Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Cardiac & Cardiovascular Systems
Pulmonary hypertension (PH) is a serious hemodynamic condition, characterized by increased pulmonary vascular resistance (PVR), leading to right heart failure (HF) and death when not properly treated. The prognosis of PH depends on etiology, hemodynamic and biochemical parameters, as well as on response to specific treatment.
- pulmonary hypertension
- chronic thromboembolic pulmonary hypertension
- pulmonary arterial hypertension
1. Introduction
Pulmonary hypertension (PH) is a progressive, heterogenous disease, characterized by increased pulmonary vascular resistance (PVR), subsequently leading to elevated pulmonary arterial pressure (PAP) and increased workload of the right ventricle (RV). The RV adapts to the pathological afterload by increasing wall thickness and contractility. However, the compensatory mechanisms may fail, resulting in right heart failure (HF) and death, if not properly treated.
Consistent with the European Society of Cardiology (ESC)/European Respiratory Society (ERS) Guidelines, there are five groups of PH, according to clinical and pathophysiological criteria. Group 1 contains idiopathic pulmonary arterial hypertension (iPAH), as well as drug-induced PAH and all heritable forms of PAH. Group 2 is PH secondary to left-sided heart failure. PH in group 3 is caused by lung disease and/or hypoxia, and group 4 is chronic thromboembolic pulmonary hypertension (CTEPH). Group 5 consists of PH due to uncertain multifactorial mechanisms. Targeted pharmacological or interventional treatment can be offered to patients diagnosed with PAH and CTEPH, respectively [1][2]. The prognosis of PH varies broadly and depends mostly on etiology of PH, but is also based on hemodynamic and biochemical parameters, which indicate the severity of right ventricular failure, as well as on response to specific treatment. Early recognition of the disease and risk stratification seem to be crucial to identifying patients at high risk and optimizing therapeutic management. Thus, biomarkers may specifically indicate the disease and provide information about the disease stage and treatment response in a relatively easily accessible and noninvasive way. The search for novel emerging biomarkers is still ongoing, resulting in a few potential biomarkers mirroring numerous pathophysiological courses. The main focus is on detecting and quantifying abnormal adaptations and remodeling of the right heart in response to chronic pulmonary circulation impairment. However, in the natural course of PH and right ventricular HF, tissue damage, fibrosis, inflammation, and endothelial dysfunction seem to be also crucial underlying mechanisms, which may be included in noninvasive biomarker assessment (Figure 1). In the present article, we review circulating biomarkers related to different mechanisms underlying the precapillary PH and describe the potential application for them, highlighting their limitations and necessity for further investigation.
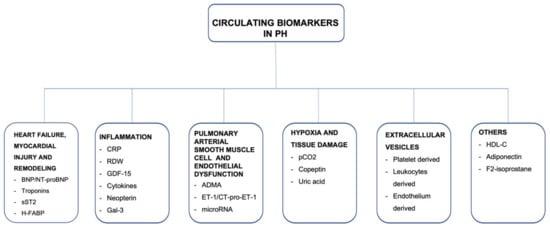
Figure 1. A summary of circulating biomarkers in precapillary pulmonary hypertension.
2. Biomarkers Related to Heart Failure, Myocardial Injury, and Remodeling
In PH, elevated PVR and PAP lead to hemodynamic stress, myocardial strain, and stretching of the heart. Consequently, this condition results in the release of the molecular mediators, indicative for numerous cardiovascular diseases with additional prognostic value. Several markers associated with HF, myocardial injury, and myocardial remodeling, such as natriuretic peptides, cardiac troponins, soluble ST2, and heart-type fatty acid-binding protein, have been investigated in a cohort of patients with precapillary PH.
2.1. Natriuretic Peptides
Brain-type natriuretic peptide (BNP) is produced as an inactive precursor (proBNP), then converted into the active form N-terminal-pro brain-type natriuretic peptide (NT-proBNP) and released from cardiomyocytes. Due to the longer half-life of NT-proBNP compared to BNP, NT-proBNP is preferred in clinical practice as a marker of heart overload and myocardial dysfunction [3][4]. NT-proBNP remains a well-established and widely used biomarker in numerous cardiovascular diseases. It is released in response to ventricular wall stress and myocardial hypoxia or ischemia. NT-proBNP is mostly used in the diagnostic process of patients with acute or chronic HF as well as in predicting prognosis of those patients [5]. In PH patients, serum NT-proBNP levels correlate with right heart dysfunction and provide prognostic information at diagnosis and during follow-up assessment [6][7][8]. However, due to the high variability of NT-proBNP levels and its possible inadequate correlation with hemodynamic parameters and exercise capacity, it should only be interpreted in the clinical context [6]. At present, NT-proBNP is a crucial element of risk stratification in PAH patients and is addressed in both the risk score developed from the REVEAL registry (Registry to Evaluate Early and Long-Term PAH Disease Management) [9][10] and in the risk stratification method proposed by ESC/ERS guidelines [2]. Consistent with REVEAL registry data, a baseline NT-proBNP level of ≤340 ng/L is a strong predictor of improved survival up to 5 years in PAH patients [10]. In slightly different terms, the ESC/ERS guidelines classify NT-proBNP concentrations as low (<5%), intermediate (5–10%), or high (>10%) risk of 1-year mortality, by using specific thresholds of 300 and 1400 ng/L [2]. A significant decrease in NT-proBNP levels among patients with PAH is associated with the response to targeted medical therapy [11][12]. In CTEPH patients, BNP may not only reflect the degree of RV dysfunction and hemodynamic severity of the disease, but also facilitate to assess the effect of pulmonary endarterectomy (PEA) [13], with estimated BNP baseline cut-off values predictive of worse postoperative survival [14]. Furthermore, both balloon pulmonary angioplasty (BPA) as well as pharmacological treatment result in a decrease of NT-proBNP concentration [15][16][17]. In patients treated with BPA, a reduction in NT-proBNP concentration is associated with a significant decrease in mean PAP and PVR, thereby indicating the procedural success of BPA [15]. Table 1 and Table 2 presents changes in BNP and NT-proBNP concentrations before and after BPA treatment in the hitherto published case series.
