Topical drug delivery has many advantages over other ways of administration, having increased patient compliance, avoiding the first-pass effect following oral drug administration or not requesting multiple doses administration. However, the skin barrier prevents the access of the applied drug, affecting its therapeutic activity. Carriers containing phospholipid soft vesicles are a new approach to enhance drug delivery into the skin and to improve the treatment outcome. These vesicles contain molecules that have the property to fluidize the phospholipid bilayers generating the soft vesicle and allowing it to penetrate into the deep skin layers. Ethosomes, glycerosomes and transethosomes are soft vesicles containing ethanol, glycerol or a mixture of ethanol and a surfactant, respectively.
- soft phospholipid vesicle
- skin disorders
- ethosomes
- glycerosomes
- transethosomes
- skin infection
- skin inflammation
- skin cancer
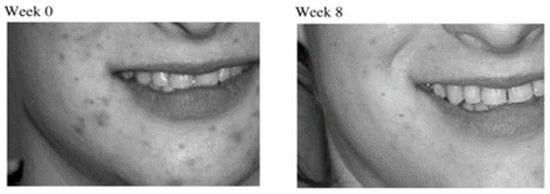
1. Treatment of Acne Vulgaris
| Treated Disorder | Investigated Vesicular Carrier | Study | Ref. |
|---|---|---|---|
| Acne Vulgaris | Clindamycin and salicylic acid ethosomal system | Clinical study on the reduction of acne vulgaris and skin tolerability of the formulation | [5] |
| Azelaic acid ethosomal system | Diffusion study through synthetic membrane | [7] | |
| Cryptotanshinone ethosomal system | In vitro skin permeation and skin deposition; in vivo anti-acne activity on rabbits | [8] | |
| Karanjin ethosomal system | In vitro skin premeation study on excised rats skin; in vivo skin irritation study on rats; in vivo anti-inflammatory and anti- acne studies on rats | [9] | |
| Viral infections | Acyclovir ethosomal system | Two-armed double-blind clinical study on subjects with recurrent herpes labialis | [10] |
| Acyclovir ethosomal system | Antiviral activity against HSV-1 by plaque reduction assay in monolayer cultures of Vero cells | [11][12] | |
| 9-[(2-hydroxyethoxy) methyl]guanine ethosomal system | Antiviral activity against HSV-1 by plaque reduction assay in monolayer cultures of Vero cells | [12] | |
| Ethosomal system of the essential oil of Melissa officinalis L. | In vitro activity against HSV type 1 in mammalian cells | [13] | |
| Bacterial infections | Bacitracin ethosomal system | Intracellular penetration and localization in fibroblasts (3T3); in vitro deposition and permeation through human cadaver skin | [14] |
| Erythromycin ethosomal system | In vivo activity in mice model of deep dermal S. aureus infection | [15] | |
| Psoralen ethosomal system | Photodynamic therapy in biofilms formed in Petri dishes | [16] | |
| Fungal infections | Voriconazole transethosomal system | In vitro skin permeation and deposition studies through mice skin; in vivo deposition study on mice | [17] |
| Griseofulvin ethosomal system | In vitro permeation and deposition study on new-born pig skin | [18] | |
| Clove oil ethosomal system | Ex vivo permeation studies on rat skin; antifungal activity in cup plate test against Candida albicans | [19] | |
| Voriconazole ethosomal system | In vitro anti-fungal activity against Asperigillus flavus colonies. In vitro skin deposition and permeation through abdominal rat skin |
[20] | |
| Econazole nitrate transethosomal system | Ex-vivo skin permeation and retention studies followed by in vitro antifungal activity against C. albicans fungus | [21] | |
| Skin inflammation | Diclofenac ethosomal system | In vitro permeation study on rat skin; in vivo anti-inflammatory activity in carrageenan-induced rat paw edema model | [22][23] |
| Ammonium glycyrrhizinate ethosomal system | In vitro permeation through human skin; clinical study to evaluate the anti-inflammatory activity in volunteers with methyl nicotinate erythema | [24] | |
| Matrine ethosomal system | In vitro percutaneous permeation study on rat skin; in vivo anti-inflammatory activity in rat measured by reflection spectrophotometery | [25] | |
