The endocannabinoids system (ECS) has garnered considerable interest as a potential therapeutic target in various carcinomas and cancer-related conditions alongside neurodegenerative diseases. Cannabinoids are implemented in several physiological processes such as appetite stimulation, energy balance, pain modulation and the control of chemotherapy-induced nausea and vomiting (CINV). However, pharmacokinetics and pharmacodynamics interactions could be perceived in drug combinations, so in this short review we tried to shed light on the potential drug interactions of medicinal cannabis. Hitherto, few data have been provided to the healthcare practitioners about the drug–drug interactions of cannabinoids with other prescription medications. In general, cannabinoids are usually well tolerated, but bidirectional effects may be expected with concomitant administered agents via affected membrane transporters (Glycoprotein p, breast cancer resistance proteins, and multidrug resistance proteins) and metabolizing enzymes (Cytochrome P450 and UDP-glucuronosyltransferases). Caution should be undertaken to closely monitor the responses of cannabis users with certain drugs to guard their safety, especially for the elderly and people with chronic diseases or kidney and liver conditions.
- Cannabis
- cannabinoids
- THC
- CBD
- drug–drug interactions
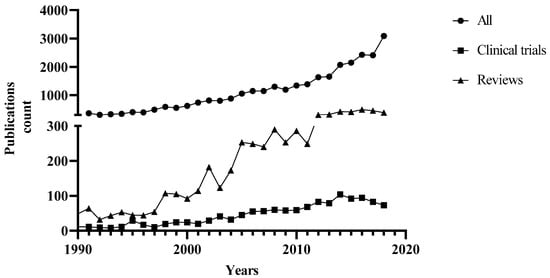
1. Introduction
| Cannabis-Based Treatment | Study Type/Location/n | Dosage/Administration | Efficacy, Tolerability and Notes | References |
|---|---|---|---|---|
| Chemotherapy-Induced Nausea and Vomiting (CINV) | ||||
| -Dronabinol [Marinol®; (-) trans Δ9-THC) alone or in combination with ondansetron (8–15 mg IV] |
-Interventional (Placebo controlled). -n = 64. -USA. |
-Capsule (2.5–20 mg). -Oral. |
-Both were effective in CINV and well tolerated while dronabinol was more effective. -Combination is not more effective. |
[27] |
| -Dronabinol [Marinol®; (-) trans Δ9-THC] |
-Interventional (retrospective). -Children with malignancy. |
-Solution administered orally (2.5–5 mg/m2 body surface every 6 h as needed). | -Positive responses were reported for 60% of patients. -Prospective trial would be needed to confirm the dronabinol effect in CINV therapy. |
[28] |
| -Nabilone with 5HT3 antagonist | -Interventional (retrospective) -n = 110 with median age 14 years with malignancy. |
-Oral. | -Adverse effect was reported with minor clinical significance. -Poor nausea control in nabilone treated group. |
[29] |
| Cancer Pain | ||||
| -Sativex® (Δ9-THC: CBD at a ratio of 27:25 mg/mL) -THC (27 mg/mL) |
-Interventional (Double-Blind, Randomized, Parallel-Group, Placebo-Controlled), n = 177. -Phase 3. -UK. |
-Oromucodal spray with maximum Δ9-THC: CBD (130:120 mg/day) or 130 mg/day Δ9-THC alone Each actuation is 100 μL. |
-Compared with the placebo, the Sativex treated group showed significant pain relief, unlike the Δ9-THC which was non-significant. -Reported adverse effects included dizziness, gastrointestinal disorders and confusion. |
[30] |
| -Sativex® (Δ9-THC: CBD at a ratio of 27:25 mg/mL) |
-Interventional (single group assignment) -Phase 3. -UK. |
-Oromucodal spray with a maximum 130:120 mg/day of Δ9-THC: CBD. | -The long-term use is well tolerated without losing pain-relieving effects in terminal cancer-related pain refectory to opioids. -Adverse effects and tolerability were assessed at the RCT withdrawal visit, 7–10 days later, then monthly, and at the withdrawal or completion of the study. |
[31] |
| - Sativex® (Δ9-THC: CBD at a ratio of 27:25 mg/mL) |
-Interventional (Double-Blind, Randomized, Parallel-Group, Placebo-Controlled). -Phase 3. -Multicentric. -n = 399. |
-Oromucodal spray (100 μL per actuation twice daily in the morning and evening with a maximum of 10 sprays for 5 weeks). | -No significant difference was reported in advanced cancer patients with chronic pain (unalleviated with opioids). -Nabiximol is still beneficial to secondary endpoints. -No evidence of abuse or misuse was reported. |
