Despite the remarkable development of the medical industry in the current era, herbal products with therapeutic potentials arise as attractive alternative treatments. Consequently, Chios mastiha, a natural, aromatic resin obtained from the trunk and brunches of the mastic tree, has recently gained increasing scientific interest due to its multiple beneficial actions. Chios mastiha is being exclusively produced on the southern part of Chios, a Greek island situated in the northern Aegean Sea, and its therapeutic properties have been known since Greek antiquity. There is now substantial evidence to suggest that mastiha demonstrates a plethora of favorable effects, mainly attributed to the anti-inflammatory and anti-oxidative properties of its components. The main use of mastiha nowadays, however, is for the production of natural chewing gum, although an approval by the European Medicines Agency for mild dyspeptic disorders and for inflammations of the skin has been given.
- mastic gum
- mastiha
- Pistacia lentiscus
- anti-inflammatory effect
- oxidative stress
- inflammatory bowel disease
- oxidized LDL
1. Anti-Inflammatory Properties
| Study | Design | Effect |
|---|---|---|
| Kaliora et al. [5] |
10 pts with active CD and 8 healthy controls 2.2 g of mastic daily for 4 weeks |
|
| Kaliora et al. [6] |
10 pts with active CD and 8 healthy controls 2.2 g of mastic daily, 4 weeks |
|
| Papada et al. [7][8] |
60 pts with IBD randomized to either 2.8 g of mastic daily for 3 months or placebo |
|
| Papada et al. [9] |
68 pts with IBD randomized to either 2.8 g of mastic daily for 6 months or placebo |
|
| Amerikanou et al. [10] |
129 pts with IBD—68 randomized to mastic group (2.8 g daily for 6 months for pts in remission and for 3 months for pts in relapse) and 61 to placebo |
|
| Dabos et al. [11] |
148 pts with functional dyspepsia randomized to either mastic 350 mg tid or placebo for 3 weeks |
|
| Kanoni et al. [12] |
98 patients with obesity (BMI ≥ 30 kg/m2) and NAFLD and randomized to either mastic 2.1 g/day or placebo for 6 months |
|
| Moudi et al. [13] |
147 postpartum women randomized to topical application of 15 g mastic for 3 days on episiotomy wound or to placebo |
|
| Triantafyllou et al. [14] |
133 subjects were randomized to either 5 g mastic powder (high dose) or mastic solution for 18 months |
|
| Kartalis et al. [15] |
156 subjects received different doses of mastic for 8 weeks |
|
| Kontogiannis et al. [16] |