| Apigenin ethosomal system | In vitro and in vivo deposition study on rat skin; evaluation of the reduction of cyclooxygenase-2 levels in mouse with skin inflammation | [26] | |
| Crocin ethosomal system | Evaluation of the anti-inflammatory activity on healthy volunteers. | [27] | |
| Diclofenac ethosomal system and Diclofenac transethosomal system | In vitro permeation and deposition studies on rat skin | [28] | |
| Diclofenac glycerosoomal system | In vitro penetration and permeation studies on new-born pig skin | [29][30][31] | |
| Paeoniflorin glycerosomal system | In vitro permeation experiments through excised rat abdominal skin; in vivo deposition in rat synovium | [32] | |
| Achillea millefolium L. extract ethosomal system | In vitro permeation strudy through fresh rat skin | [33] | |
| Psoriasis | Psoralen ethosomal system | In vitro permeation and penetration study using Franz diffusion cells and excised rat skin | [34] |
| Methotrexate and Salicylic acid ethosomal system | In vitro retention and permeation study on pig ear skin; in vivo anti-psoriatic activity in mice model with imiquimod-induced psoriasis | [35] | |
| Anthralin ethosomal system | Preparation, comparative evaluation and clinical assessment in psoriatic patients |
[36] | |
| Curcumin ethosomal system surface-modified with glycyrrhetinic acid-D-α-tocopherol acid polyethylene glycol succinate | In vitro anti-inflmmatory effect on interleukin-6-induced oxidative stress cell model; in vivo anti-psoriatic activity in mice model with imiquimod-induced psoriasis | [37] | |
| Skin cancer | 5- Fluorouracil ethosomal system containing -decyl methyl sulfoxide (Tumorep) | In vitro anti-tumor effect on five cell lines; in vivo anti-tumor effect in mice model of skin cancer | [38] |
| Paclitaxel ethosomal system | In vitro permeation study on human SC; in vitro antiproliferative effect in squamous carcinoma cells | [39] | |
| Fe-chlorophyllin transethosomal system | In vitro skin permeation and deposition studies through mice skin; in vivo evaluation of the anti-cancer effect in mice | [40] | |
| Plumbagin glycerosomal system | Ex vivo permeation study on rats skin | [41] | |
| Skin injury (wound healing) | Curcumin ethosomal system | In vivo wound healing effect in rats | [42] |
| Curcumin-propylene glycol liposomal system | In vivo wound repair effect is rats with burned skin | [43] | |
| Thymosinβ-4(Tβ-4) ethosomal system | In vitro drug release study on mice skin; in vivo pharmacokinetic and skin irritation studies on mice | [44] | |
| Skin pigmentation disorders | Linoleic acid ethosomal system | In vitro percutaneous permeation through human stratum corneum and viable epidermis membrane | [45] |
| Methoxsalen ethosomal system | Ex vivo release studies and photo- toxicity after exposure to UV light | [46] | |
| Hair loss | Minoxidil ethosomal system Minoxidil glycerosomal system |
In vitro penetration and permeation through abdominal nude mice skin | [6][47][48] |
| Ethosomal systems of plant extracts | In vivo effect on hair growth in rats with testosterone induced alopecia | [49] | |
| Skin aging | Vitamin E ethosomal system | In vitro permeation studies through skin and cultured fibroblasts | [50] |
| Curcumin ethosomal system | Clinical trial evaluating skin viscoelasticity, total deformation, biological elasticity and sagginess | [51] | |
| Rosmarinic acid ethosomal system | Ex vivo permeation studies using Franz diffusion cells and mice skin; ex vivo antioxidant activity | [52] |
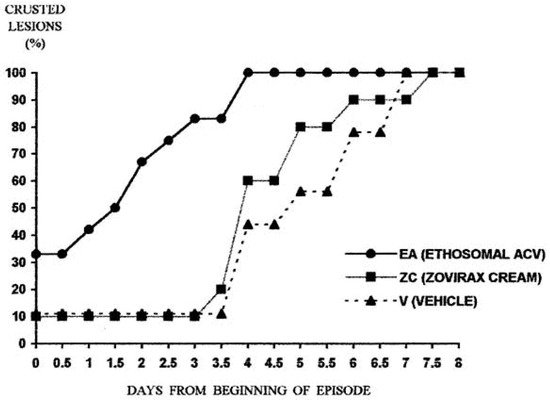
2. Treatment of Viral Skin Infections
3. Treatment of Bacterial Skin Infections
4. Treatment of Fungal Skin Infections
5. Treatment of Skin Inflammation
This entry is adapted from the peer-reviewed paper 10.3390/pharmaceutics13122129
References
- Gollnick, H.P.; Zouboulis, C.C.; Akamatsu, H.; Kurokawa, I.; Schulte, A. Pathogenesis and pathognesis-related treatment of acne. J. Dermatol. 1991, 18, 489–499.