Table 1. Changes in BNP levels in CTEPH patients before and after BPA treatment in the hitherto published case series.
| Studies | No. of Patients (n) | No. of BPA Sessions (n) | BNP before BPA (pg/mL) | BNP after BPA (pg/mL) | p |
|---|---|---|---|---|---|
| Sugimura et al. [18] | 12 | NR | 335 ± 105 | 16 ± 11 | S |
| Kimura et al. [19] | 66 | 446 | 237.7 ± 475.7 | 45.2 ± 47.6 | S |
| Ogo et al. [20] | 80 | 385 | 227 ± 282 | 48 ± 57 | S |
| Yamasaki et al. [21] | 20 | 2.7 per pt | 66.5 ± 61.3 | 33.8 ± 30.0 | S |
| Aoki et al. [22] | 24 | 113 | 112 (49–199) | 27.5 (14.6–58.4) | S |
| Inami et al. [23] | 103 | 350 | 94 (42–232) | 61 (39–150) | S |
Data are presented as mean ± SD or median and (IQR), S—p < 0.05; BPA—balloon pulmonary angioplasty; BNP—brain natriuretic peptide.
Table 2. Changes in NT-proBNP levels in CTEPH patients before and after BPA treatment in the hitherto published case series.
| Studies | No. of Patients (n) | No. of BPA Sessions (n) | NT-proBNP before BPA (pg/mL) | NT-proBNP after BPA (pg/mL) | p |
|---|---|---|---|---|---|
| Kurzyna et al. [24] | 31 | 117 | 2571 ± 2719 | 634 ± 697 | S |
| Olsson et al. [25] | 66 | 446 | 504 (233–1676) | 242 (109–555) | S |
| Araszkiewicz et al. [26] | 15 | 71 | 1554.8 ± 1541.3 | 537 ± 642.6 | S |
| Darocha et al. [27] | 70 | 377 | 1307 (510–3294) | 206 (83–531) | S |
| Gerges et al. [28] | 45 | 6 (4–10) per pt | 579 (182–1385) | 198 (70–429) | S |
Data are presented as mean ± SD or median and (IQR), S—p < 0.05; BPA—balloon pulmonary angioplasty; NT-proBNP—N-terminal-pro brain-type natriuretic peptide.
2.2. Cardiac Troponins
So far, both cardiac troponin I (cTnI) and T (cTnT) are the principal biomarkers for the detection of myocardial damage and key factors in the diagnosis of acute myocardial infarction [29]. In addition, the development of high-sensitivity assays has made it possible to detect troponin concentrations and their association with morbidity and mortality in many chronic diseases, such as heart failure, coronary artery disease, or chronic kidney disease [30][31][32][33]. Although the underlying mechanisms for troponin release in some conditions remain not completely elucidated, in most cases troponins levels correlate with markers of left heart structural abnormalities and other markers related to left HF. In contrast, the mechanism of troponin release in PH patients seems to be associated with RV pathology, seemingly caused by demand–perfusion mismatch or microcirculation impairment. These theories are supported by the results from several research studies, in which significant correlations between troponins concentration and hemodynamic parameters, including mean PAP (mPAP), mixed venous oxygen saturation (mvSatO2), and RV ejection fraction, were identified [34][35]. Moreover, both cTnT and cTnI concentrations were associated with worse outcomes in mixed cohorts of PH patients [34][36]. Thereby, ESC guidelines indicate that for comprehensive prognostic assessment and risk stratification, troponin levels should be measured at the diagnosis of PAH, then at least once a year or whenever the patient presents with clinical worsening [2]. In CTEPH patients undergoing interventional treatment with BPA, high-sensitivity cTnT concentration decreases stepwise under therapy, signifying a reduction of ongoing myocardial damage due to decreased right ventricular afterload after BPA therapy [37]. Thus, also in the CTEPH patient population, troponins can be a useful marker to monitor the progress of treatment.
2.3. Soluble ST2
Soluble ST2 (sST2) protein is another promising biomarker in PH patients. Protein ST2 belongs to the Toll interleukin 1 receptor superfamily and exists in two isoforms: transmembrane ST2 ligand (ST2L) and soluble ST2 (sST2), which circulates in the blood. The transmembrane form is expressed mainly on inflammatory cells and takes part in strengthening of the immune response of Th2 lymphocytes. However, it is also exposed in cardiomyocytes and endothelium [38]. The ligand for ST2 is interleukin 33 (IL-33), whose expression increases due to mechanical overload and ischemia of cardiomyocytes [38]. The paracrine IL-33/ST2L system plays a protective role, counteracting fibrosis and myocardial hypertrophy. The sST2 protein, which prevents IL-33 binding to the ST2L, is responsible for interrupting this protective action. The balance between both isoforms of ST2 protein ensures the correct biological effect [38][39]. The increase of sST2 concentration in plasma is associated with cardiac remodeling and hemodynamic stress [38][39][40]. Besides natriuretic peptide family and cardiac troponins, sST2 protein may be an additional biomarker for adverse outcomes in cohorts of patients with acute and chronic heart failure [41][42][43]. The sST2 level above 35 ng/mL in patients with HF is associated with higher risk of adverse events, defined as hospitalization or death in one year, in comparison to subjects with sST2 level below this value [44][45][46]. At present, there is increasing evidence of the use of the sST2 protein for risk stratification in patients with RV failure due to PH. In different types of PH, higher sST2 levels are linked to the remodeling of the RV [47]. In a study involving 100 patients diagnosed with PAH or CTEPH, significant correlations between sST2 and cardiac index (CI), mean right atrial pressure (mRAP), PVR, mvSatO2, NT-proBNP concentration, and 6 min walking distance (6MWD) were noticed [48]. These observations are consistent with those from other studies conducted in smaller populations of patients with precapillary PH [49][50]. Moreover, sST2 has been assessed as a marker of therapy response in 57 CTEPH patients, treated with BPA. In detail, the median sST2 concentration decreased to the range of control group after interventional treatment, but it was not related to the individual grade of response to BPA therapy [51]. In another study, sST2 concentration changed significantly in 37 CTEPH patients treated with BPA in the immediate postprocedural period. Interestingly, in patients who experienced complications in the postprocedural period, the baseline sST2 levels were significantly higher in comparison to those without complications. Thereby, sST2 could be beneficial for preoperative risk assessment in these patients. Furthermore, sST2 concentration significantly increased early after BPA procedure, irrespective of complications. In contrast, no analogous changes in NT-proBNP levels were noticed, which may be suggestive of an additional noncardiac source of sST2 in CTEPH patients. Therefore, in PH, sST2 as a complex biomarker may reflect not only the heart condition but also pulmonary vascular system and lung tissue [52]. Table 3 summarizes the main differences between sST2 and NT-proBNP in management of PH.
