Methylene blue (MB), as the first fully man-made medicine, has a wide range of clinical applications. Apart from its well-known applications in surgical staining, malaria, and methemoglobinemia, the anti-oxidative properties of MB recently brought new attention to this century-old drug. Mitochondrial dysfunction has been observed in systematic aging that affects many different tissues, including the brain and skin. This leads to increased oxidative stress and results in downstream phenotypes under age-related conditions. MB can bypass Complex I/III activity in mitochondria and diminish oxidative stress to some degree. MB also shows broad spectrum UV absorption capability.
- methylene blue
- mitochondria
- neurodegeneration
- skin aging
- progeria
1. Introduction
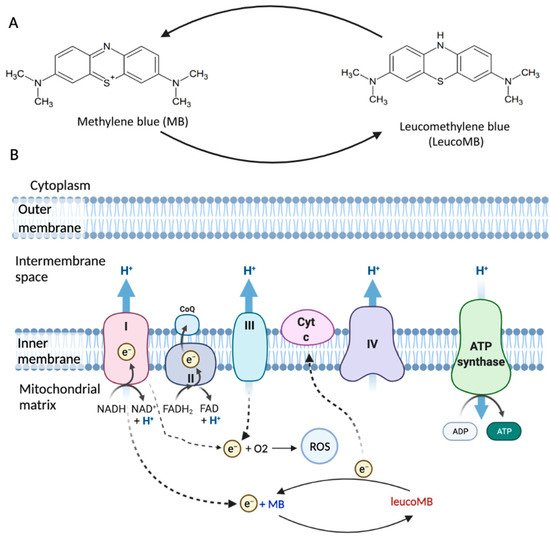
2. Anti-Aging Potentials of Methylene Blue
2.1. MB in Brain Aging
MB is highly lipophilic and able to effectively cross the blood-brain barrier (BBB) [10]. MB concentration is found to be higher in the brain than in plasma after oral administration or intravenous injection in rats [13]. Besides, it has a strong affinity for mitochondria [14]. Different from the other antioxidants such as MitoQ and MitoVitE, MB can reduce the production of free radicals by bypassing Complex I/III activity rather than scavenging free radicals [15]. In fact, the membrane potential in Complex III-inhibited mitochondria can be partially restored by MB in both mice and rats [11]. MB, acting as an electron donor, can also increase the expression of brain cytochrome oxidase and oxygen consumption in vivo [16][17]. Besides, low doses of MB were shown to effectively inhibit nitric oxide (NO), which inhibits cytochrome c oxidase activity [18][19]. All these properties make MB a promising drug candidate for brain diseases’ treatment.
Several studies suggest that there is an association between mitochondrial dysfunction and abnormal processing of Aβ and tau [20][21][22][23]. Amyloid precursor protein (APP) can be trapped in the mitochondrial membrane and impair mitochondrial function [24]. Overexpression of tau can also result in mitochondrial dysfunction by decreasing ATP production and increasing oxidative stress [25]. Conversely, the damaged mitochondrial function can induce aberrant Aβ production and promote abnormal phosphorylation of tau [26]. MB was reported to prevent Aβ and tau aggregation or dissolve existing aggregates via autophagic clearance, and therefore alleviate downstream pathological consequences [27][28][29][30]. MB could directly or indirectly target β-secretase cleavage of amyloid precursor protein (APP) and regulate the generation of Aβ [31]. MB’s role in Aβ and tau aggregation clearance may help improve mitochondrial functions in Alzheimer's Disease (AD) neurons and thus contribute to AD treatment. Besides, cytochrome oxidase activity has been shown to decline in AD [32], while MB can increase the enzymatic activity of cytochrome oxidase, which results in an increased oxidative metabolic capacity of neurons [33].
Parkinson's Disease (PD) is another neurodegenerative disease associated with aging. It is one of the most common movement diseases featured by dopaminergic neuronal damage [34]. The pathological hallmark of PD is Lewy Bodies and Lewy Neurites, intracellular aggregates of the protein α-synuclein (α-syn) [35]. A-syn can lead to progressive mitochondrial dysfunction when translocated to the mitochondria [36]. Mitochondrial dysfunction is considered the primary cause of dopaminergic apoptosis via inducing oxidative stress in PD [37]. Based on MB’s role in improving mitochondrial function, MB could be a promising treatment in PD.
