Metabolic syndrome (MetS) forms a cluster of metabolic dysregulations including insulin resistance, atherogenic dyslipidemia, central obesity, and hypertension. The pathogenesis of MetS encompasses multiple genetic and acquired entities that fall under the umbrella of insulin resistance and chronic low-grade inflammation. If left untreated, MetS is significantly associated with an increased risk of developing diabetes and cardiovascular diseases (CVDs). Given that CVDs constitute by far the leading cause of morbidity and mortality worldwide, it has become essential to investigate the role played by MetS in this context to reduce the heavy burden of the disease.
- metabolic syndrome
- insulin resistance
- nutraceuticals
1. Introduction
1.1. History
| Clinical Measure | Criteria | Diagnosis | ||||
|---|---|---|---|---|---|---|
| Central Obesity | Blood Glc | High TG | Low HDL | High BP | ||
| AHA/ NHLBI (2009) [4] |
>40” (men) or >35” (women) |
|
|
|
|
≥3 criteria |
| IDF (2005) [5,6] |
>32” (women) or
|
≥3 criteria one of which should be central obesity | ||||
| ATPIII (2001) [7] |
WC >40” (men) or >35” (women) |
|
|
|
≥3 criteria | |
| EGIR (1999) [8] |
>32” (women) |
|
|
|
≥3 criteria one of which should be IR * | |
| WHO (1998) [1] |
|
|
|
|
≥3 criteria one of which should be IR ** | |
1.2. Epidemiology
2. Management
2.1. Mediterranean Diet
2.2. Nutraceuticals
| Source | Action |
|---|---|
| Turmeric (Curcuma longa) Active ingredient: diferuloylmethane in curcumin |
Suppress NF-kB activation-> ↓ expression of pro-inflammatory cytokines-↓ TNF-α expression, ↓ expression of plasminogen activator inhibitor type-1-> ↓ inflammation [202]—antioxidant effect [201]—curcumin hinders Wnt/β-catenin pathway associated with obesity [203] |
| Garlic (Allium sativum) Active ingredient: allicin |
Anti-inflammatory effect from the organosulfur compounds in its derivatives. Antioxidant action due to thiol groups—Antithrombotic effect [201]—increases insulin sensitivity [204] |
| Cinnamon (Cinnamomum verum) Active ingredient: polyphenols |
Antithrombotic–antioxidant–anti-inflammatory effects—increases insulin sensitivity—regulates blood glucose and blood pressure [201] |
| Rhizoma coptidis Active ingredient: berberine |
Improves body weight, triglyceride levels—increases insulin sensitivity—downregulation of genes involved in lipogenesis [205]—reduction in blood pressure [206] |
| Neem (Azadirachta indica) Active ingredient: neem oil |