2.4. Heart-Type Fatty Acid-Binding Protein
Heart-type fatty acid-binding protein (H-FABP) is a low-molecular-weight protein, which is expressed in the cytosol of cardiomyocytes. H-FABP appears to be a marker of injury of cardiomyocytes and is also considered as additional biomarker for early diagnosis of acute coronary syndrome [53]. Of note, Puls et al. described H-FABP as a suitable marker for risk assessment in patients with acute pulmonary embolism [54]. However, there are only limited data about the application of H-FABP in PH patients. Lankeit et al. examined the role of H-FABP in risk stratification in CTEPH patients. The results of the study revealed H-FABP as an independent marker of adverse outcomes, defined as persistent PH after PEA, CTEPH-related death, or lung transplantation [55]. In contrast, Mirna et al. identified H-FABP as an indicator of postcapillary, but not precapillary PH [56]. Although these initial reports appear promising, further studies enrolling a larger population are needed in order to evaluate existing discrepancies.
3. Markers of Inflammation
There is increasing evidence that inflammation processes have great significance in the pathophysiology of PH, being involved in pulmonary arterial remodeling. However, the inflammatory component could also mirror organs distress caused by a certain degree of ischemia and elevated sympathetic drive as a consequence of limited cardiac output. A variety of both anti- and proinflammatory molecules have been investigated as potential biomarkers in cohorts of PH patients.
3.1. C-Reactive Protein
C-reactive protein (CRP), a widely used marker of inflammation, is broadly established as a predictor of numerous cardiovascular diseases, including different types of PH. In PAH, significant correlations between CRP and RAP, 6MWD as well as NYHA class were revealed [57]. Scognamiglio et al. observed that in patients with congenital heart disease-associated PAH (CHD-PAH), CRP concentration was commonly increased and the CRP elevation above 10 mg/mL was associated with around four times greater risk of death [58]. Wynants et al. examined CRP effects on pulmonary vascular cells in CTEPH patients. They revealed that CRP could play a role in chronic obstruction of pulmonary arteries by stimulating endothelial dysfunction, vascular remodeling, and in situ thrombosis [59]. In CTEPH patients, plasma CRP concentrations were related to tissue factor (TF) antigen, suggesting the connection between thrombosis and inflammatory processes in the pathogenesis of CTEPH [60]. Moreover, Quarck et al. observed that CRP levels were elevated among CTEPH patients and significantly decreased 12 months after PEA [57]. However, due to the reported elevated CRP levels in many clinical conditions, including various cardiovascular diseases, its potential use in the diagnosis and monitoring of PH patients remains limited.
3.2. Red Blood Cell Distribution Width
Red blood cell distribution width (RDW) is a laboratory biomarker of heterogeneity, regularly measured in standard blood analyses. Elevated RDW levels are the sign of anisocytosis, which is linked with underlying inflammatory processes [61]. So far it is known that RDW may be a predictor of survival in various cardiovascular diseases, such as coronary artery disease [62], chronic heart failure [63], or acute pulmonary embolism [64]. Moreover, RDW is a prognostic marker of PH of different etiologies, and an association with mortality in a cohort of PH patients was noticed [65][66]. In study involving 77 inoperable CTEPH and PAH patients, the decrease in RDW level after initiation or escalation of specific treatment was linked with good treatment response and improved prognosis [67]. Similar results were previously obtained by Wang et al. in 56 CTEPH patients [68]. However, there is a need for prospective studies to better assess the prognostic value of RDW in cohorts of patients with precapillary PH.
3.3. Growth Differentiation Factor-15
Growth differentiation factor-15 (GDF-15) is a member of the TGFβ superfamily. GDF-15 is exposed in various types of cells in response to tissue damage, ischemia, or shear stress [69][70]. So far, GDF-15 has been indicated as a nonspecific marker of systemic stress in several cardiovascular diseases [71]. In PH, GDF-15 is present in the plexiform lesions in the pulmonary vascular bed and may thus affect both apoptosis and proliferation of endothelial cells [69]. Nickel et al. revealed that in 22 patients with iPAH, GDF-15 levels were associated with hemodynamic parameters such as RAP and pulmonary capillary wedge pressure (PCWP), as well as with biochemical parameters, such as NT-proBNP concentration. However, there were no significant changes in median GDF-15 levels measured prior to beginning of specific therapy and at three- or six-month follow-up [72]. Furthermore, in a study by Meadows et al. in patients with scleroderma and associated PAH, GDF-15 was a marker of reduced survival and correlated with NT-proBNP levels and right ventricular systolic pressure assessed by transthoracic echocardiography [73]. The observations mentioned above brightly propose that GDF-15 could be a prognostic factor in PAH. GDF-15 was also assessed as a marker in therapy response in CTEPH patients treated with BPA. Kriechbaum et al. revealed no significant changes before and after BPA treatment, but there was a correlation between delta change in GDF-15 levels and the change in CI and RAP. In addition, a low concentration of GDF-15 measured at baseline indicated responders to the BPA therapy at the follow-up [51].