In addition to its beneficial role in age-related brain disorders, MB is a promising memory enhancer. Since metabolic derangement is observed in old brains, and mitochondrial impairment accumulates over time, improving mitochondria may help neurons maintain their health and improve their functions [38][39][40].
2.2. MB in Skin Aging
Oxidative stress is involved in both intrinsic and extrinsic skin aging [83 ]. Especially when skin is exposed to certain environmental risk factors such as UV radiation, increasing oxidative damage will decrease collagen synthesis and increase collagen breakdown, leading to accelerated aging. Antioxidants such as MB can protect skin and slow down the aging process (Figure 2).
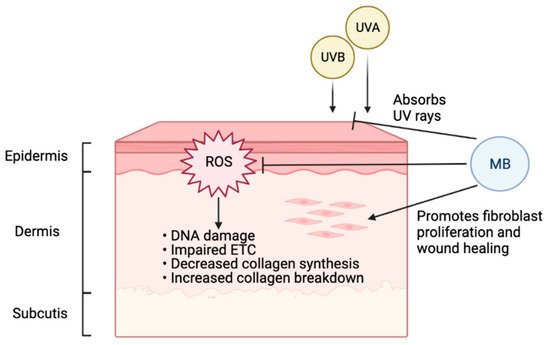
A previous study has shown that MB treatment in normal fibroblasts could increase lifespan and cell proliferation while reducing aging markers [41]. In that study, MB increased cytochrome oxidase by 30%, enhanced oxygen consumption by 37–70%, and reversed premature senescence caused by H2O2 or cadmium, and the ratio MB/cytochrome c could be important for MB’s protective role. Interestingly, when MB was compared with common antioxidants used in skincare, including vitamin C and retinol (vitamin A), MB-treated skin cells outperformed both of them significantly in terms of promoting cell proliferation and reducing age-related markers [42][43]. In addition to its potent antioxidant function, MB treatment in skin fibroblasts could stimulate the expression of ECM proteins, including upregulation of elastin and collagen 2A1, the two vital proteins for healthy, youthful skin [42]. Another study showed that MB provides broad-spectrum absorption of UV rays and mitigates DNA double-strand breaks caused by UVB irradiation in human keratinocytes [43]. Together, this evidence supports MB as protective and beneficial to human skin, suggesting that the inclusion of MB in daily skincare may effectively delay photoaging.
MB can facilitate wound healing. During aging, the proliferation and migration of fibroblasts are often decreased, and collagen and elastin in the ECM are degraded [44]. Therefore, the repair capabilities of the skin decline due to structural and functional changes. Our study indicated that MB treatment could promote fibroblast migration and proliferation in the wound healing process [42]. In skin survival burn models in rats, MB treatment could reduce necrosis progression, which might be mediated by decreasing oxidative stress through blocking nitric oxide (NO) [45]. Moreover, MB can facilitate wound healing by reducing antimicrobial burden and decreasing hyper-granulation. MB also has a drying effect without harming healthy cells [46][47][48]. Overall, MB was shown to improve tissue viability with little to no irritation in laboratory models.
2.3. MB in Progeria
This entry is adapted from the peer-reviewed paper 10.3390/cells10123379
References
- Schirmer, R.H.; Adler, H.; Pickhardt, M.; Mandelkow, E. “Lest we forget you—Methylene blue…”. Neurobiol. Aging 2011, 32, 2325.e7–2325.e16.
- Oz, M.; Lorke, D.E.; Hasan, M.; Petroianu, G.A. Cellular and molecular actions of Methylene Blue in the nervous system. Med. Res. Rev. 2011, 31, 93–117.
- Harman, D. Aging: A Theory Based on Free Radical and Radiation Chemistry. J. Gerontol. 1956, 11, 298–300.
- Murphy, M.P. How mitochondria produce reactive oxygen species. Biochem. J. 2009, 417, 1–13.
- Chistiakov, D.A.; Sobenin, I.A.; Revin, V.V.; Orekhov, A.N.; Bobryshev, Y.V. Mitochondrial aging and age-related dysfunction of mitochondria. Biomed. Res. Int. 2014, 2014, 238463.
- Guo, C.Y.; Sun, L.; Chen, X.P.; Zhang, D.S. Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regen. Res. 2013, 8, 2003–2014.