[32] |
| -No significant difference was reported in advanced cancer patients with chronic uncontrolled pain. | [33] | |||
| -Nabiximols (Sativex®; Δ9-THC: CBD at a ratio of 27:25 mg/mL) | -Interventional (Double-Blind, Randomized, Parallel-Group, Placebo-Controlled). -Phase 2. -USA. -n = 360. |
-Oromucodal spray in low (1–4 sprays/day), medium (6–10 sprays/day) and high (11–16 sprays/day) doses. | -Efficacy and safety were reported at low and medium doses against advanced cancer pain. -The adverse effects at high doses. |
[34] |
| -Nabiximols (Sativex®, Δ9-THC: CBD at a ratio of 27:25 mg/mL) | -Interventional (Double-Blind, Placebo-controlled, Crossover Pilot trial). -n = 16. |
-Sublingual spray (7.5–30 mg/day). | -No significant difference was reported against chemotherapy-induced neuropathy. -Two-fold reduction of the pain in the responder group with adverse effects. |
[35] |
| Cannabis cigarettes (3.56% Δ9-THC) in combination with opiates | -Interventional (open label). | -Pulmonary administration for chronic pain, including cancer patients. | -Declined chronic pain around 27% in patients receiving oxycodone or morphine analgesics. -No serious adverse effects were reported. |
[36] |
2. Potential Drug Interactions
3. Effects of Cannabis on Drug Metabolizing Enzymes and Related Drug Interactions
| Cannabinoid Based Treatment and Interactions | Affected Transporters and/or Metabolic Enzymes | Experimental Results, Notes and Outcomes | References |
|---|---|---|---|
| Cannabis, THC, CBD, CBN with either chemotherapy, abuse drugs or medications | -Membrane transporters ABC superfamily (glycoprotein P; P-gp, Breast cancer-resistance protein; BCRP, and multidrug resistance protein; MRP1, 2, 3 and 4) -Cytochrome P450 (3A, 2D6, 2C9, 1A1, 1A2, 1B1, 2B6 and 2C8) -UDP-glucuronosyltransferases (UGTs) |
-P-gp, BCRP, and MRP1-4 transporters expression were dysregulated by cannabinoids, but in higher concentrations than that usually measured in cannabis smokers. -CYP3A was competitively inhibited by THC, CBD and CBN, with CBD being the most potent in a concentration compatible with that in usual cannabis inhalation. -CYP2D6 was inhibited by THC, CBD and CBN, with CBD being the most potent in a higher concentration than that in usual cannabis consumption. -CYP2C9 was inhibited by THC, CBD and CBN, with CBD inhibitory effect being dependent on the used substrates. -CYP1A1, 1A2, 1B1, 2B6, 2C19, 3A4 and 2C8 were strongly inhibited by CBD. -UGT1A9, and 2B7 were inhibited by CBD. -UGT1A7, 1A8, and 1A9 were inhibited by CBN. -UGT2B7 was activated by CBN.
|
[19][54][55] |
| Δ9-THC, CBD and marijuana inhalation with psychotropic agents | -Cytochrome P450 | -CYP2C9 and CYP3A4 were inhibited by Δ9-THC. -CYP2C19 and CYP3A4 were inhibited by CBD. -CYP1A1 and CYP1A2 were induced by marijuana inhalation.
|
[56] |
| Cannabinoids on other drugs | Cytochrome P450 | -CYP3A4 inhibitors and stimulators affect the elimination of Δ9-THC and CBD.
|
[57] |
| CBD with antiepileptic drugs | Cytochrome P450 or unknown | Clinical studies of DDI: -Non-significant increase of the clobazam plasma level administered with CBD (n = 13 children) due to potent inhibition of CYP2C19. -Significant change of plasma level of N-desmethylclobazam by CBD co-administration while no significant change in the level of valproate, stiripentol and levetiracetam (n = 24 open label trial). -All patients showed significant changes of the plasma levels of clobazam, N-desmethylclobazam, rufinamide, and topiramate by increasing CBD doses. The mean therapeutic range was exceeded for clobazam and N-desmethylclobazam; the plasma levels of eslicarbazepine and zonisamide were increased in adults only (n = 39 adults and 42 children).
