Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Materials Science, Biomaterials
The integrated development of innovative nanoparticle (NP) based approaches will be a key to address one of the main issues in both radiotherapy and chemotherapy: normal tissue toxicity. Among other inorganic NP systems, gold nanoparticle (GNP) based systems offer the means to further improve chemotherapy through controlled delivery of chemotherapeutics, while local radiotherapy dose can be enhanced by targeting the GNPs to the tumor.
- Gold nanoparticles
- radiation
- chemoradiotherapy
- chemotherapy
- drug delivery system
1. Introduction
The interaction of high-Z materials with therapeutic X-ray photons results in an increase in the production of cell damaging species, such as free radicals and low energy electrons [19,20]. Inorganic NP systems such as gold nanoparticles (GNPs), silver NPs, gadolinium-based NPs, lanthanide-based NPs, and titanium oxide nanotubes have been reported as radiosensitizers [21,22,23,24,25,26,27]. Gadolinium-based NPs offer an innovative approach because of their capacity to act as a radiosensitizer as well as a powerful contrast agent in magnetic resonance imaging [26]. The high Z-nature of silver-based NPs along with their antimicrobial properties made them a good candidate in radiotherapy [27]. However, GNPs are the most widely used NP system in radiotherapy due to their ease of production, high Z-nature, advantageous surface chemistry, and biocompatibility [25,28,29,30].
There are different gold-based nanotherapeutic systems available, such as spherical GNPs, gold nanorods, gold nanoshells, gold nanoclusters, and GNP-incorporated liposomal nanoparticles, with many new anisotropic geometries being developed regularly. Spherical GNPs are the most commonly used gold-based nanotherapeutic, as their production is relatively simple and alteration of size and surface chemistry, such as conjugation with polyethylene glycol, is easily achieved [31,32]. Further, GNPs are heavily studied for use in the treatment of cancer through X-ray irradiation and as an anticancer drug carrier [33]. The use of gold nano-rods and gold nanoshells for the treatment of cancer involves the induction of hyperthermia, due to their larger cross-section at near-infrared (NIR) frequencies [34,35]. A comprehensive review of the use of gold-based nanomaterials such as gold nanoshells and gold nanorods in photothermal therapy has been described previously by Vines et al. [36]. It has also recently been shown that gold-based nanotherapeutics can absorb radiofrequency (RF) frequencies and generate heat, opening an avenue to treat more deep-set tumors with the use of gold and hyperthermia-based options [37]. Although more research must be completed, the use of RF waves with gold nanomaterials is very promising. Furthermore, due to the surface plasmon resonance effect present in GNPs, visible light irradiation can also allow for hyperthermia via photothermal therapy, recently shown by Mendes et al. with a green laser light in combination with 14 nm GNPs and doxorubicin [38]. However, the penetration depth of green light is even less than NIR and is thus limited in applicability [39]. Due to their theranostic benefits, such as imaging and biosensing, along with therapeutic properties such as drug delivery, gold nanoclusters have emerged as a useful tool [40,41]. The use of gold nanoclusters can allow for molecular imaging, improving diagnostics and imaging in the future [42]. Ultrasmall gold nanoclusters have also emerged as a useful technology due to their near 100% renal clearance, allowing for the improved probing of disease when utilized as a biosensor [43]. Lipid-based nanoparticles are an avenue that is being explored due to their ability to encapsulate GNPs for radiosensitization purposes and simultaneously act as a drug delivery platform [44]. Utilizing liposomal nanoparticles as a ‘smart’ drug carrier can allow for controlled release of the internalized cargo, such as in response a NIR light source, allowing more control over the treatment process [45].