- Gabrielli, D.; Belisle, E.; Severino, D.; Kowaltowski, A.J.; Baptista, M.S. Binding, Aggregation and Photochemical Properties of Methylene Blue in Mitochondrial Suspensions. Photochem. Photobiol. 2004, 79, 227.
- Atamna, H.; Kumar, R. Protective role of methylene blue in Alzheimer’s disease via mitochondria and cytochrome c oxidase. J. Alzheimers Dis. 2010, 20, S439–S452.
- Lee, S.K.; Mills, A. Novel photochemistry of leuco-Methylene Blue. Chem. Commun. 2003, 3, 2366–2367.
- Rojas, J.C.; Bruchey, A.K.; Gonzalez-Lima, F. Neurometabolic mechanisms for memory enhancement and neuroprotection of methylene blue. Prog. Neurobiol. 2012, 96, 32–45.
- Sváb, G.; Kokas, M.; Sipos, I.; Ambrus, A.; Tretter, L. Methylene blue bridges the inhibition and produces unusual respiratory changes in complex iii-inhibited mitochondria. Studies on rats, mice and guinea pigs. Antioxidants 2021, 10, 305.
- Tretter, L.; Horvath, G.; Hölgyesi, A.; Essek, F.; Adam-Vizi, V. Enhanced hydrogen peroxide generation accompanies the beneficial bioenergetic effects of methylene blue in isolated brain mitochondria. Free Radic. Biol. Med. 2014, 77, 317–330.
- Peter, C.; Hongwan, D.; Küpfer, A.; Lauterburg, B.H. Pharmacokinetics and organ distribution of intravenous and oral methylene blue. Eur. J. Clin. Pharmacol. 2000, 56, 247–250.
- Klosowski, E.M.; de Souza, B.T.L.; Mito, M.S.; Constantin, R.P.; Mantovanelli, G.C.; Mewes, J.M.; Bizerra, P.F.V.; Menezes, P.V.M.D.C.; Gilglioni, E.H.; Utsunomiya, K.S.; et al. The photodynamic and direct actions of methylene blue on mitochondrial energy metabolism: A balance of the useful and harmful effects of this photosensitizer. Free Radic. Biol. Med. 2020, 153, 34–53.
- Biju, K.C.; Evans, R.C.; Shrestha, K.; Carlisle, D.C.B.; Gelfond, J.; Clark, R.A. Methylene Blue Ameliorates Olfactory Dysfunction and Motor Deficits in a Chronic MPTP/Probenecid Mouse Model of Parkinson’s Disease. Neuroscience 2018, 380, 111–122.
- Gonzalez-Lima, F.; Barksdale, B.R.; Rojas, J.C. Mitochondrial respiration as a target for neuroprotection and cognitive enhancement. Biochem. Pharmacol. 2014, 88, 584–593.
- Riha, P.D.; Bruchey, A.K.; Echevarria, D.J.; Gonzalez-Lima, F. Memory facilitation by methylene blue: Dose-dependent effect on behavior and brain oxygen consumption. Eur. J. Pharmacol. 2005, 511, 151–158.
- Mayer, B.; Brunner, F.; Schmidt, K. Inhibition of nitric oxide synthesis by methylene blue. Biochem. Pharmacol. 1993, 45, 367–374.
- Brown, G.C.; Borutaite, V. Nitric oxide, cytochrome c and mitochondria. Biochem. Soc. Symp. 1999, 66, 17–25.
- Hansson Petersen, C.A.; Alikhani, N.; Behbahani, H.; Wiehager, B.; Pavlov, P.F.; Alafuzoff, I.; Leinonen, V.; Ito, A.; Winblad, B.; Glaser, E.; et al. The amyloid β-peptide is imported into mitochondria via the TOM import machinery and localized to mitochondrial cristae. Proc. Natl. Acad. Sci. USA 2008, 105, 13145–13150.
- Eckert, A.; Pagani, L. Amyloid-beta interaction with mitochondria. Int. J. Alzheimers Dis. 2011, 2011, 1–12.
- Cheng, Y.; Bai, F. The association of tau with mitochondrial dysfunction in Alzheimer’s disease. Front. Neurosci. 2018, 12, 2014–2019.
- Szabo, L.; Eckert, A.; Grimm, A. Insights into disease-associated Tau impact on mitochondria. Int. J. Mol. Sci. 2020, 21, 6344.