Increases glucose tolerance via reduction of intestinal and pancreatic glucosidase activity → improves post-prandial hyperglycemia [207]—regenerates pancreatic beta cells → ↑ insulin secretion [201] |
| Bergamot orange (Citrus bergamia) Active ingredient: bergamot essential oil |
Anticancer–anti-inflammatory–antimicrobial–antioxidant–antianxiety properties—↓ ROS formation—↓ lectin-like LDL receptor-1 expression [208] |
| Grapes (Vitus vinifera) Active ingredient: resveratrol, (3,5,4′-trihydroxystilbene) |
↓ Adipogenesis—↑ lipolysis—inhibits cyclooxygenase → antioxidant action [209]—enhances insulin sensitivity, glucose tolerance, overall weight, and BMI—[210] |
| Onions (Allium cepa) Active ingredient: quercetin |
Anti-inflammatory—antioxidant—↓ blood pressure—↓ cholesterol levels—↓ insulin resistance [211] |
| Fish oils (omega fatty acids) Active ingredient: polyunsaturated fatty acids |
↓ Lipogenesis—↑ fatty acid oxidation in liver and adipose tissue—regulates peroxisome proliferator—activates receptor gamma [212] |
| Broccoli (Brassica oleracea) Active ingredient: sulforaphrane |
Anti-inflammatory properties—activates nuclear factor erythroid 2-related factor 2, an antioxidant transcription factor → antioxidant properties—role against hypertension, hyperlipidemia, and diabetes [213] |
| Ginger Active ingredient: gingerols, shogaols, parasols |
Anti-inflammatory—↓ cyclooxygenase-2—↓ 5-lipoxygenase—↓ systolic blood pressure [201] |
| Cumin (Cuminum cyaminum) Active ingredient: cuminaldehyde |
↓ Lipid levels—↓ glycemia [201] |
| Cynara cardunculus (L.) subsp. scolymus Hayek-based Altilix® |
Modulates the expression of PPAR-γ and inhibits fatty acid synthase activity—↓ in body weight, waist circumference, HbA1c, plasma lipids, hepatic transaminases, flow-mediated dilation, carotid intima-media thickness [214]. |
| Monascus purpureus, red yeast rice | Reversible inhibition of 3-hydroxy-3-methyl-glutaryl-CoA reductase [215] |
2.3. Butyrate
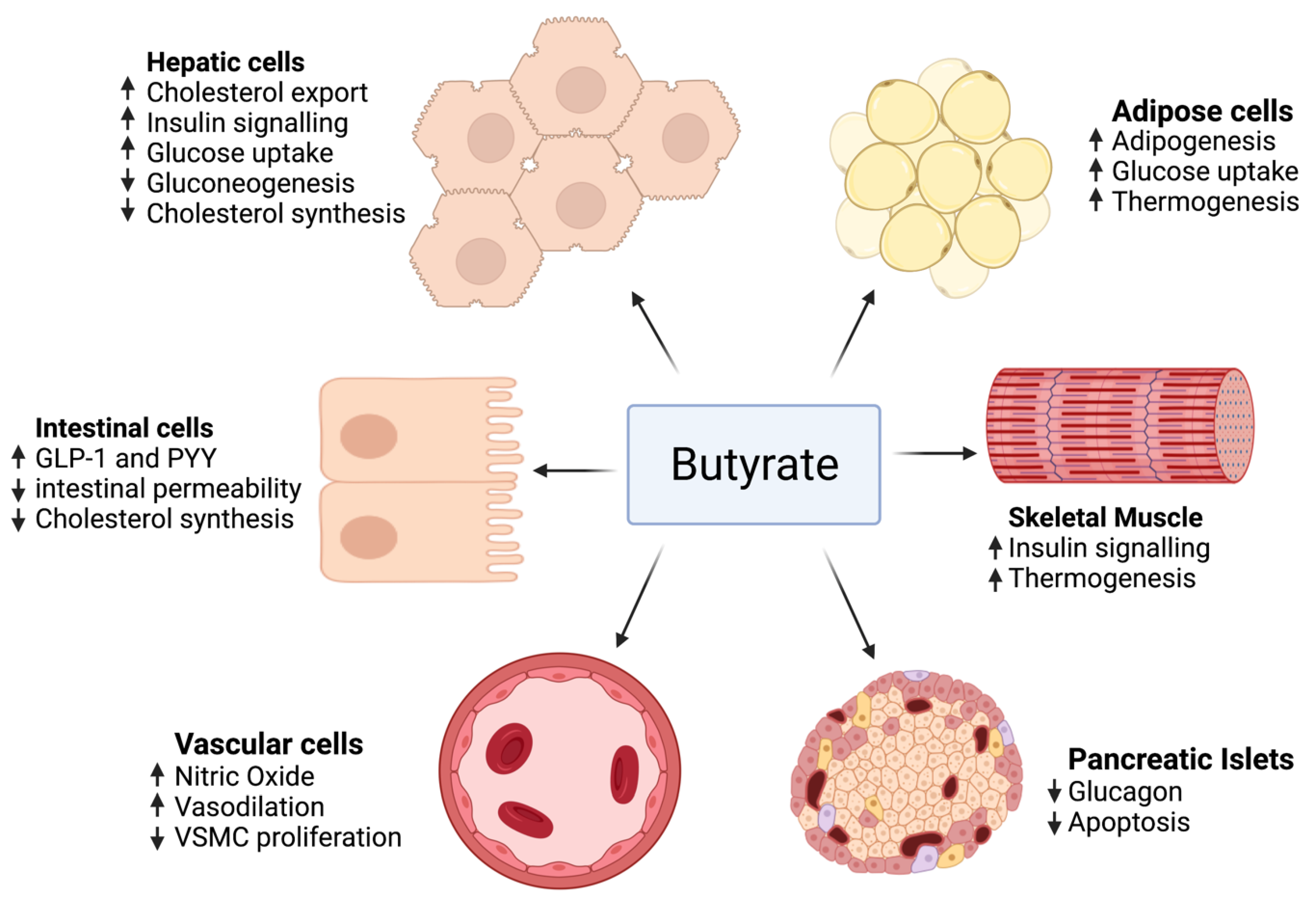
2.4. Probiotics
| Reference | Sample Size (n) | Age Range Probiotic Strain Period of Intervention (Weeks) Probiotic Dose |
Primary Outcomes | Secondary Outcomes |
|---|---|---|---|---|
| [247] | 28 | Control group: 54.5 ± 8.9 Probiotic group: 51.5 ± 11.4 Lactobacillus casei Shirota 12 milk (65 mL bottles × 3/day) 108 cells/mL |
