Marine sponges were among the first multicellular organisms on our planet and have survived to this day thanks to their unique mechanisms of chemical defense and the specific design of their skeletons, which have been optimized over millions of years of evolution to effectively inhabit the aquatic environment. Ianthella basta (Pallas, 1766) is one of the largest, fan-shaped marine demosponges that can reach up to 2.5 m both in height; and the body of which is a micro-reticular, durable structure that determines the ideal filtration function of this organism. Calcite biomineral is responsible for nano-tuning the chitinous skeletal fibers of this sponge species.
- biological materials
- demospongiae
- biomineralization
- calcite
- chitin
- Micro X-ray Fluorescence
- NEXAFS
1. Introduction

Figure 1. Marine verongiid demosponge Ianthella basta possess a huge chitin-based, a network-like skeleton that is stiff enough to withstand underwater currents due to its natural rigidity.
2. Identification of Ca and Br Localization Using Three-Dimensional Confocal Micro X-ray Fluorescence (CµXRF)

Figure 2. CµXRF analysis results of selected I. basta skeletal samples after treatment with distilled water (a), 3 M HCl (b), and 10% NaOH at 40 °C (c). The 3D distribution of Ca (red) and Br (green) is illustrated in the figure for an analyzed sample volume of 1.5 × 1.5 × 1.5 mm3 (and locations with both elements are shown in a yellow coloration). By HCl treatment the Ca is removed from the skeletal structure (b), whereas by NaOH treatment both elements, Ca and Br, remain within the demosponge, however thin proteinaceous tissue layers between the sponge’s skeleton are dissolved (c) and characteristic 3D chitinous construct remains present [14]. In figures (d,e) the sponge structure of (a,b) is depicted without the above-mentioned thin tissue layers, which was obtained by omitting the regions with very low signal intensity from data processing.

Figure 3. SEM images of the selected fragment treated with a distilled fragment of I. basta sponge skeletal fiber (a) show the presence of sphere-like nanoparticles (b–d).
3. Visualization of the Presence of Mineral Phase Using Electron Microscopy Methods
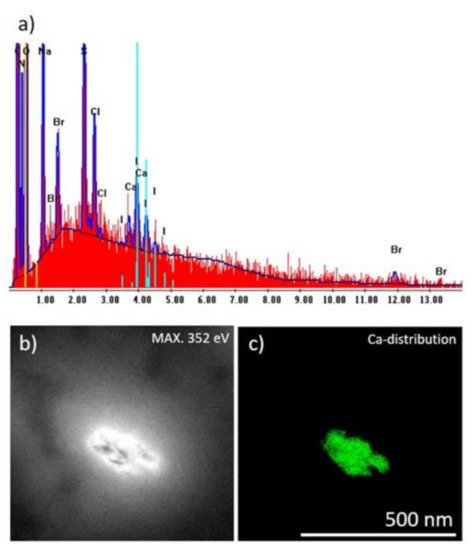
Figure 4. (a) EDS analysis of the I. basta skeletal fiber washed with distilled water demonstrating the atomic composition of this structure; (b) bright-field image of corresponding TEM image and (c) Ca distribution within the corresponding sample visualized in green color (colorization provided by the software to highlight electron diffraction signals of Ca atoms).

