Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Pancreatic cancer (PC) is one of the leading causes of death and is the fourth most malignant tumor in men. The epigenetic and genetic alterations appear to be responsible for development of PC. Small interfering RNA (siRNA) is a powerful genetic tool that can bind to its target and reduces expression level of a specific gene. The various critical genes involved in PC progression can be effectively targeted using diverse siRNAs.
- pancreatic cancer
- small interfering RNA (siRNA)
1. Proliferation and Growth
The uncontrolled growth of PC cells makes it difficult to manage this life-threatening disease. Several different factors and pathways are responsible for rapid proliferation of PC cells. A number of recent studies have focused on various oncogenic signaling networks involved in the progression of PC cells. For instance, pancreatic stellate cells are able to secrete exosomes containing miRNA-5703 to induce PI3K/Akt signaling and promote PC growth rate [1]. The β-catenin is another pathway that its up-regulation by Mind Bomb 1 leads to PC growth [2]. On the other hand, tumor-suppressor factors such as miRNA-573 suppress PC growth via TSPAN1 down-regulation [3]. Hence, the process of PC growth seems to be complicated and each gene can target various downstream targets to modulate PC progression [4]. The aim of current section is to show that how siRNAs can be applied in targeting pathways related to the aberrant growth of PC cells.
Precursor of nerve growth factor (proNGF) is a new and potential therapeutic target in cancer therapy. ProNGF expression undergoes up-regulation in PC and mediates metastasis of tumor cells. The lncRNA OIP5-AS1 enhances ProNGF expression via down-regulating miRNA-186-5p expression to increase PC invasion [5]. On the other hand, anoikis is a kind of apoptotic response stimulated by loss of adhesion to substrate. Reversing anoikis resistance is of importance in PC therapy [6]. Furthermore, proNGF can induce anoikis resistance [7][8]. In another study, it was found that proNGF-siRNA promotes anoikis induction in PC cells, and significantly reduces their proliferation. Following proNGF down-regulation by siRNA, autophagy inducers including autophagy-related gene 5 (ATG5) and Beclin-1 can undergo inhibition, thereby showing that apoptosis induction and autophagy inhibition can occur by proNGF-siRNA [9]. This study provides new insight about siRNA capacity in affecting interaction among programmed cell death (PCD) pathways. The apoptosis and autophagy interaction can be considered a determining factor in cancer. Inhibiting pro-survival autophagy can sensitize tumor cells to apoptosis [10][11][12]. The previous study clearly revealed that proNGF-siRNA could be beneficial in apoptosis induction via preventing autophagy [9].
NUF2 (NUF2 Component of NDC80 Kinetochore Complex) is a linker between kinetochore attachment site and tubulin subunits [13]. It has been reported that NUF2 down-regulation by RNAi leads to an impairment in attachment of kinetochore to spindle microtubules and can effectively suppress cell proliferation at prometaphase [14]. In vitro and in vivo experiments have shown that NUF2 down-regulation by siRNA leads to decreased PC cell growth. Colony formation was stopped and cell cycle arrest at G0/G1 phase occurs due to down-regulation of cyclin B1, Cdc2 and Cdc25A [15].
Receptor-associated protein 80 (RAP80) shows overexpression in PC and can mediate its progression as well as proliferation [16]. RAP80 is involved in DNA repair process via binding to BRCA1 and recruiting it to DNA damage sites [17][18][19][20][21][22]. Therefore, RAP80-siRNA can be of significant importance in reducing PC proliferation. In this case, RAP80 down-regulation by siRNA can lead to apoptosis induction via Bax up-regulation and Bcl-2 down-regulation. Caspase-8 as an executor of apoptosis was stimulated, while no changes were observed in survivin levels [23]. One of the features of cancer cells is their hypoxic microenvironment that facilitates their progression and malignancy. Hypoxia inducible factor-1α (HIF-1α) up-regulation is responsible for immunosuppression [24], radio-resistance [24], chemoresistance (gemcitabine) [25] and proliferation (Warburg effect) [26] in PC. HIF-1α-siRNA leads to a decrease in mRNA and protein levels of HIF-1α that can remarkably diminish the proliferation and induce apoptosis in PC cells [27]. Therefore, using siRNA can act as a potential strategy in suppressing PC proliferation [28]. Cell cycle arrest and apoptosis induction are major outcomes of targeting tumor-promoting factors by siRNA in PC therapy [29].
SnoN gene is a key member of Skt family with tumor-promoting role. This gene was first recognized due to its similarity in sequence with v-Ski and further investigation revealed that SnoN could induce growth of chicken and quail embryo fibroblasts [30][31]. The overexpression of SnoN occurs in human cancers that may result from gene amplification, transcriptional activation and enhanced protein stability [32][33][34]. In respect to the involvement of SnoN in cancer survival, its down-regulation can be considered to be a promising strategy in PC therapy. Therefore, siRNA has been introduced into PC cells for down-regulating SnoN gene expression. Upon SnoN down-regulation, PC cells undergo apoptosis, and their proliferation was interrupted [35]. It appears that anti-apoptotic proteins can be directly affected for triggering apoptosis in PC cells, instead of targeting molecular pathways that can promote PC progression. For instance, in mitochondrial pathway of apoptosis, the expression of Bcl-2 as an anti-apoptotic factor decreases. Overexpression of Bcl-2 protects cancer cells against cell death. By introducing Bcl-2-siRNA into PC cells, apoptosis can be induced [36]. The interesting point is that a variety of molecular pathways can result in increased proliferation and survival of PC cells. Nek2 is a serine-threonine kinase with potential role in both splitting centrosome and spindle formation in mammalian cells [37]. Nek2 up-regulation provides chromosome instability and aneuploidy in cancers [38]. The Nek2 inhibition appears to be advantageous in decreasing expression level of PD-L1 to enhance lymphocyte infiltration and promote anti-tumor immunity in PC suppression [39]. Hence, targeting Nek2 is of importance in suppressing cancer progression. For this reason, Nek2-siRNA has been applied for PC suppression in vitro and in vivo. Noteworthy, in mouse model of PC, siRNA has been introduced via a catheter. The Nek2-siRNA can impair the proliferation of PC cells and it promotes survival of xenograft mouse model. Furthermore, Nek2-siRNA can prevent liver metastasis of PC cells [40].
In addition, it has been reported that RPL1, as a ribosomal protein can be targeted in PC therapy. Moreover, down-regulation of RPL1 by siRNA leads to apoptosis and cell cycle arrest at G1 phase, and suppress DNA replication [41]. These studies advocated the fact that first step in PC therapy is identifcation of the various tumor-promoting factors. Thus, siRNAs can be designed for specific targeting of tumor cells to suppress PC proliferation and induce apoptosis [42].
Mammalian histone deacetylases (HDAC) are grouped into four different categories (I–IV) [43][44]. They participate in regulating biological processes including chromatin remodeling, gene repression, regulating cell cycle, and differentiation [43][45][46][47]. HDAC dysregulation is associated with transcription repression and inhibiting expression of tumor-suppressor genes [48][49]. HDAC1 plays a significant role in PC progression. It has been shown that HDAC1 down-regulation can lead to cancer proliferation suppression [50]. HDAC1 and HIF-1α can produce a complex in binding to hypoxia response elements (HRE) on the miR-548an promoter, down-regulating its expression and enhancing carcinogenesis in PC [51]. HDAC1 recruitment can lead to PC metastasis via reducing E-cadherin levels [52]. It has been established that HDAC1 undergoes up-regulation in PC tissues compared to normal ones. The expression of HDAC1 in PC tissues was 56.4%, while this expression was reduced significantly to 6.7% in the normal tissues. HDAC1-siRNA leads to down-regulation of this tumor-promoting factor, therefore paving the way for up-regulation of p21 and Bax in apoptosis induction in PC [53].
