The immune system has a crucial role in skin wound healing and the application of specific cell-laden immunomodulating biomaterials emerged as a possible treatment option to drive skin tissue regeneration. Cell-laden tissue-engineered skin substitutes have the ability to activate immune pathways, even in the absence of other immune-stimulating signals. In particular, mesenchymal stem cells with their immunomodulatory properties can create a specific immune microenvironment to reduce inflammation, scarring, and support skin regeneration.
1. Introduction
The skin is the largest organ in the human body performing essential functions, including body protection against the external environment, prevention of water loss from the body, temperature regulation, and ultraviolet (UV) absorption from sunlight to produce vitamin D [
1,
2]. Skin regeneration undergoes a dynamic and complex multistep process characterized by homeostasis, inflammation, proliferation, re-epithelization, and fibrosis. The wound healing is possible due to various platelets and immune skin cells secreted/activated by the number of cytokines [
2,
3,
4,
5,
6].
Whereas minor superficial skin injuries usually heal by epithelialization alone, large and deep skin defects require a skin substitution to heal properly. Therefore, skin replacement therapies have evolved tremendously over the last few years [
7]. Given the limited donor sites as it is in severely burned patients, tissue-engineered skin substitutes offer a promising treatment option for those cases [
4,
5,
6,
7,
8,
9,
10,
11,
12,
13,
14,
15,
16,
17].
2. Skin
Human skin is composed of three layers, the epidermis, dermis, and hypodermis with complex cells, nerve, and blood supply [
17,
18].
The epidermis is the outermost layer which is composed mostly of keratinocytes, arranging a stratified epithelium with basal keratinocytes at the innermost layer and the keratinized stratified squamous epithelium, which is known as the stratum corneum [
19]. Moreover, melanocytes are present in the epidermal basal layer to form melanin being important for skin pigmentation and especially for the protection against harmful ultraviolet (UV) light [
18]. Additionally, there are Langerhans cells present in the epidermis which act as a network of immune system sentinels. The main duty of these immune cells is to distinguish the appropriate adaptive immune response by interpreting the microenvironmental context in which they face foreign substances [
20].
The dermis located below the epidermis is the thickest layer of skin. This layer is mainly composed of extracellular matrix (ECM) containing collagen type I, elastin, and glycosaminoglycans (GAGs), produced by fibroblasts [
21]. The dermis has a major role in the biomechanics of the skin thereby providing mechanical strength and elasticity.
The hypodermis contains adipose tissue that is well vascularized and aids both the thermoregulatory and mechanical properties of the skin [
18].
The stability and continuity of this complex multilayer organ can be disturbed and destroyed by wounds caused by different internal and physical or thermal external factors. A wound is a damage of the skin integrity and its function [
22]. Wounds are usually classified based on the area of affected skin nature; the injured skin layers or the nature of the skin repair process [
23,
24]. Injuries that affect only the epidermal skin layer are called superficial wound, while injuries that damaged both the epidermis and dermal layers are referred to as partial-thickness wound. Full-thickness wounds are injuries of the epidermis, dermis including the sweat glands and hair follicles, and the underlying subcutaneous fat or deeper tissues as well [
25].
Depending on the nature of the repair process, wounds are categorized into acute and chronic wounds. Acute wounds usually heal in an expected time frame (8–12 weeks) with minimal scarring [
26]. Mechanical injuries which are sustained by abrasions and cuts (penetrating of knives or sharp objects) and surgical wounds are the primary sources of acute wounds. Moreover, burns and chemical injuries caused by radiation, corrosive chemicals, electricity, and thermal sources, are another class of acute wounds [
26]. On the other hand, chronic wounds are tissue injuries that heal slowly and often show delayed healing as 12 weeks from the injury. Different factors such as diabetes, malignancies and ongoing immunosuppressive treatment, poor primary wound treatment, and persistent infections of wounds can impair the healing process, leading to chronic nonhealing wounds [
25].
