In this work, a novel heterojunction based on ZnSnO3/ZnO nanofibers was prepared by a simple electrospinning method. The crystal, structural, and surface compositional properties of ZnSnO3 and ZnSnO3/ZnO composite nanofibers were investigated by X-ray diffractometer (XRD), scanning electron microscope (SEM), transmission electron microscope (TEM), X-ray photoelectron spectrometer (XPS), and Brunauer–Emmett–Teller (BET). Compared to pure ZnSnO3 nanofibers, the ZnSnO3/ZnO heterostructure nanofibers had high sensitivity and selectivity response with the fast response toward ethanol gas at low operational temperature.
- electrospinning
- ZnSnO3/ZnO nanofibers
- sensing performance
- n-n heterojunction
1. Introduction
2. Experimental Section
2.1. Material Preparation
2.2. Characterization
2.3. Gas-Sensitive Performance Measurement
3. Results and Discussion
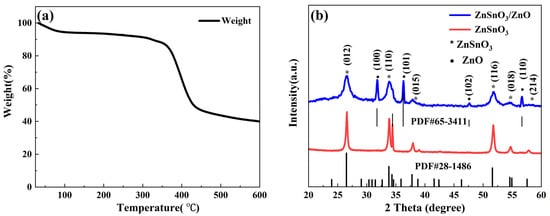
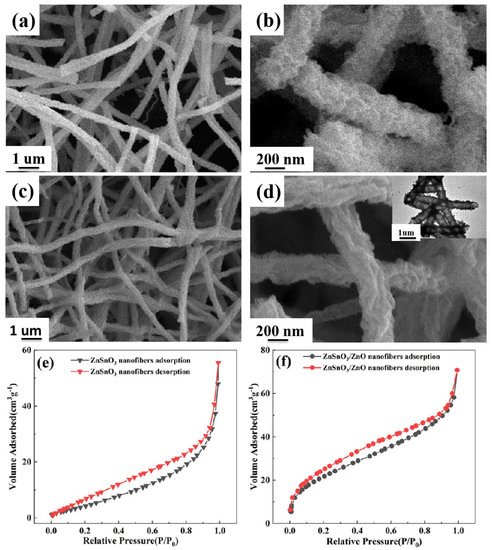
3.1. Microstructure and Morphology
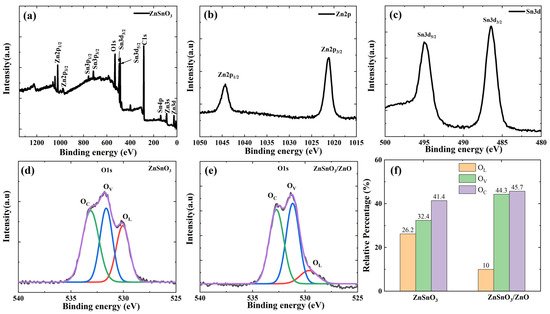
| Sample | OL (%) | OV (%) | OC (%) |
|---|---|---|---|
| ZnSnO3 | 26.2% | 32.4% | 41.4% |
| ZnSnO3/ZnO | 10% | 44.3% | 45.7% |
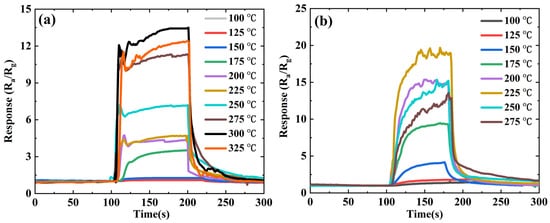
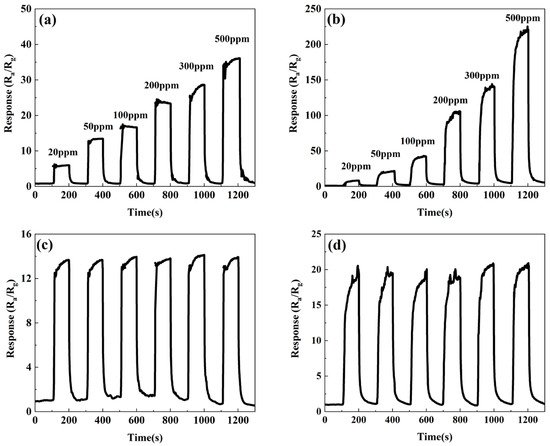
3.2. Gas-Sensitive Performances of ZnSnO3/ZnO Nanofibers
This entry is adapted from the peer-reviewed paper 10.3390/met12020196
References
- Ma, J.; Xiao, X.; Zou, Y.; Ren, Y.; Zhou, X.; Yang, X.; Cheng, X.; Deng, Y. A general and straightforward route to noble metal-decorated mesoporous transition-metal oxides with enhanced gas sensing performance. Adv. Funct. Mater. 2019, 15, e1904240.