|
[51][58] |
| Synthetic and Phyto-cannabinoids | -Cytochrome P450 -UGTs |
-CYP1A catalysed MROD activity was weakly inhibited by MAM-2201, JWH-019, STS-135, and UR-144. -CYP2C8 catalysed amodiaquine N-deethylase was strongly inhibited by AM-2201, MAM-2201, and EAM-2201. -CYP2C9 catalysed diclofenac hydroxylation and CYP3A-catalyzed midazolam 1′-hydroxylation were inhibited by AM-2201 and MAM-2201. -CYP2C9 catalysed diclofenac 4′-hydroxylation, CYP2C19-catalyzed [S] -mephenytoin 4′-hydroxylation, and CYP3A-catalyzed midazolam 1′ hydroxylation were strongly inhibited by EAM-2201 (time-dependent inhibition). -CYP2B6 and CYP2C9 were strongly inhibited by THC, CBN and CBD. -CYP2A6 was inhibited by THC and CBN (mechanism-based inhibition). -CYP2D6 was competitively inhibited by CBD. -CYP1A1 mRNA expression was increased by THC in Hepa-1 cells, but EROD activity in CYP1A1 supersomes was inhibited by THC. -CYP1A1, CYP1A2, and CYP1B1 were strongly inhibited by CBD (mechanism-based inhibition). -CYP3A was inhibited by CBD in human liver microsomes. -CYP2C19-catalyzed [S] -mephenytoin hydroxylation was inhibited by (CBD and THC (Mixed-type inhibition). -UGT1A9- and UGT2B7 catalysed ethanol glucuronidation were non-competitively inhibited by CBD, and unlike the inclined ethanol glucuronidation in human liver microsome by CBN (dose-dependent). -UGT1A3 catalysed chenodeoxycholic acid 24-acyl glucuronidation was strongly competitively inhibited by AM-2201, MAM-2201, and EAM-2201. -UGT2B7-mediated naloxone 3β-D-glucuronidation was competitively inhibited by AM-2201.
|
[59][60] |
4. Other Potential Drug Interactions
5. Conclusions
Funding
This work was partially supported by the Maxwell Family Foundation and Cairo University via the mission's sector at the Ministry of higher education, Cairo, Egypt.
This entry is adapted from the peer-reviewed paper 10.3390/medicines6010003
References
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018.
- Corroon, J.; Kight, R. Regulatory Status of Cannabidiol in the United States: A Perspective. Cannabis Cannabinoid Res. 2018, 3, 190–194.
- Wessam H. Abd-Elsalam; Muhammad A. Alsherbiny; Janice Y. Kung; David W. Pate; Raimar Löbenberg; LC–MS/MS quantitation of phytocannabinoids and their metabolites in biological matrices. Talanta, 2019, 204, 846-867, https://doi.org/10.1016/j.talanta.2019.06.053.
- Brown, A. Novel cannabinoid receptors. Br. J. Pharmacol. 2007, 152, 567–575.
- De Petrocellis, L.; Di Marzo, V. Non-CB 1, non-CB 2 receptors for endocannabinoids, plant cannabinoids, and synthetic cannabimimetics: Focus on G-protein-coupled receptors and transient receptor potential channels. J. Neuroimmune Pharmacol. 2010, 5, 103–121.
- Pertwee, R.G.; Howlett, A.; Abood, M.E.; Alexander, S.; Di Marzo, V.; Elphick, M.; Greasley, P.; Hansen, H.S.; Kunos, G.; Mackie, K. International Union of Basic and Clinical Pharmacology. LXXIX. Cannabinoid receptors and their ligands: Beyond CB1 and CB2. Pharmacol. Rev. 2010, 62, 588–631.
- Matsuda, L.A.; Lolait, S.J.; Brownstein, M.J.; Young, A.C.; Bonner, T.I. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 1990, 346, 561.
- Munro, S.; Thomas, K.L.; Abu-Shaar, M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 1993, 365, 61.