3. Skin Wound Healing Process
Skin wound healing is a complex dynamic and multistage process initiated by an injury. It requires the activation, recruitment or activity of numerous cell types such as keratinocytes, endothelial, fibroblast and inflammatory cells as well as growth factors, cytokines and chemokines [
27]. In adults, the healing skin process is more restrictive than in children or embryo [
27,
28].
There are two main categories—primary and secondary wound healing. Primary wound healing requires straight, aseptic wound borders, which are close to each other. In contrast, during secondary wound healing, the wound borders are away from each other so that the wound has to be closed by granulation tissue [
30].
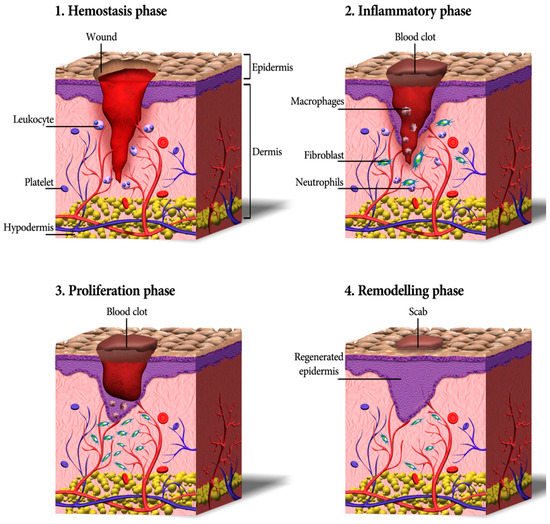
Cutaneous wound healing consists of a sequence of molecular and cellular events which occur after the onset of a tissue lesion in order to restore the damaged tissue. This process can be subdivided into four phases namely hemostasis, inflammation, proliferation, and remodeling (
Figure 1) [
17,
31].
Figure 1. A schematic depicting the process of wound healing, including four continuous phases—homeostasis, inflammation, proliferation, and remodeling. Firstly, blood platelets are activated to form a blood clot and have a role in leukocyte recruitment. Next, neutrophils and macrophages clean the wound site from dead cells, bacteria, and other pathogens or debris. Then, fibroblasts migrate, proliferate, and activate the angiogenesis process. Finally, granulation tissue is formed, the deposition of extracellular matrix proteins occurs to reconstitute the dermal tissue, and the epidermis is regenerated. Eventually, many of the formed capillaries and fibroblasts disappear [
17].
Hemostasis and Inflammation Phases
Most skin injuries, even superficial wounds, caused bleeding. Blood loss and the risk of infection is reduced by the clotting mechanism in which fibrinogen produced in the exudate stimulates a fibrin clot formation and seals the wound site. Therefore, hemostasis provides a protective barrier and contributes to successful wound healing [
32]. The chemokines generated during hemostasis attract inflammatory cells and encourage resident immune cells. All those cells together start the inflammatory phase of wound healing within only minutes after the injury [
33,
34]. First, the vessels are dilated and the capillary permeability is enhanced. Then, local edema occurs and leukocytes such as macrophages and neutrophil granulocytes migrate into the wound. Cell debris is eliminated via phagocytosis to cleanse the skin wound [
35]. The latter produces growth factors, cytokines, and other soluble mediators that activate keratinocytes, endothelial cells, fibroblasts, inflammatory cells, and other cells present in the wound. Neutrophils also produce antimicrobial peptides, reactive oxygen species (ROS), and proteases to kill and degrade potential pathogens [
36]. It is worthy to mention that oxygen is critical in this step as ROS is generated in leukocytes by the nicotinamide adenine dinucleotide phosphate (NADPH) oxidase. Further, the formation of ROS has an active role in cytokine release [
37].
