Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Medicine, Research & Experimental
|
Oncology
Cancer immunotherapy, a promising and widely applied mode of oncotherapy, makes use of immune stimulants and modulators to overcome the immune dysregulation present in cancer, and leverage the host’s immune capacity to eliminate tumors.
- cancer
- immunotherapy
- liposomal nanosystems
- synthesis
- immune reagents
- immunoliposomes
- cancer vaccine
- nano-immunotherapy
1. Introduction
1.1. Background
Cancer immunotherapy differs from traditional methods of directly removing a tumor via radiation, surgery, or chemotherapeutic cell kill. In many ways, it is an indirect method, where agents introduced to the body or site of cancer aim at stimulation or harnessing of the immune system for removal of cancer cells, or reversal of characteristic immune dysregulation in cancers [1]. This approach aims to leverage what is available (chemotherapy, radiotherapy, surgery, and other effective treatment modalities) to the scientific and medical community and add to those modalities that can strengthen treatment outcomes.
Cancer immunotherapy lay dormant for many years after its inception in 1892, owing to poor clinical outcomes; hence, tentative adoption in the clinic [2]. However, recent years have seen immunotherapy gaining considerable ground in the clinic, building on increased plausibility and translationality in preclinical investigations, becoming one of the pillars in cancer therapy [3]. These moves necessitate accurate, critical, and contextual review of current literature to encourage further development of this field, pregnant as it is with possibility. Cancer immunotherapy, conceptually, promises lower toxicity, increased quality of life, and importantly, longer-term survival than chemotherapy, with cells gaining immune memory against a type or types of cancerous cells [4]. Immunotherapy has previously consisted of free, unbound agents being introduced to the body systemically or directly into the site of cancerous growth [5], with a view to induce immunogenic cell death (ICD), prevent formation of essential structures to tumor growth [2], or to block negative immune regulation [6]. This poses many challenges, most of which stem from the chemical or biological nature of the active compounds. For this reason, it is desirable to co-administer immunogenic agents in the form of adjuvants, combination therapy, as well as agents which work to target compounds/drugs to the site of disease [7], together lowering systemic toxicity [2].
Nanomaterials have recently seen increased application as drug delivery and targeting agents. This is especially useful in combination therapy, where separate administration of multiple compounds in combination brings into play each compound’s individual pharmacokinetic profile, posing a challenge regarding timing of dosage and inter-individual metabolic differences. Here, nanomaterials add value: packaging agents together can ensure synergistic activity and annul the effects of individual pharmacokinetics of administered agents. This also alleviates the effects of the narrow therapeutic window within which traditional chemotherapeutics operate, by lowering systemic toxicity [8].
1.2. Defining “Smart” Nanomaterials
Smart nanomaterials, particularly liposomal nanoparticles, have become popular topics of study within this context in recent years [9]. “Smart” seems to be something of a buzzword in this field, sounding impressive to laypeople, but often having loose relevance or no meaning at all. However, we wish to clarify what is meant by “smart”, and all the applications and considerations that go with that descriptor.
“Smart” can refer to a compound responding to internal (antibody-receptor binding, changes in pH, and increased concentration of reactive oxygen species (ROS)) or external stimuli (change to temperature or exposure to radiation or ultrasound) [10,11,12]. In other cases, “smart” has referred to multifunctionality, tailorability, or an element of control being present regarding release [9]. An extensive summary of smart liposomal therapies is provided in a review by Alavizadeh et al. [12]. We define “smart” as a descriptor of the manipulable and multifunctional drug delivery characteristics of nanomedicines. These characteristics afford researchers the ability to steer, control, or image nanomaterials and their contents or conjugates, from introduction to fate, within a single delivery system. Smart modalities are poised to become a collection of new means of improving treatment outcomes, quality of life, and survival in various cancers, which remain burdensome with persistent high mortality [1].
2. Tumor Immunotherapy
2.1. Tumor Immune Response and Escape
The cancer immunosurveillance concept, i.e., the opinion that the immune system could recognize and destroy nascent malignant tumor cells, was first conceptualized by Burnet and Thomas in the twentieth century [14]. However, due to a lack of convincing evidence, this theory was largely ignored for the decades that followed. With better understanding of tumor antigens and the establishment of a completely immunodeficient mouse model, an abundant body of data was generated, which supported the existence of immunosurveillance, rekindling interest in this process. Further research showed that tumors were being modified by the immune environment, which resulted in tumor cells of high immunogenicity being killed by the immune system, but the weakly immunogenic ones being ignored, while simultaneously acquiring other mechanisms for immune evasion. These processes may be due to the inherent genetic instability and rapid adaptability of tumors [15]. New tumor cell variants with reduced immunogenicity are continuously produced and cleared to form a kind of equilibrium. Eventually, the immune system is impaired by the constant stimulation of tumor cells and the balance is upset. It is during this stage that tumor escape occurs and a tumor may progress to clinical relevance. Dunn et al. concluded that three sequential phases exist during interactions between tumors, immune cells, and the tumor microenvironment (TME): elimination, equilibrium, and escape [15,16]. These breakthroughs in the comprehension of tumor immune biology have opened a new chapter in the war against cancer in the new millennium.
