In the early 1970s, environmental conservationists were becoming concerned that a reduction in the thickness of the atmospheric ozone layer would lead to increased levels of ultraviolet (UV) radiation at ground level, resulting in higher population exposure to UV and subsequent harm, especially a rise in skin cancer. At the time, no measurements had been reported on the normal levels of solar UV radiation which populations received in their usual environment, so this lack of data, coupled with increasing concerns about the impact to human health, led to the development of simple devices that monitored personal UV exposure.
1. Fundamental Requirements of Personal Ultraviolet Dosimeters
Whilst radiometers and spectroradiometers are used to monitor ambient solar UV radiation, these instruments are not suited to determining the UV exposure of individuals, especially at multiple sites over the body, because of their bulk and generally high cost. Ideally, UV dosimeters designed for personal use should have the following characteristics:
-
The dosimeter should be easy to handle and not impose restrictions on the activities of the wearers.
-
In physical dosimeters, the output electrical signal should increase linearly with irradiance and have a wide dynamic range.
-
In chemical or biological dosimeters, the change produced in the system should increase linearly with UV dose. If not, the dose response curve should at least be monotonic; that is, any given dosimeter response is the result of only one radiation dose.
-
The dosimeter should exhibit photoaddition; in other words, each wavelength acts independently, and the effect of polychromatic radiation is the sum of the effects of all wavelengths involved.
-
The spectral sensitivity of the dosimeter should, ideally, match the action spectrum of the photobiological effect being monitored, most commonly erythema.
-
The dosimeter response should be independent of temperature and humidity, exhibit no ‘dark effect’ (continuing response when radiation exposure is terminated), and be stable in long-term storage.
-
The dosimeter should not require laborious processing, and it should be easy to convert the physical, chemical, or biological response to a measure of UV exposure dose.
-
The cost per dosimeter should be low so that large scale monitoring is feasible.
The most widely used personal UV dosimeter over the past 40 years has been the plastic film, polysulphone. The narrative that follows is told from the perspective of someone who was there at the beginning of the development of personal UV dosimeters and who has contributed to several studies on the measurement of human exposure to solar UV radiation throughout this period.
2. The Birth of Polysulphone as a Dosimeter for Measuring Personal Solar UV Exposure
While evaluating the weathering characteristics of the polymers polysulphone (PSP) and polyphenylene oxide (PPO), Tony Davis and colleagues, based at a UK defense establishment, found that they both darkened when exposed to UV radiation [
1]. They realized the potential of these polymers as UV monitors and used PPO to continuously monitor solar ultraviolet radiation at forty sites throughout the world [
2].
At a chance meeting in 1975, I suggested to Tony Davis that maybe PPO or PSP might be useful for monitoring personal exposure to UV, analogous to film badges worn by people exposed to ionizing radiation in hospital X-ray departments, for example. An important requirement for any UV dosimeter used to monitor biologically effective UV is that the wavelength response of the dosimeter should be similar to the action spectrum pertinent to the biological effect of interest, which is usually UV-induced erythema in human skin [
3].
PPO film shows spectral sensitivity up to 400 nm, unlike PSP, which is not responsive above about 340 nm. Additionally, because PSP is about 10 times more sensitive than PPO, it was more suited for use as a personal UV dosimeter. Therefore, we focused our attention on PSP film (40 μm thick) with a view to developing the material as a possible personal dosimeter to measure exposure of the skin to UV radiation.
When exposed to UV radiation, PSP film degrades and loses tensile properties. At the same time, the initial broad absorption band in the near UV extends with dose towards the visible. As a measure of the degree of degradation of the polymer, we took the change in absorbance pre- and post-exposure measured at 330 nm (∆A
330), as a measure of UV dose. This absorbance change was investigated as a function of incident UV exposure for wavelengths ranging from 254 nm to 335 nm [
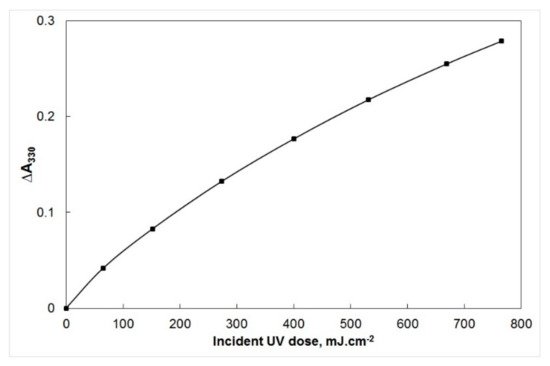
4]. The first dose-response curve, obtained for PSP film irradiation at 297 ± 5 nm, is shown in
Figure 1.
Figure 1. The first dose-response curve of polysulphone (PSP) film, showing the variation of ∆A
330 with incident ultraviolet exposure at a wavelength of 297 nm [
4].
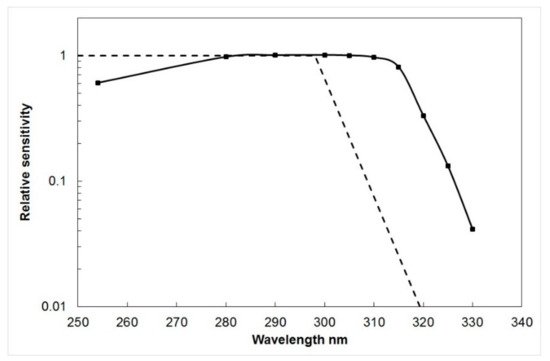
To derive the wavelength response of the film, the reciprocal of the dose required to produce a ∆A
330 of 0.1 at each wavelength was plotted as a function of wavelength, normalized to unity at the most sensitive wavelength (
Figure 2). In
Figure 2, the erythemal action spectrum of human skin [
3] is compared with the spectral sensitivity of polysulphone; and although the agreement is not ideal, it is possible to allow for this, if the relative spectral distribution of the incident radiation is known (see below).