The injury including damage of blood vessels triggers coagulation and is linked to the release of transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF) and VEGF. Those cytokines trigger tissue edema and inflammation. VEGF activates the migration of endothelial cells, leukocytes and epithelial cells to the wound [
38]. Activated TGF-β provides rapid chemotaxis of neutrophils and monocytes to the wound site [
38]. In addition, TGF-β induces leukocytes and fibroblasts to secrete TNF-α, IL-1, PDGF, and different pro-inflammatory chemokines. Pro-inflammatory chemokines are produced by cells primarily to recruit leukocytes to the site of injury [
39].
As inflammation is settled, the wound advances into angiogenesis, matrix formation, and remodeling. Angiogenesis is triggered by mesenchymal stem cells secreting insulin-like growth factor 1 (IGF-1), PDGF-BB, VEGF, angiopoietin-1, and FGF.
Proliferation
Activated resident fibroblasts, endothelial cells, and keratinocytes are crucial cellular players facilitating the re-epithelialization and the formation of granulation tissue. These are two major processes occurring concomitantly during the proliferation phase [
40].
First, the granulation tissue and blood vessels are formed by the in-growth of blood capillaries and lymphatic vessels into the wound site, viable endothelial cells at the wound borders start proliferation, and blood vessels start growing into the wound forming an organized microvascular network. Those processes are essential because the wound needs to be provided with oxygen and nutrients [
41,
42]. Then, a new vasculature is followed by the production of the extracellular matrix (ECM). Fibroblasts degrade the provisional matrix and synthesize new ECM in order to replace the injured tissue with a connective tissue scar. Thus, is mediated by different cytokines and growth factors starting with TGF-β that recruits fibroblasts synthesizing collagen I, III, and V, proteoglycans, and fibronectin [
43]. Leukocytes are engaged in phagocytosis of debris and microbes and degradation of matrix. Recruitment of other blood cells as neutrophils and monocytes is enhanced by release of pro-inflammatory chemokines and cytokines, in particular TNF-α, IL-1, and IL-6. The newly synthesized ECM contains initially an enormous amount of hyaluronan that creates a structure enabling other migrating cells to penetrate into the wound area. When the granulation tissue is formed, some fibroblasts transform into myofibroblasts. This differentiation can be induced by TGF-β1. In addition to the presence of a soluble stimulus such as TGF-β1 found in inflammatory zone 1 (FIZZ1), other cytokines and mechanical stress are also needed to complete the differentiation [
44]. Myofibroblasts generate a force that pulls the surrounding dermal and adipose tissue toward the wound. The formation of granulation tissue is a crucial part of wound healing [
45,
46]. An impaired granulation results in impaired wound healing and, in contrast, excessive granulation leads to a delayed re-epithelization [
47,
48]. On the other hand, epithelial cells respond to wounding by extensive changes in transcription, and the phenotype allows initiation of movement within a few hours after the injury [
49].
Re-epithelialization is a critical phase of skin healing and it is characterized by replication and migration of epithelial cells across the skin edges in response to some growth factors such as FGF, epidermal growth factor (EGF), and keratinocyte growth factor (KGF). Moreover, matrix metalloproteinases (MMPs) such as MMP-1, 9, 10, and MMP-13 have been implicated in re-epithelialization. MMP-1 and MMP-9 promote human keratinocyte migration, the former tending to guide keratinocytes on fibrillar collagen in the basement membrane, allowing basal keratinocytes to detach and migrate toward the wound. MMP-10 is expressed by epidermal cells three days after wounding and it aids keratinocytes expression. MMP-13 also promotes re-epithelialization indirectly by affecting wound contraction and inducing keratinocyte migration [
50,
51]. As soon as the wound is covered with a monolayer of keratinocytes, their migration stops, and the formation of a (stratified) epidermis starts [
52,
53].
Remodeling (Maturation)
During the final phase of wound healing, remodeling, collagen is deposited in an organized and well-mannered network. Macrophages release some MMPs that trigger production of type VIII collagen, which is critical for local tissue integrity [
34]. Net collagen synthesis will continue for at least 4 to 5 weeks after wounding. The increased rate of collagen synthesis during wound healing can be because of an increase in the number of fibroblasts and also from a net increase in the collagen production per cell [
54]. Additionally, collagen type III is replaced by stronger collagen type I, which is followed by cleavage and cross-linking of fibrillar collagen [
55]. In the remodeling stage, oxygen is required for mature collagen formation and fibroblast accumulation; it is necessary in the hydroxylation reaction of proline and lysine from procollagen chains in order to stabilize the triple helices of collagen [
37].