Malignant cells escape from host immune responses via complex mechanisms (Figure 1). Tumor-specific antigens (TSAs) are regarded as vital cornerstones in immunotherapy, consisting of tumor-associated antigens (TAAs), oncogenic viral antigens, and neoantigens, that can sufficiently induce both humoral and cell-mediated immunity. The “ideal cancer antigen” should have highly specific overexpression only in tumor cells, and immunogenicity which could mount a robust immune response. Alas, internal tumor antigens exist, which are usually weakly immunogenic, or develop mechanisms to avoid presentation on the surface of antigen presenting cells (APCs) [17], which fail to activate specific cytotoxic T lymphocytes (CTLs) against tumors. Apart from this, several suppressive factors in the TME can cause immune tolerance and immune deviation, such as: upregulation of immune checkpoints (ICPs) on the tumor cell and immune cell surfaces, low secretion of immune costimulatory molecules, imbalance of the T helper lymphocyte (Th) 1:Th2 ratio, and production of immunosuppressive cytokines. Further to this, immunosuppressive cells can be recruited. These can include regulatory T cells (Tregs), tumor-associated macrophages (TAMs) and myeloid-derived suppressor cells (MDSCs). In addition, chronic inflammation could also stimulate tumor progression [18]. Nutrient competition, including for glucose, amino acids, and fatty acids in the TME can also impact T-cell function and cause T-cell hyporesponsiveness [19,20].
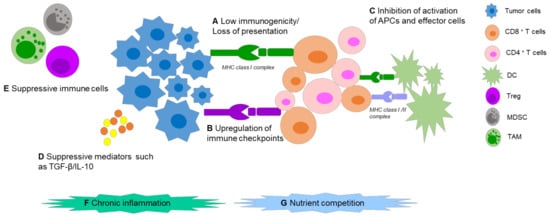
Figure 1. Mechanisms of tumor immune escape. (A) Tumor-associated antigens show low immunogenicity and/or develop several antigen presentation problems. (B) Upregulation of immune checkpoints like PD-1 on T-cell surface or its ligand PD-L1 on tumor surface can cause T-cell anergy and inhibit the antitumor immune response. (C) Low secretion of immune stimulatory molecules in the TME, like IL-2, which inhibits the activation of APCs and other effector cells such as CD8+/CD4+ T cells. (D,E) Production of immunosuppressive cytokines by tumor cells, such as TGF-β and IL-10 and recruitment of several immunosuppressive cells like regulatory T cells (Tregs), Tumor-associated macrophages (TAMs) and myeloid-derived suppressor cells (MDSCs), are able to attenuate antitumor immune response. (F,G) Chronic inflammation and nutrient competition promote immune dysfunction. Abbreviations: MHC (major histocompatibility complex); CD (cluster of differentiation).
2.2. Shift of Immunotherapy Strategy
The immune system and cancer progression are both highly intricate and heterogeneous, increasing the complexity in immunotherapy. Early on, scientists paid more attention to enhancing the antitumor immune response in quantity and/or quality. This strategy could be subdivided into two groups. The first approach is passive immunotherapy, which utilizes effector cells and/or molecules of the immune system for direct tumor attack, such as monoclonal antibody and antibody-drug conjugate therapy (ADC), adoptive cell therapy, and genetically engineered T cells (chimeric antigen receptor [CAR]-T, T-cell receptor [TCR]). These passive strategies can boost host immune responses against tumor at a high level. The second approach is active immunotherapy, which can enhance endogenous immune response against tumors. The representatives are antigen/adjuvant vaccines and dendritic cell (DC) vaccines, which can enhance antigen uptake, processing, and presentation to T cells by APCs. This could also extend to cytokines or agents that promote the activity of APCs, such as type I interferons (IFNs), Toll-like receptor (TLR) agonists, and stimulators of interferon genes (STINGs). Although a broad range of applications of the above strategies against various cancers have acquired Food and Drug Administration (FDA) approval, there is only a small proportion of patients, mainly suffering from classical immunogenic tumors, who can benefit from these therapies. Moreover, these immune-enhancing treatments can lead to serious immune-related adverse events (irAEs), such as some off-target toxicities and acute cytokine release syndrome, since the immune system is in supraphysiological level [20,21]. Introduction of these new compounds is accompanied by new side effects which are often mild or moderate, but may be severe and life-threatening, demanding adjustment and development of better therapies or strategies to overcome adverse effects [22,23].
With a deeper understanding of immune escape mechanisms, more explorations emphasize correction of the particular defects or dysfunctions present during tumor progression, and aim to correct or modify blocked antitumor immune responses in comparison with enhancement strategy. Sanmamed and Chen described this shift of immunotherapy strategy as “the age of normalization cancer immunotherapy”. Anti-programmed death receptor (PD) therapy is a typical example of undoing the immune suppression within the TME and has been approved by the FDA for the treatment of various malignant tumors beyond those classically considered immunogenic tumors. Clinical studies show more frequent objective tumor responses than severe irAEs. These exemplify the move within novel cancer immunotherapy strategies, from traditional immune enhancement approaches to immune normalization [21].