GNP-based platforms are being researched and have been tested extensively in the field of cancer nanomedicine [46]. For example, a novel nanomedicine that conjugated human tumor necrosis factor alpha (rhTNF) and thiolated PEG onto the surface of colloidal GNPs (named CYT-6091) has been tested in phase 1 clinical trial in cancer patients [47]. The results from the CYT-6091 trial showed that doses up to 600 µg/g of rhTNF were administered without encountering dose-limiting toxicity and was less toxic than a treatment with just rhTNF, as evidenced by a lack of hypertension in patients. Furthermore, the GNPs had gathered in the tumor and mostly avoided healthy tissue. Other phase 1 clinical trials involved the use of PEGylated gold nanoshells around a silica nanoparticle, called AuroLase®, in head and neck, lung, and prostate cancer, with laser irradiation [48,49,50]. Results have, however, not translated to an effective treatment outcome. Another early phase 1 clinical trial involves the use of NU-0129, a platform consisting of nucleic acids attached to the surface of spherical GNPs [51]. The goal of this study is to use the conjugated nucleic acids to bypass the blood-brain barrier and target the BcL2L12 gene present in recurrent glioblastoma. If successful, this platform could supress this gene, which would lead to reduced proliferation and containing the spread of the tumor. However, translation of GNPs to the clinic is still in progress, and further optimization of protocols will have to be elucidated before the majority of research can move out of the preclinical stage, as described in the extensive review by Schuemann et al. [52].
For patients with locally advanced disease, a combination of treatments, such as surgery with chemotherapy and/or radiotherapy is being used. A combination of chemotherapy and radiotherapy (referred to as chemoradiation) is a logical and reasonable approach that has greatly improved the cure rates of solid tumor [8,53]. This combined treatment modality provides local control of the primary tumor mass through radiation while tumor metastasis is suppressed through anticancer drugs [8]. One of the major limitations of chemoradiation as a treatment option is the normal-tissue toxicity, as either radiotherapy or chemotherapy can cause major normal tissue toxicity, as described previously. In order to overcome the normal tissue toxicity in current cancer treatment modalities mentioned previously, NPs are being used to enhance either the local radiation dose or improve delivery of anticancer drugs, or both, as seen in Figure 1. GNPs are one of the materials extensively tested for both radiotherapy and chemotherapy. Therefore, this review article will be focused on prospects of GNP-mediated cancer therapeutics.
Figure 1. Gold nanoparticle-based cancer therapeutics. Radiotherapy and chemotherapy are the two main modalities, besides surgery, in treating cancer. However, normal tissue toxicity in both methods remains a large issue in limiting the effective dose to the tumor. Thus, gold nanomaterials have been introduced to improve the locally deposited dose into tumors and act as a drug delivery system. The combination of radiotherapy and chemotherapy, called chemoradiotherapy, allows for an optimum platform for eradicating the tumor and improving cancer therapeutics.
2. Gold Nanoparticles as Radiosensitizers
The use of high atomic number (Z) material to enhance radiation dose has been studied for more than 50 years. The interest in using high-Z material stems from the production of secondary electrons, such as photoelectrons, Auger electrons, and Compton electrons. These secondary products are effective at damaging DNA as well as ionizing surrounding water molecules, forming free radicals [110]. While the atomic number of tissue is approximately Z~7.5, materials with a higher atomic number used in the past such as Iodine (Z = 53) and gold (Z = 79) have a larger cross-section for absorption of radiation. For example, it was demonstrated in vitro that incorporating iodine into cellular DNA using iododeoxyuridine enhanced radiosensitivity at keV ranges by a factor of three [111]. The outcome of the in vitro study was also seen in an in vivo study, where an intratumoral injection of iodine and 200 kVp X-ray radiation suppressed the tumor growth by 80% [112]. In addition to having a great difference in mass attenuation between gold and soft tissue, gold has been shown to be biocompatible, simple, and economical to manufacture in many different shapes and sizes [113].