The remodeling of the ECM components is the final and longest phase during the cutaneous wound healing. The granulation tissue matures forming a scar [
56]. The ECM remodeling and the final scar formation can last up to 2 years after the wound is closed [
25].
5. Application of Stem Cells in Skin Substitutes
According to the findings obtained by McCulloch and Till, based on [93,94], stem cells can be defined two prominent features: (1) they are undifferentiated and renew themselves for the entire life span and (2) they have an extraordinary potential to develop from a common precursor into multiple cell types with particular functions [15]. Stem cell-based therapies have the potential to enhance cutaneous regeneration due to their ability to secrete proregenerative cytokines modulating immune response, making them an appreciated option for the treatment of chronic wounds [95]. However, stem cell therapies are limited by the need for invasive harvesting techniques, immunogenicity, and limited cell survival in vivo [96]. Embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs), and adult stem cells are among the main sources of cells that have been used in various experimental research for wound treatment and regeneration of injured skin [97].
5.1. Induced Pluripotent Stem Cells
iPSCs are able to reproduce all types of adult cells in the course of their differentiation and they have an unlimited self-renewal capacity [
98]. Before studying the iPSCs, ESCs were the only well-studied source of pluripotent stem cells. ESCs can be obtained from the inner cell mass and/or epiblast of blastocysts [
99]. Although there are some protocols for the preparation of various cell derivatives from human ESCs, there are restrictions for ESC use in cell replacement therapy, such as the incompatibility between the donor cells and the recipient, which can result in the rejection of transplanted cells [
100]. Then, iPSCs were obtained later by reprogramming animals [
101,
102,
103,
104] and human differentiated cells by induced expression of transcription factors including Oct4/Sox2/c-Myc/KLF4 or Oct4/Sox2/NANOG/LIN28 [
105,
106]. iPSCs closely resemble ESCs in a broad spectrum of properties, closely such as cell morphology and proliferation, sensitivity to growth factors, and signaling molecules. Moreover, similar to ESCs, iPSCs are able to differentiate in vitro into derivatives of all three primary germ layers (ectoderm, mesoderm, and endoderm) and form teratomas following their subcutaneous injection into immunodeficient mice [
100,
105,
106]. Therefore, iPSCs can be an appropriate alternative for ESCs in the area of clinical application of cell replacement therapy.
iPSCs are derived from adult cells by in vitro induction of pluripotency with noninvasively harvesting, and can be transplanted autologous, reducing immune rejection. Importantly, iPSCs are the only cell type capable of being differentiated into all cell types of normal skin, and therefore they have been widely utilized in wound healing applications. Owing to their high differentiation capacity into descendants of all three germ layers, iPSC-derived cells have the potential to enhance each of the phases of diabetic wound healing through their paracrine and direct cellular effects [
96]. During the inflammatory phase, iPSC-derived cells secrete growth factors and cytokines, counteracting the suppressed cytokine secretion profile seen in diabetic patients [
107,
108]. This process eventuates in the recruitment of macrophages and proliferative cells including fibroblasts and keratinocytes, which are known to be deficient in chronic wounds [
109,
110,
111,
112]. Direct application of stem cells into the wound bed also mitigates the impaired homing potential of progenitor cells into diabetic wounds [
113]. In the proliferative phase, different cells including endothelial, fibroblasts, pericytes, smooth muscle, keratinocytes, or MSCs are derived from potential iPSC [
96,
114,
115], afterward increasing angiogenesis and promoting collagen deposition [
110,
111]. Since the remodeling phase is extremely dependent on functional myofibroblasts, their recruitment during the proliferative phase is vital to the last stage of wound healing. Eventually, iPSCs retain the ability to differentiate into keratinocytes [
114]. In a review by Gorecka et al. [
96], the potential and limitations of iPSCs in wound healing applications were fully examined.