Figure 5. (a) low-resolution TEM image of the selected micro-fragment of the I. basta skeletal fiber-containing nanoparticles and (b) high-resolution TEM images showing their crystalline nature.
4. Identification of Calcite within I. basta Skeletal Fibres
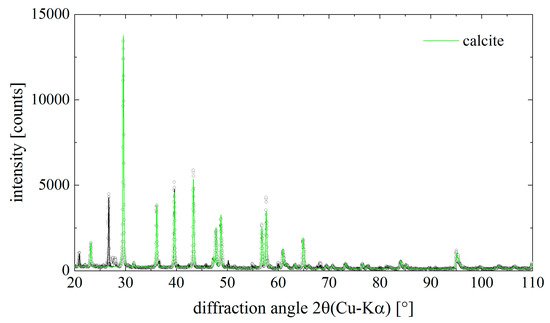
Figure 6. Diffraction pattern of the mineral phase isolated from chitinous skeletal fibers of I. basta demosponge. Dots are measured data points, the solid lines result from a Rietveld- (Pawley)-fit of the data. The contribution of calcite to the total diffraction pattern is highlighted in green color. Calcite is the main component in the sample.
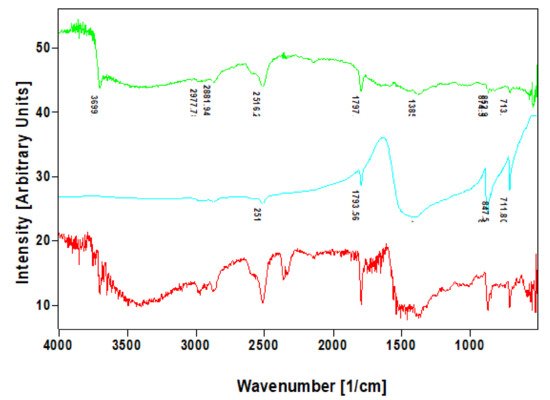
Figure 7. Comparative FTIR spectra of calcite standard (blue line) with that of nanocrystalline phases isolated from skeletal fibers of I. basta demosponges collected near Guam (USA) (red line) and in coastal waters of Australia (green line).
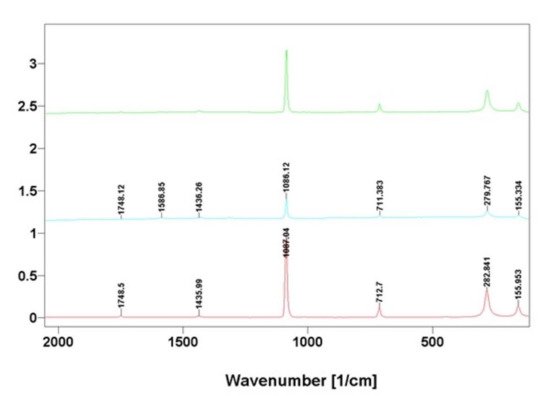
Figure 8. Comparative Raman spectra of calcite standard (blue line) with that of nanocrystalline phases isolated from skeletal fibers of I. basta demosponges collected near Guam (USA) (red line) and in coastal waters of Australia (green line).
5. Near Edge X-ray Fine Structure (NEXAFS) Features of Identified Mineral Phase