Autophagy is another important form of PCD that can exhibit both pro-survival and pro-death functions in cancers and inhibiting pro-survival autophagy can boost apoptosis induction in PC cells. The siRNAs have been used for down-regulating expression levels of proNGF, NUF2, RAP80, HIF-1α and SnoN to effectively impair the growth of PC cells and induce apoptosis. There are several other oncogenic pathways involved in PC progression including Wnt/β-catenin, STAT3 and NF-κB that can be focus of future studies.
2. Metastasis and Angiogenesis
The stimulation of MAPK/EscientistsRK axis by A-Raf was found to be vital in elevating migration of PC cells [54]. Furthermore, interactions occurring in tumor microenvironment can lead to PC metastasis. The recruitment of macrophages and their M2 polarization can secrete IL-6 that subsequently can induce STAT3 signaling for promoting PC migration and invasion via EMT induction [55]. STAT3 signaling can stimulate the growth and proliferation of cancer cells [56][57][58]. Both Akt and ERK1/2 molecular pathways can participate in PC metastasis and their stimulation occurs by FGF19 [59]. The metformin administration, as anti-tumor agent, substantially can reduce HNF4G expression via AMPK up-regulation to impair metastasis of PC cells [60]. Hence, PC migration is an increasing challenge and can significantly promotes aggressiveness of PC cells [61]. This section has been allocated to discuss application of siRNAs in disrupting PC invasion.
Cyclooxygenase-2 (COX-2) is an enzyme of metabolic process of arachidonic acid that can actively participate in carcinogenesis [62][63][64]. COX-2 induces angiogenesis in PC through up-regulating epidermal growth factor receptor (EGFR) [65]. COX-2 overexpression demonstrates poor prognosis in PC patients [66]. COX-2 inhibitors have been applied in PC therapy due to their efficacy in angiogenesis inhibition via vascular endothelial growth factor (VEGF) down-regulation and suppressing growth [67][68]. COX-2-siRNA can trigger apoptosis and cell cycle arrest in PC cells. In tumor xenografts, COX-2-siRNA can significantly attenuate volume and weight of tumors, thus showing the efficiency of gene silencing in vivo [69].
One of the potential therapeutic targets in cancer therapy is c-Src, an important non-receptor protein tyrosine kinase. Increasing evidence demonstrates tumor-promoting role of c-Src in cancer [70][71][72]. It can promote carcinogenesis via glycolysis induction [73]. Cell adhesion molecule 1 (CADM1) as a tumor-suppressor, can down-regulate expression of c-Src in suppressing colon tumorigenesis [74]. The self-renewal capacity of breast cancer cells is also regulated by c-Src [75]. These studies highlight the role of c-Src, as a tumor-promoting factor. It seems that c-Src down-regulation by siRNA impairs progression of PC cells via inhibiting angiogenesis. The activation of angiogenesis can occur by EGFR up-regulation, a process involved in cancer metastasis and migration to the distant sites [76][77][78]. The transfection efficiency of siRNA in PC cells was more than 90% and expression of c-Src was reduced by 86.1%. Following c-Src down-regulation by siRNA, the expression of VEGF was inhibited, thus suppressing angiogenesis and cancer progression [79].
Although there have been efforts in using siRNA for reducing migration of PC cells and various factors such as COX-2 and c-Src have been affected, there is still a long way before naked siRNAs can be applied for regulating PC progression. There are several other factors such as EMT and matrix metalloproteinases (MMPs) that can also lead to metastasis of PC cells. However, there are no studies reported about using siRNAs for modulating their upstream regulators such as ZEB1/2, TGF-β and Snail, among others.
3. Immune Regulation
Transforming growth factor-β (TGF-β) is considered as a novel target in cancer therapy [80][81]. Under physiological conditions, TGF-β regulates proliferation, differentiation, survival and cell adhesion to preserve tissue homeostasis. In cancer cells, TGF-β acts as a positive factor for metastasis via EMT induction [82][83][84]. On the other hand, retinoic acid-inducible gene I (RIG-I) is affected in cancer immunotherapy by enhancing the levels of interferon and apoptosis induction [85][86][87]. A bifunctional siRNA for down-regulating TGF-β and enhancing RIG-I expression has also been designed. This improvement in function can be obtained by introducing triphosphate group at the 5′ end of siRNA. Following TGF-β down-regulation, an increase occurs in survival time of xenograft models and metastasis and invasion of PC cells undergo down-regulation. Due to RIG-I activation, immune system can be activated that can promote the levels of interferon and RIG-I and induces apoptosis [88]. In addition, TGF-β down-regulation and RIG-I up-regulation increase potential of cancer immunotherapy [89]. However, an experiment has only investigated role of siRNA in cancer immunotherapy and as immune evasion is a common phenomenon in PC [90], more studies are required to show how such genetic tools can be employed in PC treatment and activating anti-tumor immunity. Although only TGF-β signaling has been targeted in improving anti-tumor immunity in PC, there is another well-known molecular pathway, known as PD-L1/PD-1 axis that is involved in triggering immune evasion [91]. Hence, further experiments can focus on using siRNA for down-regulating PD-1 expression and promoting anti-tumor immunity against PC cells.
4. Therapy Response and Synergistic Therapy
Another potential of siRNAs lies in improving potential of chemotherapeutic agents in suppressing tumor progression [92][93]. Therefore, combination cancer therapy with siRNA-anticancer drug could be designed. For instance, L-ascorbate is not capable of preventing PC migration. Co-application of siRNA-HIF-1α and L-ascorbate resulted in ansynergistic effect in suppressing PC invasion [94]. siRNA could serve as a potential adjuvant for promoting anti-tumor activity of compounds in PC therapy. Surgery is not considered a successful option in PC therapy due to diagnosis of PC at advanced stages. Therefore, chemotherapy has been primarily used for the treatment of PC patients. However, drug resistance emerges in PC and different molecular pathways including long non-coding RNAs [95], SRPX2 [96] and USP7 [97], among others can participate in the resistance of PC cells to chemotherapy. siRNAs can be potentially beneficial in impairing tumor proliferation as well as metastasis and causing subsequent increase in chemosensitivity of PC cells.
Ribonucleotide reductase (RR) is a rate-limiting enzyme vital for DNA synthesis and replication [98]. Celastrol can suppress progression of PC cells via down-regulating RRM2 expression, showing tumor-promoting function of this pathway [99]. Exposing PC cells to RRM2-siRNA leads to an increase in apoptosis and cell growth inhibition. Noteworthy, co-administration of RRM2-siRNA and doxorubicin (DOX) can lead to synergistic effect and a four-fold increase in anti-tumor activity [100]. Gemcitabine (GEM) is another chemotherapeutic agent that its potential in PC treatment has been reduced due to emergence of drug resistance [101]. The hTERT-siRNA can increase the number of PC cells undergoing apoptosis. The hTERT down-regulation induces cell cycle arrest at G0/G1 phase and enhances number of PC cells in S and G2/M phases [102]. Following hTERT down-regulation by siRNA, expressions of Bcl-2 and COX-2, as tumor-promoting factors undergo inhibition that is of importance for inducing apoptosis in PC cells [103], and enhancing their sensitivity to chemotherapy.