3. Immune-Inducing Liposomes: Formulation and Characterization
3.1. Nanoparticles in Immunotherapy
Nano-immunotherapeutics can have a range of targets and applications due to a large amount of tailorability. Shi and Lammers documented three primary ways to achieve tumor reduction via cancer nano-immunotherapy, over and above direct cytotoxicity [2]. These include cancer cell targeting, immune microenvironment alteration, and peripheral immune system targeting. It is agreed that recent progress made in all three of these areas is indicative of the significant potential held by combinational nano-immunotherapy to improve treatment outcomes in patients [24]. Below, the common applications of nanomaterials in immune therapy are summarized (Figure 2). Excellent reviews exist on the topic [25,26,27], but are beyond the scope of this paper, so only an overview will be provided. We wish to emphasize that the areas of application of liposomes are ideal for cancer immunotherapy: immune induction, synergistic therapy, and drug delivery and targeting [26]. These major strengths are further bolstered by low inherent toxicity [28], earning much clinical interest in recent years [29].
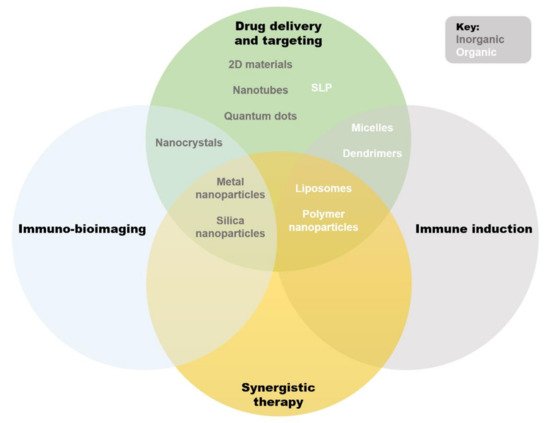
Figure 2. Qualitative Venn diagram showing an overview of primary applications and overlaps of available nanosystems used in immunotherapy. Abbreviations: SLNs (solid lipid nanoparticles).
3.2. Liposomes
Liposomes are uni- or multilamellar lipid-based vesicular nanoparticles, made up of a phospholipid bilayer encapsulating an internal milieu [30]. Liposomes were first described in 1964 [31], and utilized as drug delivery systems in 1971 [32]. By virtue of the structures of utilized phospholipids, there exist distinct hydrophobic and hydrophilic regions in the core of the liposome and within the lipid bilayer, respectively. Phospholipid bilayers tend to measure approximately 4 nm and liposomes are known by different classifications based on the size and geometry thereof [33]. If multiple lamellae are enclosed in an outer membrane, a liposome is known as a multilamellar vesicle (MLV), whereas single-lamella liposomes, enclosing only a hydrophilic internal milieu, are known as small unilamellar vesicles (SUVs), large unilamellar vesicles (LUVs), or giant unilamellar vesicles (GUVs) due to size; <100 nm, >100 nm, and >1 µm, respectively. This article will address the application of immunoliposomes in the treatment of cancer; therefore, the term liposome, for the purposes of this paper, refers primarily to SUVs. Advantages and disadvantages of liposomes are summarized in Table 1.
Table 1. Advantages and disadvantages of liposomes.
| Advantage | Explanation | References |
|---|---|---|
| Biocompatibility | Non-toxic, biodegradable, non-immunogenic | [30] |
| Amphiphilicity | High solubility of various compounds (both hydrophobic and hydrophilic, and compatible with many physiological cavities) | |
| Smart | Stimuli-responsive and multifunctional | [8] |
| Easy, accessible formulation procedures | With simple and inexpensive equipment, liposomes can be synthesized and customized in any laboratory | [10] |
| Rational design, customization is an option (flexibility) | For example, attaching fluorophores, polyethylene glycosylation (PEGylation) | |
| Drug packaging/protection | Reduced toxicity and clearance of encapsulated agent, controlled release Reduced unwanted exposure to normal tissues |
[8] |
| Applicable in multiple therapies | Including oncology, infectious diseases, and vaccinations | [34,35,36,37,38] |
| Ab conjugation and other functionalization possibilities | Active targeting Therapeutic immune induction |
[8] |
| Deeper penetration to physiological compartments |
e.g., blood–brain tumor barrier, deeper tissues | [39] |
| Disadvantage | ||
| Lack of specificity in delivery applications | The enhanced permeation and retention effect is often relied upon, with varying success | [9] |
| Stability/half-life | Low stability can lead to leakage of encapsulated drugs or premature degradation | [8] |
| Unreliable drug packaging based on low loading efficiency and drug leakage | In some cases loading efficiency and leakage can prevent therapeutic progression | [40] |
| Certain cell layers not transversable | Stratum corneum cannot be crossed, blood–brain barrier can only be crossed with modifications (still being tested) | [41,42] |
| Rigidity | Can cause insufficient drug release | [30,41] |
| Upscaling challenges | Scale up requires stringent quality control and is labor intensive | [8] |
This entry is adapted from the peer-reviewed paper 10.3390/pharmaceutics14010026
This entry is offline, you can click here to edit this entry!