- Gomaa, M.M.; Sayed, M.H.; Patil, V.L.; Boshta, M.; Patil, P.S. Gas sensing performance of sprayed NiO thin films toward NO2 gas. J. Alloys Compd. 2021, 885, 160908.
- Wang, Y.; Zeng, Y.; Wang, L.; Lou, Z.; Qiao, L.; Tian, H.; Zheng, W. Ultrathin nanorod-assembled SnO2 hollow cubes for high sensitive n-butanol detection. Sens. Actuators B Chem. 2019, 283, 693–704.
- Kou, X.; Xie, N.; Chen, F.; Wang, T.; Guo, L.; Wang, C.; Wang, Q.; Ma, J.; Sun, Y.; Zhang, H.; et al. Superior acetone gas sensor based on electrospun SnO2 nanofibers by Rh doping. Sens. Actuators B Chem. 2018, 256, 861–869.
- Kim, J.H.; Mirzaei, A.; Kim, H.W.; Kim, S.S. Low voltage driven sensors based on ZnO nanowires for room temperature detection of NO2 and CO gases. ACS Appl. Mater. Interfaces 2019, 1, 24172–24183.
- Geng, W.; Ma, Z.; Yang, J.; Duan, L.; Li, F.; Zhang, Q. Pore size dependent acetic acid gas sensing performance of mesoporous CuO. Sens. Actuators B Chem. 2021, 334, 129639.
- Jain, K.; Pant, R.P.; Lakshmi-kumar, S.T. Effect of Ni doping on thick film SnO2 gas sensor. Sens. Actuators B Chem. 2006, 113, 823–829.
- Yin, Y.; Shen, Y.; Zhou, P.; Lu, R.; Li, A.; Zhao, S.; Liu, W.; Wei, D.; Wei, K. Fabrication, characterization and n-propanol sensing properties of perovskite-type ZnSnO3 nanospheres based gas sensor. Appl. Surf. Sci. 2020, 509, 145335.
- Wang, Z.; Miao, J.; Zhang, H.; Wang, D.; Sun, J. Hollow cubic ZnSnO3 with abundant oxygen vacancies for H2S gas sensing. J. Hazard. Mater. 2020, 391, 122226.
- Feng, G.Q.; Che, Y.H.; Song, C.W.; Xiao, J.K.; Fan, X.F.; Sun, S.; Huang, G.H.; Ma, Y.C. Morphology-controlled synthesis of ZnSnO3 hollow spheres and their n-butanol gas-sensing performance. Ceram. Int. 2021, 47, 2471–2482.
- Wang, X.Y.; Zhu, X.T.; Tao, T.; Leng, B.X.; Xu, W.; Mao, L.H. Structural inheritance and change from ZnSn(OH)6 to ZnSnO3 compounds used for ethanol sensors: Effects of oxygen vacancies, temperature and UV on gas-sensing properties. J. Alloys Compd. 2020, 829, 154445.
- Wang, X.; Liu, Y.; Ding, B.; Li, H.; Zhu, X.; Xia, M. Influence of the addition of nano-TiO2 and ZnO on the sensing performance of micro-ZnSnO3 ethanol sensors under UV illumination. Sens. Actuators B Chem. 2018, 276, 211–221.
- Xun, H.; Zhang, Z.; Yu, A.; Yi, J. Remarkably enhanced hydrogen sensing of highly-ordered SnO2-decorated TiO2 nanotubes. Sens. Actuators B Chem. 2018, 273, 983–990.
- Shao, S.; Chen, X.; Chen, Y.; Lai, M.; Che, L. Ultrasensitive and highly selective detection of acetone based on 3-SnO2 corrugated nanofibers. Appl. Surf. Sci. 2019, 473, 902–911.
