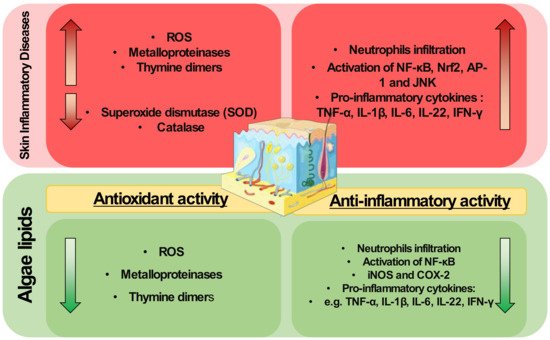
Lipids from algae have been scarcely applied to modulate skin diseases, but they are well known antioxidant and anti-inflammatory agents. They have shown scavenging activities and can modulate redox homeostasis enzymes. They can also downmodulate key inflammatory signaling pathways and transcription factors such as NF-κB, decreasing the expression of pro-inflammatory mediators. Thus, the exploitation of algae lipids as therapeutical agents for the treatment of inflammatory skin diseases is highly attractive.
- skin diseases
- inflammation
- oxidative stress
- lipidomics
- bioactive lipids
- anti-inflammatory
- antioxidant
- macroalgae
- microalgae
1. Introduction
2. Algae Lipids with Antioxidant Activity
| Studies | Mechanism | Assay | Identified Lipids | Algae Species | Ref. |
|---|---|---|---|---|---|
| in chemico | Free radical scavenging | ABTS, DPPH, hydroxyl radical, superoxide anion | Polar lipids, neutral lipids, FAME | Macroalgae: Bifurcaria bifurcata, Codium tomentosum, Fucus vesiculosus, Gracilaria gracilis Grateloupia turuturu, Palmaria palmata, Porphyra dioica Sargassum muticum, Solieria chordalis, Ulva rigida Microalgae: Chlorella vulgaris, Chlorococcum amblystomatis, Nannochloropsis oceanica, Phaeodactylum tricornutum, Scenedesmus intermedius Scenedesmus obliquus, Spirulina sp., Tetraselmis chui |
[69,70,82,83] |
| in vitro | Detoxify intracellular ROS |
Increased the expression of Nrf2 in irradiated HaCat cells Upregulate target antioxidant enzymes Cu/Zn SOD, CAT, and HO-1 |
Crude ethanolic extract | Macroalga: Carpomitra costata | [84] |
| Free radical scavenging | Superoxide generation on peritoneal leukocytes | Sulfoquinovosylacylglycerols | Microalgae: Porphyridium cruentum | [86] | |
| Inhibition of ROS | Photoprotective against UVB in NHDF | Crude ethyl acetate extract | Microalga: Ettlia sp. YC001 | [85] | |
| Enzyme/protein expression | Downregulation of expression of MMPs | Crude ethanolic extract | Microalga: Arthrospira platensis | [87] | |
| Enzyme/protein expression | Downregulation of expression of MMPs, IL-6 and TGF-1 in human dermal fibroblast Modulate MAPK in irradiated HaCat cells |
Fucosterol | Macroalga: Sargassum fusiforme | [88,89] |
The pathophysiology of inflammatory skin diseases is associated with unregulated elevated levels of ROS and the activity of enzymes and proteins involved in the regulation of oxidative stress [30]. In cells, mitochondria metabolize oxygen, producing ROS. During the oxidative phosphorylation in mitochondria, oxygen is converted to O2•−, which can be transformed in H2O2 by superoxide dismutase, and then to water by glutathione peroxidase (GPX) or peroxiredoxin III (PRX III) radical [90]. Under normal conditions, the mitochondria ROS production is balanced by the production of a variety of antioxidants. However, oxidative stress occurs when there is an imbalance between ROS and antioxidants production. An imbalance in ROS production leads to redox signaling from cellular organelles, causing mitochondrial damage and dysfunction in several conditions [91]. However, the application of algae lipids to prevent mitochondrial dysfunction and modulate the oxidative status is little understood and requires in-depth study to understand the mechanisms underlying this potential antioxidant role. The use of crude extracts from algae may reduce ROS levels induced by UVB and impair the expression of MMPs and thymine dimers formation due to UVB exposure in skin cells [87]. These studies were performed using complex crude extracts rich in lipids and not with isolated lipids or fractions. This hinders the understanding of the mechanisms of action of algal lipids as antioxidants, and more work is needed to determine the potential protective role of algal lipids in skin diseases. A better understanding of this antioxidant action is needed, for example, there is a lack of knowledge about the impact of specific lipid classes or lipid molecules in the enzymes and proteins involved in the regulation of oxidative stress, such as metalloproteinases, HO-1, catalase, or superoxide dismutase.