Now, it is obvious that when cancer cells demonstrate malignant behavior in terms of proliferation and migration, they can obtain chemoresistance [104][105][106][107][108][109][110]. Therefore, in order to provide effective cancer chemotherapy, it is vital to suppress the various pathways that lead to cancer migration and growth [107][111]. It appears that an overexpression of heterogeneous nuclear ribonucleoprotein A2/B1 (hnRNP A2/B1) is in favor of PC growth [16]. As RNA-binding proteins, hnRNP A2/B1 participates in mRNA processing and telomere biogenesis [112]. Clinical study evaluating 42 patients with PC has shown role of hnRNP A2/B1 in PC and its association with E-cadherin, an important epithelial marker [113]. The exposure of PC cells to hnRNP A2/B1 can lead to apoptosis induction. A combination of hnRNP A2/B1 and chemotherapeutic agents such as 5-fluorouracil (5-FU), oxaliplatin and GEM can stimulate synergistic effect against PC cells. In fact, by suppressing PC growth, hnRNA A2/B1-siRNA enhances sensitivity of PC cells to chemotherapy. This combination leads to Bcl-2 down-regulation and Bax up-regulation, providing apoptotic cell death. Moreover, expression of P-glycoprotein, as a drug transporter that induces chemoresistance [114], decreases following this combination that is of importance in enhancing chemosensitivity [115].
Increasing evidence demonstrates that RR and thymidylate synthase (TS) can induce chemoresistance in cancer cells [116][117]. It has been reported that RRM2-siRNA, as a subunit of RR can promote GEM sensitivity, in vitro and in vivo [117][118]. A combination of GEM and RR- and TS-siRNA can stimulate apoptosis in PC cells and reduces their proliferation. This combination inhibits NF-κB activation following GEM administration and enhances TNF-related apoptosis-inducing ligand (TRAIL)-mediated cell death in PC cells [119]. In the previous section, it was mentioned that HIF-1α is a desirable factor for progression of PC cells. Moreover, HIF-1α down-regulation by siRNA leads to an increase in chemosensitivity of PC cells [120]. A set of tumor-promoting factors such as HIF-1α, ARNT, PFKFB4, and RBKS can be down-regulated by siRNA in inducing apoptosis and enhancing their sensitivity to chemotherapeutic agents including DOX and GEM (Figure 1 and Figure 2) [121]. The interesting point of this section is that prior studies have considered role of both molecular pathways and drug transporters in triggering drug resistance in PC and these molecular pathways and mechanisms can be markedly suppressed using siRNAs as an effective tool.
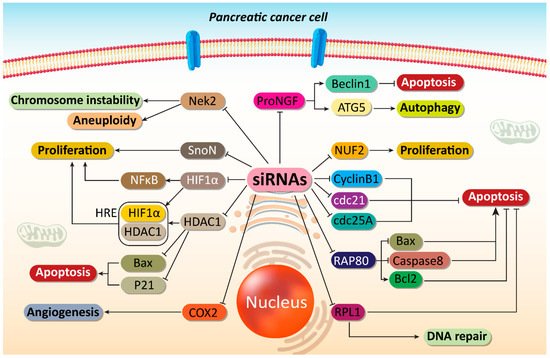
Figure 1. Impairing proliferation and angiogenesis of PC cells. Major molecular pathways can be targeted by siRNAs to induce apoptosis and DNA damage in PC cells. Moreover, angiogenesis responsible for PC progression can be suppressed by siRNAs in PC therapy. ProNGF, precursor of nerve growth factor; ATG, autophagy-related gene; COX2, cyclooxygenase-2; HDAC1, histone deacetylase 1; HIF-1α, hypoxia inducible factor-1α; NF-κB, nuclear factor-kappaB; siRNA, small interfering RNA; PC, prostate cancer.
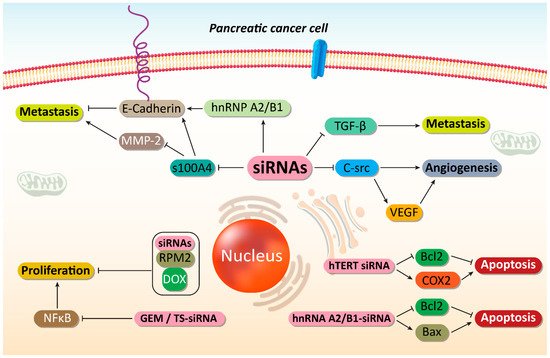
Figure 2. Suppressing PC metastasis and increasing their sensitivity to chemotherapy. By disrupting cancer proliferation and metastasis, as well as triggering apoptotic cell death, an increase occurs in sensitivity of PC cells to chemotherapy. SiRNAs play an important role in mediating these anticancer effects. MMp-2, matrix melloproteinase-2; hnRNP A2/B1, heterogeneous nuclear ribonucleoprotein A2/B1; TGF-β, transforming growth factor-beta; VEGF, vascular endothelial growth factor; COX-2, cyclooxygenase-2; GEM, gemcitabine; NF-κB, nuclear factor-kappaB; DOX, doxorubicin, siRNA, small interfering RNA; PC, prostate cancer.
This entry is adapted from the peer-reviewed paper 10.3390/cells10123348
References
- Li, M.; Guo, H.; Wang, Q.; Chen, K.; Marko, K.; Tian, X.; Yang, Y. Pancreatic Stellate Cells Derived Exosomal miR-5703 Promotes Pancreatic Cancer by Downregulating CMTM4 and Activating PI3K/Akt Pathway. Cancer Lett. 2020, 490, 20–30.
- Fu, X.; Tang, N.; Xie, W.; Mao, L.; Qiu, Y. Mind Bomb 1 Promotes Pancreatic Cancer Proliferation by Activating β-Catenin Signaling. J. Nanosci. Nanotechnol. 2020, 20, 7276–7282.
- Wang, L.; Gao, P.; Yuan, P.; Zhou, P.; Fan, H.; Lin, X.; Yuan, X.; Zhu, M.; Fan, X.; Lu, Y.; et al. miR-573 Suppresses Pancreatic Cancer Cell Proliferation, Migration, and Invasion through Targeting TSPAN1. Strahlenther. Onkol. 2021, 197, 438–448.
- Swami, P.; Thiyagarajan, S.; Vidger, A.; Indurthi, V.S.K.; Vetter, S.W.; Leclerc, E. RAGE up-Regulation Differently Affects Cell Proliferation and Migration in Pancreatic Cancer Cells. Int. J. Mol. Sci. 2020, 21, 7723.
- Li, A.; Feng, L.; Niu, X.; Zeng, Q.; Li, B.; You, Z. Downregulation of OIP5-AS1 Affects proNGF-Induced Pancreatic Cancer Metastasis by Inhibiting p75NTR Levels. Aging (Albany NY) 2021, 13, 10688–10702.
- Fofaria, N.M.; Srivastava, S.K. STAT3 Induces Anoikis Resistance, Promotes Cell Invasion and Metastatic Potential in Pancreatic Cancer Cells. Carcinogenesis 2015, 36, 142–150.
- Lagadec, C.; Romon, R.; Tastet, C.; Meignan, S.; Com, E.; Page, A.; Bidaux, G.; Hondermarck, H.; Le Bourhis, X. Ku86 Is Important for TrkA Overexpression-Induced Breast Cancer Cell Invasion. Proteomics. Clin. Appl. 2010, 4, 580–590.
- Bao, X.; Shi, J.; Xie, F.; Liu, Z.; Yu, J.; Chen, W.; Zhang, Z.; Xu, Q. Proteolytic Release of the p75NTR Intracellular Domain by ADAM10 Promotes Metastasis and Resistance to Anoikis. Cancer Res. 2018, 78, 2262–2276.
- Xu, J.; Song, J.; Yang, X.; Guo, J.; Wang, T.; Zhuo, W. ProNGF siRNA inhibits cell proliferation and invasion of pancreatic cancer cells and promotes anoikis. Biomed Pharmacother 2019, 111, 1066–1073.