Figure 2. The spectral sensitivity of PSP film (solid curve) [
4] and the erythema action spectrum (broken curve) [
3].
The absorbance of PSP film before and after exposure can be measured with any standard UV spectrophotometer and takes about 5–10 s per badge, depending on the dexterity of the operator. However, the task can be tedious, especially when several hundred badges have to be processed. To address this problem, a device based on a commercial slide projector was described [
5], that read and printed the absorbance of up to 60 badges at a time.
Polysulphone film largely meets the requirements for a personal dosimeter, as listed in the previous section, but there are some shortcomings. The spectral sensitivity is not ideal for a dosimeter designed to measure erythemal radiation (Figure 2); although this limitation can be accounted for, to some extent, in the calibration procedure (see below). The dynamic range is somewhat restricted as a result of saturation effects, necessitating the replacement of film badges on a daily to a weekly basis, depending on outdoor behavior of subjects.
Finally, PSP film does exhibit a dark reaction, in that the ∆A
330 measured immediately after exposure is about 8% less than that measured 24 h later, and 10% less than that measured one week later [
6]. However, this can be accounted for if the measurement was made at the same time after exposure, e.g., 24 h, that was used during calibration.
3. Calibration of Polysulphone Film as a UV Dosimeter
Because the spectral sensitivity of PSP film is not an exact match to the erythema action spectrum (
Figure 2), it is important that the badges are calibrated for each type of UV source to which they are exposed, ideally by spectroradiometric methods [
6].
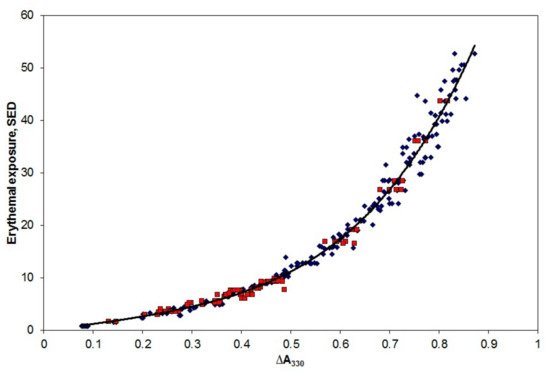
Most applications measure personal exposure to solar UV radiation, and Figure 3 shows the results of calibrating the film using Australian sunlight. Briefly, the spectral irradiance from 290 to 400 nm, in steps of 1 nm, was measured using a scanning spectroradiometer on a flat, unshaded surface every half hour throughout a day in spring in Queensland, and a summer’s day in Victoria. Pairs of PSP dosimeters, placed adjacent to the spectroradiometer, were exposed for varying combinations of time intervals during the day. By this means, it was possible to expose several pairs of dosimeters to approximately the same erythemally effective UV dose, but for differing time periods.
Figure 3. Exposure of PSP film badges to sunlight in Queensland (blue diamonds) and Victoria (red squares). The solid black curve is a quartic fit to the data.
The following day, the optical absorbance of each dosimeter at 330 nm was determined in a spectrophotometer, and the difference (∆A
330) between this post-exposure absorbance and the absorbance determined prior to the exposure was determined for each dosimeter. The erythemally effective dose received by a given film badge during a specified time interval was estimated from the spectral irradiance determined every 30 min, combined with the erythema action spectrum [
3]. The observed ∆A
330 for each film badge is plotted in
Figure 3.
The solid line is an equation of the form:
The erythemal dose is expressed in SED (Standard Erythema Dose), where 1 SED is equivalent to an erythemal effective radiant exposure of 100 Jm
−2 [
3].
The mismatch between the spectral response of PSP and the erythema action spectrum (
Figure 2) will lead to calibration errors that are a function of solar altitude and ozone amount. In principle, it is possible to correct such errors using the procedures described by Seckmeyer et al. [
7]. Without such corrections, however, it is important to determine the calibration curve under similar atmospheric conditions to the exposure of population groups [
8].
4. Using Polysulphone Film as a Personal UV Dosimeter
The very first PSP film badges were made by securing rectangles of the film, approximately 40 μm thick, into standard 35 mm transparency mounts, resulting in a film badge of 50 × 50 mm with an open aperture of 25 × 35 mm (Figure 4a). These mounts were slotted into specially constructed metal holders which were fastened by a safety pin to the lapel site of volunteers as a proxy for facial solar UV exposure. By comparison, personal ionizing radiation monitors utilizing photographic film are normally worn on the torso to give an indication of whole-body dose.
Figure 4. Three different PSP film badge holders.
These film badges were not especially liked by volunteers due to their prominent size and so were soon replaced with a cardboard holder measuring 30 mm × 30 mm, with the film mounted in a central aperture of 10 mm × 15 mm (
Figure 4b). With this single aperture badge, only a small fraction of the exposed surface area is used for spectroradiometry. In order to increase reliability, Gibbs et al. [
9] described a film badge of the same overall dimension incorporating four circular apertures, each of 6 mm diameter (
Figure 4c).
However, these four-aperture mounts never became popular due to the increased time required to measure the pre- and post-exposure absorbances, and so for the vast majority of studies of personal exposure reported in the literature, the single aperture mount (Figure 4b) has been the film badge of choice.
This entry is adapted from the peer-reviewed paper 10.3390/atmos11020125