Radiation dose enhancement due to GNPs was first demonstrated using 1.9 nm GNPs in a mouse model, in one of the pioneering studies in GNP-mediated radiation dose enhancement by Hainfeld et al. [29]. A radiation dose of 30 Gy with 250 kVp X-rays to subcutaneous tumors in mice resulted in a significant decrease in tumor volume. However, the concentration of gold in this study was considerably high, at 2.7 g Au/kg body weight, which is not clinically feasible. Furthermore, the use of kV energies, while allowing for prominent photoelectric absorption in gold, is hindered due to the reduced penetration for deep-set tumors. Thus, as previously discussed, optimization of the internalization of the GNPs into the tumor cells, both in-vitro and in-vivo, is required for ideal efficacy. Whenever gold was internalized in vitro, radiosensitization was achievable at MV energy ranges, at concentrations as low as 1 ng/g [25,114,115,116]. This was demonstrated by Chithrani et al. in Figure 2A–C, which found a 17% increase in radiosensitization at 6 MV with 50 nm spherical GNPs [25]. When moving to an in vivo environment, radiosensitization was seen at a delivered dose of 10 μg/g of body weight [117]. This was accomplished by Wolfe et al. using targeted GNRs, as seen in Figure 2D–F, where there was a 36% increase in radiosensitization in vitro in PC3 cells, and a significantly enhanced tumor-growth delay when treated in vivo [117]. The treated dose is a ~1×106
improvement over the original treatment seen in Hainfeld’s pioneering study. The addition of targeting and improvements in the optimization of uptake has allowed significant progress in facilitating the progress of gold nanomaterials to the clinic.
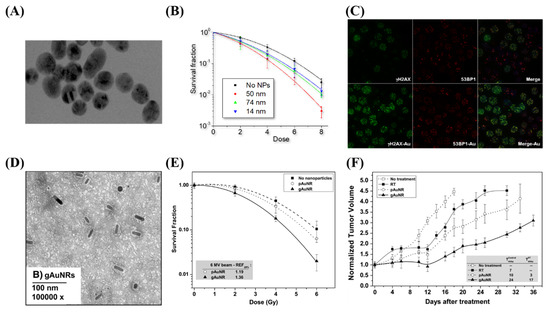
Figure 2. Radiosensitization due to gold nanoparticles. (A–C) Spheroidal GNPs improve radiosensitization in vitro, with the largest effect occurring with 50 nm GNPs, as they have the optimum uptake. This can be seen both through clonogenic assays as well as through imaging of double strand break foci with confocal microscopy. (D–F) GNRs displayed increased radiosensitization when targeted towards prostate cancer cell lines both in vitro through a clonogenic assay as well as in vivo through tumor volume measurements in a mouse model. Reproduced with permission from [117]. Copyright Elsevier, 2015.
GNPs localized intracellularly increases the probability of ionization events leading to local enhanced deposition of energy causing more damage to tumor cells [25]. The physical mechanism of GNP radiosensitization, seen in Figure 3A, occurs within the first nanoseconds of exposure, and is based on the difference in mass energy absorption coefficients between gold and soft tissue, enabling dose enhancement. The range of electrons released from GNPs is short, only a few micrometers, causing highly localized ionizing events. Thus, to achieve any enhancement from GNPs in radiation therapy, GNPs must be delivered and internalized specifically by tumor cells.
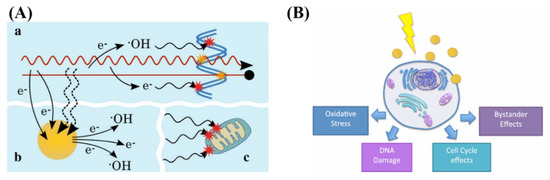
Figure 3. Radiosensitization and radiobiological effects. (A) Schematic showing chemical mechanism of GNP radiosensitization. While the radiation causes direct and indirect damage (yellow and red stars, respectively), there can be induction of secondary electrons and reactive oxygen species through gold nanoparticles. This can lead to damage to the DNA as well as secondary parts of the cell, such as the mitochondria. (B) GNPs can influence the cell through generation of reactive oxygen species, DNA damage, as well as cell cycle and bystander effects. Reproduced with permission from [110,118]. Copyright Springer Nature, 2016, 2017.
The chemical mechanism of GNP radiosensitization occurs through the radiochemical sensitization of DNA by increasing catalytic surface activity and increasing radical generation from the GNP surface [60]. Despite the prevailing notion that GNPs are chemically inert, there is a growing body of evidence that, due to the electronically active surface of GNPs, they are capable of catalyzing chemical reactions [119]. Catalysis by GNPs occurs mainly through surface interaction with molecular oxygen, generating free radicals [60]. This seems more evident in small GNPs (<5 nm in diameter) where surface to volume ratio is greater [120]. When combined with irradiation, the catalytic effects appear to be enhanced, with smaller GNPs with larger surface areas yielding more ROS [121]. However, it has been shown that at all energy levels, the dose enhancement observed cannot be simply explained by physical or chemical mechanisms [122]. To explain this, a radiobiological effect must be occurring.