5.2. Bone Marrow-Derived Mesenchymal Stem/Stromal Cells
Bone marrow is an important compartment of bone regulating its homeostasis. Bone marrow itself can be considered as an immune organ containing distinct cell types, which secrete a large number of cytokines and growth factors that can have angiogenic, anti-inflammatory, anti-apoptotic and immunomodulatory effects [
121,
122]. Moreover, bone marrow is a metabolic organ and has been demonstrated to regulate whole-body energy metabolism. Thus, the cellular composition of bone marrow can change with age, gender, and metabolic activity [
122,
123]. The adult bone marrow is composed of hematopoietic cells and the associated supporting stroma. The stroma consists of cells with multipotent differentiation capacities that are usually considered mesenchymal stem cells [
124].
Bone marrow-derived mesenchymal stromal cells (BMSCs) are multipotent stem cells capable to differentiate into numerous cell types, including fibroblasts, endothelial cells, cartilage, bone, muscle, and neuronal cells. BMSCs also secrete a large number of growth factors and cytokines that are critical for the repair of injured tissues [
125,
126]. Because BMSCs are able to differentiate into multiple cell types and produce tissue repair factors, BMSCs skin substitutes provide an alternative to conventional treatments for skin repair [
127]. In numerous animal and human studies, BMSCs were directly injected into skin wounds [
128,
129,
130,
131]. The results indicate that both autologous and allogeneic BMSCs could induce tissue regeneration and accelerate wound closure. However, the direct injection of cells showed only a small therapeutic efficacy with side effects such as invasive procedure with attendant risks, and accumulation of cells for a long time [
132,
133,
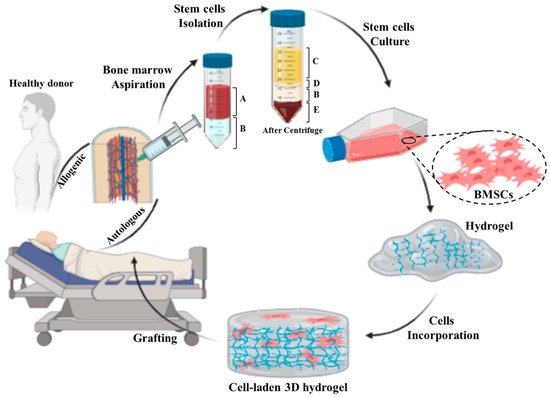
134]. Therefore, the integration of stem cells into engineered scaffolds such as hydrogel networks offers controllable mechanical, physical, and chemical properties, which can improve the integration of a skin substitute into the host tissue (
Figure 3). Thus, the integration of BMSCs into an appropriate scaffold can sustainably support the healing process by creating a proregenerative microenvironment in the wound area [
135].
Figure 3. Schematic representing preparation process of an autologous/allogenic stem cell-laden hydrogel for skin regenerative approaches: the bone marrow is aspirated from donor or patient own and then the stem cells are isolated from bone marrow. Afterward, the BMSCs are cultured and incorporated/encapsulated in a hydrogel solution. Finally, the cell-laden hydrogel is pre/post-crosslinked and grafted to the injured site. (A: whole blood, B: Ficoll gradient, C: plasma, D: mononuclear cell, and E: red blood cells).