- Ashrafizadeh, M.; Zarrabi, A.; Orouei, S.; Hushmandi, K.; Hakimi, A.; Zabolian, A.; Daneshi, S.; Samarghandian, S.; Baradaran, B.; Najafi, M. MicroRNA-Mediated Autophagy Regulation in Cancer Therapy: The Role in Chemoresistance/Chemosensitivity. Eur. J. Pharmacol. 2021, 892, 173660.
- Ashrafizadeh, M.; Ahmadi, Z.; Farkhondeh, T.; Samarghandian, S. Autophagy Regulation Using Luteolin: New Insight into Its Anti-Tumor Activity. Cancer Cell Int. 2020, 20, 537.
- Patra, S.; Mishra, S.R.; Behera, B.P.; Mahapatra, K.K.; Panigrahi, D.P.; Bhol, C.S.; Praharaj, P.P.; Sethi, G.; Patra, S.K.; Bhutia, S.K. Autophagy-Modulating Phytochemicals in Cancer Therapeutics: Current Evidences and Future Perspectives. Semin. Cancer Biol. 2020, S1044-1579X(1020)30104-30108.
- Nabetani, A.; Koujin, T.; Tsutsumi, C.; Haraguchi, T.; Hiraoka, Y. A Conserved Protein, Nuf2, is Implicated in Connecting the Centromere to the Spindle during Chromosome Segregation: A Link between the Kinetochore Function and the Spindle Checkpoint. Chromosoma 2001, 110, 322–334.
- DeLuca, J.G.; Moree, B.; Hickey, J.M.; Kilmartin, J.V.; Salmon, E.D. hNuf2 Inhibition Blocks Stable Kinetochore-Microtubule Attachment and Induces Mitotic Cell Death in HeLa Cells. J. Cell Biol. 2002, 159, 549–555.
- Hu, P.; Chen, X.; Sun, J.; Bie, P.; Zhang, L.D. siRNA-Mediated Knockdown against NUF2 Suppresses Pancreatic Cancer Proliferation in Vitro and in Vivo. Biosci. Rep. 2015, 35, e00170.
- Wang, L.; Liu, H.L.; Li, Y.; Yuan, P. Proteomic Analysis of Pancreatic Intraepithelial Neoplasia and Pancreatic Carcinoma in Rat Models. World J. Gastroenterol. 2011, 17, 1434.
- Yan, Z.; Kim, Y.S.; Jetten, A.M. RAP80, a Novel Nuclear Protein That Interacts With the Retinoid-Related Testis-Associated Receptor. J. Biol. Chem. 2002, 277, 32379–32388.
- Di Fiore, P.P.; Polo, S.; Hofmann, K. When Ubiquitin Meets Ubiquitin Receptors: A Signalling Connection. Nat. Rev. Mol. Cell Biol. 2003, 4, 491–497.
- Hoeller, D.; Crosetto, N.; Blagoev, B.; Raiborg, C.; Tikkanen, R.; Wagner, S.; Kowanetz, K.; Breitling, R.; Mann, M.; Stenmark, H. Regulation of Ubiquitin-Binding Proteins by Monoubiquitination. Nat. Cell Biol. 2006, 8, 163–169.
- Yan, J.; Kim, Y.S.; Yang, X.P.; Li, L.P.; Liao, G.; Xia, F.; Jetten, A.M. The Ubiquitin-Interacting Motif–Containing Protein RAP80 Interacts with BRCA1 and Functions in DNA Damage Repair Response. Cancer Res. 2007, 67, 6647–6656.
- Wang, B.; Matsuoka, S.; Ballif, B.A.; Zhang, D.; Smogorzewska, A.; Gygi, S.P.; Elledge, S.J. Abraxas and RAP80 Form a BRCA1 Protein Complex Required for the DNA Damage Response. Science 2007, 316, 1194–1198.
- Kim, H.; Chen, J.; Yu, X. Ubiquitin-Binding Protein RAP80 Mediates BRCA1-Dependent DNA Damage Response. Science 2007, 316, 1202–1205.
- Li, Y.; Gu, W.J.; Liu, H.L. Induction of Pancreatic Cancer Cell Apoptosis and Enhancement of Gemcitabine Sensitivity by RAP80 siRNA. Dig. Dis. Sci. 2012, 57, 2072–2078.
- Yang, X.; Lu, Y.; Hang, J.; Zhang, J.; Zhang, T.; Huo, Y.; Liu, J.; Lai, S.; Luo, D.; Wang, L.; et al. Lactate-Modulated Immunosuppression of Myeloid-Derived Suppressor Cells Contributes to the Radioresistance of Pancreatic Cancer. Cancer Immunol. Res. 2020, 8, 1440–1451.
- Liu, Y.F.; Luo, D.; Li, X.; Li, Z.Q.; Yu, X.; Zhu, H.W. PVT1 Knockdown Inhibits Autophagy and Improves Gemcitabine Sensitivity by Regulating the MiR-143/HIF-1α/VMP1 Axis in Pancreatic Cancer. Pancreas 2021, 50, 227–234.
- Liu, A.; Xu, J. Circ_03955 Promotes Pancreatic Cancer Tumorigenesis and Warburg Effect by Targeting the miR-3662/HIF-1α Axis. Clin. Transl. Oncol. 2021, 23, 1905–1914.
- Chen, C.; Yu, Z. siRNA Targeting HIF-1alpha Induces Apoptosis of Pancreatic Cancer Cells through NF-kappaB-Independent and-Dependent Pathways under Hypoxic Conditions. Anticancer Res. 2009, 29, 1367–1372.
- Yan, C.Q.; Zhao, Y.P. The Significance of VEGF siRNA and bFGF siRNA in Invasion and Proliferation of Pancreatic Carcinoma cell. Zhonghua Wai Ke Za Zhi 2010, 48, 610–614.
- Xu, M.; Gao, J.; Du, Y.Q.; Gao, D.J.; Zhang, Y.Q.; Li, Z.S.; Zhang, Y.L.; Gong, Y.F.; Xu, P. Reduction of Pancreatic Cancer Cell Viability and Induction of Apoptosis Mediated by siRNA Targeting DNMT1 through Suppression of Total DNA Methyltransferase Activity. Mol. Med. Rep. 2010, 3, 699–704.
- Boyer, P.L.; Colmenares, C.; Stavnezer, E.; Hughes, S.H. Sequence and Biological Activity of Chicken snoN cDNA Clones. Oncogene 1993, 8, 457–466.
- Pearson-White, S.; Crittenden, R. Proto-Oncogene Sno Expression, Alternative Isoforms and Immediate Early Serum Response. Nucleic Acids Res. 1997, 25, 2930–2937.
- Imoto, I.; Pimkhaokham, A.; Fukuda, Y.; Yang, Z.Q.; Shimada, Y.; Nomura, N.; Hirai, H.; Imamura, M.; Inazawa, J. SNO is a Probable Target for Gene Amplification at 3q26 in Squamous-Cell Carcinomas of the Esophagus. Biochem. Biophys. Res. Commun. 2001, 286, 559–565.
- Bonni, S.; Wang, H.R.; Causing, C.G.; Kavsak, P.; Stroschein, S.L.; Luo, K.; Wrana, J.L. TGF-Beta Induces Assembly of a Smad2-Smurf2 Ubiquitin Ligase Complex that Targets SnoN for Degradation. Nat. Cell Biol. 2001, 3, 587–595.
- Stroschein, S.L.; Wang, W.; Zhou, S.; Zhou, Q.; Luo, K. Negative Feedback Regulation of TGF-Beta Signaling by the SnoN Oncoprotein. Science 1999, 286, 771–774.