27 subject (13 hypertensive) randomized to receive one dose of 2.8 g mastic |
|
| Kottakis et al. [17] |
5 pts with H. Pylori infection and 3 controls treated with 1 g of mastic daily for 2 months |
|
| Dabos et al. [18] |
52 pts with H. Pylori randomized to receive either 350 mg tid of mastic for 14 days (Group A), or 1.05 g tid of mastic (Group B) for 14 days, or pantoprazole 20 mg bd plus mastic 350 mg tid for 14 days (Group C) or pantoprazole 20 mg bd plus amoxicillin 1 g bd plus clarithromycin 500 mg bd for 10 days (Group D) |
|
| Bebb et al. [19] |
8 pts with H. Pylori 1 g mastic four times daily for 14 days |
|
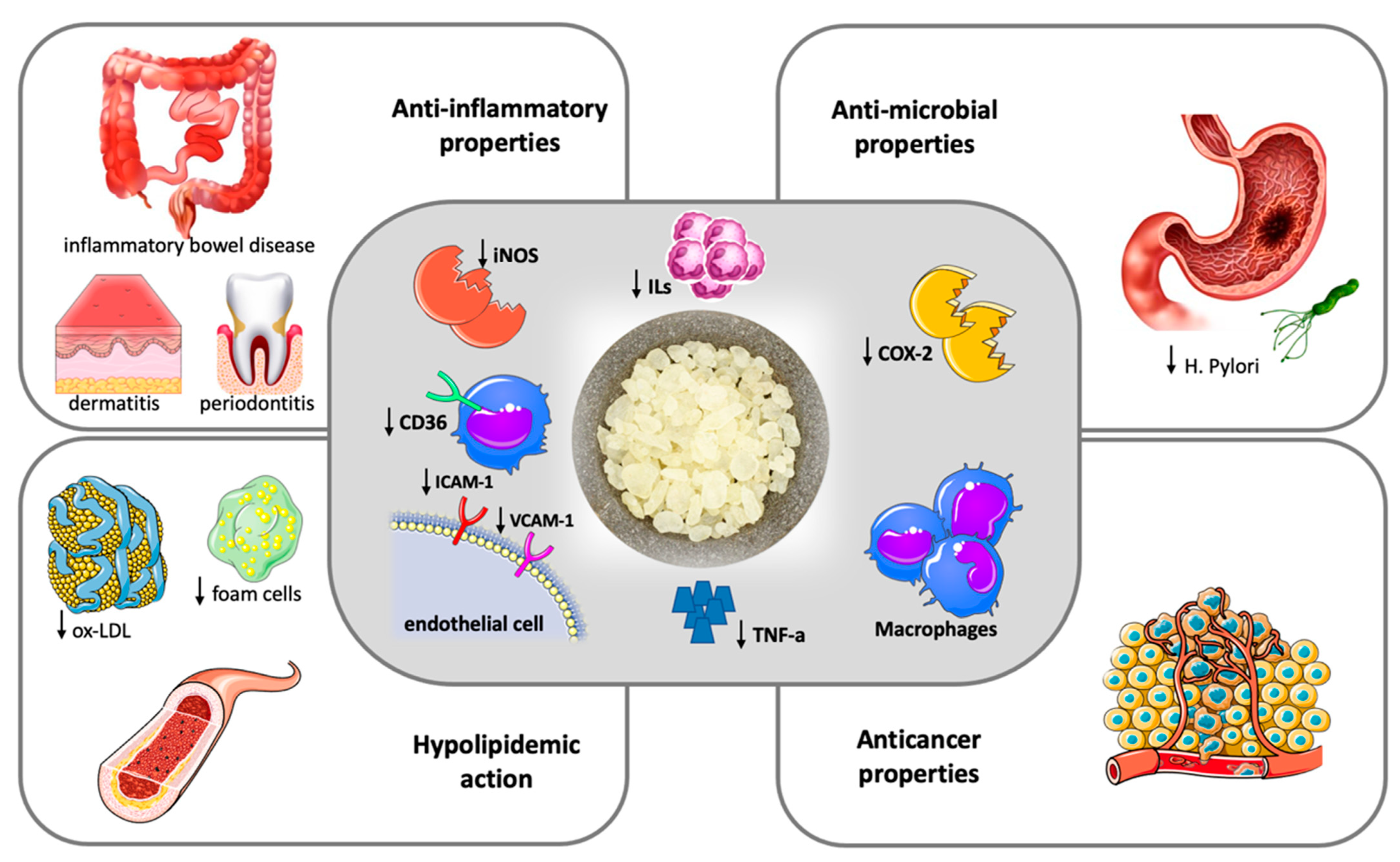 Figure 1. Therapeutic potentials of Chios Mastic.
Figure 1. Therapeutic potentials of Chios Mastic.2. Anti-Oxidant Properties
3. Anti-Atherogenic Properties
4. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/nu14030590
References
- Cronkite, D.A.; Strutt, T.M. The regulation of Inflammation by innate and adaptive lymphocytes. J. Immunol. Res. 2018, 2018, 1467538.
- Libby, P. Inflammatory mechanisms: The molecular basis of inflammation and disease. Nutr. Rev. 2007, 65, S140–S146.
- Zhou, L.; Satoh, K.; Takahashi, K.; Watanabe, S.; Nakamura, W.; Maki, J.; Hatano, H.; Takekawa, F.; Shimada, C.; Sakagami, H. Re-evaluation of anti-inflammatory activity of mastic using activated macrophages. Vivo 2009, 23, 583–590.
- Loizou, S.; Paraschos, S.; Mitakou, S.; Chrousos, G.P.; Lekakis, I.; Moutsatsou, P. Chios mastic gum extract and isolated phytosterol tirucallol exhibit anti-inflammatory activity in human aortic endothelial cells. Exp. Biol. Med. 2009, 234, 553–561.
- Kaliora, A.C.; Stathopoulou, M.G.; Triantafillidis, J.K.; Dedoussis, G.V.; Andrikopoulos, N.K. Chios mastic treatment of patients with active Crohn’s disease clinical research. World J. Gastroenterol. 2007, 13, 748–753.
- Kaliora, A.C.; Stathopoulou, M.G.; Triantafillidis, J.K.; Dedoussis, G.V.; Andrikopoulos, N.K. Alterations in the function of circulating mononuclear cells derived from patients with Crohn’s disease treated withmastic. World J. Gastroenterol. 2007, 13, 6031–6036.
- Papada, E.; Gioxari, A.; Amerikanou, C.; Forbes, A.; Tzavara, C.; Smyrnioudis, I.; Kaliora, A.C. Regulation of faecal biomarkers in inflammatory bowel disease patients treated with oral mastiha (Pistacia lentiscus) supplement: A double-blind and placebo-controlled randomised trial. Phyther. Res. 2019, 33, 360–369.