- Leyden, J.J. New understanding of the pathogenesis of acne. J. Am. Acad. Dermatol. 1995, 32, 515–525.
- Gollnick, H.P.; Krautheim, A. Topical treatment in acne: Current status and future aspects. Dermatology 2003, 206, 29–36.
- Strauss, J.S.; Krowchuk, D.P.; Leyden, J.J.; Lucky, A.W.; Shalita, A.R.; Siegfried, E.C.; Thiboutot, D.M.; Van Voorhees, A.S.; Beutner, K.A.; Sieck, C.K.; et al. American Academy of Dermatology/American Academy of Dermatology Association. Guidelines of care for acne vulgaris management. J. Am. Acad. Dermatol. 2007, 56, 651–663.
- Touitou, E.; Godin, B.; Shumilov, M.; Bishouty, N.; Ainbinder, D.; Shouval, R.; Ingber, A.; Leibovici, V. Efficacy and tolerability of clindamycin phosphate and salicylic acid gel in the treatment of mild to moderate acne vulgaris. J. Eur. Acad. Dermatol. Venereol. 2008, 22, 629–631.
- Touitou, E.; Dayan, N.; Bergelson, L.; Godin, B.; Eliaz, M. Ethosomes- novel vesicular carriers for enhanced delivery: Characterization and skin penetration properties. J. Control. Release 2000, 65, 403–418.
- Esposito, E.; Menegatti, E.; Cortesi, R. Ethosomes and liposomes as topical vehicles for azelaic acid: A preformulation study. Int. J. Cosmet. Sci. 2004, 55, 253–264.
- Yu, Z.; Lv, H.; Han, G.; Ma, K. Ethosomes loaded with cryptotanshinone for acne treatment through topical gel formulation. PLoS ONE 2016, 11, e0159967.
- Ansari, S.A.; Qadir, A.; Warsi, M.H.; Mujeeb, M.; Aqil, M.; Mir, S.R.; Sharma, S. Ethosomes-based gel formulation of karanjin for treatment of acne vulgaris: In vitro investigations and preclinical assessment. 3 Biotech 2021, 11, 1–14.
- Horwitz, E.; Pisanty, S.; Czerninski, R.; Helser, M.; Eliav, E.; Touitou, E. A clinical evaluation of a novel liposomal carrier for acyclovir in the topical treatment of recurrent herpes labialis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1999, 87, 700–705.
- Cortesi, R.; Ravani, L.A.; Zaid, A.N.A.; Menegatti, E.A.; Romagnoli, R.A.; Drechsler, M.A.; Esposito, E.A. Ethosomes for the delivery of anti-HSV-1 molecules: Preparation, characterization and in vitro activity. Pharmazie 2010, 65, 743–749.
- Cortesi, R.; Ravani, L.; Menegatti, E.; Drechsler, M.; Esposito, E. Colloidal dispersions for the delivery of acyclovir: A comparative study. Indian J. Pharm. Sci. 2011, 73, 687–693.
- Vanti, G.; Ntallis, S.G.; Panagiotidis, C.A.; Dourdouni, V.; Patsoura, C.; Bergonzi, M.C.; Lazari, D.; Bilia, A.R. Glycerosome of Melissa officinalis L. Essential Oil for Effective Anti-HSV Type 1. Molecules 2020, 25, 3111.
- Godin, B.; Touitou, E. Mechanism of bacitracin permeation enhancement through the skin and cellular membranes from an ethosomal carrier. J. Control. Release 2004, 94, 365–379.
- Godin, B.; Touitou, E. Erythromycin ethosomal systems: Physicochemical characterization and enhanced antibacterial activity. Curr. Drug Deliv. 2005, 2, 269–275.
- Bagchi, D.; Dutta, S.; Singh, P.; Chaudhuri, S.; Pal, S.K. Essential dynamics of an effective phototherapeutic drug in a nanoscopic delivery vehicle: Psoralen in ethosomes for biofilm treatment. ACS Omega 2017, 2, 1850–1857.
- Song, C.K.; Balakrishnan, P.; Shim, C.K.; Chung, S.J.; Chong, S.; Kim, D.D. A novel vesicular carrier, transethosome, for enhanced skin delivery of voriconazole: Characterization and in vitro/in vivo evaluation. Colloids Surf. B Biointerfaces 2012, 92, 299–304.