3.4. Cytokines
Various cytokines are considered crucial inflammatory mediators in numerous conditions, including PH. In a study conducted by Soon et al., serum levels of several interleukins (IL), such as IL-1, IL-2, IL-4, IL-6, IL-8, Il-10, and IL-12p70, and tumor necrosis factor-α (TNFα) were higher in patients with PAH in comparison to a group of healthy controls. From the ILs mentioned above, IL-6, IL-8, IL-10, and IL-12p70 were prognostic factors of poor survival in iPAH and familial PAH [74]. These data are consistent with results obtained by Selimovic et al., which revealed significantly higher levels of IL-6, transforming growth factor β1 (TGFβ1), platelet-derived growth factor (PDGF), and vascular endothelial growth factor (VEGF) in PAH patients compared to controls. Moreover, in this study, a significant association between IL-6 and mortality was observed [75]. Similar observations were previously revealed by Langer et al. in a cohort of CTEPH patients [76]. Elevated levels of IL-6, IL-8, and TNFα were observed in CTEPH patients before PEA. Hence, both IL-6 and Il-8 presented a noticeable peak immediately after PEA, whereas TNFα levels significantly decreased within 24 h after the procedure [76]. What is more, in a study conducted by Zabini et al., significant correlations of IL-6 and hemodynamic parameters and exercise capacity were observed [77].
This entry is adapted from the peer-reviewed paper 10.3390/jcm11020383
References
- Galie, N.; McLaughlin, V.V.; Rubin, L.J.; Simonneau, G. An overview of the 6th World Symposium on Pulmonary Hypertension. Eur. Respir. J. 2019, 53, 1802148.
- Galie, N.; Humbert, M.; Vachiery, J.L.; Gibbs, S.; Lang, I.; Torbicki, A.; Simonneau, G.; Peacock, A.; Noordegraaf, A.V.; Beghetti, M.; et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur. Respir. J. 2015, 46, 903–975.
- Leuchte, H.H.; El Nounou, M.; Tuerpe, J.C.; Hartmann, B.; Baumgartner, R.A.; Vogeser, M.; Muehling, O.; Behr, J. N-terminal pro-brain natriuretic peptide and renal insufficiency as predictors of mortality in pulmonary hypertension. Chest 2007, 131, 402–409.
- Williams, M.H.; Handler, C.E.; Akram, R.; Smith, C.J.; Das, C.; Smee, J.; Nair, D.; Denton, C.P.; Black, C.M.; Coghlan, J.G. Role of N-terminal brain natriuretic peptide (N-TproBNP) in scleroderma-associated pulmonary arterial hypertension. Eur. Heart J. 2006, 27, 1485–1494.
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.; Coats, A.J.; Falk, V.; Gonzalez-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. J. Heart Fail. 2016, 18, 891–975.
- Berghaus, T.M.; Kutsch, J.; Faul, C.; von Scheidt, W.; Schwaiblmair, M. The association of N-terminal pro-brain-type natriuretic peptide with hemodynamics and functional capacity in therapy-naive precapillary pulmonary hypertension: Results from a cohort study. BMC Pulm. Med. 2017, 17, 167.
- Souza, R.; Jardim, C.; Fernandes, C.J.C.; Lapa, M.S.; Rabelo, R.; Humbert, M. NT-proBNP as a tool to stratify disease severity in pulmonary arterial hypertension. Respir. Med. 2007, 101, 69–75.
- Fijalkowska, A.; Kurzyna, M.; Torbicki, A.; Szewczyk, G.; Florczyk, M.; Pruszczyk, P.; Szturmowicz, M. Serum N-terminal brain natriuretic peptide as a prognostic parameter in patients with pulmonary hypertension. Chest 2006, 129, 1313–1321.
- Benza, R.L.; Gomberg-Maitland, M.; Elliott, C.G.; Farber, H.W.; Foreman, A.J.; Frost, A.E.; McGoon, M.D.; Pasta, D.J.; Selej, M.; Burger, C.D.; et al. Predicting Survival in Patients With Pulmonary Arterial Hypertension: The REVEAL Risk Score Calculator 2.0 and Comparison With ESC/ERS-Based Risk Assessment Strategies. Chest 2019, 156, 323–337.
- Benza, R.L.; Miller, D.P.; Barst, R.J.; Badesch, D.B.; Frost, A.E.; McGoon, M.D. An evaluation of long-term survival from time of diagnosis in pulmonary arterial hypertension from the REVEAL Registry. Chest 2012, 142, 448–456.
- Chin, K.M.; Rubin, L.J.; Channick, R.; Di Scala, L.; Gaine, S.; Galie, N.; Ghofrani, H.A.; Hoeper, M.M.; Lang, I.M.; McLaughlin, V.V.; et al. Association of N-Terminal Pro Brain Natriuretic Peptide and Long-Term Outcome in Patients With Pulmonary Arterial Hypertension. Circulation 2019, 139, 2440–2450.
- Galie, N.; Jansa, P.; Pulido, T.; Channick, R.N.; Delcroix, M.; Ghofrani, H.A.; Le Brun, F.O.; Mehta, S.; Perchenet, L.; Rubin, L.J.; et al. SERAPHIN haemodynamic substudy: The effect of the dual endothelin receptor antagonist macitentan on haemodynamic parameters and NT-proBNP levels and their association with disease progression in patients with pulmonary arterial hypertension. Eur. Heart J. 2017, 38, 1147–1155.
- Nagaya, N.; Ando, M.; Oya, H.; Ohkita, Y.; Kyotani, S.; Sakamaki, F.; Nakanishi, N. Plasma brain natriuretic peptide as a noninvasive marker for efficacy of pulmonary thromboendarterectomy. Ann. Thorac. Surg. 2002, 74, 180–184.
