediate meiotic recombination initiation was mostly drawn from studies in yeast. In S. cerevisiae, the activation of Cdc28 by its two B-type cyclin partners, Clb5 and Clb6, stimulates the phosphorylation on Mer2 during pre-meiotic DNA replication (Figure 1A) [115,116,117,118,119]; subsequently, phosphorylated Mer2 recruits other Spo11-accessary proteins to initiate DSB formation (Figure 1B) [45,120,121]. Similarly, in S. pombe, the association between Cdc2 and any of the three cyclins, Crs1, Cig1 and Cig2, is crucial for DSB formation [114].
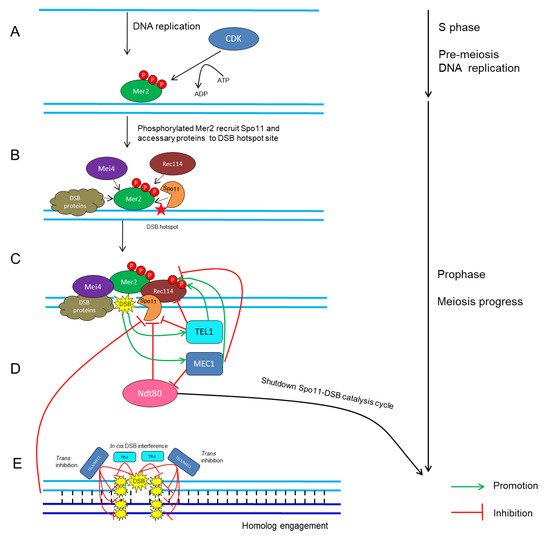
Figure 1. Schematic network of CDK- and ATM/ATR-mediated regulatory cycles of meiotic DSB timing and number in S. cerevisiae. (A) CDK phosphorylates Mer2 during pre-meiotic DNA replication [114,115,120]. (B) Phosphorylated Mer2 recruits Rec114, Mei4, Spo11 and other DSB proteins to DSB hotspot sites [45,120,121]. (C) DSB formation catalyzed by Spo11 and accessary proteins [4,66]. (D) Recurrent DSB formation activates TEL1/MEC1-dependent positive- and negative-feedback loops, which then restrains Spo11 activity and regulates the rate and number of DSB formation [136,140,147,156]. (E) Cis DSB interference mediated by TEL1 reduces the frequency of coincident DSB formation at the region adjacent to an already-formed DSB [143,149]. Trans inhibition mediated by TEL1 and MEC1 describes the ability of a DSB formation on one chromosome to suppress DSB formation on its homolog and sister chromatid at the same or adjacent regions [141].
In Arabidopsis, there are at least five types of cell-cycle CDKs (CDKA;1, CDKB;1, CDKB1;2, CDKB2;1 and CDKB2;2) and more than 50 cyclins [122,123,124,125,126,127,128,129,130,131], of which a few have been characterized as functioning during meiosis. CDKA;1 was originally identified as the key regulator for both mitosis and meiosis progression in Arabidopsis [129,131,132], and very recently, CDKA;1 was identified as a major regulator of meiotic recombination by mediating the number and placement of crossovers of homologous chromosomes [124,131]. However, CDKA;1 seems not to regulate meiotic prophase I although the exact influence on DSB formation was not determined in that study [124]. SDS, a cyclin protein, has been shown to control the formation of crossovers, but not DSBs, which differs from the necessity of its rice homologs in DSB formation [87,127,132,133]. TAM, an A-type cyclin (CYCA1;2), is indispensable for meiosis termination at the end of the first meiotic division [134,135]. Therefore, the core CDKs or related cyclins that directly regulate DSB formation remain to be explored in plants.
3.2. Tel1/ATM and Mec1/ATR
Once DSBs are made, there are several mechanisms to maintain the proper number of DSBs within a chromosome region [95,111,136,137,138,139,140,141]. This phenomenon, termed DSB homeostasis, prevents the severe effects caused by excess or insufficient Spo11 activity [136,137,140,142,143]. In S. cerevisiae, Tel1 and the related protein kinase Mec1 act as key factors that synergistically fine-tune the number of DSBs [141,142,144]. Lack of Tel1 leads to a considerable increase in the level of Spo11-oligonucleotide complexes, which are the by-product of meiotic DSB formation, indicating that Tel1 negatively regulates DSB formation (Figure 1C) [95]. In contrast, a defect in Mec1 activity causes a dramatic reduction in DSB number via inhibition of Ndt80, which triggers the exit from pachytene and shuts down Spo11-DSB catalysis of the cell cycle [145,146]. Mec1 ensures persistent Spo11 activity and indirectly promotes DSB formation, revealing that Mec1 positively regulates DSB formation (Figure 1C) [147,148]. In addition, recent studies in S. cerevisiae have revealed a cis-regulatory machinery, termed DSB interference, that reduces the frequency of coincident DSBs at the region adjacent to the preexisting DSB [136,142,143], and this process is dependent on TEL1 over short distances of ~70–100 kb (Figure 1D) [95,149]. On the contrary, the other machinery, known as trans-inhibition, defines the ability of a DSB to occur on a single chromosome, and this suppresses DSB designation on its homolog and sister chromatid at the same locus [141]. This mechanism relies on both Tel1 and Mec1, ensuring that an interhomolog interaction surrounding a DSB will not occur twice at the allelic or two nearby chromosomal positions so that DSBs are constrained to one per pair of homologs (Figure 1E) [141]. Interestingly, homolog engagement can also restrict the number of DSBs by inhibiting Spo11 activity (Figure 1E) [101,150]. Moreover, Tel1 and/or Mec1 can phosphorylate Rec114 directly, which limits its interaction with DSB hotspots and consequently reduces DSB formation genome wide (Figure 1C) [140].
In Arabidopsis, Tel1 and Mec1 homologs exist, termed ATM and ATR, respectively. However, ATM and ATR play synergetic roles in maintenance of genomic stability in meiotic cells by processing Spo11-dependent DSBs rather than influencing DSB formation [151,152,153,154,155]. Therefore, either ATM/ATR-mediated signaling pathway is evolutionarily divergent across different species or the effects of ATM/ATR on DSB formation is too subtle to be detected by conventional methods.
This entry is adapted from the peer-reviewed paper 10.3390/ijms20194718
This entry is offline, you can click here to edit this entry!