- Marto, J.; Vitor, C.; Guerreiro, A.; Severino, C.; Eleutério, C.; Ascenso, A.; Simões, S. Ethosomes for enhanced skin delivery of griseofulvin. Colloids Surf. B Biointerfaces 2016, 146, 616–623.
- Shetty, S.; Jose, J.; Kumar, L.; Charyulu, R.N. Novel ethosomal gel of clove oil for the treatment of cutaneous candidiasis. J. Cosmet. Dermatol. 2018, 18, 862–869.
- Faisal, W.; Soliman, G.M.; Hamdan, A.M. Enhanced skin deposition and delivery of voriconazole using ethosomal preparations. J. Liposome Res. 2018, 28, 14–21.
- Verma, S.; Utreja, P. Transethosomes of Econazole Nitrate for Transdermal Delivery: Development, In-vitro Characterization, and Ex-vivo Assessment. Pharm. Nanotechnol. 2018, 6, 171–179.
- Touitou, E. Compositions for Applying Active Substances to or through the Skin. U.S. Patent 5,716,638, 10 February 1998.
- Jain, S.; Patel, N.; Madan, P.; Lin, S. Formulation and rheological evaluation of ethosome-loaded carbopol hydrogel for transdermal application. Drug Dev. Ind. Pharm. 2016, 42, 1315–1324.
- Paolino, D.; Lucania, G.; Mardente, D.; Alhaique, F.; Fresta, M. Ethosomes for skin delivery of ammonium glycyrrhizinate: In vitro percutaneous permeation through human skin and in vivo anti inflammatory activity on human volunteers. J. Control. Release 2005, 106, 99–110.
- Zhaowu, Z.; Xiaoli, W.; Yangde, Z.; Nianfeng, L. Preparation of matrine ethosome, its percutaneous permeation in vitro and anti- inflammatory activity in vivo in rats. J. Liposome Res. 2009, 19, 155–162.
- Shen, L.N.; Zhang, Y.T.; Wang, Q.; Xu, L.; Feng, N.P. Enhanced in vitro and in vivo skin deposition of apigenin delivered using ethosomes. Int. J. Pharm. 2014, 460, 280–288.
- Esposito, E.; Drechsler, M.; Huang, N.; Pavoni, G.; Cortesi, R.; Santonocito, D.; Puglia, C. Ethosomes and organogels for cutaneous administration of crocin. Biomed. Microdevices 2016, 18, 108.
- Ghanbarzadeh, S.; Arami, S. Enhanced transdermal delivery of diclofenac sodium via conventional liposomes, ethosomes, and transfersomes. BioMed Res. Int. 2013, 2013, 616810.
- Manca, M.L.; Zaru, M.; Manconi, M.; Lai, F.; Valenti, D.; Sinico, C.; Fadda, A.M. Glycerosomes: A new tool for effective dermal and transdermal drug delivery. Int. J. Pharm. 2013, 455, 66–74.
- Manca, M.L.; Cencetti, C.; Matricardi, P.; Castangia, I.; Zaru, M.; Sales, O.D.; Nacher, A.; Valenti, D.; Maccioni, A.M.; Fadda, A.M.; et al. Glycerosomes: Use of hydrogenated soy phosphatidylcholine mixture and its effect on vesicle features and diclofenac skin penetration. Int. J. Pharm. 2016, 511, 198–204.
- Manca, M.L.; Manconi, M.; Zaru, M.; Valenti, D.; Peris, J.E.; Matricardi, P.; Maccioni, A.M.; Fadda, A.M. Glycerosomes: Investigation of role of 1, 2-dimyristoyl-sn-glycero-3-phosphatidycholine (DMPC) on the assembling and skin delivery performances. Int. J. Pharm. 2017, 532, 401–407.
- Zhang, K.; Zhang, Y.; Li, Z.; Li, N.; Feng, N. Essential oil-mediated glycerosomes increase transdermal paeoniflorin delivery: Optimization, characterization, and evaluation in vitro and in vivo. Int. J. Nanomed. 2017, 12, 3521–3532.
- Andleeb, M.; Shoaib Khan, H.M.; Daniyal, M. Development, Characterization and Stability Evaluation of Topical Gel Loaded with Ethosomes Containing Achillea millefolium L. Extract. Front. Pharmacol. 2021, 12, 336.