To develop stem cell-based scaffolds, stem cells need to be first harvested from animals or humans and then loaded into the prepared scaffold. Hence, to avoid repeated harvesting of BMSCs for each treatment and cell loading into scaffolds, cryopreservation is vitally important [
138,
139]. Recently, an animal study was conducted to compare the efficiency of freshly isolated and cryopreserved BMSCs on wound healing [
138]. For this, both fresh and 30-day preserved BMSCs were cultured on monofilament polypropylene scaffolds and used for therapeutic purposes in guinea pigs. There was no significant difference in population doubling time between fresh and cryopreserved BMSCs and both of them expressed cell surface markers (CD73, CD90, and CD105) and mRNA without significant difference. Additionally, both pre- and post-thaw BMSCs were successfully differentiated into three different cell lineages including chondrogenic, osteogenic, and adipogenic lineages. Moreover, a significant difference was detected in wound contraction between cell-treated wounds and control groups, while there was no difference observed among scaffold-augmented MSCs, both pre- and post-thaw, and the MSCs-only group.
To summarize, BMSCs are capable to differentiate into various cell types and secrete numerous bioactive factors that improve granulation tissue formation, angiogenesis, and decrease inflammation. Furthermore, they can be easily cryopreserved to be used for later applications. Therefore, BMSCs-based skin substitutes represent one of the promising approaches to heal hard-to-heal wounds.
5.3. Adipose-Derived Stromal/Stem Cells
Recently, adipose tissue has been highlighted as a promising source in the field of cell therapy and regenerative medicine. Adipose tissue is composed of multiple cell types such as mature adipocytes and the stromal vascular fraction (SVF), which is a source of adipose-derived stromal/stem cells (ASCs), endothelial cells, pre-adipocytes, lymphocytes, and adipose-resident macrophages [
4,
140].
In contrast to bone marrow, adipose tissue represents advantages in terms of its accessibility and abundance for use in tissue engineering and regenerative medical applications [
16,
141]. The potential utility of ASCs has been demonstrated in multiple preclinical animal models [
142,
143]. Similar to BMSCs, the ASCs have been characterized based on their immunophenotypic and differentiation properties [
144,
145,
146,
147]. Because large volumes of adipose tissue can be easily obtained from individual donors, it is possible to obtain a high yield of 375 ± 147 × 10
3 cells per mL of lipoaspirate [
146]. The clinical application of adipose-derived cells has developed considerably in the past two decades. Both the heterogeneous adipose stromal vascular fraction (SVF) and the more homogeneous adipose stromal cells (ASCs) offer distinctive opportunities as novel cell-based treatments for tissue regeneration [
148]. Additionally, adipose-derived cells have shown great potential in various therapeutic fields such as osteoarthritis, scleroderma, multiple sclerosis, renal insufficiency, erectile dysfunction, and wound healing [
148,
149].
6. Immunomodulatory Properties of MSCs: ASCs and BMSCs
It is known that MSCs can modulate the immune system and regulate skin tissue regeneration. Importantly, MSCs can mitogen- and allo-activated lymphocyte proliferation [170,171]. This effect is heavily dependent on some factors; for example, MSCs inhibit lymphocyte proliferation mainly via the secretion of TGF-β1, IL-10, HLA-G, nitric oxide, and hepatocyte growth factor, as well as due to the expression of indoleamine 2,3-dioxygenase (IDO enzyme) [172,173,174]. Further, MSCs secrete trophic factors which are critical for vasculogenesis and angiogenesis, and aid tissue regeneration [175,176]. Additionally, MSCs have been administrated to the site of wound in animal models by encapsulation in gelatin microspheres and microcryogels or loading into a 3D graphene foam [177]. It has been shown that 3D graphene foam loaded with MSCs released prostaglandin E2 (PGE2), which suppresses the release of pro-inflammatory cytokines including TNF-α, IFN-γ, IL-6, IL-8, and IL-12p70, and increases the release of anti-inflammatory cytokines such as IL-10 and IL-12p40, and TGF-β1 by macrophages [178,179,180]. Additionally, PGE2 reduced the proliferation of T cells in the wound and is a cofactor in the transition from TH1 to TH2 cells, which decrease tissue inflammation and, further, tissue regeneration [179]. Moreover, higher levels of IL-10 expressed by T cells and macrophages in response to PGE2 can limit or reduce the inflammatory mechanism of immune cells. IL-10, which is an important anti-inflammatory cytokine, inhibits further neutrophil invasion and respiratory burst [180]. IL-10 also affects fibrosis by downregulating the release of TGF-β1 in T cells and macrophages, and remodeling ECM by reprogramming wound fibroblasts. IL-10 has direct effects on the prevention of excessive collagen deposition by reducing the expression of pro-inflammatory cytokines such as IL-6 and IL-8 in the wound environment [181]. Finally, IL-10 expression results in resolution of inflammatory stage and rushing of the wound into the proliferation stage and over-expression of IL-10 can produce an environment in which wound healing tends to occur without scar formation [182,183,184].