3. Algae Lipids with Anti-Inflammatory Activity
| Studies | Action | Model | Identified Lipids | Algae Species | Ref. |
|---|---|---|---|---|---|
| In chemico | COX-2 inhibition | COX-2 kit assay | Polar lipids | Macroalgae: Codium tomentosum, Fucus vesiculosus Gracilaria gracilis, Palmaria palmata, Porphyra dioica, Ulva rigida, Microalgae: Chlorella vulgaris, Chlorococcum amblystomatis, Gloeothece sp., Skeletonema sp., Tetraselmis sp. mutants |
[69,92,93,94,95] |
| In vitro | NO inhibition | Raw 264.7 | Polar and non-polar lipids; PC, PG, DGDG, DGTS, MGDG, MGMG, SQDG classes; Free and ethyl esterified DGLA |
Macroalgae: Chondrus crispus, Lobophora sp.Palmaria palmata, Microalgae: Chlorella sorokiniana Lobosphaera incisa, Nannochloropsis granulata, Tetraselmis chui, |
[96,97,98,99,100,101,102,103] |
| Decrease in PGE2 Downregulation of COX-2 | Raw 264.7; White blood cells; Epidermal cells |
Crude ethanolic extracts; lipid extracts rich in PC; free and ethyl esterified DGLA |
Macroalgae: Laminaria ochroleuca Microalgae: Chlorella vulgaris, Chloromonas reticulata, Lobosphaera incisa Micractinium sp., Phaeodactylum tricornutum, |
[101,104,105,106,107,119] | |
| Downregulation of mRNA expression of pro-inflammatory cytokines Downregulation of cytokines levels: TNF-α, IL-6, IL-1α, and IL-1β |
THP-1; PBMC; Epidermal cells; HaCaT cells |
Crude ethanolic extracts; lipid extracts; lipid extracts rich in MGDG, DGDG and SQDG; Lipid extracts rich in PC; LPC(16:0); oxylipins; ergosterol and 7-dehydroporiferasterol; free and ethyl esterified DGLA |
Macroalgae: Chondrus crispus, Laminaria ochroleuca, Palmaria palmata, Porphyra dioica, Prasiola japonica Microalgae: Aurantiochytrium mangrovei, Chlamydomonas debaryana, Chlorella vulgaris, Chloromonas reticulata, Cylindrotheca closterium, Dunaliella tertiolecta, Micratinium sp., Nannochloropsis gaditana, Nitzschia palea, Phaeodactylum tricornutum, Lobosphaera incisa Spirulina maxima, Pavlova lutheri, Tetraselmis suecica, |
[84,101,104,105,106,107,110,111,112,113,114,115,116,118,119,121,122] | |
| Inhibition of pro-inflammatory signaling pathways mediated by TLR and NF-κB | THP-1 | Lipid extracts rich in MGDG, DGDG, and SQDG | Macroalgae: Chondrus crispus, Palmaria palmata, Porphyra dioica Microalgae: Pavlova lutheri |
[110] | |
| In vivo | Attenuation of ear oedema | PLA2 kit assay; Mice with ear oedema; DNFB-induced in naive C57BL/6 mice |
MMHDA; Lipid extracts rich in PC; MGDG, DGDG, and SQDG fractions |
Macroalgae: Ishige okamurae, Laminaria ochroleuca Microalgae: ETS-05 cyanobacterium. |
[119,123,127] |
| Neutrophil gathering in the wound region | Wounded zebrafish model | Glycolipids rich in γ-linolenic acid | Microlagae: Spirulina platensis | [124] | |
| Inhibition of pro-inflammatory cytokines production: TNF-α, IL-6, IL-8, IFN- γ, IL-1β, IL-17 | db/db and CD1 mice model of diabetes mellitus; TNBS-induced colitis rats; BALB/c mice skin; TPA-induced hyperplasia murine model |
Crude ethanolic extract; omega-3 fatty acids; oxylipins; MGDG cream |
Macroalgae: Sargassum cristaefolium Microalgae: Chlamydomonas debaryana, Isochrysis galbana |
[125,126,128,129] | |
| Downregulation of iNOS and COX-2, and decrease in NO and PGE2 production | TNBS-induced colitis rat; BALB/c mice skin; TPA-induced hyperplasia murine model |
Crude ethanolic extract; oxylipins; MGDG cream | Macroalgae: Sargassum cristaefolium Microalgae: Chlamydomonas debaryana, Isochrysis galbana |
[126,128,129] |
It was demonstrated that methoxylated fatty acids (MMHDA) isolated from macroalga Ishige okamurae [127] and MGDG, DGDG, and SQDG fractions from microalga ETS-05 cyanobacterium [123] presented anti-inflammatory activity by reducing ear oedema (swelling) in a mouse model. The anti-inflammatory action of MMHDA has been associated with the inhibition of phospholipase A2 (PLA2), the enzyme responsible for the hydrolysis of the sn-2 position of membrane glycerophospholipids to liberate arachidonic acid (AA). The reduction in neutrophils was observed in the wound region of a zebrafish model when glycolipids rich in γ-linolenic acid from the microalga Spirulina platensis were used [124]. Extracts with omega-3 FA isolated from microalgae promoted the reduction of CD4+ T cells production of the pro-inflammatory mediators IFN-γ, TNF-α, and IL-4 and increased the secretion of IL-17A, IL-14, and TGF-β in a db/db and CD1 mouse model of diabetes Mellitus [125]. Downregulation of TNF-α was also observed, as well as decreased expression of iNOS and COX-2, when 2,4,6-trinitrobenzene sulfonic acid (TNBS)-induced colitis rats were supplemented with oxylipins extracted from Chlamydomonas debaryana [126].
4. Concluding Remarks
This entry is adapted from the peer-reviewed paper 10.3390/metabo12020096
