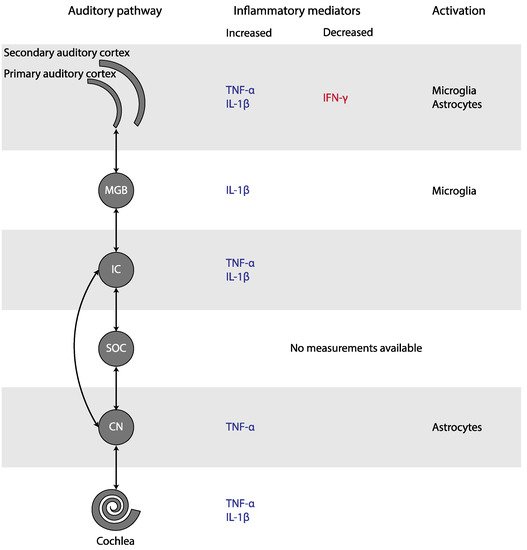
Experimental studies found an increase of TNF-α in the cochlea, the CN, the IC, and the AC, as well as an increase of IL-1β in the cochlea, the IC, the MGB, and the AC of animals with tinnitus (Figure 3). In addition, microglial markers were increased in the MGB and AC, but decreased in the CN. Astrocytic markers were elevated in the CN and AC. Only human CBC measurements could be meta-analyzed. MPV and PDW in blood samples were increased only if outliers/influential studies were removed. Together, these results indicate that inflammation may play a role in tinnitus.
Figure 3. Changes in inflammatory mediators and glial activation in tinnitus, depicted in the simplified auditory pathway. Abbreviations: CN, cochlear nucleus; IC, inferior colliculus; IFN, interferon; IL, Interleukin; MGB, medial geniculate body; SOC, superior olivary complex; TNF, Tumor Necrosis Factor.
2.2. Neuroglial Involvement in Tinnitus
Microglial activation is characterized by the increased expression of specific markers, such as Iba-1, and morphological changes from the ramified shape to a non-ramified shape. The number of Iba-1 positive microglia and their activation state were increased in the MGB and in the primary AC, but decreased in the CN. After activation, microglia produce proinflammatory cytokines such as TNF-α. These cytokines further activate the microglia, forming a self-stimulating loop [
16]. The central role of microglia is further supported by the observation that microglial depletion prevented an increase in TNF-α expression and noise-induced tinnitus [
16]. Similar to microglial activation, astrocytic activation is characterized by increased expression of specific markers, such as GFAP, and deramification of their processes [
51]. The number of GFAP-positive astrocytes and their activation state were increased in the ventral CN and primary AC, but not in the MGB. Astrocytes are important in regulating levels of neurotransmitter and ion concentrations, controlling synapse formation and function, and repairing the nervous system [
52,
53]. In response to neuronal degeneration, they promote synaptic regrowth and axonal sprouting [
22,
54]. This may lead to enhanced synaptic activity. Moreover, astrocytes can directly excite neighboring neurons through a calcium-dependent glutamate release and promote neural synchrony mediated by extra-synaptic excitatory receptors [
55,
56]. Furthermore, they can produce the proinflammatory cytokines implicated in tinnitus [
57]. Thus, both microglia and astrocytes may play an important role in tinnitus.
2.3. Platelet Involvement in Tinnitus
In humans with tinnitus, MPV was increased in six out of ten studies, but this was only significant when outliers or influential cases were removed. PDW was increased in three out of four studies that examined this marker, but like MPV this was only significant when an influential study was removed. MPV indicates the size of platelets, whereas PDW reflects variation of platelet size distribution. Since platelets change their shape when activated, increased MPV and PDW can both be used as a sign of activated platelets. In line with these results, Chrbolka et al. (2020) showed that platelet activity was increased in patients with tinnitus [
36]. Platelet count and NLR seem to remain unchanged.
Platelets may be involved in the development of tinnitus in several ways. Platelets release granule-stored cytokines such as IL-6, IL-8 and TNF-α within seconds to minutes after activation. In addition, they produce various chemokines and cytokines, such as IL-1β, within hours after activation [
58,
59]. These cytokines, and thus platelets, are potent inducers of the acute phase response [
60]. Therefore, platelets are an important source of cytokines in the inflammatory response. On the other hand, increased platelet volumes may be an indicator of a prothrombic condition or even cause thrombosis, e.g., in the internal auditory artery leading to hypoperfusion of the cochlea, impairing its function and contributing to the development of tinnitus [
28,
38,
42]. Finally, platelets are involved in glutamate uptake. Reduced systemic glutamate uptake by platelets, as well as glutamate excitotoxicity, has been demonstrated in various neurodegenerative disorders, such as Alzheimer’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis [
61]. Although speculative, reduced systemic glutamate uptake by platelets could play a role in tinnitus as well, given the elevated glutamate concentration in tinnitus [
62].
2.4. Inflammation in the Pathophysiology of Tinnitus
The question remains how inflammation would be involved in the pathophysiology of tinnitus. Tinnitus has been associated with an increase in excitatory and a decrease in inhibitory neurotransmission [
62]. Inflammation, on the other hand, may lead to alterations in synaptic transmission and synaptic organization.