- Surie, S.; Reesink, H.J.; van der Plas, M.N.; Hardziyenka, M.; Kloek, J.J.; Zwinderman, A.H.; Bresser, P. Plasma brain natriuretic peptide as a biomarker for haemodynamic outcome and mortality following pulmonary endarterectomy for chronic thromboembolic pulmonary hypertension. Interact. Cardiovasc. Thorac. Surg. 2012, 15, 973–978.
- Kriechbaum, S.D.; Wiedenroth, C.B.; Wolter, J.S.; Hutz, R.; Haas, M.; Breithecker, A.; Roller, F.C.; Keller, T.; Guth, S.; Rolf, A.; et al. N-terminal pro-B-type natriuretic peptide for monitoring after balloon pulmonary angioplasty for chronic thromboembolic pulmonary hypertension. J. Heart Lung Transplant. 2018, 37, 639–646.
- Sadushi-Kolici, R.; Jansa, P.; Kopec, G.; Torbicki, A.; Skoro-Sajer, N.; Campean, I.A.; Halank, M.; Simkova, I.; Karlocai, K.; Steringer-Mascherbauer, R.; et al. Subcutaneous treprostinil for the treatment of severe non-operable chronic thromboembolic pulmonary hypertension (CTREPH): A double-blind, phase 3, randomised controlled trial. Lancet Respir. Med. 2019, 7, 239–248.
- Ghofrani, H.A.; Galie, N.; Grimminger, F.; Grunig, E.; Humbert, M.; Jing, Z.C.; Keogh, A.M.; Langleben, D.; Kilama, M.O.; Fritsch, A.; et al. Riociguat for the treatment of pulmonary arterial hypertension. N. Engl. J. Med. 2013, 369, 330–340.
- Sugimura, K.; Fukumoto, Y.; Satoh, K.; Nochioka, K.; Miura, Y.; Aoki, T.; Tatebe, S.; Miyamichi-Yamamoto, S.; Shimokawa, H. Percutaneous transluminal pulmonary angioplasty markedly improves pulmonary hemodynamics and long-term prognosis in patients with chronic thromboembolic pulmonary hypertension. Circ. J. 2012, 76, 485–488.
- Kimura, M.; Kohno, T.; Kawakami, T.; Kataoka, M.; Inohara, T.; Takei, M.; Tsugu, T.; Murata, M.; Maekawa, Y.; Fukuda, K. Balloon pulmonary angioplasty attenuates ongoing myocardial damage in patients with chronic thromboembolic pulmonary hypertension. Int. J. Cardiol. 2016, 207, 387–389.
- Ogo, T.; Fukuda, T.; Tsuji, A.; Fukui, S.; Ueda, J.; Sanda, Y.; Morita, Y.; Asano, R.; Konagai, N.; Yasuda, S. Efficacy and safety of balloon pulmonary angioplasty for chronic thromboembolic pulmonary hypertension guided by cone-beam computed tomography and electrocardiogram-gated area detector computed tomography. Eur. J. Radiol. 2017, 89, 270–276.
- Yamasaki, Y.; Nagao, M.; Abe, K.; Hosokawa, K.; Kawanami, S.; Kamitani, T.; Yamanouchi, T.; Horimoto, K.; Yabuuchi, H.; Honda, H. Balloon pulmonary angioplasty improves interventricular dyssynchrony in patients with inoperable chronic thromboembolic pulmonary hypertension: A cardiac MR imaging study. Int. J. Cardiovasc. Imaging 2017, 33, 229–239.
- Aoki, T.; Sugimura, K.; Nochioka, K.; Miura, M.; Tatebe, S.; Yamamoto, S.; Yaoita, N.; Suzuki, H.; Sato, H.; Kozu, K.; et al. Effects of Balloon Pulmonary Angioplasty on Oxygenation in Patients With Chronic Thromboembolic Pulmonary Hypertension- Importance of Intrapulmonary Shunt. Circ. J. 2016, 80, 2227–2234.
- Inami, T.; Kataoka, M.; Shimura, N.; Ishiguro, H.; Yanagisawa, R.; Fukuda, K.; Yoshino, H.; Satoh, T. Pressure-wire-guided percutaneous transluminal pulmonary angioplasty: A breakthrough in catheter-interventional therapy for chronic thromboembolic pulmonary hypertension. JACC Cardiovasc. Interv. 2014, 7, 1297–1306.
- Kurzyna, M.; Darocha, S.; Pietura, R.; Pietrasik, A.; Norwa, J.; Manczak, R.; Wieteska, M.; Biederman, A.; Matsubara, H.; Torbicki, A. Changing the strategy of balloon pulmonary angioplasty resulted in a reduced complication rate in patients with chronic thromboembolic pulmonary hypertension. A single-centre European experience. Kardiol. Pol. 2017, 75, 645–654.
- Olsson, K.M.; Wiedenroth, C.B.; Kamp, J.C.; Breithecker, A.; Fuge, J.; Krombach, G.A.; Haas, M.; Hamm, C.; Kramm, T.; Guth, S.; et al. Balloon pulmonary angioplasty for inoperable patients with chronic thromboembolic pulmonary hypertension: The initial German experience. Eur. Respir. J. 2017, 49, 1602409.
- Araszkiewicz, A.; Darocha, S.; Pietrasik, A.; Pietura, R.; Jankiewicz, S.; Banaszkiewicz, M.; Slawek-Szmyt, S.; Biederman, A.; Mularek-Kubzdela, T.; Lesiak, M.; et al. Balloon pulmonary angioplasty for the treatment of residual or recurrent pulmonary hypertension after pulmonary endarterectomy. Int. J. Cardiol. 2019, 278, 232–237.
- Darocha, S.; Araszkiewicz, A.; Kurzyna, M.; Banaszkiewicz, M.; Jankiewicz, S.; Dobosiewicz, A.; Slawek-Szmyt, S.; Janus, M.; Grymuza, M.; Pietrasik, A.; et al. Balloon Pulmonary Angioplasty in Technically Operable and Technically Inoperable Chronic Thromboembolic Pulmonary Hypertension. J. Clin. Med. 2021, 10, 1038.