The main radiobiological mechanisms involved in the cell’s response to irradiated GNPs results are the production of reactive oxygen species (ROS), oxidative stress, DNA damage induction, potential bystander effects, and cell cycle effects, as explained by Rosa et al. in Figure 6B [118]. Oxidative stress can cause cellular damage to the cell, including the oxidation of lipids, proteins, and DNA, which can result in apoptotic and necrotic cell death [123]. The mitochondria do appear to play a role, and the data indicate loss of function due to high intracellular ROS levels. It has been shown that the use of 1.4-nm triphenyl monosulfonate (TPPMS)-coated GNPs resulted in a loss of mitochondrial potential through elevated oxidative stress, resulting in necrotic cell death [122]. There have also been studies suggesting that GNPs may cause cell cycle disruptions and induce apoptosis. Radiosensitivity varies throughout the cell cycle with S phase being where a cell is most radioresistant and G2/M phase being most sensitive [124]. This could also depend on cell type, expression of cyclin kinases, and NP characteristics such as coating and size. For example, the use of 1.9 nm GNPs in DU-145 and MDA-MB-231 resulted in an increase in sub-G1 population in DU-145 population but not in MDA-MB-231 [125].
The biocompatibility of GNPs has already been tested in a phase I clinical trial. Furthermore, both in vitro and in vivo studies have shown the possibility of using GNPs as a radiosensitizer at clinically feasible concentrations, as discussed previously. Radiotherapy can also be combined with chemotherapy (chemoradiation) in cases where the tumor is not localized anymore, but metastasized as well, or to reduce potential micro-metastases spread. We will discuss the recent research conducted towards adding GNPs to this chemoradiation protocol in the next section.
3. Chemotherapy and combined therapy
While radiotherapy is mainly used to control the tumor locally, chemotherapy is used to control the tumor metastasis. Therefore, a combination of chemotherapy and radiotherapy (chemoradiation) is being practiced in the clinic to treat patients with locally advanced disease. Considering the variety of drugs available for cancer treatment, the possible choice of sequencing of combined chemotherapy and radiation therapy is countless, and the treatment plan differs between each patient. The standard treatment sequence refers to chemotherapy regimen before a traditional external beam radiation therapy treatment [53]. Chemotherapy used prior to irradiation is expected to cause maximal tumor regression for locally advanced tumors. The major limitation of combining chemotherapy and radiation therapy is normal tissue toxicity, since either modality can cause major normal tissue toxicity [8]. The main problems currently associated with chemotherapy are the biodistribution of pharmaceuticals, the lack of drug-specific affinity towards the tumor, limited plasma half-life, poor solubility and stability in physiological fluids, and nonspecific toxicity [126]. GNPs, due to their high surface area-to-volume ratio, as well as a large number of surface bio conjugation possibilities, are an ideal platform for delivering pharmaceutics for chemotherapy [46,127,128,129]. The use of GNPs as drug delivery system (DDS) can improve the pharmacokinetics, the pharmacodynamics, and the biodistribution of various drugs, as well as allow for improved targeting to reduce normal tissue toxicity. Beyond being an effective radiosensitizer, GNPs allow for a 100- to 1000-fold increase in ligand density compared to that of liposomal or polymeric DDSs [55]. Thus, the combination of the GNPs with radiotherapy and chemotherapy is part of the natural progression of the exploration of GNPs as a complete treatment modality.
The conjugation of moieties such as chemotherapeutic agents to the surface of the GNPs can be done using various techniques. The most common method is through the use of thiol group-containing biomolecules [130]. The use of thiolated biomolecules allows for functionalization of the GNPs with various agents, such as DNA, peptides, antibodies, and proteins [131,132]. This is a very robust method, as the majority of anticancer drugs can be thiolated, so as to be compatible with GNPs as a DDS [133,134]. Furthermore, capping agents, such as carboxyl terminated PEG molecules, with a thiol bond can allow for further functionalization techniques [131,135]. A general overview of various drug loading techniques using GNPs was explored by Fratoddi et al. [136]. GNPs, due to their favorable surface chemistry, are a suitable drug carrier for use in chemotherapeutics, and may be available for use in a wide range of drug delivery applications.