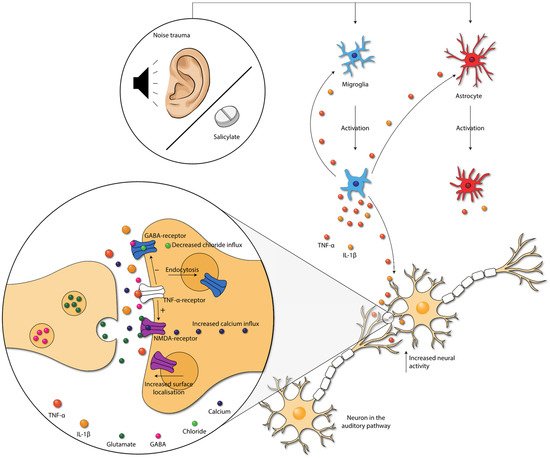
Salicylate-induced tinnitus is associated with the increased activity of the NMDA (N-methyl-D-aspartate) receptor, one of the excitatory glutamate receptors [
63] (
Figure 4). Some animal studies included here also studied the (gene) expression of NR2A and NR2B, two NMDA receptor subunits. Both NR2A and NR2B (gene) expression were consistently elevated in the IC, the AC, and the cochlea in animals with tinnitus after salicylate injection. Moreover, a positive association between NR2B and tinnitus scores was present [
17,
18,
19,
20,
23,
46]. When tinnitus was diminished, (gene) expression of both NR2A and NR2B returned to normal as well. Thus, in animals with salicylate-induced tinnitus, NMDA (gene) expression is increased. Interestingly, this increase was consistently coexistent with increases in TNF-α and IL-1β in animals with tinnitus throughout the entire auditory pathway [
17,
18,
19,
46]. The specific effect of TNF-α on the NMDA receptor is still a matter of debate as both an increase and decrease of NMDA receptor currents have been demonstrated [
64,
65,
66,
67]. However, most studies show an increase. Additionally, TNF-α enhances surface localization of NMDA receptor subunits [
67]. Last, Wang et al. (2019) showed that tinnitus was associated with TNF-α-dependent increased excitatory and decreased inhibitory synaptic currents [
16]. Besides TNF-α, IL-1β has also been shown to enhance NMDA-induced currents [
66,
68]. In sum, the increased expression of TNF-α, and possibly also IL-1β, leads to increased NMDA receptor-dependent calcium influx and enhanced post-synaptic currents. This leads to increased neural activity.
Figure 4. Damage caused by noise exposure and salicylate administration leads to the activation of neuroglia. Neuroglia produce cytokines that further activate the neuroglia. Moreover, cytokines influence neurotransmission within the auditory pathway. TNF-α increases the NMDA-receptor and decreases the GABAA-receptor activity, via the TNF-α-receptor. Moreover, NMDA receptor surface localization increases, and GABA-receptor surface localization decreases. These changes lead to increased neural activity in the auditory pathway. Abbreviations: GABA, gamma-aminobutyric acid; IL, interleukin; NMDA, N-methyl-D-aspartate; TNF, tumor necrosis factor.
The gamma-aminobutyric acid (GABA)-receptor is one of the major inhibitory neurotransmitter receptors. A potent tinnitus inducer, sodium salicylate, has been shown to suppress GABA
(A)-induced currents, and tinnitus was completely eliminated after oral administration of the GABA agonist Vigabatrin, which suggests the involvement of decreased inhibitory neurotransmission in tinnitus [
69,
70,
71]. Interestingly, inhibitory neurotransmission is also impacted by cytokines. TNF-α expression influences inhibitory GABA
A (gamma-aminobutyric acid) receptors by causing an endocytosis of GABA
A receptors, resulting in fewer surface GABA
A receptors and a decrease in inhibitory synaptic strength [
72]. Additionally, IL-1β has been shown to suppress GABA-induced currents [
66]. So, besides increasing excitatory neurotransmission, TNF-α and IL-1β presumably also suppress inhibitory neurotransmission in tinnitus.
Vice versa, the abnormal release and/or uptake of neurotransmitters can also result in inflammation [
57]. Intraperitoneal injections of memantine, a non-competitive NMDA receptor antagonist, for seven consecutive days was successful in decreasing NR2B expression and attenuating tinnitus. It also decreased expression of TNF-α [
46]. Therefore, it seems probable that salicylate and noise exposure cause both an imbalance in neurotransmission and neuroinflammation, leading to tinnitus. This could lead to a vicious circle in which inflammation and neurotransmitter imbalance reinforce each other, exacerbating tinnitus.
Changes in the activity of receptors can lead to synaptic plasticity, which is the ability of synapses to strengthen or weaken over time [
73]. Therefore, the cytokine-induced changes in excitatory and inhibitory neurotransmission could influence neural plasticity associated with tinnitus [
74]. Moreover, it has been stated that the proinflammatory cytokines as TNF-α and IL-1β itself may also directly alter long-term synaptic plasticity [
66,
75,
76,
77]. Synaptic plasticity may occur throughout the whole auditory pathway, both ascending and descending. The detailed, specific mechanisms and types of neuroplasticity in tinnitus are beyond the scope of this review. Therefore, we refer to the reviews of Roberts (2018) and Wu et al. (2016) on this topic [
78,
79]. Concluding, synaptic plasticity caused by inflammation could explain the long-term effects after the induction of tinnitus.
Various studies have reported that psychological stress increases the bothersomeness and loudness of tinnitus [
80]. Baigi et al. (2011) even suggested that stress is a key factor in modulating the severity of tinnitus [
81]. Acute stress stimulates the inflammatory response by increasing the amount of circulating IL-6, IL-1β, TNF-α and IL-10 [
82]. On the other hand, chronic stress is also associated with increases in the amount of circulating cytokines, such as IL-6 and TNF-α [
82]. Therefore, it is not implausible that the effect of stress on the severity of tinnitus is effectuated via inflammatory pathways.
In conclusion, damage caused by noise exposure and salicylate administration leads to inflammation. In turn, proinflammatory cytokines seem to influence synaptic transmission by either increasing excitatory synaptic transmission and/or decreasing inhibitory synaptic transmission throughout the whole auditory tract. Neuroplasticity occurs because of the altered activity of these receptors. This could explain the long-term effects of tinnitus.