- Fraguas-Sánchez, A.I.; Torres-Suárez, A.I. Medical Use of Cannabinoids. Drugs 2018, 78, 1665–1703.
- Peres, F.F.; Diana, M.C.; Levin, R.; Suiama, M.A.; Almeida, V.; Vendramini, A.M.; Santos, C.M.; Zuardi, A.W.; Hallak, J.E.; Crippa, J.A. Cannabidiol administered during peri-adolescence prevents behavioral abnormalities in an animal model of schizophrenia. Front. Pharmacol. 2018, 9.
- Saravia, R.; Ten-Blanco, M.; Julià-Hernández, M.; Gagliano, H.; Andero, R.; Armario, A.; Maldonado, R.; Berrendero, F. Concomitant THC and stress adolescent exposure induces impaired fear extinction and related neurobiological changes in adulthood. Neuropharmacology 2018, 144, 345–357.
- Wade, N.E.; Wallace, A.L.; Swartz, A.M.; Lisdahl, K.M. Aerobic Fitness Level Moderates the Association Between Cannabis Use and Executive Functioning and Psychomotor Speed Following Abstinence in Adolescents and Young Adults. J. Int. Neuropsychol. Soc. 2018, 1–12.
- Guzmán, M. Cannabinoids: Potential anticancer agents. Nat. Rev. Cancer 2003, 3, 745–755.
- Bogdanovic, V.; Mrdjanovic, J.; Borisev, I. A Review of the Therapeutic Antitumor Potential of Cannabinoids. J. Altern. Compl. Med. 2017, 23, 831–836.
- Vuger, A.T.; Šeparović, R.; Silovski, T.; Pavlović, M.; Pavlica, V.; Knežević, S.V. Cannabis in oncology. Libri Oncol. 2016, 44, 51–57.
- Davis, M.P. Cannabinoids for Symptom Management and Cancer Therapy: The Evidence. J. Natl. Comprehen. Cancer Netw. 2016, 14, 915–922.
- Velasco, G.; Hernández-Tiedra, S.; Dávila, D.; Lorente, M. The use of cannabinoids as anticancer agents. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2016, 64, 259–266.
- Alsherbiny, M.A.; Bhuyan, D.J.; Low, M.N.; Chang, D.; Li, C.G.; Synergistic Interactions of Cannabidiol with Chemotherapeutic Drugs in MCF7 Cells: Mode of Interaction and Proteomics Analysis of Mechanisms. International Journal of Molecular Sciences 2021, 22(18):10103, 1-36, https://doi.org/10.3390/ijms221810103.
- Bouquié, R.; Deslandes, G.; Mazaré, H.; Cogné, M.; Mahé, J.; Grégoire, M.; Jolliet, P. Cannabis and anticancer drugs: Societal usage and expected pharmacological interactions—A review. Fundam. Clin. Pharmacol. 2018, 32, 462–484.
- Xie, L.; Xie, L.; Kinnings, S.L.; Bourne, P.E. Novel computational approaches to polypharmacology as a means to define responses to individual drugs. Annu. Rev. Pharmacol. Toxicol. 2012, 52, 361–379.
- Liu, H.; Wang, J.; Zhou, W.; Wang, Y.; Yang, L. Systems approaches and polypharmacology for drug discovery from herbal medicines: An example using licorice. J. Ethnopharmacol. 2013, 146, 773–793.
- Alsherbiny, M.A.; Abd-Elsalam, W.H.; El badawy, S.A.; Taher, E.; Fares, M.; Torres, A.; Chang, D.; Guang Li, C. Ameliorative and protective effects of ginger and its main constituents against natural, chemical and radiation-induced toxicities: A comprehensive review. Food Chem. Toxicol. 2018.
- Alsherbiny, M.A.; Ezzat, S.M.; Elsakhawy, F.S.; Kamel, G.M.; Abdel-Kawy, M.A. Impact of certain Solanum speciess natural products as potent cytotoxic and anti-Inflammatory agents. J. Med. Plants Res. 2015, 9, 779–786.
- Ho, T.T.; Tran, Q.T.; Chai, C.L. The polypharmacology of natural products. Futur. Med. Chem. 2018, 10, 1361–1368.