- Papada, E.; Forbes, A.; Amerikanou, C.; Torović, L.; Kalogeropoulos, N.; Tzavara, C.; Triantafillidis, J.K.; Kaliora, A.C. Antioxidative efficacy of a Pistacia lentiscus supplement and its effect on the plasma amino acid profile in inflammatory bowel disease: A Randomised, double-blind, placebo-controlled trial. Nutrients 2018, 10, 1779.
- Papada, E.; Amerikanou, C.; Torović, L.; Kalogeropoulos, N.; Tzavara, C.; Forbes, A.; Kaliora, A.C. Plasma free amino acid profile in quiescent Inflammatory Bowel Disease patients orally administered with Mastiha (Pistacia lentiscus); A randomised clinical trial. Phytomedicine 2019, 56, 40–47.
- Amerikanou, C.; Dimitropoulou, E.; Gioxari, A.; Papada, E.; Tanaini, A.; Fotakis, C.; Zoumpoulakis, P.; Kaliora, A.C. Linking the IL-17A immune response with NMR-based faecal metabolic profile in IBD patients treated with mastiha. Biomed. Pharmacother. 2021, 138, 111535.
- Dabos, K.J.; Sfika, E.; Vlatta, L.J.; Frantzi, D.; Amygdalos, G.I.; Giannikopoulos, G. Is Chios mastic gum effective in the treatment of functional dyspepsia? A prospective randomised double-blind placebo controlled trial. J. Ethnopharmacol. 2010, 127, 205–209.
- Kanoni, S.; Kumar, S.; Amerikanou, C.; Kurth, M.J.; Stathopoulou, M.G.; Bourgeois, S.; Masson, C.; Kannt, A.; Cesarini, L.; Kontoe, M.S.; et al. Nutrigenetic interactions might modulate the antioxidant and anti-inflammatory status in mastiha-supplemented patients with NAFLD. Front. Immunol. 2021, 12, 1688.
- Moudi, Z.; Edozahi, M.; Emami, S.A.; Asili, J.; Pour, M.S. Effects of mastic oleoresin on wound healing and episiotomy pain: A mixed methods study. J. Ethnopharmacol. 2018, 214, 225–231.
- Triantafyllou, A.; Bikineyeva, A.; Dikalova, A.; Nazarewicz, R.; Lerakis, S.; Dikalov, S. Anti-inflammatory activity of Chios mastic gum is associated with inhibition of TNF-alpha induced oxidative stress. Nutr. J. 2011, 10, 1–9.
- Kartalis, A.; Didagelos, M.; Georgiadis, I.; Benetos, G.; Smyrnioudis, N.; Marmaras, H.; Voutas, P.; Zotika, C.; Garoufalis, S.; Andrikopoulos, G. Effects of Chios mastic gum on cholesterol and glucose levels of healthy volunteers: A prospective, randomized, placebo-controlled, pilot study (chios-mastiha). Eur. J. Prev. Cardiol. 2016, 23, 722–729.
- Kontogiannis, C.; Georgiopoulos, G.; Loukas, K.; Papanagnou, E.D.; Pachi, V.K.; Bakogianni, I.; Laina, A.; Kouzoupis, A.; Karatzi, K.; Trougakos, I.P.; et al. Chios mastic improves blood pressure haemodynamics in patients with arterial hypertension: Implications for regulation of proteostatic pathways. Eur. J. Prev. Cardiol. 2019, 26, 328–331.
- Kottakis, F.; Kouzi-Koliakou, K.; Pendas, S.; Kountouras, J.; Choli-Papadopoulou, T. Effects of mastic gum Pistacia lentiscus var. Chia on innate cellular immune effectors. Eur. J. Gastroenterol. Hepatol. 2009, 21, 143–149.
- Dabos, K.J.; Sfika, E.; Vlatta, L.J.; Giannikopoulos, G. The effect of mastic gum on Helicobacter pylori: A randomized pilot study. Phytomedicine 2010, 17, 296–299.
- Bebb, J.R.; Bailey-Flitter, N.; Ala’Aldeen, D.; Atherton, J.C. Mastic gum has no effect on Helicobacter pylori load in vivo. J. Antimicrob. Chemother. 2003, 52, 522–523.
- Zahouani, Y.; Ben Rhouma, K.; Kacem, K.; Sebai, H.; Sakly, M. Aqueous leaf extract of Pistacia lentiscus improves acute acetic acid-induced colitis in rats by reducing inflammation and oxidative stress. J. Med. Food 2021, 24, 697–708.
- Gioxari, A.; Kaliora, A.C.; Papalois, A.; Agrogiannis, G.; Triantafillidis, J.K.; Andrikopoulos, N.K. Pistacia lentiscus resin regulates intestinal damage and inflammation in trinitrobenzene sulfonic acid-induced colitis. J. Med. Food 2011, 14, 1403–1411.