- Liu, C.; Zhang, H.; Zang, X.; Wang, C.; Kong, Y.; Zhang, H. The Influence of SnoN Gene Silencing by siRNA on the Cell Proliferation and Apoptosis of Human Pancreatic Cancer Cells. Diagn. Pathol. 2015, 10, 30.
- Ocker, M.; Neureiter, D.; Lueders, M.; Zopf, S.; Ganslmayer, M.; Hahn, E.G.; Herold, C.; Schuppan, D. Variants of bcl-2 Specific siRNA for Silencing Antiapoptotic bcl-2 in Pancreatic Cancer. Gut 2005, 54, 1298–1308.
- Fry, A.M.; Meraldi, P.; Nigg, E.A. A Centrosomal Function for the Human Nek2 Protein Kinase, a Member of the NIMA Family of Cell Cycle Regulators. EMBO J. 1998, 17, 470–481.
- Hayward, D.G.; Fry, A.M. Nek2 Kinase in Chromosome Instability and Cancer. Cancer Lett. 2006, 237, 155–166.
- Zhang, X.; Huang, X.; Xu, J.; Li, E.; Lao, M.; Tang, T.; Zhang, G.; Guo, C.; Zhang, X.; Chen, W.; et al. NEK2 Inhibition Triggers Anti-Pancreatic Cancer Immunity by Targeting PD-L1. Nat. Commun. 2021, 12, 4536.
- Kokuryo, T.; Hibino, S.; Suzuki, K.; Watanabe, K.; Yokoyama, Y.; Nagino, M.; Senga, T.; Hamaguchi, M. Nek2 siRNA Therapy Using a Portal Venous Port-Catheter System for Liver Metastasis in Pancreatic Cancer. Cancer Sci. 2016, 107, 1315–1320.
- Li, C.; Ge, M.; Chen, D.; Sun, T.; Jiang, H.; Xie, Y.; Lu, H.; Zhang, B.; Han, L.; Chen, J.; et al. RPL21 siRNA blocks proliferation in Pancreatic Cancer Cells by Inhibiting DNA Replication and Inducing G1 Arrest and Apoptosis. Front. Oncol. 2020, 10, 1730.
- Yu, Q.; Qiu, Y.; Chen, X.; Wang, X.; Mei, L.; Wu, H.; Liu, K.; Liu, Y.; Li, M.; Zhang, Z.; et al. Chemotherapy Priming of the Pancreatic Tumor Microenvironment Promotes Delivery and Anti-Metastasis Efficacy of Intravenous Low-Molecular-Weight Heparin-Coated lipid-siRNA Complex. Theranostics 2019, 9, 355–368.
- Merarchi, M.; Sethi, G.; Shanmugam, M.K.; Fan, L.; Arfuso, F.; Ahn, K.S. Role of Natural Products in Modulating Histone Deacetylases in Cancer. Molecules 2019, 24, 1047.
- Wang, L.; Syn, N.L.; Subhash, V.V.; Any, Y.; Thuya, W.L.; Cheow, E.S.H.; Kong, L.; Yu, F.; Peethala, P.C.; Wong, A.L.; et al. Pan-HDAC Inhibition by Panobinostat Mediates Chemosensitization to Carboplatin in Non-Small Cell Lung Cancer via Attenuation of EGFR Signaling. Cancer Lett. 2018, 417, 152–160.
- Zhang, Y.; LeRoy, G.; Seelig, H.P.; Lane, W.S.; Reinberg, D. The Dermatomyositis-Specific Autoantigen Mi2 is a Component of a Complex Containing Histone Deacetylase and Nucleosome Remodeling Activities. Cell 1998, 95, 279–289.
- McKinsey, T.A.; Zhang, C.L.; Lu, J.; Olson, E.N. Signal-Dependent Nuclear Export of a Histone Deacetylase Regulates Muscle Differentiation. Nature 2000, 408, 106–111.
- Shanmugam, M.K.; Arfuso, F.; Arumugam, S.; Chinnathambi, A.; Jinsong, B.; Warrier, S.; Wang, L.Z.; Kumar, A.P.; Ahn, K.S.; Sethi, G.; et al. Role of Novel Histone Modifications in Cancer. Oncotarget 2018, 9, 11414–11426.
- Johnstone, R.W. Histone-Deacetylase Inhibitors: Novel Drugs for the Treatment of Cancer. Nat. Rev. Drug Discov. 2002, 1, 287–299.
- Ng, H.H.; Bird, A. Histone Deacetylases: Silencers For hire. Trends Biochem. Sci. 2000, 25, 121–126.
- Sixto-López, Y.; Gómez-Vidal, J.A.; de Pedro, N.; Bello, M.; Rosales-Hernández, M.C.; Correa-Basurto, J. Hydroxamic Acid Derivatives as HDAC1, HDAC6 and HDAC8 Inhibitors with Antiproliferative Activity in Cancer Cell Lines. Sci. Rep. 2020, 10, 10462.
- Zhu, S.; He, C.; Deng, S.; Li, X.; Cui, S.; Zeng, Z.; Liu, M.; Zhao, S.; Chen, J.; Jin, Y.; et al. MiR-548an, Transcriptionally Downregulated by HIF1α/HDAC1, Suppresses Tumorigenesis of Pancreatic Cancer by Targeting Vimentin Expression. Mol. Cancer Ther. 2016, 15, 2209–2219.
- Aghdassi, A.; Sendler, M.; Guenther, A.; Mayerle, J.; Behn, C.O.; Heidecke, C.D.; Friess, H.; Büchler, M.; Evert, M.; Lerch, M.M.; et al. Recruitment of Histone Deacetylases HDAC1 and HDAC2 by the Transcriptional Repressor ZEB1 Downregulates E-Cadherin Expression in Pancreatic Cancer. Gut 2012, 61, 439–448.
- Gao, D.J.; Xu, M.; Zhang, Y.Q.; Du, Y.Q.; Gao, J.; Gong, Y.F.; Man, X.H.; Wu, H.Y.; Jin, J.; Xu, G.M.; et al. Upregulated Histone Deacetylase 1 Expression in Pancreatic Ductal Adenocarcinoma and Specific siRNA Inhibits the Growth of Cancer Cells. Pancreas 2010, 39, 994–1001.
- Meng, L.D.; Shi, G.D.; Ge, W.L.; Huang, X.M.; Chen, Q.; Yuan, H.; Wu, P.F.; Lu, Y.C.; Shen, P.; Zhang, Y.H.; et al. Linc01232 Promotes the Metastasis of Pancreatic Cancer by Suppressing the Ubiquitin-Mediated Degradation of HNRNPA2B1 and Activating the A-Raf-induced MAPK/ERK Signaling Pathway. Cancer Lett. 2020, 494, 107–120.
- Geng, Y.; Fan, J.; Chen, L.; Zhang, C.; Qu, C.; Qian, L.; Chen, K.; Meng, Z.; Chen, Z.; Wang, P. A Notch-Dependent Inflammatory Feedback Circuit between Macrophages and Cancer Cells Regulates Pancreatic Cancer Metastasis. Cancer Res. 2021, 81, 64–76.
- Lee, J.H.; Chiang, S.Y.; Nam, D.; Chung, W.S.; Lee, J.; Na, Y.S.; Sethi, G.; Ahn, K.S. Capillarisin Inhibits Constitutive and Inducible STAT3 Activation through Induction of SHP-1 and SHP-2 Tyrosine Phosphatases. Cancer Lett. 2014, 345, 140–148.
- Kim, S.M.; Lee, J.H.; Sethi, G.; Kim, C.; Baek, S.H.; Nam, D.; Chung, W.S.; Kim, S.H.; Shim, B.S.; Ahn, K.S. Bergamottin, a Natural Furanocoumarin Obtained from Grapefruit Juice Induces Chemosensitization and Apoptosis through the Inhibition of STAT3 Signaling Pathway IN Tumor Cells. Cancer Lett. 2014, 354, 153–163.