GNPs have been conjugated to a large variety of cytotoxic, anticancer drugs, and combined with radiation for improved efficacy. This includes paclitaxel, methotrexate, gemcitabine, doxorubicin, docetaxel, bleomycin, and platinum-based drugs like cisplatin [133,137,138,139,140,141]. Many different drugs can be used for different purposes, a few of which we will expand on. For instance, the antitumor activity of cisplatin was first discovered by Rosenberg and co-workers in 1960s, when they were examining whether electrical currents affect cellular division [142]. The researchers discovered that the inhibition of cellular division observed in the study was not due to electrical current, but platinum hydrolysis products formed from platinum electrodes. They reported that cis-tetrachlorodiammineplatinum (IV), cis-[PtCl4 (NH3)2], was the potent agent responsible for the inhibition. Cisplatin is now used to treat various types of cancers (i.e., cervical cancer, non-small-cell lung cancer, ovarian cancer, germ cell tumors, osteosarcomas, etc.), with a cure rate as high as 90% in testicular cancer [143]. However, long term cisplatin usage results in drug resistance [144]. To counteract this resistance, very high systemic doses of cisplatin should be administered, which results in severe systemic toxicity and poor patient compliance, limiting its clinical use [144,145,146]. It was shown recently that GNPs can be used to enhance damage caused by platinum-based anticancer drugs [147,148]. Comenge et al. conjugated cisplatin to GNPs and tested the efficacy of this DDS compared to the free drug along, as shown in Figure 4A–C [147]. They found that the use of GNPs led to 300 times more platinum being encapsulated in A549 cells in vitro, and while moving to in vivo, found similar efficacy but largely absent normal tissue toxicity. Yang et al. instead used free cisplatin along with GNPs in a combined chemoradiotherapy modality in vitro, seen in Figure 4D–F [116]. An additive relationship was discovered when treated with GNPs, cisplatin, and radiation in MDA-MB-231 cells. The use of GNPs may be an important avenue to explore when integrating cisplatin into chemoradiation protocols.
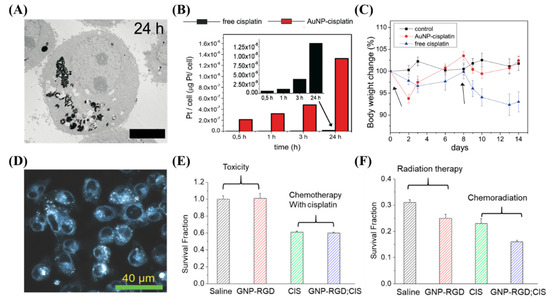
Figure 4. Cisplatin and gold nanoparticles. (A–C) Cisplatin conjugated GNPs lead to an increased deposition of platinum into A549 cells compared to the free drug in vitro, which led to less side effects in vivo with similar efficacy. Scale bar is 4 μm. (D–F) The use of free cisplatin to synergize with GNPs in a chemoradiation modality in vitro lead to a synergistic effect. Scale bar is 40 μm. Reproduced with permission from [116,147]. Copyright Public Library of Science, 2012; Copyright Multidisciplinary Digital Publishing Institute, 2018.
The use of GNPs as chemotherapeutic DDSs is increasing, and the combination of chemotherapy with radiotherapy remains one of the most effective treatment modalities available in the clinic. GNPs in combination with anticancer drugs and radiation is still limited in literature; however, the published work thus far shows a trend of improved dose response to the tumor coupled with reduced normal tissue toxicity. Further studies need to be completed, however, before translation to the clinic.