- Bhuyan, D.J.; Perera, S.; Kaur, K.; Alsherbiny; M.A., Low; M., Seto, S.W.; Li, C.G; Zhou, X.; Synergistic Effects of Chinese Herbal Medicine and Biological Networks. In: Bizzarri M. (eds) Approaching Complex Diseases. uman Perspectives in Health Sciences and Technology 2020, 2, 393-436, https://doi.org/10.1007/978-3-030-32857-3_17.
- Alsherbiny, Muhammad; Radwan, Ibrahim; Moustafa, Nour; Bhuyan, Deep; El-Waisi, Muath; Chang, Dennis; Li, Chun Guang.; Trustworthy Deep Neural Network for Inferring Anticancer Synergistic Combinations. IEEE Journal of Biomedical and Health Informatics 2021, (), 1-9, 10.1109/JBHI.2021.3126339.
- Meiri, E.; Jhangiani, H.; Vredenburgh, J.J.; Barbato, L.M.; Carter, F.J.; Yang, H.-M.; Baranowski, V. Efficacy of dronabinol alone and in combination with ondansetron versus ondansetron alone for delayed chemotherapy-induced nausea and vomiting. Curr. Med. Res. Opin. 2007, 23, 533–543.
- Elder, J.J.; Knoderer, H.M. Characterization of dronabinol usage in a pediatric oncology population. J. Pediatr. Pharmacol. Ther. 2015, 20, 462–467.
- Polito, S.; MacDonald, T.; Romanick, M.; Jupp, J.; Wiernikowski, J.; Vennettilli, A.; Khanna, M.; Patel, P.; Ning, W.; Sung, L.; et al. Safety and efficacy of nabilone for acute chemotherapy-induced vomiting prophylaxis in pediatric patients: A multicenter, retrospective review. Pediatr. Blood Cancer 2018, 65, e27374.
- Johnson, J.R.; Burnell-Nugent, M.; Lossignol, D.; Ganae-Motan, E.D.; Potts, R.; Fallon, M.T. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancer-related pain. J. Pain Symp. Manag. 2010, 39, 167–179.
- Fallon, M.T.; Albert Lux, E.; McQuade, R.; Rossetti, S.; Sanchez, R.; Sun, W.; Wright, S.; Lichtman, A.H.; Kornyeyeva, E. Sativex oromucosal spray as adjunctive therapy in advanced cancer patients with chronic pain unalleviated by optimized opioid therapy: Two double-blind, randomized, placebo-controlled phase 3 studies. Br. J. Pain 2017, 11, 119–133.
- Lichtman, A.H.; Lux, E.A.; McQuade, R.; Rossetti, S.; Sanchez, R.; Sun, W.; Wright, S.; Kornyeyeva, E.; Fallon, M.T. Results of a Double-Blind, Randomized, Placebo-Controlled Study of Nabiximols Oromucosal Spray as an Adjunctive Therapy in Advanced Cancer Patients with Chronic Uncontrolled Pain. J. Pain Symp. Manag. 2018, 55, 179–188.
- Johnson, J.R.; Lossignol, D.; Burnell-Nugent, M.; Fallon, M.T. An open-label extension study to investigate the long-term safety and tolerability of THC/CBD oromucosal spray and oromucosal THC spray in patients with terminal cancer-related pain refractory to strong opioid analgesics. J. Pain Symp. Manag. 2013, 46, 207–218.
- Portenoy, R.K.; Ganae-Motan, E.D.; Allende, S.; Yanagihara, R.; Shaiova, L.; Weinstein, S.; McQuade, R.; Wright, S.; Fallon, M.T. Nabiximols for opioid-treated cancer patients with poorly-controlled chronic pain: A randomized, placebo-controlled, graded-dose trial. J. Pain Off. J. Am. Pain Soc. 2012, 13, 438–449.
- Lynch, M.E.; Cesar-Rittenberg, P.; Hohmann, A.G. A Double-Blind, Placebo-Controlled, Crossover Pilot Trial With Extension Using an Oral Mucosal Cannabinoid Extract for Treatment of Chemotherapy-Induced Neuropathic Pain. J. Pain Symp. Manag. 2014, 47, 166–173.
- Abrams, D.I.; Couey, P.; Shade, S.B.; Kelly, M.E.; Benowitz, N.L. Cannabinoid–Opioid Interaction in Chronic Pain. Clin. Pharmacol. Ther. 2011, 90, 844–851.