- Zhang, J.; Ahn, K.S.; Kim, C.; Shanmugam, M.K.; Siveen, K.S.; Arfuso, F.; Samym, R.P.; Deivasigamanim, A.; Lim, L.H.; Wang, L.; et al. Nimbolide-Induced Oxidative Stress Abrogates STAT3 Signaling Cascade and Inhibits Tumor Growth in Transgenic Adenocarcinoma of Mouse Prostate Model. Antioxid. Redox. Signal 2016, 24, 575–589.
- Fang, D.; Zhang, C.; Xu, P.; Liu, Y.; Mo, X.; Sun, Q.; Abdelatty, A.; Hu, C.; Xu, H.; Zhou, G.; et al. S100A16 Promotes Metastasis and Progression of Pancreatic Cancer through FGF19-Mediated AKT and ERK1/2 Pathways. Cell Biol. Toxicol. 2021, 37, 555–571.
- Wang, C.; Zhang, T.; Liao, Q.; Dai, M.; Guo, J.; Yang, X.; Tan, W.; Lin, D.; Wu, C.; Zhao, Y. Metformin Inhibits Pancreatic Cancer Metastasis Caused by SMAD4 Deficiency and Consequent HNF4G Upregulation. Protein Cell 2021, 12, 128–144.
- Xu, S.; Lei, S.L.; Liu, K.J.; Yi, S.G.; Yang, Z.L.; Yao, H.L. circSFMBT1 Promotes Pancreatic Cancer Growth and Metastasis via Targeting miR-330-5p/PAK1 Axis. Cancer Gene Ther. 2021, 28, 234–249.
- Yarla, N.S.; Bishayee, A.; Sethi, G.; Reddanna, P.; Kalle, A.M.; Dhananjaya, B.L.; Dowluru, K.S.; Chintala, R.; Duddukuri, G.R. Targeting Arachidonic Acid Pathway by Natural Products for Cancer Prevention and Therapy. Semin. Cancer Biol. 2016, 40–41, 48–81.
- Shanmugam, M.K.; Ong, T.H.; Kumar, A.P.; Lun, C.K.; Ho, P.C.; Wong, P.T.; Hui, K.M.; Sethi, G. Ursolic Acid Inhibits the Initiation, Progression of Prostate Cancer and Prolongs the Survival of TRAMP Mice by Modulating Pro-Inflammatory Pathways. PLoS ONE 2012, 7, e32476.
- Li, F.; Shanmugam, M.K.; Chen, L.; Chatterjee, S.; Basha, J.; Kumar, A.P.; Kundu, T.K.; Sethi, G. Garcinol, a Polyisoprenylated Benzophenone Modulates Multiple Proinflammatory Signaling Cascades Leading to the Suppression of Growth and Survival of Head and Neck Carcinoma. Cancer Prev. Res. 2013, 6, 843–854.
- Hu, H.; Han, T.; Zhuo, M.; Wu, L.L.; Yuan, C.; Wu, L.; Lei, W.; Jiao, F.; Wang, L.W. Elevated COX-2 Expression Promotes Angiogenesis through EGFR/p38-MAPK/Sp1-Dependent Signalling in Pancreatic Cancer. Sci. Rep. 2017, 7, 470.
- Matsubayashi, H.; Infante, J.R.; Winter, J.; Klein, A.P.; Schulick, R.; Hruban, R.; Visvanathan, K.; Goggins, M. Tumor COX-2 Expression and Prognosis of Patients with Resectable Pancreatic Cancer. Cancer Biol. Ther. 2007, 6, 1569–1575.
- Eibl, G.; Takata, Y.; Boros, L.G.; Liu, J.; Okada, Y.; Reber, H.A.; Hines, O.J. Growth Stimulation of COX-2-Negative Pancreatic Cancer by a Selective COX-2 Inhibitor. Cancer Res. 2005, 65, 982–990.
- Shanmugam, M.K.; Warrier, S.; Kumar, A.P.; Sethi, G.; Arfuso, F. Potential Role of Natural Compounds as Anti-Angiogenic Agents in Cancer. Curr. Vasc. Pharmacol. 2017, 15, 503–519.
- Zhong, Y.; Xia, Z.; Liu, J.; Lin, Y.; Zan, H. The Effects of Cyclooxygenase-2 Gene Silencing by siRNA on Cell Proliferation, Cell Apoptosis, Cell Cycle and Tumorigenicity of Capan-2 Human Pancreatic Cancer Cells. Oncol. Rep. 2012, 27, 1003–1010.
- Jung, Y.Y.; Lee, J.H.; Nam, D.; Narula, A.S.; Namjoshi, O.A.; Blough, B.E.; Um, J.Y.; Sethi, G.; Ahn, K.S. Anti-Myeloma Effects of Icariin are Mediated through the Attenuation of JAK/STAT3-Dependent Signaling Cascade. Front. Pharmacol. 2018, 9, 531.
- Arora, L.; Kumar, A.P.; Arfuso, F.; Chng, W.J.; Sethi, G. The Role of Signal Transducer and Activator of Transcription 3 (Stat3) and Its Targeted Inhibition in Hematological Malignancies. Cancers (Basel) 2018, 10, 327.
- Kim, C.; Lee, S.G.; Yang, W.M.; Arfuso, F.; Um, J.Y.; Kumar, A.P.; Bian, J.; Sethi, G.; Ahn, K.S. Formononetin-induced Oxidative Stress Abrogates the Activation of STAT3/5 Signaling Axis and Suppresses the Tumor Growth in Multiple Myeloma Preclinical Model. Cancer Lett. 2018, 431, 123–141.
- Ma, H.; Zhang, J.; Zhou, L.; Wen, S.; Tang, H.Y.; Jiang, B.; Zhang, F.; Suleman, M.; Sun, D.; Chen, A.; et al. c-Src Promotes Tumorigenesis and Tumor Progression by Activating PFKFB3. Cell Rep. 2020, 30, 4235-4249.e6.
- Tsuboi, Y.; Oyama, M.; Kozuka-Hata, H.; Ito, A.; Matsubara, D.; Murakami, Y. CADM1 Suppresses c-Src Activation by Binding with Cbp on Membrane Lipid Rafts and Intervenes Colon Carcinogenesis. Biochem. Biophys. Res. Commun. 2020, 529, 854–860.
- Mayoral-Varo, V.; Calcabrini, A.; Sánchez-Bailón, M.P.; Martínez-Costa, Ó.H.; González-Páramos, C.; Ciordia, S.; Hardisson, D.; Aragón, J.J.; Fernández-Moreno, M.; Martín-Pérez, J. c-Src Functionality Controls self-Renewal and Glucose Metabolism in MCF7 Breast Cancer Stem Cells. PLoS ONE 2020, 15, e0235850.
- Zhang, S.; Li, G.; Liu, C.; Lu, S.; Jing, Q.; Chen, X.; Zheng, H.; Ma, H.; Zhang, D.; Ren, S.; et al. miR-30e-5p Represses Angiogenesis and Metastasis by Directly Targeting AEG-1 in Squamous Cell Carcinoma of the Head and Neck. Cancer Sci. 2020, 111, 356–368.
- Chen, R.Y.; Yen, C.J.; Liu, Y.W.; Guo, C.G.; Weng, C.Y.; Lai, C.H.; Wang, J.M.; Lin, Y.J.; Hung, L.Y. CPAP Promotes Angiogenesis and Metastasis by Enhancing STAT3 Activity. Cell Death Differ. 2020, 27, 1259–1273.