- Anandatheerthavarada, H.K.; Biswas, G.; Robin, M.A.; Avadhani, N.G. Mitochondrial targeting and a novel transmembrane arrest of Alzheimer’s amyloid precursor protein impairs mitochondrial function in neuronal cells. J. Cell Biol. 2003, 161, 41–54.
- Li, X.C.; Hu, Y.; Wang, Z.H.; Luo, Y.; Zhang, Y.; Liu, X.P.; Feng, Q.; Wang, Q.; Ye, K.; Liu, G.P.; et al. Human wild-type full-length tau accumulation disrupts mitochondrial dynamics and the functions via increasing mitofusins. Sci. Rep. 2016, 6, 1–10.
- Swerdlow, R.H. Mitochondria and Mitochondrial Cascades in Alzheimer’s Disease. J. Alzheimers Dis. 2018, 62, 1403–1416.
- Necula, M.; Breydo, L.; Milton, S.; Kayed, R.; Van Der Veer, W.E.; Tone, P.; Glabe, C.G. Methylene blue inhibits amyloid Aβ oligomerization by promoting fibrillization. Biochemistry 2007, 46, 8850–8860.
- Medina, D.X.; Caccamo, A.; Oddo, S. Methylene blue reduces Aβ levels and rescues early cognitive deficit by increasing proteasome activity. Brain Pathol. 2011, 21, 140–149.
- Hosokawa, M.; Arai, T.; Masuda-Suzukake, M.; Nonaka, T.; Yamashita, M.; Akiyama, H.; Hasegawa, M. Methylene Blue Reduced Abnormal Tau Accumulation in P301L Tau Transgenic Mice. PLoS ONE 2012, 7, e52389.
- Lee, B., II; Suh, Y.S.; Chung, Y.J.; Yu, K.; Park, C.B. Shedding Light on Alzheimer’s β-Amyloidosis: Photosensitized Methylene Blue Inhibits Self-Assembly of β-Amyloid Peptides and Disintegrates Their Aggregates. Sci. Rep. 2017, 7, 1–10.
- Mori, T.; Koyama, N.; Segawa, T.; Maeda, M.; Maruyama, N.; Kinoshita, N.; Hou, H.; Tan, J.; Town, T. Methylene Blue Modulates β-Secretase, Reverses Cerebral Amyloidosis, and Improves Cognition in Transgenic Mice. J. Biol. Chem. 2014, 289, 30303–30317.
- Praticò, D. Oxidative stress hypothesis in Alzheimer’s disease: A reappraisal. Trends Pharmacol. Sci. 2008, 29, 609–615.
- Oz, M.; Lorke, D.E.; Petroianu, G.A. Methylene blue and Alzheimer’s disease. Biochem. Pharmacol. 2009, 78, 927–932.
- Surmeier, D.J. Determinants of dopaminergic neuron loss in Parkinson’s disease. FEBS J. 2018, 285, 3657–3668.
- Dickson, D.W. Parkinson’s Disease and Parkinsonism: Neuropathology. Cold Spring Harb. Perspect. Med. 2012, 2, a009258.
- Ganjam, G.K.; Bolte, K.; Matschke, L.A.; Neitemeier, S.; Dolga, A.M.; Höllerhage, M.; Höglinger, G.U.; Adamczyk, A.; Decher, N.; Oertel, W.H.; et al. Mitochondrial damage by α-synuclein causes cell death in human dopaminergic neurons. Cell Death Dis. 2019, 10, 1–16.
- Subramaniam, S.R.; Chesselet, M.F. Mitochondrial dysfunction and oxidative stress in Parkinson’s disease. Prog. Neurobiol. 2013, 106–107, 17–32.
- Pérez, M.J.; Jara, C.; Quintanilla, R.A. Contribution of Tau pathology to mitochondrial impairment in neurodegeneration. Front. Neurosci. 2018, 12, 1–14.
- Onyango, I.G.; Lu, J.; Rodova, M.; Lezi, E.; Crafter, A.B.; Swerdlow, R.H. Regulation of neuron mitochondrial biogenesis and relevance to brain health. Biochim. Biophys. Acta-Mol. Basis Dis. 2010, 1802, 228–234.