No changes were found in BMI, BP, waist circumference, triacylglycerols, TC, and fasting glucose levels. | High-sensitive CRP (1.86 mg/L in the probiotic group vs. −1.60 mg/L in the placebo group, p = 0.016) and LBP levels (5827 ng/mL in the probiotic group vs. −1510 ng/mL in the placebo group, p = 0.023) increased within the probiotic group |
| [248] | 40 | Control group: 51.7 ± 12.1 Probiotic group: 52 ± 10.9 Lactobacillus plantarum TENSIA 3 cheese (50 g/day) 1.5 × 1011 CFU/g |
BMI was significantly reduced in the probiotic group (BMI variation in probiotic group −2 vs. −1.6 kg/m2 in the placebo group, p = 0.031). |
A positive association was detected between TENSIA colonization and the extent of change of morning diastolic BP (r = 0.617, p = 0.0248) |
| [249] | 28 | Control group: 55 ± 9 Probiotic group: 51 ± 11 Lactobacillus casei Shirota 12 milk (65 mL bottles × 3/day) 108 cells/mL |
No changes were found in BMI, fasting plasma glucose levels, and HOMA-IR index. | Probiotic supplementation resulted in a significant reduction in sVCAM-1 level (−195 ng/mL in the probiotic group vs. 30 ng/mL in the placebo group, p = 0.008) and a significant increase in high-sensitive CRP level (1.86 mg/L in the probiotic group vs. −1.60 mg/L in the placebo group, p = 0.002) |
| [247] | 24 | Control group: 63 ± 7.6 Probiotic group: 62 ± 4.35 Lactobacillus plantarum 12 milk (80 mL bottles × 1/day) 107 CFU/g |
Glucose levels showed a significant reduction in the FM group compared with the NFM group (glucose variation in FM −10.5 vs. −3 mg/dL in NFM group, p = 0.037). | Homocysteine levels showed a significant reduction in the FM group compared with the NFM group p = 0.019). |
| [250] | 28 | Control group: 55 ± 9 Probiotic group: 51 ± 11 Lactobacillus casei Shirota 12 milk (65 mL bottles × 3/day) 108 cells/mL |
No changes were found in BMI, BP, waist circumference, triacylglycerols, and TC blood levels. | LcS administration was associated with subtle microbiota changes at a genus level (enrichment of Parabacteroidetes) |
| [251] | 51 | No data Bifidobacterium lactis HN019 6 milk (80 mL bottle × 1/day) 3.4 × 108 CFU/mL |
Significant differences in BMI variation (probiotic group −1.3 vs. −0.3 kg/m2 in control group, p = 0.017); TC variation (probiotic group −15 vs. 6 mg/dL in control group, p = 0.09) and LDLc variation (probiotic group −17.5 vs. −2 mg/dL in control group, p = 0.08). | Significant decrease in TNFα and IL−6 (p < 0.05) in the probiotic group |