4. Future Considerations
A large issue that is plaguing nanotechnology in general is that a very low (~0.7%) portion of the administered dose is being delivered to the tumor [177]. While Wilhelm et al. describe the issue as improving understanding of the processes present in the body that inhibit the uptake of NPs, and then optimizing those processes, a more personalized approach could be introduced. Personalized medicine involves the analysis of a patient’s genetic code in order to have a better understanding of the potential response to treatment [178]. This is a very important avenue to explore, as it has been estimated that any class of anticancer drug used is ineffective in 75% of patients [179]. This is a result of no two cancers from different patients being the same. Beyond using genomic information to improve each individual’s cancer treatments, the use of an in vitro model that can better mimic the in vivo environment present in a patient may allow for the actual testing of treatment prior to administration. This can be achieved through the use of three-dimensional organoid models [180].
Organoid models have many advantages if implemented into a personalized medicine protocol. The use of a patient’s own cells, as described in Figure 5 by Fan et al., allows for the maintenance of the heterogeneity present [181]. Furthermore, normal spheroids can be engineered to have similar genetic alterations present in a patient, as discovered using their genomic information, through the use of gene-editing [182]. There are many other advantages as well, including low-cost generation, and quick (~4 weeks) results can be obtained. The capability of organoids to enable drug screening in an in vitro environment is being widely explored [180]. Furthermore, the use of organoid models has recently been tested on a chemoradiotherapy treatment of advanced rectal cancer and was able to accurately predict the response [183]. In the future, the use of organoids to screen chemoradiotherapy protocols with GNPs may enable an accurate assessment of response and allow for tailored, personalized medical care.
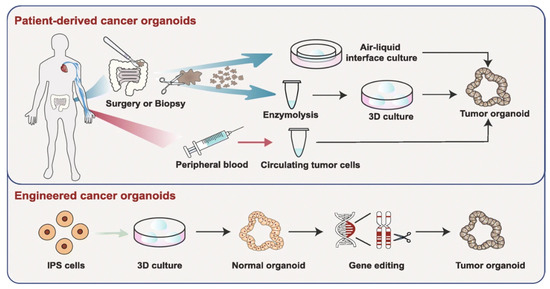
Figure 5. Organoid models toward personalized medicine. Patient-derived cancer organoids can be derived from surgically resected/biopsied tissues and circulating tumor cells. Furthermore, using gene-editing, normal spheroids can be mutated into tumor organoids. Reproduced with permission from [180]. Copyright Springer Nature, 2019.
Spheroids, and patient derived organoids, should be seen as avascular tumors, with limitations. To move towards personalized medical care with GNPs—using organoids as an assessment tool—certain strategies will have to be employed and obstacles overcome [184]. First, many more preclinical studies will have to be undertaken involving the use of GNPs with spheroids and their ability to accurately predict tumor response. This will have to be a large expanse of research, with many different types of treatment options including chemotherapy and radiotherapy as well as combined modalities. Second, scalability is very important: a high throughput method of testing efficacy of drugs and radiation modalities will have to be elucidated, as well as producing the organoids in a large-scale manner. Work is under way to improve production of spheroids as well as protocols for high throughput drug and radiation testing [185,186,187]. However, translation to the clinic will require more work at the bio–nano interface.
The use of GNPs in this workflow has limited published work, with most research focusing on individual treatment modalities, and not overall high throughput methods for translation to personalized medicine. Towards this, cheap and efficient GNP systems that can be easily functionalized with various moieties such as anticancer drugs or targeting ligands need to be designed for mass-scale production. Furthermore, comparisons of GNP-treated spheroids and organoids with in vivo models must be undertaken for improved confidence for translation to the clinic. Finally, it must be accepted the spheroids are a simplistic model when compared to an in vivo environment and will not be able to predict everything. However, despite this, the use of GNPs with organoid models for personalized medicine may be able to help save lives and improve the quality of lives in the future.
5. Conclusions
In the pursuit of improved cancer therapeutics, the use of GNPs offers the potential of improving on many different facets of the treatment process. Despite progress, the translation of GNPs to clinical practice has been limited due to the lack of coordination between researchers and clinicians. Many advances covered in this review aim to address issues that have arisen in the past, including targeted therapy, and the combination of radiotherapy and chemotherapy paired with GNPs for improved efficacy. However, it is still important to improve upon the current research so that translation to the clinic can be expedited.
This entry is adapted from the peer-reviewed paper 10.3390/nano10091671
This entry is offline, you can click here to edit this entry!