- Zhang, Y.T.; Shen, L.N.; Wu, Z.H.; Zhao, J.H.; Feng, N.P. Comparison of ethosomes and liposomes for skin delivery of psoralen for psoriasis therapy. Int. J. Pharm. 2014, 471, 449–452.
- Chandra, A.; Aggarwal, G.; Manchanda, S.; Narula, A. Development of topical gel of methotrexate incorporated ethosomes and salicylic acid for the treatment of psoriasis. Pharm. Nanotechnol. 2019, 7, 362–374.
- Fathalla, D.; Youssef, E.M.K.; Soliman, G.M. Liposomal and Ethosomal Gels for the Topical Delivery of Anthralin: Preparation, Comparative Evaluation and Clinical Assessment in Psoriatic Patients. Pharmaceutics 2020, 12, 446.
- Guo, T.; Lu, J.; Fan, Y.; Zhang, Y.; Yin, S.; Sha, X.; Feng, N. TPGS assists the percutaneous administration of curcumin and glycyrrhetinic acid coloaded functionalized ethosomes for the synergistic treatment of psoriasis. Int. J. Pharm. 2021, 604, 120762.
- Ainbinder, D.; Touitou, E. A new approach for skin tumor treatment: From delivery system characterization to in vivo evaluation. Drug Deliv. Trans. Res. 2011, 1, 53–65.
- Paolino, D.; Celia, C.; Trapasso, E.; Cilurzo, F.; Fresta, M. Paclitaxel-loaded ethosomes®: Potential treatment of squamous cell carcinoma, a malignant transformation of actinic keratoses. Eur. J. Pharm. Biopharm. 2012, 81, 102–112.
- Rady, M.; Gomaa, I.; Afifi, N.; Abdel-Kader, M. Dermal delivery of Fe-chlorophyllin via ultradeformable nanovesicles for photodynamic therapy in melanoma animal model. Int. J. Pharm. 2018, 548, 480–490.
- Md, S.; Alhakamy, N.A.; Aldawsari, H.M.; Husain, M.; Khan, N.; Alfaleh, M.A.; Asfour, H.Z.; Riadi, Y.; Bilgrami, A.L.; Akhter, M.H. Plumbagin-Loaded Glycerosome Gel as Topical Delivery System for Skin Cancer Therapy. Polymers 2021, 13, 923.
- Partoazar, A.; Kianvash, N.; Darvishi, M.H.; Nasoohi, S.; Rezayat, S.M.; Bahador, A. Ethosomal curcumin promoted wound healing and reduced bacterial flora in second degree burn in rat. Drug Res. 2016, 66, 660–665.
- Kianvash, N.; Bahador, A.; Pourhajibagher, M.; Ghafari, H.; Nikoui, V.; Rezayat, S.M.; Dehpour, A.R.; Partoazar, A. Evaluation of propylene glycol nanoliposomes containing curcumin on burn wound model in rat: Biocompatibility, wound healing, and anti-bacterial effects. Drug Deliv. Transl. Res. 2017, 7, 654–663.
- Fu, X.; Shi, Y.; Wang, H.; Zhao, X.; Sun, Q.; Huang, Y.; Qi, T.; Lin, G. Ethosomal Gel for Improving Transdermal Delivery of Thymosin β-4. Int. J. Nanomed. 2019, 14, 9275–9284.
- Celia, C.; Cilurzo, F.; Trapasso, E.; Cosco, D.; Fresta, M.; Paolino, D. Ethosomes® and transfersomes® containing linoleic acid: Physicochemical and technological features of topical drug delivery carriers for the potential treatment of melasma disorders. Biomed. Microdevices 2012, 14, 119–130.
- Garg, B.J.; Garg, N.K.; Beg, S.; Singh, B.; Katare, O.P. Nanosized ethosomes-based hydrogel formulations of methoxsalen for enhanced topical delivery against vitiligo: Formulation optimization, in vitro evaluation and preclinical assessment. J. Drug Target 2016, 24, 233–246.
- Meidan, V.M.; Touitou, E. Treatments for Androgenetic Alopecia and Alopecia Areata. Drugs 2001, 61, 53–69.
- Rani, D.; Sharma, V.; Manchanda, R.; Chaurasia, H. Formulation, Design and Optimization of Glycerosomes for Topical Delivery of Minoxidil. Res. J. Pharmacol. Technol. 2021, 14, 2367–2374.
- Madhunithya, E.; Venkatesh, G.; Shyamala, G.; Manjari, V.; Ramesh, S.; Karuppaiah, A.; Sankar, V. Development of ethosome comprising combined herbal extracts and its effect on hair growth. Adv. Tradit. Med. 2021, 21, 131–141.