| [252] | 81 | Control group: 58.72 ± 7.25 Low-dose group: 56.38 ± 6.55 High-dose group: 55.16 ± 6.87 Bifidobacterium bifidum W23, Bifidobacterium lactis W51, Bifidobacterium lactis W52, Lactobacillus acidophilus W37, Lactobacillus brevis W63, Lactobacillus casei W56, Lactobacillus salivarius W24, Lactococcus lactis W19, and Lactococcus lactis W58 12 lyophilisate powder low dose (2.5 × 109 CFU/day) or high dose (1 × 1010 CFU/day) |
Significant differences were found in glucose variation (HD vs. placebo −0.61 mg/dL, p= 0.0272; HD vs. LD −0.72 mg/dL, p = 0.0043), insulin (HD vs. placebo −0.83 UI/L, p = 0.0002; HD vs. LD −0.40 UI/L, p = 0.0155), and HOMA-IR (HD vs. placebo −0.90, p = 0.0005; HD vs. LD −0.54 mg/dL, p = 0.0127). | Significant differences were found in uric acid (HD vs. placebo −0.73 mmol/L, p = 0.0109; HD vs. LD −0.92 mmol/L, p = 0.0016) and LPS levels (HD vs. placebo −0.99 ng/mL, p = 0.001) |
| [253] | 81 | Control group: 58.72 ± 7.25 Low-dose group: 56.38 ± 6.55 High-dose group: 55.16 ± 6.87 Bifidobacterium bifidum W23, Bifidobacterium lactis W51, Bifidobacterium lactis W52, Lactobacillus acidophilus W37, Lactobacillus brevis W63, Lactobacillus casei W56, Lactobacillus salivarius W24, Lactococcus lactis W19, and Lactococcus lactis W58 12 lyophilisate powder low dose (2.5 × 109 CFU/day) or high dose (1 × 1010 CFU/day) |
No changes were found in BMI and BP. | Significant differences were found in the pulse wave analysis systolic pressure (HD vs. placebo −1 mmHg, p = 0.0054; HD vs. LD −0.91 mmHg, p = 0.0057), the pulse wave analysis augmentation index (HD vs. placebo −0.55, p = 0.0079), the pulse wave velocity (HD vs. placebo −0.82 m/s, p = 0.0045; HD vs. LD −0.55 m/s, p = 0.0189), VEGF (HD vs. placebo −1.09 pg/mL, p = 0.0001; HD vs. LD −1.10 pg/mL, p = 0.0007), TNFα (HD vs. placebo −1.03 pg/mL, p = 0.0009; HD vs. LD −0.68 pg/mL, p = 0.0471), and thrombomodulin levels (HD vs. placebo −0.78 ng/mL, p= 0.0194) |
| [254] | 44 | Control group: 44.55 ± 5.70 Probiotic group: 44.05 ± 6.60 Lactobacillus acidophilus La5, Bifidobacterium lactis Bb12 8 yogurt containing 6.45 × 106 CFU/g of L. acidophilus and 4.94 × 106 CFU/g of B. lactis Bb12 |
Consumption of probiotic yogurt resulted in a significant reduction in the level of blood glucose (mean difference: −3.80, p = 0.01). | Consumption of probiotic yogurt resulted in a significant reduction in the level of VCAM-1 (mean difference −463.39, p = 0.001) |
2.5. Coconut Oil Effect on Asymmetric Dimethylarginine (ADMA)
3. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/ijms23020786