- Yan, Y.; Liu, J.Y.; Zhang, H.S.; Song, D.L.; Li, J.Q.; Yang, P.P.; Zhang, M.L.; Wang, J. One-pot synthesis of cubic ZnSnO3/ZnO heterostructure composite and enhanced gas-sensing performance. J. Alloys. Compd. 2019, 780, 193–201.
- Yu, S.W.; Jia, X.H.; Yang, J.; Wang, S.Z.; Li, Y.; Song, H.J. Highly sensitive ethanol gas sensor based on CuO/ZnSnO3 heterojunction composites. Mater. Lett. 2021, 291, 129531.
- Zhang, J.T.; Jia, X.H.; Lian, D.D.; Yang, J.; Wang, S.Z.; Li, Y.; Song, H.J. Enhanced selective acetone gas sensing performance by fabricating ZnSnO3/SnO2 concave microcube. Appl. Surf. Sci. 2021, 542, 148555.
- Cheng, P.; Lv, L.; Wang, Y.; Zhang, B.; Zhang, Y.; Zhang, Y.; Lei, Z.; Xu, L. SnO2/ZnSnO3 double-shelled hollow microspheres based high-performance acetone gas sensor. Sens. Actuators B Chem. 2021, 332, 129212.
- Chen, Y.; Yu, L.; Li, Q.; Wu, Y.; Li, Q.; Wang, T. An evolution from 3D face-centered-cubic ZnSnO3 nanocubes to 2D orthorhombic ZnSnO3 nanosheets with excellent gas sensing performance. Nanotechnology 2012, 23, 415501.
- Zhang, D.; Zhang, Y.; Fan, Y.; Luo, N.; Cheng, Z.; Xu, J. Micro-spherical ZnSnO3 material prepared by microwave-assisted method and its ethanol sensing properties. Chin. Chem. Lett. 2020, 31, 2087–2090.
- Li, L.; Peng, S.; Wang, J.; Cheah, Y.L.; Teh, P.; Ko, Y.; Wong, C.; Srinivasan, M. Facile Approach to Prepare Porous CaSnO3 Nanotubes via a Single Spinneret Electrospinning Technique as Anodes for Lithium Ion Batteries. ACS Appl. Mater. Int. 2012, 4, 6005.
- Qin, Y.-L.; Zhang, F.-F.; Du, X.-C.; Huang, G.; Liu, Y.-C.; Wang, L.-M. Controllable synthesis of cube-like ZnSnO3@TiO2 nanostructures as lithium ion battery anodes. J. Mater. Chem. A 2015, 3, 2985–2990.
- Duan, J.-F.; Hou, S.-C.; Chen, S.-G.; Duan, H.-G. Synthesis of amorphous ZnSnO3 hollow nanoboxes and their lithium storage properties. Mater. Lett. 2014, 122, 261–264.
- Chen, O.; Wang, Y.; Wang, M.; Ma, S.; Wang, P.; Zhang, G.; Chen, W.; Jiao, H.; Liu, L.; Xu, X. Enhanced acetone sensor based on Au functionalized In-doped ZnSnO3 nanofibers synthesized by electrospinning method. J. Colloid Interf. Sci. 2019, 543, 285–299.
- Lian, D.; Shi, B.; Dai, R.; Jia, X.H.; Wu, X.Y. Synthesis and enhanced acetone gas-sensing performance of ZnSnO3/SnO2 hollow urchin nanostructures. J. Nanopart. Res. 2017, 19, 401.
- Liu, J.; Wang, T.; Wang, B.; Sun, P.; Yang, Q.; Liang, X.; Song, H.; Lu, G. Highly sensitive and low detection limit of ethanol gas sensor based on hollow ZnO/SnO2 spheres composite material. Sens. Actuators B Chem. 2017, 245, 551–559.
- Bai, S.L.; Tian, Y.; Zhao, Y.H.; Fu, H.; Tang, P.G.; Luo, R.X.; Li, D.Q.; Chen, A.F.; Liu, C.C. Construction of 3 hierarchical microspheres decorated with NiO nanosheets for formaldehyde sensing. Sens. Actuators B Chem. 2018, 259, 908–916.
- Al-Hashem, M.; Akbar, S.; Morris, P. Role of Oxygen Vacancies in Nanostructured Metal-Oxide Gas Sensors: A Review. Sens. Actuators B Chem. 2019, 301, 126845.