- Gerges, C.; Friewald, R.; Gerges, M.; Shafran, I.; Sadushi-Kolici, R.; Skoro-Sajer, N.; Moser, B.; Taghavi, S.; Klepetko, W.; Lang, I.M. Efficacy and Safety of Percutaneous Pulmonary Artery Subtotal Occlusion and Chronic Total Occlusion Intervention in Chronic Thromboembolic Pulmonary Hypertension. Circ. Cardiovasc. Interv. 2021, 14, e010243.
- Collet, J.P.; Thiele, H.; Barbato, E.; Barthelemy, O.; Bauersachs, J.; Bhatt, D.L.; Dendale, P.; Dorobantu, M.; Edvardsen, T.; Folliguet, T.; et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Rev. Esp. Cardiol. 2021, 74, 544.
- Eggers, K.M.; Lagerqvist, B.; Venge, P.; Wallentin, L.; Lindahl, B. Persistent cardiac troponin I elevation in stabilized patients after an episode of acute coronary syndrome predicts long-term mortality. Circulation 2007, 116, 1907–1914.
- de Lemos, J.A.; Drazner, M.H.; Omland, T.; Ayers, C.R.; Khera, A.; Rohatgi, A.; Hashim, I.; Berry, J.D.; Das, S.R.; Morrow, D.A.; et al. Association of troponin T detected with a highly sensitive assay and cardiac structure and mortality risk in the general population. JAMA 2010, 304, 2503–2512.
- Apple, F.S.; Murakami, M.M.; Pearce, L.A.; Herzog, C.A. Predictive value of cardiac troponin I and T for subsequent death in end-stage renal disease. Circulation 2002, 106, 2941–2945.
- Horwich, T.B.; Patel, J.; MacLellan, W.R.; Fonarow, G.C. Cardiac troponin I is associated with impaired hemodynamics, progressive left ventricular dysfunction, and increased mortality rates in advanced heart failure. Circulation 2003, 108, 833–838.
- Velez-Martinez, M.; Ayers, C.; Mishkin, J.D.; Bartolome, S.B.; Garcia, C.K.; Torres, F.; Drazner, M.H.; de Lemos, J.A.; Turer, A.T.; Chin, K.M. Association of cardiac troponin I with disease severity and outcomes in patients with pulmonary hypertension. Am. J. Cardiol. 2013, 111, 1812–1817.
- Torbicki, A.; Kurzyna, M.; Kuca, P.; Fijalkowska, A.; Sikora, J.; Florczyk, M.; Pruszczyk, P.; Burakowski, J.; Wawrzynska, L. Detectable serum cardiac troponin T as a marker of poor prognosis among patients with chronic precapillary pulmonary hypertension. Circulation 2003, 108, 844–848.
- Heresi, G.A.; Tang, W.H.; Aytekin, M.; Hammel, J.; Hazen, S.L.; Dweik, R.A. Sensitive cardiac troponin I predicts poor outcomes in pulmonary arterial hypertension. Eur. Respir. J. 2012, 39, 939–944.
- Kriechbaum, S.D.; Wiedenroth, C.B.; Keller, T.; Wolter, J.S.; Ajnwojner, R.; Peters, K.; Haas, M.A.; Roller, F.C.; Breithecker, A.; Rieth, A.J.; et al. Dynamics of high-sensitivity cardiac troponin T during therapy with balloon pulmonary angioplasty for chronic thromboembolic pulmonary hypertension. PLoS ONE 2018, 13, e0204683.
- Weinberg, E.O.; Shimpo, M.; De Keulenaer, G.W.; MacGillivray, C.; Tominaga, S.; Solomon, S.D.; Rouleau, J.L.; Lee, R.T. Expression and regulation of ST2, an interleukin-1 receptor family member, in cardiomyocytes and myocardial infarction. Circulation 2002, 106, 2961–2966.
- Sanada, S.; Hakuno, D.; Higgins, L.J.; Schreiter, E.R.; McKenzie, A.N.; Lee, R.T. IL-33 and ST2 comprise a critical biomechanically induced and cardioprotective signaling system. J. Clin. Investig. 2007, 117, 1538–1549.
- Januzzi, J.L., Jr.; Peacock, W.F.; Maisel, A.S.; Chae, C.U.; Jesse, R.L.; Baggish, A.L.; O’Donoghue, M.; Sakhuja, R.; Chen, A.A.; van Kimmenade, R.R.; et al. Measurement of the interleukin family member ST2 in patients with acute dyspnea: Results from the PRIDE (Pro-Brain Natriuretic Peptide Investigation of Dyspnea in the Emergency Department) study. J. Am. Coll. Cardiol. 2007, 50, 607–613.
- Dieplinger, B.; Mueller, T. Soluble ST2 in heart failure. Clin. Chim. Acta 2015, 443, 57–70.
- Ky, B.; French, B.; McCloskey, K.; Rame, J.E.; McIntosh, E.; Shahi, P.; Dries, D.L.; Tang, W.H.; Wu, A.H.; Fang, J.C.; et al. High-sensitivity ST2 for prediction of adverse outcomes in chronic heart failure. Circ. Heart Fail. 2011, 4, 180–187.
- Pascual-Figal, D.A.; Manzano-Fernandez, S.; Boronat, M.; Casas, T.; Garrido, I.P.; Bonaque, J.C.; Pastor-Perez, F.; Valdes, M.; Januzzi, J.L. Soluble ST2, high-sensitivity troponin T- and N-terminal pro-B-type natriuretic peptide: Complementary role for risk stratification in acutely decompensated heart failure. Eur. J. Heart Fail. 2011, 13, 718–725.