- Shanmugam, M.K.; Ahn, K.S.; Hsu, A.; Woo, C.C.; Yuan, Y.; Tan, K.H.B.; Chinnathambi, A.; Alahmadi, T.A.; Alharbi, S.A.; Koh, A.P.F.; et al. Thymoquinone Inhibits Bone Metastasis of Breast Cancer Cells through Abrogation of the CXCR4 Signaling Axis. Front. Pharmacol. 2018, 9, 1294.
- Zhao, X.; Li, D.C.; Zhao, H.; Li, Z.; Wang, J.X.; Zhu, D.M.; Zhou, J.; Cen, J.N. A Study of the Suppressive Effect on Human Pancreatic Adenocarcinoma Cell Proliferation and Angiogenesis by Stable Plasmid-Based siRNA Silencing of c-Src Gene Expression. Oncol. Rep. 2012, 27, 628–636.
- Ashrafizadeh, M.; Najafi, M.; Orouei, S.; Zabolian, A.; Saleki, H.; Azami, N.; Sharifi, N.; Hushmandi, K.; Zarrabi, A.; Ahn, K.S. Resveratrol Modulates Transforming Growth Factor-Beta (tgf-β) Signaling Pathway for Disease Therapy: A New Insight into Its Pharmacological Activities. Biomedicines 2020, 8, 261.
- Ashrafizadeh, M.; Zarrabi, A.; Hushmandi, K.; Zarrin, V.; Moghadam, E.R.; Hashemi, F.; Makvandi, P.; Samarghandian, S.; Khan, H.; Hashemi, F. Toward Regulatory Effects of Curcumin on Transforming Growth Factor-Beta across Different Diseases: A Review. Front. Pharmacol. 2020, 11, 1785.
- Yoshimoto, S.; Tanaka, F.; Morita, H.; Hiraki, A.; Hashimoto, S. Hypoxia-Induced HIF-1α and ZEB1 Are Critical for the Malignant Transformation of Ameloblastoma via TGF-β-Dependent EMT. Cancer Med. 2019, 8, 7822–7832.
- Cheng, J.T.; Wang, L.; Wang, H.; Tang, F.R.; Cai, W.Q.; Sethi, G.; Xin, H.W.; Ma, Z. Insights into Biological Role of LncRNAs in Epithelial-Mesenchymal Transition. Cells 2019, 8, 1178.
- Lee, J.H.; Chinnathambi, A.; Alharbi, S.A.; Shair, O.H.M.; Sethi, G.; Ahn, K.S. Farnesol Abrogates Epithelial to Mesenchymal Transition Process through Regulating Akt/mTOR Pathway. Pharmacol. Res. 2019, 150, 104504.
- Yoneyama, M.; Fujita, T. RNA Recognition and Signal Transduction by RIG-I-like Receptors. Immunol. Rev. 2009, 227, 54–65.
- Besch, R.; Poeck, H.; Hohenauer, T.; Senft, D.; Häcker, G.; Berking, C.; Hornung, V.; Endres, S.; Ruzicka, T.; Rothenfusser, S.; et al. Proapoptotic Signaling Induced by RIG-I and MDA-5 Results in Type I Interferon-Independent Apoptosis in Human Melanoma Cells. J. Clin. Investig. 2009, 119, 2399–2411.
- Poeck, H.; Besch, R.; Maihoefer, C.; Renn, M.; Tormo, D.; Morskaya, S.S.; Kirschnek, S.; Gaffal, E.; Landsberg, J.; Hellmuth, J. 5′-Triphosphate-siRNA: Turning Gene Silencing and Rig-I Activation against Melanoma. Nat. Med. 2008, 14, 1256–1263.
- Ellermeier, J.; Wei, J.; Duewell, P.; Hoves, S.; Stieg, M.R.; Adunka, T.; Noerenberg, D.; Anders, H.J.; Mayr, D.; Poeck, H.; et al. Therapeutic Efficacy of Bifunctional siRNA Combining TGF-β1 Silencing with RIG-I Activation in Pancreatic Cancer. Cancer Res. 2013, 73, 1709–1720.
- Schnurr, M.; Duewell, P. Breaking Tumor-Induced Immunosuppression with 5′-Triphosphate siRNA Silencing TGFβ and Activating RIG-I. Oncoimmunology 2013, 2, e24170.
- Ruan, Z.; Liang, M.; Shang, L.; Lai, M.; Deng, X.; Su, X. Shikonin-Mediated PD-L1 Degradation Suppresses Immune Evasion in Pancreatic Cancer by Inhibiting NF-κB/STAT3 and NF-κB/CSN5 Signaling Pathways. Pancreatology 2021, 21, 630–664.
- Ashrafizadeh, M.; Zarrabi, A.; Hushmandi, K.; Zarrin, V.; Moghadam, E.R.; Zabolian, A.; Tavakol, S.; Samarghandian, S.; Najafi, M. PD-1/PD-L1 axis regulation in cancer therapy: The role of long non-coding RNAs and microRNAs. Life Sci. 2020, 256, 117899.
- Manu, K.A.; Shanmugam, M.K.; Ramachandran, L.; Li, F.; Siveen, K.S.; Chinnathambi, A.; Zayed, M.E.; Alharbi, S.A.; Arfuso, F.; Kumar, A.P.; et al. Isorhamnetin Augments the Anti-Tumor Effect of Capecitabine through the Negative Regulation of NF-κB Signaling Cascade in Gastric Cancer. Cancer Lett. 2015, 363, 28–36.
- Manu, K.A.; Shanmugam, M.K.; Li, F.; Chen, L.; Siveen, K.S.; Ahn, K.S.; Kumar, A.P.; Sethi, G. Simvastatin Sensitizes Human Gastric Cancer Xenograft in Nude Mice to Capecitabine by Suppressing Nuclear Factor-Kappa B-Regulated Gene Products. J. Mol. Med. (Berl) 2014, 92, 267–276.
- Chen, C.; Tang, P.; Yue, J.; Ren, P.; Liu, X.; Zhao, X.; Yu, Z. Effect of siRNA Targeting HIF-1alpha Combined L-Ascorbate on Biological Behavior of Hypoxic MiaPaCa2 Cells. Technol. Cancer Res. Treat 2009, 8, 235–240.
- Xie, W.; Chu, M.; Song, G.; Zuo, Z.; Han, Z.; Chen, C.; Li, Y.; Wang, Z.W. Emerging Roles of Long Noncoding RNAs in Chemoresistance of Pancreatic Cancer. Semin. Cancer Biol. 2020, S1044-1579X(1020)30222-30224.
- Gao, Z.; Wu, J.; Wu, X.; Zheng, J.; Ou, Y. SRPX2 Boosts Pancreatic Cancer Chemoresistance by Activating PI3K/AKT Axis. Open Med. (Wars) 2020, 15, 1072–1082.
- Chen, H.; Zhu, X.; Sun, R.; Ma, P.; Zhang, E.; Wang, Z.; Fan, Y.; Zhou, G.; Mao, R. Ubiquitin-Specific Protease 7 Is a Druggable Target That Is Essential for Pancreatic Cancer Growth And Chemoresistance. Investig. New Drugs 2020, 38, 1707–1716.
- Elledge, S.J.; Zhou, Z.; Allen, J.B. Ribonucleotide Reductase: Regulation, Regulation, Regulation. Trends Biochem. Sci. 1992, 17, 119–123.
- Youns, M.; Askoura, M.; Abbas, H.A.; Attia, G.H.; Khayyat, A.N.; Goda, R.M.; Almalki, A.J.; Khafagy, E.S.; Hegazy, W.A.H. Celastrol Modulates Multiple Signaling Pathways to Inhibit Proliferation of Pancreatic Cancer via DDIT3 and ATF3 up-Regulation and RRM2 and MCM4 Down-Regulation. Onco Targets Ther. 2021, 14, 3849–3860.