- Li, C.G.; Yang, L.; Zhou, S.-F. Interactions between Chinese herbal medicines and drugs. Aust. J. Acupunct. Chin. Med. 2007, 2, 17.
- Damkier, P.; Lassen, D.; Christensen, M.M.H.; Madsen, K.G.; Hellfritzsch, M.; Pottegård, A. Interaction between warfarin and cannabis. Basic Clin. Pharmacol. Toxicol. 2018.
- Holland, M.; Lau, D.; Allen, J.; Arnold, J. The multidrug transporter ABCG2 (BCRP) is inhibited by plant-derived cannabinoids. Br. J. Pharmacol. 2007, 152, 815–824.
- Feinshtein, V.; Erez, O.; Ben-Zvi, Z.; Eshkoli, T.; Sheizaf, B.; Sheiner, E.; Holcberg, G. Cannabidiol enhances xenobiotic permeability through the human placental barrier by direct inhibition of breast cancer resistance protein: An ex vivo study. Am. J. Obstet. Gynecol. 2013, 209, 573.e1–573.e15.
- Holland, M.; Panetta, J.; Hoskins, J.; Bebawy, M.; Roufogalis, B.; Allen, J.; Arnold, J. The effects of cannabinoids on P-glycoprotein transport and expression in multidrug resistant cells. Biochem. Pharmacol. 2006, 71, 1146–1154.
- Zhu, H.-J.; Wang, J.-S.; Markowitz, J.S.; Donovan, J.L.; Gibson, B.B.; Gefroh, H.A.; DeVane, C.L. Characterization of P-glycoprotein inhibition by major cannabinoids from marijuana. J. Pharmacol. Exp. Ther. 2006, 317, 850–857.
- Tournier, N.; Chevillard, L.; Megarbane, B.; Pirnay, S.; Scherrmann, J.-M.; Decleves, X. Interaction of drugs of abuse and maintenance treatments with human P-glycoprotein (ABCB1) and breast cancer resistance protein (ABCG2). Int. J. Neuropsychopharmacol. 2010, 13, 905–915.
- Arnold, J.C.; Hone, P.; Holland, M.L.; Allen, J.D. CB2 and TRPV1 receptors mediate cannabinoid actions on MDR1 expression in multidrug resistant cells. Pharmacol. Rep. 2012, 64, 751–757.
- Feinshtein, V.; Erez, O.; Ben-Zvi, Z.; Erez, N.; Eshkoli, T.; Sheizaf, B.; Sheiner, E.; Huleihel, M.; Holcberg, G. Cannabidiol changes P-gp and BCRP expression in trophoblast cell lines. PeerJ 2013, 1, e153.
- Marquez, B.; Van Bambeke, F. ABC multidrug transporters: Target for modulation of drug pharmacokinetics and drug-drug interactions. Curr. Drug Targets 2011, 12, 600–620.
- Wittgen, H.G.; van den Heuvel, J.J.; van den Broek, P.H.; Dinter-Heidorn, H.; Koenderink, J.B.; Russel, F.G. Cannabinoid CB1 receptor antagonists modulate transport activity of multidrug resistance-associated proteins MRP1, MRP2, MRP3, and MRP4. Drug Metab. Dispos. 2011.
- Holland, M.L.; Allen, J.D.; Arnold, J.C. Interaction of plant cannabinoids with the multidrug transporter ABCC1 (MRP1). Eur. J. Pharmacol. 2008, 591, 128–131.
- Engels, F.K.; De Jong, F.A.; Sparreboom, A.; Mathot, R.A.; Loos, W.J.; Kitzen, J.J.; De Bruijn, P.; Verweij, J.; Mathijssen, R.H. Medicinal cannabis does not influence the clinical pharmacokinetics of irinotecan and docetaxel. Oncologist 2007, 12, 291–300.
- Geffrey, A.L.; Pollack, S.F.; Bruno, P.L.; Thiele, E.A. Drug–drug interaction between clobazam and cannabidiol in children with refractory epilepsy. Epilepsia 2015, 56, 1246–1251.
- Gaston, T.E.; Bebin, E.M.; Cutter, G.R.; Liu, Y.; Szaflarski, J.P.; Program, U.C. Interactions between cannabidiol and commonly used antiepileptic drugs. Epilepsia 2017, 58, 1586–1592.