- Rosenkranz, S.C.; Shaposhnykov, A.A.; Träger, S.; Engler, J.B.; Witte, M.E.; Roth, V.; Vieira, V.; Paauw, N.; Bauer, S.; Schwencke-Westphal, C.; et al. Enhancing mitochondrial activity in neurons protects against neurodegeneration in a mouse model of multiple sclerosis. Elife 2021, 10, 1–60.
- Atamna, H.; Nguyen, A.; Schultz, C.; Boyle, K.; Newberry, J.; Kato, H.; Ames, B.N. Methylene blue delays cellular senescence and enhances key mitochondrial biochemical pathways. FASEB J. 2008, 22, 703–712.
- Mei Xiong, Z.; O’Donovan, M.; Sun, L.; Young Choi, J.; Ren, M.; Cao, K. Anti-Aging Potentials of Methylene Blue for Human Skin Longevity. Sci. Rep. 2017, 7, 1–12.
- Xiong, Z.M.; Mao, X.; Trappio, M.; Arya, C.; el Kordi, J.; Cao, K. Ultraviolet radiation protection potentials of Methylene Blue for human skin and coral reef health. Sci. Rep. 2021, 11, 1–9.
- Gosain, A.; DiPietro, L.A. Aging and Wound Healing. World J. Surg. 2004, 28, 321–326.
- Rosique, M.J.; Rosique, R.G.; Faria, F.M.; Oliveira, C.C.; Farina, J.A.; Évora, P.R.B. Methylene blue reduces progression of burn and increases skin survival in an experimental rat model. Burns 2017, 43, 1702–1708.
- Edwards, K. New Twist on an Old Favorite: Gentian Violet and Methylene Blue Antibacterial Foams. Adv. Wound Care 2016, 5, 11–18.
- Woo, K.Y.; Heil, J. A prospective evaluation of methylene blue and gentian violet dressing for management of chronic wounds with local infection. Int. Wound J. 2017, 14, 1029–1035.
- Pérez, M.; Robres, P.; Moreno, B.; Bolea, R.; Verde, M.T.; Pérez-Laguna, V.; Aspiroz, C.; Gilaberte, Y.; Rezusta, A. Comparison of Antibacterial Activity and Wound Healing in a Superficial Abrasion Mouse Model of Staphylococcus aureus Skin Infection Using Photodynamic Therapy Based on Methylene Blue or Mupirocin or Both. Front. Med. 2021, 8, 673408.
- Gonzalo, S.; Kreienkamp, R.; Askjaer, P. Hutchinson-Gilford Progeria Syndrome: A premature aging disease caused by LMNA gene mutations. Ageing Res. Rev. 2017, 33, 18–29.
- Capell, B.C.; Erdos, M.R.; Madigan, J.P.; Fiordalisi, J.J.; Varga, R.; Conneely, K.N.; Gordon, L.B.; Der, C.J.; Cox, A.D.; Collins, F.S. Inhibiting farnesylation of progerin prevents the characteristic nuclear blebbing of Hutchinson-Gilford progeria syndrome. Proc. Natl. Acad. Sci. USA 2005, 102, 12879–12884.
- Scaffidi, P. Lamin A-Dependent Nuclear Defects in Human Aging. Science 2006, 312, 1059–1063.
- Köhler, F.; Bormann, F.; Raddatz, G.; Gutekunst, J.; Corless, S.; Musch, T.; Lonsdorf, A.S.; Erhardt, S.; Lyko, F.; Rodríguez-Paredes, M. Epigenetic deregulation of lamina-associated domains in Hutchinson-Gilford progeria syndrome. Genome Med. 2020, 12, 1–16.
- Viteri, G.; Chung, Y.W.; Stadtman, E.R. Effect of progerin on the accumulation of oxidized proteins in fibroblasts from Hutchinson Gilford progeria patients. Mech. Ageing Dev. 2010, 131, 2–8.
- Lattanzi, G.; Marmiroli, S.; Facchini, A.; Maraldi, N.M. Nuclear damages and oxidative stress: New Perspectives for laminopathies. Eur. J. Histochem. 2012, 56.
- Xiong, Z.M.; Choi, J.Y.; Wang, K.; Zhang, H.; Tariq, Z.; Wu, D.; Ko, E.; Ladana, C.; Sesaki, H.; Cao, K. Methylene blue alleviates nuclear and mitochondrial abnormalities in progeria. Aging Cell 2016, 15, 279–290.