- Felker, G.M.; Fiuzat, M.; Thompson, V.; Shaw, L.K.; Neely, M.L.; Adams, K.F.; Whellan, D.J.; Donahue, M.P.; Ahmad, T.; Kitzman, D.W.; et al. Soluble ST2 in ambulatory patients with heart failure: Association with functional capacity and long-term outcomes. Circ. Heart Fail. 2013, 6, 1172–1179.
- Lassus, J.; Gayat, E.; Mueller, C.; Peacock, W.F.; Spinar, J.; Harjola, V.P.; van Kimmenade, R.; Pathak, A.; Mueller, T.; Disomma, S.; et al. Incremental value of biomarkers to clinical variables for mortality prediction in acutely decompensated heart failure: The Multinational Observational Cohort on Acute Heart Failure (MOCA) study. Int. J. Cardiol. 2013, 168, 2186–2194.
- Lu, J.; Snider, J.V.; Grenache, D.G. Establishment of reference intervals for soluble ST2 from a United States population. Clin. Chim. Acta 2010, 411, 1825–1826.
- Carlomagno, G.; Messalli, G.; Melillo, R.M.; Stanziola, A.A.; Visciano, C.; Mercurio, V.; Imbriaco, M.; Ghio, S.; Sofia, M.; Bonaduce, D.; et al. Serum soluble ST2 and interleukin-33 levels in patients with pulmonary arterial hypertension. Int. J. Cardiol. 2013, 168, 1545–1547.
- Banaszkiewicz, M.; Pietrasik, A.; Darocha, S.; Piłka, M.; Florczyk, M.; Dobosiewicz, A.; Kędzierski, P.; Pędzich-Placha, E.; Kochman, J.; Opolski, G.; et al. Soluble ST2 protein as a new biomarker in patientswith precapillary pulmonary hypertension. Arch. Med. Sci. 2020.
- Geenen, L.W.; Baggen, V.J.M.; Kauling, R.M.; Koudstaal, T.; Boomars, K.A.; Boersma, E.; Roos-Hesselink, J.W.; van den Bosch, A.E. The Prognostic Value of Soluble ST2 in Adults with Pulmonary Hypertension. J. Clin. Med. 2019, 8, 1517.
- Zheng, Y.G.; Yang, T.; He, J.G.; Chen, G.; Liu, Z.H.; Xiong, C.M.; Gu, Q.; Ni, X.H.; Zhao, Z.H. Plasma soluble ST2 levels correlate with disease severity and predict clinical worsening in patients with pulmonary arterial hypertension. Clin. Cardiol. 2014, 37, 365–370.
- Kriechbaum, S.D.; Wiedenroth, C.B.; Peters, K.; Barde, M.A.; Ajnwojner, R.; Wolter, J.S.; Haas, M.; Roller, F.C.; Guth, S.; Rieth, A.J.; et al. Galectin-3, GDF-15, and sST2 for the assessment of disease severity and therapy response in patients suffering from inoperable chronic thromboembolic pulmonary hypertension. Biomarkers 2020, 25, 578–586.
- Banaszkiewicz, M.; Pietrasik, A.; Florczyk, M.; Kedzierski, P.; Pilka, M.; Manczak, R.; Kochman, J.; Opolski, G.; Torbicki, A.; Kurzyna, M.; et al. Soluble ST2 as a Biomarker for Early Complications in Patients with Chronic Thromboembolic Pulmonary Hypertension Treated with Balloon Pulmonary Angioplasty. Diagnostics 2021, 11, 133.
- Carroll, C.; Al Khalaf, M.; Stevens, J.W.; Leaviss, J.; Goodacre, S.; Collinson, P.O.; Wang, J. Heart-type fatty acid binding protein as an early marker for myocardial infarction: Systematic review and meta-analysis. Emerg. Med. J. 2013, 30, 280–286.
- Puls, M.; Dellas, C.; Lankeit, M.; Olschewski, M.; Binder, L.; Geibel, A.; Reiner, C.; Schafer, K.; Hasenfuss, G.; Konstantinides, S. Heart-type fatty acid-binding protein permits early risk stratification of pulmonary embolism. Eur. Heart J. 2007, 28, 224–229.
- Lankeit, M.; Dellas, C.; Panzenbock, A.; Skoro-Sajer, N.; Bonderman, D.; Olschewski, M.; Schafer, K.; Puls, M.; Konstantinides, S.; Lang, I.M. Heart-type fatty acid-binding protein for risk assessment of chronic thromboembolic pulmonary hypertension. Eur. Respir. J. 2008, 31, 1024–1029.
- Mirna, M.; Rohm, I.; Jirak, P.; Wernly, B.; Baz, L.; Paar, V.; Kretzschmar, D.; Hoppe, U.C.; Schulze, P.C.; Lichtenauer, M.; et al. Analysis of Novel Cardiovascular Biomarkers in Patients With Pulmonary Hypertension (PH). Heart Lung Circ. 2020, 29, 337–344.
- Quarck, R.; Nawrot, T.; Meyns, B.; Delcroix, M. C-reactive protein: A new predictor of adverse outcome in pulmonary arterial hypertension. J. Am. Coll. Cardiol. 2009, 53, 1211–1218.
- Scognamiglio, G.; Kempny, A.; Price, L.C.; Alonso-Gonzalez, R.; Marino, P.; Swan, L.; D’Alto, M.; Hooper, J.; Gatzoulis, M.A.; Dimopoulos, K.; et al. C-reactive protein in adults with pulmonary arterial hypertension associated with congenital heart disease and its prognostic value. Heart 2014, 100, 1335–1341.
- Wynants, M.; Quarck, R.; Ronisz, A.; Alfaro-Moreno, E.; Van Raemdonck, D.; Meyns, B.; Delcroix, M. Effects of C-reactive protein on human pulmonary vascular cells in chronic thromboembolic pulmonary hypertension. Eur. Respir. J. 2012, 40, 886–894.