Therefore, allogeneic MSCs have been utilized for treatment of different diseases, especially for scarless skin regeneration. MSCs from different sources are similar in a range of phenotypic and functional features. However, there are subtle differences that may result from the microenvironmental niche, the local, and the ontogenetic age or induced by the isolation and culture procedure [
185]. Therefore, the immunomodulatory properties of BMSCs and ASCs, in particular due to their paracrine effects, have also been characterized in detail [
186,
187,
188,
189].
ASCs represent an interesting source of MSCs, which can be easily obtained and used for autologous applications as they reduce apoptosis and improve tissue repair and angiogenesis and affect immunoregulation [
190,
191].
BMSCs are another immunoregulatory MSCs. The immunomodulatory properties of BMSCs are facilitated by their interactions with immune cells such as macrophages, T cells, B cells, and DCs in a context and microenvironment-dependent manner. These cells can also inhibit natural killer (NK) cells activity, B cell proliferation, and DC function and differentiation. Moreover, BMSCs are considered to immunosuppress the local environment by secretions of cytokines and growth factors and cell–cell interaction [
207]. For instance, soluble factors such as growth factors and cytokines, namely PGE2, IDO, IL-6, and M-CSF, have been evaluated in various clinical studies and the cell-based properties have been explored in many T cell-mediated diseases. Moreover, the evidence demonstrated that both undifferentiated and differentiated BMSCs have a suppressive impression on mitogen-stimulated and alloantigen lymphocyte proliferation followed by a concomitant reduction in the production of pro-inflammatory cytokines such as TNF-α and IFN-γ [
208,
209]. Therefore, the clinical applications of human BMSCs are ranging from transplantation, immune-related disorders including autoimmune disorders and cell replacement [
210].
Functional characterization of BMSCs and ASCs has revealed that both cell types are able to suppress lymphocyte reactivity in mixed lymphocyte response (MLR) assays and decrease the production of inflammatory cytokines in vivo [
200,
202]. Particularly, additional recent studies reported that delivering ASCs in polyhydroxybutyrate-co-hydroxy valerate constructs achieves a similar outcome to BMSCs role in wound healing and may be more clinically appropriate since the yield following isolation is much higher than BMSCs [
177,
211].
7. Immunomodulatory Skin Scaffolds
Different physical and chemical properties of the biomaterial such as stiffness, topography, roughness, pore size and pore distribution, degradation rate and its debris, surface charge, ligand presentation, and surface functional groups influence the behavior of host cells [215,216]. However, the effects of such biophysical and biochemical characteristics on immune cells, especially when a skin substitute is implanted, are still not elucidated. A biomaterial should be designed to minimize the deleterious host body responses [217,218,219,220,221]. The host immune system response after implantation of an engineered skin substitute is called foreign body reaction (FBR), which can cause significant problems for patients through excessive inflammation and adverse effects on tissue repair processes. Therefore, controlling the biomaterial interaction with the host tissue or FBR is of crucial importance in the field of regenerative medicine and tissue engineering [222,223,224]. In this respect, the term “bioinert implant” refers to any material that is placed in the human body and demonstrates a minimal interaction with its surrounding tissue. Specifically, an acellular fibrous capsule is formed at the interface between tissues and bioinert biomaterials [225]. However, novel biomaterials are being designed to stimulate specific cellular responses at the cellular level to trigger desired immunological outcomes, thereby supporting the wound healing process [212,226].