- Touitou, E.; Godin, B. Skin nonpenetrating sunscreens for cosmetic and pharmaceutical formulations. Clin. Dermatol. 2008, 26, 375–379.
- Jeswani, G.; Saraf, S. Topical Delivery of Curcuma longa extract loaded nanosized ethosomes to combat facial wrinkles. J. Pharm. Drug Deliv. Res. 2014, 3, 1–8.
- Yücel, Ç.; Şeker Karatoprak, G.; Değim, İ.T. Anti-aging formulation of rosmarinic acid-loaded ethosomes and liposomes. J. Microencapsul. 2019, 36, 180–191.
- Ramdass, P.; Mullick, S.; Farber, H.F. Viral skin diseases. Prim. Care Clin. Off. 2015, 42, 517–567.
- Brady, R.C.; Bernstein, D.I. Treatment of herpes simplex virus infections. Antivir. Res. 2004, 61, 73–81.
- Spruance, S.L.; Crumpacker, C.S. Topical 5% acyclovir in polyethylene-glycol for herpes simplex labialis: Antiviral effect without clinical benefit. Am. J. Med. 1982, 73, 315–319.
- Raborn, G.W.; McGaw, W.T.; Grace, M.; Houle, L. Herpes labialis treatment with acyclovir 5% ointment. J. Can. Dent. Assoc. 1989, 55, 135–137.
- Fiddian, A.P.; Ivanyi, L. Topical acyclovir in the management of herpes labialis. Br. J. Dermatol. 1983, 109, 321–326.
- Raborn, G.W.; McGaw, W.T.; Grace, M.; Percy, J.; Samuels, S. Herpes labialis treatment with acyclovir 5% modified aqueous cream: A double blind randomized trial. Oral Surg. Oral Med. Oral Pathol. 1989, 67, 676–679.
- Touitou, E.; Godin, B.; Dayan, N.; Weiss, C.; Piliponsky, A.; Levi-Schaffer, F. Intracellular delivery mediated by an ethosomal carrier. Biomaterials 2001, 22, 3053–3059.
- Naimi, T.S.; LeDell, K.H.; Como-Sabetti, K. Comparison of community- and health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA 2003, 290, 2976–2984.
- Fung, H.B.; Chang, J.Y.; Kuczynski, S. A practical guide to the treatment of complicated skin and soft tissue infections. Drugs 2003, 63, 1459–1480.
- O’Dell, M.L. Skin and wound infections: An overview. Am. Fam. Physician 1998, 57, 2424–2432.
- Nichols, R.L. Optimal treatment of complicated skin and skin structure infections. J. Antimicrob. Chemother. 1999, 44, 19–23.
- Eldridge, M.L.; Chambers, C.J.; Sharon, V.R.; Thompson, G.R., III. Fungal infections of the skin and nail: New treatment options. Expert Rev. Anti Infect. Ther. 2014, 12, 1389–1405.
- Zhang, L.; Li, X.; Zhu, S.; Zhang, T.; Maimaiti, A.; Ding, M.; Shi, S. Dermal Targeting Delivery of Terbinafine Hydrochloride Using Novel Multi-Ethosomes: A New Approach to Fungal Infection Treatment. Coatings 2020, 10, 304.
- Ferrero-Miliani, L.; Nielsen, O.H.; Andersen, P.S.; Girardin, S.E. Chronic inflammation: Importance of NOD2 and NALP3 in interleukin-1beta generation. Clin. Exp. Immunol. 2007, 147, 227–235.
- Kim, J.; Kim, B.E.; Leung, D.Y. Pathophysiology of atopic dermatitis: Clinical implications. Allergy Asthma Proc. 2019, 40, 84.
- Eichenfield, L.F.; Tom, W.L.; Berger, T.G.; Krol, A.; Paller, A.S.; Schwarzenberger, K.; Bergman, J.N.; Chamlin, S.L.; Cohen, D.E.; Cooper, K.D.; et al. Guidelines of care for the management of atopic dermatitis: Section 2. Management and treatment of atopic dermatitis with topical therapies. J. Am. Acad. Dermatol. 2014, 71, 116–132.
- Chen, J.G.; Liu, Y.F.; Gao, T.W. Preparation and anti-inflammatory activity of triptolide ethosomes in an erythema model. J. Liposome Res. 2010, 20, 297–303.