- Yang, M.; Deng, C.; Wu, D.; Zhong, Z.; Lv, X.; Huang, Z.; Lian, N.; Liu, K.; Zhang, Q. The role of mononuclear cell tissue factor and inflammatory cytokines in patients with chronic thromboembolic pulmonary hypertension. J. Thromb. Thrombolysis 2016, 42, 38–45.
- Lippi, G.; Targher, G.; Montagnana, M.; Salvagno, G.L.; Zoppini, G.; Guidi, G.C. Relation between red blood cell distribution width and inflammatory biomarkers in a large cohort of unselected outpatients. Arch. Pathol. Lab. Med. 2009, 133, 628–632.
- Osadnik, T.; Strzelczyk, J.; Hawranek, M.; Lekston, A.; Wasilewski, J.; Kurek, A.; Gutowski, A.R.; Wilczek, K.; Dyrbus, K.; Gierlotka, M.; et al. Red cell distribution width is associated with long-term prognosis in patients with stable coronary artery disease. BMC Cardiovasc. Disord. 2013, 13, 113.
- Felker, G.M.; Allen, L.A.; Pocock, S.J.; Shaw, L.K.; McMurray, J.J.; Pfeffer, M.A.; Swedberg, K.; Wang, D.; Yusuf, S.; Michelson, E.L.; et al. Red cell distribution width as a novel prognostic marker in heart failure: Data from the CHARM Program and the Duke Databank. J. Am. Coll. Cardiol. 2007, 50, 40–47.
- Zorlu, A.; Bektasoglu, G.; Guven, F.M.; Dogan, O.T.; Gucuk, E.; Ege, M.R.; Altay, H.; Cinar, Z.; Tandogan, I.; Yilmaz, M.B. Usefulness of admission red cell distribution width as a predictor of early mortality in patients with acute pulmonary embolism. Am. J. Cardiol. 2012, 109, 128–134.
- Rhodes, C.J.; Wharton, J.; Howard, L.S.; Gibbs, J.S.; Wilkins, M.R. Red cell distribution width outperforms other potential circulating biomarkers in predicting survival in idiopathic pulmonary arterial hypertension. Heart 2011, 97, 1054–1060.
- Hampole, C.V.; Mehrotra, A.K.; Thenappan, T.; Gomberg-Maitland, M.; Shah, S.J. Usefulness of red cell distribution width as a prognostic marker in pulmonary hypertension. Am. J. Cardiol. 2009, 104, 868–872.
- Smukowska-Gorynia, A.; Tomaszewska, I.; Malaczynska-Rajpold, K.; Marcinkowska, J.; Komosa, A.; Janus, M.; Olasinska-Wisniewska, A.; Slawek, S.; Araszkiewicz, A.; Jankiewicz, S.; et al. Red Blood Cells Distribution Width as a Potential Prognostic Biomarker in Patients With Pulmonary Arterial Hypertension and Chronic Thromboembolic Pulmonary Hypertension. Heart Lung Circ. 2018, 27, 842–848.
- Wang, W.; Liu, J.; Yang, Y.H.; Zhai, Z.G.; Wang, C.; Wang, J. Red cell distribution width is increased in chronic thromboembolic pulmonary hypertension. Clin. Respir. J. 2016, 10, 54–60.
- Nickel, N.; Jonigk, D.; Kempf, T.; Bockmeyer, C.L.; Maegel, L.; Rische, J.; Laenger, F.; Lehmann, U.; Sauer, C.; Greer, M.; et al. GDF-15 is abundantly expressed in plexiform lesions in patients with pulmonary arterial hypertension and affects proliferation and apoptosis of pulmonary endothelial cells. Respir. Res. 2011, 12, 62.
- Mueller, T.; Leitner, I.; Egger, M.; Haltmayer, M.; Dieplinger, B. Association of the biomarkers soluble ST2, galectin-3 and growth-differentiation factor-15 with heart failure and other non-cardiac diseases. Clin. Chim. Acta 2015, 445, 155–160.
- Wollert, K.C.; Kempf, T.; Wallentin, L. Growth Differentiation Factor 15 as a Biomarker in Cardiovascular Disease. Clin. Chim. 2017, 63, 140–151.
- Nickel, N.; Kempf, T.; Tapken, H.; Tongers, J.; Laenger, F.; Lehmann, U.; Golpon, H.; Olsson, K.; Wilkins, M.R.; Gibbs, J.S.; et al. Growth differentiation factor-15 in idiopathic pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 2008, 178, 534–541.
- Meadows, C.A.; Risbano, M.G.; Zhang, L.; Geraci, M.W.; Tuder, R.M.; Collier, D.H.; Bull, T.M. Increased expression of growth differentiation factor-15 in systemic sclerosis-associated pulmonary arterial hypertension. Chest 2011, 139, 994–1002.
- Soon, E.; Holmes, A.M.; Treacy, C.M.; Doughty, N.J.; Southgate, L.; Machado, R.D.; Trembath, R.C.; Jennings, S.; Barker, L.; Nicklin, P.; et al. Elevated levels of inflammatory cytokines predict survival in idiopathic and familial pulmonary arterial hypertension. Circulation 2010, 122, 920–927.
- Selimovic, N.; Bergh, C.H.; Andersson, B.; Sakiniene, E.; Carlsten, H.; Rundqvist, B. Growth factors and interleukin-6 across the lung circulation in pulmonary hypertension. Eur. Respir. J. 2009, 34, 662–668.
- Langer, F.; Schramm, R.; Bauer, M.; Tscholl, D.; Kunihara, T.; Schafers, H.J. Cytokine response to pulmonary thromboendarterectomy. Chest 2004, 126, 135–141.
- Zabini, D.; Heinemann, A.; Foris, V.; Nagaraj, C.; Nierlich, P.; Balint, Z.; Kwapiszewska, G.; Lang, I.M.; Klepetko, W.; Olschewski, H.; et al. Comprehensive analysis of inflammatory markers in chronic thromboembolic pulmonary hypertension patients. Eur. Respir. J. 2014, 44, 951–962.
This entry is offline, you can click here to edit this entry!