- Zheng, S.; Wang, X.; Weng, Y.H.; Jin, X.; Ji, J.L.; Guo, L.; Hu, B.; Liu, N.; Cheng, Q.; Zhang, J.; et al. siRNA Knockdown of RRM2 Effectively Suppressed Pancreatic Tumor Growth Alone or Synergistically with Doxorubicin. Mol. Ther. Nucleic Acids 2018, 12, 805–816.
- Guo, H.; Liu, F.; Yang, S.; Xue, T. Emodin Alleviates Gemcitabine Resistance in Pancreatic Cancer by Inhibiting MDR1/P-Glycoprotein and MRPs Expression. Oncol Lett. 2020, 20, 167.
- Tan, J.; Zhou, X.; Zhu, H. hTERT-siRNA Could Potentiate the Cytotoxic Effect of Gemcitabine to Pancreatic Cancer cells Bxpc-3. Exp. Clin. Transplant. 2012, 10, 386–393.
- Zhong, Y.Q.; Xia, Z.S.; Fu, Y.R.; Zhu, Z.H. Knockdown of hTERT by SiRNA Suppresses Growth of Capan-2 Human Pancreatic Cancer Cell via the Inhibition of Expressions of Bcl-2 and COX-2. J. Dig. Dis. 2010, 11, 176–184.
- Ashrafizadeh, M.; Zarrabi, A.; Hushmandi, K.; Kalantari, M.; Mohammadinejad, R.; Javaheri, T.; Sethi, G. Association of the Epithelial-Mesenchymal Transition (EMT) with Cisplatin Resistance. Int. J. Mol. Sci. 2020, 21, 4002.
- Mirzaei, S.; Gholami, M.H.; Hashemi, F.; Zabolian, A.; Farahani, M.V.; Hushmandi, K.; Zarrabi, A.; Goldman, A.; Ashrafizadeh, M.; Orive, G. Advances in Understanding the Role of P-gp in Doxorubicin Resistance: Molecular Pathways, Therapeutic Strategies, and Prospects. Drug Discov. Today 2021, S1359-6446(21)00428-1.
- Paskeh, M.D.A.; Mirzaei, S.; Orouei, S.; Zabolian, A.; Saleki, H.; Azami, N.; Hushmandi, K.; Baradaran, B.; Hashmi, M.; Aref, A.R. Revealing the Role of miRNA-489 as a New Onco-Suppressor Factor in Different Cancers Based on Pre-Clinical and Clinical Evidence. Int. J. Biol. Macromol. 2021, 191, 727–737.
- Ashrafizadeh, M.; Mirzaei, S.; Gholami, M.H.; Hashemi, F.; Zabolian, A.; Raei, M.; Hushmandi, K.; Zarrabi, A.; Voelcker, N.H.; Aref, A.R. Hyaluronic acid-Based Nanoplatforms for Doxorubicin: A Review of Stimuli-Responsive Carriers, Co-Delivery And Resistance Suppression. Carbohydr. Polym. 2021, 272, 118491.
- Mirzaei, S.; Gholami, M.H.; Zabolian, A.; Saleki, H.; Farahani, M.V.; Hamzehlou, S.; Far, F.B.; Sharifzadeh, S.O.; Samarghandian, S.; Khan, H. Caffeic Acid and Its Derivatives as Potential Modulators of Oncogenic Molecular Pathways: New Hope in the Fight against Cancer. Pharmacol. Res. 2021, 171, 105759.
- Chopra, P.; Sethi, G.; Dastidar, S.G.; Ray, A. Polo-like Kinase Inhibitors: An Emerging Opportunity for Cancer Therapeutics. Expert Opin. Investig. Drugs 2010, 19, 27–43.
- Mishra, S.; Verma, S.S.; Rai, V.; Awasthee, N.; Chava, S.; Hui, K.M.; Kumar, A.P.; Challagundla, K.B.; Sethi, G.; Gupta, S.C. Long Non-Coding RNAs Are Emerging Targets of Phytochemicals for Cancer and Other Chronic Diseases. Cell Mol. Life Sci. 2019, 76, 1947–1966.
- Ashrafizaveh, S.; Ashrafizadeh, M.; Zarrabi, A.; Husmandi, K.; Zabolian, A.; Shahinozzaman, M.; Aref, A.R.; Hamblin, M.R.; Nabavi, N.; Crea, F. Long Non-Coding RNA in the Doxorubicin Resistance of Cancer Cells. Cancer Lett. 2021, 508, 104–114.
- Martinez-Contreras, R.; Cloutier, P.; Shkreta, L.; Fisette, J.F.; Revil, T.; Chabot, B. hnRNP Proteins and Splicing Control. Adv. Exp. Med. Biol. 2007, 623, 123–147.
- Gu, W.; Liu, W.; Shen, X.; Shi, Y.; Wang, L.; Liu, H. Emergence of Heterogeneous Nuclear Ribonucleoprotein A2/B1 vs. Loss of E-Cadherin: Their Reciprocal Immunoexpression Profiles in Human Pancreatic Cancer. Ann. Diagn. Pathol. 2013, 17, 14–17.
- Begines, P.; Sevilla-Horrillo, L.; Puerta, A.; Puckett, R.; Bayort, S.; Lagunes, I.; Maya, I.; Padrón, J.M.; López, Ó.; Fernández-Bolaños, J.G. Masked Phenolic-Selenium Conjugates: Potent and Selective Antiproliferative Agents Overcoming P-gp Resistance. Pharmaceuticals 2020, 13, 358.
- Gu, W.J.; Liu, H.L. Induction of Pancreatic Cancer Cell Apoptosis, Invasion, Migration, and Enhancement of Chemotherapy Sensitivity of Gemcitabine, 5-FU, and Oxaliplatin by hnRNP A2/B1 siRNA. Anticancer Drugs 2013, 24, 566–576.
- Goan, Y.G.; Zhou, B.; Hu, E.; Mi, S.; Yen, Y. Overexpression of Ribonucleotide Reductase as a Mechanism of Resistance to 2,2-Difluorodeoxycytidine in the Human KB Cancer Cell Line. Cancer Res. 1999, 59, 4204–4207.
- Eriksson, S.; Martin, D.W.J. Ribonucleotide Reductase in Cultured Mouse Lymphoma Cells. Cell Cycle-Dependent Variation in the Activity of Subunit Protein M2. J. Biol. Chem. 1981, 256, 9436–9440.
- Duxbury, M.S.; Ito, H.; Zinner, M.J.; Ashley, S.W.; Whang, E.E. RNA Interference Targeting the M2 Subunit of Ribonucleotide Reductase Enhances Pancreatic Adenocarcinoma Chemosensitivity to Gemcitabine. Oncogene 2004, 23, 1539–1548.
- Réjiba, S.; Bigand, C.; Parmentier, C.; Hajri, A. Gemcitabine-Based Chemogene Therapy for Pancreatic Cancer Using Ad-dCK::UMK GDEPT and TS/RR siRNA Strategies. Neoplasia 2009, 11, 637–650.
- Yang, L.; Kang, W.K. The Effect of HIF-1alpha siRNA on Growth and Chemosensitivity of MIA-Paca Cell Line. Yonsei Med. J. 2008, 49, 295–300.
- Al Hanjori, A.S.; Alshaer, W.; Anati, B.; Wehaibi, S.; Zihlif, M. Studying Antitumor Effects of siRNA Gene Silencing of Some Metabolic Genes in Pancreatic Ductal Adenocarcinoma. Curr.Mol. Pharmacol. 2020, 14, 604–619.
This entry is offline, you can click here to edit this entry!