- Ostovan, M.; Fazljou, S.M.B.; Khazraei, H.; Khodaei, M.A.; Torbati, M. The anti-inflammatory effect of Pistacia lentiscus in a rat model of colitis. J. Inflamm. Res. 2020, 13, 369.
- Kannt, A.; Papada, E.; Kammermeier, C.; D’Auria, G.; Jiménez-Hernández, N.; Stephan, M.; Schwahn, U.; Madsen, A.N.; Østergaard, M.V.; Dedoussis, G.; et al. Mastiha (Pistacia lentiscus) improves gut microbiota diversity, hepatic steatosis, and disease activity in a biopsy-confirmed mouse model of advanced non-alcoholic steatohepatitis and fibrosis. Mol. Nutr. Food Res. 2019, 63, 1688.
- Georgiadis, I.; Karatzas, T.; Korou, L.M.; Katsilambros, N.; Perrea, D. Beneficial health effects of chios gum mastic and peroxisome proliferator-activated receptors: Indications of common mechanisms. J. Med. Food 2015, 18, 1–10.
- Kishimoto, R.; Kato, N.; Koike, M.; Iwashita, N.; Takagi, Y.; Fukuyama, T. Topical treatment with mastic (resin from Pistacia lentiscus) elicits anti-inflammatory and anti-pruritic responses by modulating keratinocyte activation in a mouse model of allergic dermatitis. Phytomedicine 2021, 91, 153679.
- Khedir, S.B.; Bardaa, S.; Chabchoub, N.; Moalla, D.; Sahnoun, Z.; Rebai, T. The healing effect of Pistaciaï lentiscus fruit oil on laser burn. Pharm. Biol. 2017, 55, 1407–1414.
- Sharifi-Rad, M.; Anil Kumar, N.V.; Zucca, P.; Varoni, E.M.; Dini, L.; Panzarini, E.; Rajkovic, J.; Tsouh Fokou, P.V.; Azzini, E.; Peluso, I.; et al. Lifestyle, oxidative stress, and antioxidants: Back and forth in the pathophysiology of chronic diseases. Front. Physiol. 2020, 11, 694.
- Dayem, A.A.; Hossain, M.K.; Lee, S.B.; Kim, K.; Saha, S.K.; Yang, G.M.; Choi, H.Y.; Cho, S.G. The role of reactive oxygen species (ROS) in the biological activities of metallic nanoparticles. Int. J. Mol. Sci. 2017, 18, 120.
- Poznyak, A.V.; Nikiforov, N.G.; Markin, A.M.; Kashirskikh, D.A.; Myasoedova, V.A.; Gerasimova, E.V.; Orekhov, A.N.; Pandey, A.K.; Woolard, M.D.; Eid, A.H. Overview of OxLDL and its impact on cardiovascular health: Focus on atherosclerosis. Front. Pharmacol. 2021, 11, 2248.
- Andrikopoulos, N.K.; Kaliora, A.C.; Assimopoulou, A.N.; Papapeorgiou, V.P. Biological activity of some naturally occurring resins, gums and pigments against in vitro LDL oxidation. Phytother. Res. 2003, 17, 501–507.
- Cosentino-Gomes, D.; Rocco-Machado, N.; Meyer-Fernandes, J.R. Cell signaling through protein kinase C oxidation and activation. Int. J. Mol. Sci. 2012, 13, 10697.
- Dedoussis, G.V.Z.; Kaliora, A.C.; Psarras, S.; Chiou, A.; Mylona, A.; Papadopoulos, N.G.; Andrikopoulos, N.K. Antiatherogenic effect of Pistacia lentiscus via GSH restoration and downregulation of CD36 mRNA expression. Atherosclerosis 2004, 174, 293–303.
- Feng, J.; Han, J.; Freida, S.; Pearce, A.; Silverstein, R.L.; Gotto, A.M.; Hajjar, D.P.; Nicholson, A.C.; Feng, J.; Han, J.; et al. Induction of CD36 expression by oxidized LDL and IL-4 by a common signaling pathway dependent on protein kinase C and PPAR-γ. J. Lipid Res. 2000, 41, 688–696.
- Andreadou, I.; Mitakou, S.; Paraschos, S.; Efentakis, P.; Magiatis, P.; Kaklamanis, L.; Halabalaki, M.; Skaltsounis, L.; Iliodromitis, E.K. “Pistacia lentiscus L.” reduces the infarct size in normal fed anesthetized rabbits and possess antiatheromatic and hypolipidemic activity in cholesterol fed rabbits. Phytomedicine 2016, 23, 1220–1226.
- Triantafyllou, A.; Chaviaras, N.; Sergentanis, T.N.; Protopapa, E.; Tsaknis, J. Chios mastic gum modulates serum biochemical parameters in a human population. J. Ethnopharmacol. 2007, 111, 43–49.