When a biomaterial is implanted into a vascularized wound bed, the natural innate body response is that plasma proteins are immediately adsorbed onto the implanted biomaterial surface. Factor XII (FXII) and tissue factor (TF) are the initiators of the intrinsic and extrinsic system of the coagulation cascade, respectively, leading to the formation of a blood clot. This leads to infiltration and adherence of cells such as platelets, monocytes, and macrophages through the interaction of adhesion receptors with the adsorbed proteins [212,227]. Adhered cells release growth factors and chemokines, which are able to recruit cells of the innate immune system to the injury/implantation site. Finally, deposition and organization of collagen matrix arise from fibroblasts and MSC activities [230].
Macrophages exhibit a heterogeneous and temporally regulated polarization during skin wound healing and the phenotype changes during healing from a more pro-inflammatory (M1) profile in early stages after injury, to a less inflammatory, pro-healing (M2) phenotype in later phases. Many studies have proved that a high M2:M1 ratio in the surrounding environment of implanted biomaterials results in better remodeling outcomes [
235,
236,
237,
238]. During skin wound healing M1 macrophages are replaced by M2 polarized cells [
239]. Both M1 and M2 cells exert specific functions in this process [
9,
240]. It has been suggested that the long-term presence of M1 macrophages leads prolonged inflammation phase and impairs finally the wound healing outcome [
8,
158,
232,
239,
241]. The same is true for M2 cells, which can lead over time to the formation of detrimental foreign body giant cells [
227,
242]. Therefore, the control of the M1:M2: ratio is vitally important when designing an immune-active scaffold to promote tissue remodeling as well as its integration and regeneration in vivo [
158].
Therefore, a considerable effort has been made to employ particular ECM components and natural polymers, which are able to mimic the ECM structure to design appropriate immunomodulatory biomaterials for tissue engineering and regenerative medicine. By modulating the immune system, these biomaterials could be mainly applied to treat a broad spectrum of immune-related skin diseases in the future. Table 2 presents the summary of some papers conducted on natural hydrogels for skin immunomodulation.
Table 2. Summary of research studies conducted on natural hydrogel-based materials used for immunomodulation of skin regeneration.
| Hydrogel Type |
Main Characteristic |
Immunomodulatory Role in Skin Regeneration |
Ref. |
| Decellularized ECM |
Containing proteins, glycoproteins, and polysaccharides |
Binding to the specific surface receptors of immune cells, the ECM composition can affect immunomodulation, promoting anti-inflammatory phenotype polarization in some cases |
[230,243,244,245] |
| Collagen |
Protein-based material and the most abundant ECM component |
Binding to LAIR-1 receptors on immune cells, suppressing immune cell activity, and resolving post-wound inflammation |
[246,247] |
| Fibrin |
The main component of the haemostatic clot |
Decreasing TNF-α cytokine secretion, decreasing macrophage motility, reduction of neutrophil recruitment, and extending pro-healing effects |
[248,249,250,251,252] |
| Hyaluronic acid |
Glycosaminoglycans material with various MWs |
Modulation of leukocyte function, immunomodulatory effect dependent on MW, suppressing immune cell activity by high-MW HA, enhancing an inflammatory by low-MW HA, delaying inflammation by High-MW HA |
[253,254] |
| Chitosan |
A natural polysaccharide |
Employing leukocytes and macrophages to the wound site, decreasing the inflammatory cells, and TNF-α and MMP-9 levels, and affecting macrophage polarization |
[255,256,257,258] |
| Carrageenan |
A natural marine polysaccharide from red seaweed |
Stimulating IL-10 expression, prohibiting cytotoxic T cell responses, and delaying neutrophil activation |
[259,260,261] |
| L-arginine |
Amino acid-based material |
Decreasing nitric oxide production, stimulating macrophages to express both TNF-α and NO in combination with chitosan |
[262,263,264] |
This entry is adapted from the peer-reviewed paper 10.3390/biomedicines10010118