- Manini, A.F.; Yiannoulos, G.; Bergamaschi, M.M.; Hernandez, S.; Olmedo, R.; Barnes, A.J.; Winkel, G.; Sinha, R.; Jutras-Aswad, D.; Huestis, M.A. Safety and pharmacokinetics of oral cannabidiol when administered concomitantly with intravenous fentanyl in humans. J. Addict. Med. 2015, 9, 204.
- Stout, S.M.; Cimino, N.M. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: A systematic review. Drug Metab. Rev. 2014, 46, 86–95.
- Arellano, A.L.; Papaseit, E.; Romaguera, A.; Torrens, M.; Farre, M. Neuropsychiatric and General Interactions of Natural and Synthetic Cannabinoids with Drugs of Abuse and Medicines. CNS Neurol. Disord. Drug Targets 2017, 16, 554–566.
- Zendulka, O.; Dovrtelová, G.; Nosková, K.; Turjap, M.; Sulcová, A.; Hanus, L.; Jurica, J. Cannabinoids and cytochrome P450 interactions. Curr. Drug Metab. 2016, 17, 206–226.
- Rong, C.; Carmona, N.E.; Lee, Y.L.; Ragguett, R.M.; Pan, Z.; Rosenblat, J.D.; Subramaniapillai, M.; Shekotikhina, M.; Almatham, F.; Alageel, A.; et al. Drug-drug interactions as a result of co-administering Delta(9)-THC and CBD with other psychotropic agents. Expert Opin. Drug Saf. 2018, 17, 51–54.
- Anderson, G.D.; Chan, L.N. Pharmacokinetic Drug Interactions with Tobacco, Cannabinoids and Smoking Cessation Products. Clin. Pharm. 2016, 55, 1353–1368.
- Gaston, T.E.; Szaflarski, J.P. Cannabis for the Treatment of Epilepsy: An Update. Curr. Neurol. Neurosci. Rep. 2018, 18, 73.
- Kong, T.Y.; Kim, J.H.; Kim, D.K.; Lee, H.S. Synthetic cannabinoids are substrates and inhibitors of multiple drug-metabolizing enzymes. Arch. Pharm. Res. 2018, 41, 691–710.
- Tai, S.; Fantegrossi, W.E. Pharmacological and Toxicological Effects of Synthetic Cannabinoids and Their Metabolites. In Neuropharmacology of New Psychoactive Substances (NPS): The Science Behind the Headlines; Baumann, M.H., Glennon, R.A., Wiley, J.L., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 249–262.
- Haroutounian, S.; Ratz, Y.; Ginosar, Y.; Furmanov, K.; Saifi, F.; Meidan, R.; Davidson, E. The effect of medicinal cannabis on pain and quality-of-life outcomes in chronic pain. Clin. J. Pain 2016, 32, 1036–1043.
- Olfson, M.; Wall, M.M.; Liu, S.-M.; Blanco, C. Cannabis use and risk of prescription opioid use disorder in the United States. Am. J. Psychiatry 2017, 175, 47–53.
- Ronen, A.; Chassidim, H.S.; Gershon, P.; Parmet, Y.; Rabinovich, A.; Bar-Hamburger, R.; Cassuto, Y.; Shinar, D. The effect of alcohol, THC and their combination on perceived effects, willingness to drive and performance of driving and non-driving tasks. Accid. Anal. Prev. 2010, 42, 1855–1865.
- Hartman, R.L.; Brown, T.L.; Milavetz, G.; Spurgin, A.; Gorelick, D.A.; Gaffney, G.; Huestis, M.A. Controlled cannabis vaporizer administration: Blood and plasma cannabinoids with and without alcohol. Clin. Chem. 2015, 61, 850–869.
- Atwal, N.; Casey, S.L.; Mitchell, V.A.; Vaughan, C.W. THC and gabapentin interactions in a mouse neuropathic pain model. Neuropharmacology 2019, 144, 115–121.
- Russo, E.B. Current therapeutic cannabis controversies and clinical trial design issues. Front. Pharmacol. 2016, 7, 309.
- MacCallum, C.A.; Russo, E.B. Practical considerations in medical cannabis administration and dosing. Eur. J. Intern. Med. 2018.