Figure 9. NEXAFS C1s (left) and Ca2p (right) spectra of I. basta skeletal fibers before and after NaOH treatment in comparison with calcite standard.
This entry is adapted from the peer-reviewed paper 10.3390/ijms222212588
References
- Ehrlich, H.; Bailey, E.; Wysokowski, M.; Jesionowski, T. Forced Biomineralization: A Review. Biomimetics 2021, 6, 46.
- Ehrlich, H.; Brunner, E.; Simon, P.; Bazhenov, V.V.; Botting, J.P.; Tabachnick, K.R.; Springer, A.; Kummer, K.; Vyalikh, D.V.; Molodtsov, S.L.; et al. Calcite Reinforced Silica-Silica Joints in the Biocomposite Skeleton of Deep-Sea Glass Sponges. Adv. Funct. Mater. 2011, 21, 3473–3481.
- Ehrlich, H.; Simon, P.; Carrillo-Cabrera, W.; Bazhenov, V.V.; Botting, J.P.; Ilan, M.; Ereskovsky, A.V.; Muricy, G.; Worch, H.; Mensch, A.; et al. Insights into Chemistry of Biological Materials: Newly Discovered Silica-Aragonite-Chitin Biocomposites in Demosponges. Chem. Mater. 2010, 22, 1462–1471.
- Sethmann, I.; Wörheide, G. Structure and Composition of Calcareous Sponge Spicules: A Review and Comparison to Structurally Related Biominerals. Micron 2008, 39, 209–228.
- Voigt, O.; Adamska, M.; Adamski, M.; Kittelmann, A.; Wencker, L.; Wörheide, G. Spicule Formation in Calcareous Sponges: Coordinated Expression of Biomineralization Genes and Spicule-Type Specific Genes. Sci. Rep. 2017, 7, 45658.
- Shimizu, K.; Cha, J.; Stucky, G.D.; Morse, D.E. Silicatein Alpha: Cathepsin L-like Protein in Sponge Biosilica. Proc. Natl. Acad. Sci. USA 1998, 95, 6234–6238.
- Shimizu, K.; Amano, T.; Bari, M.R.; Weaver, J.C.; Arima, J.; Mori, N. Glassin, a Histidine-Rich Protein from the Siliceous Skeletal System of the Marine Sponge Euplectella, Directs Silica Polycondensation. Proc. Natl. Acad. Sci. USA 2015, 112, 11449–11454.
- Povarova, N.V.; Barinov, N.A.; Baranov, M.S.; Markina, N.M.; Varizhuk, A.M.; Pozmogova, G.E.; Klinov, D.V.; Kozhemyako, V.B.; Lukyanov, K.A. Efficient Silica Synthesis from Tetra(Glycerol)Orthosilicate with Cathepsin- and Silicatein-like Proteins. Sci. Rep. 2018, 8, 16759.
- Ehrlich, H. Chitin and collagen as universal and alternative templates in biomineralization. Int. Geol. Rev. 2010, 52, 661–699.
- Ehrlich, H.; Krautter, M.; Hanke, T.; Simon, P.; Knieb, C.; Heinemann, S.; Worch, H. First Evidence of the Presence of Chitin in Skeletons of Marine Sponges. Part II. Glass Sponges (Hexactinellida: Porifera). J. Exp. Zool. B Mol. Dev. Evol. 2007, 308, 473–483.
- Wysokowski, M.; Jesionowski, T.; Ehrlich, H. Biosilica as a Source for Inspiration in Biological Materials Science. Am. Mineral. 2018, 103, 665–691.
- Tsurkan, D.; Simon, P.; Schimpf, C.; Motylenko, M.; Rafaja, D.; Roth, F.; Inosov, D.S.; Makarova, A.A.; Stepniak, I.; Petrenko, I.; et al. Extreme Biomimetics: Designing of the First Nanostructured 3D Spongin–Atacamite Composite and Its Application. Adv. Mater. 2021, 33, 2101682.
- Muzychka, L.; Voronkina, A.; Kovalchuk, V.; Smolii, O.B.; Wysokowski, M.; Petrenko, I.; Youssef, D.T.A.; Ehrlich, I.; Ehrlich, H. Marine Biomimetics: Bromotyrosines Loaded Chitinous Skeleton as Source of Antibacterial Agents. Appl. Phys. A 2021, 127, 15.
- Brunner, E.; Ehrlich, H.; Schupp, P.; Hedrich, R.; Hunoldt, S.; Kammer, M.; Machill, S.; Paasch, S.; Bazhenov, V.V.; Kurek, D.V.; et al. Chitin-Based Scaffolds Are an Integral Part of the Skeleton of the Marine Demosponge Ianthella basta. J. Struct. Biol. 2009, 168, 539–547.
- Sun, J.; Wu, Z.; Cheng, H.; Zhang, Z.; Frost, R.L. A Raman Spectroscopic Comparison of Calcite and Dolomite. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 117, 158–162.
- Li, H.; Sun, C.-Y.; Fang, Y.; Carlson, C.M.; Xu, H.; Ješovnik, A.; Sosa-Calvo, J.; Zarnowski, R.; Bechtel, H.A.; Fournelle, J.H.; et al. Biomineral Armor in Leaf-Cutter Ants. Nat. Commun. 2020, 11, 5792.
- Falini, G.; Fermani, S.; Gazzano, M.; Ripamonti, A. Structure and Morphology of Synthetic Magnesium Calcite. J. Mater. Chem. 1998, 8, 1061–1065.
- Urmos, J.; Sharma, S.K.; Mackenzie, F.T. Characterization of Some Biogenic Carbonates with Raman Spectroscopy. Am. Mineral. 1991, 76, 641–646.
- De La Pierre, M.; Carteret, C.; Maschio, L.; André, E.; Orlando, R.; Dovesi, R. The Raman Spectrum of CaCO3 Polymorphs Calcite and Aragonite: A Combined Experimental and Computational Study. J. Chem. Phys. 2014, 140, 164509.
- Stöhr, J. NEXAFS Spectroscopy; Springer: Berlin/Heidelberg, Germany, 1992.
- Benzerara, K.; Yoon, T.H.; Tyliszczak, T.; Constantz, B.; Spormann, A.M.; Brown, G.E. Scanning Transmission X-ray Microscopy Study of Microbial Calcification. Geobiology 2004, 2, 249–259.
- Benzerara, K.; Menguy, N.; López-García, P.; Yoon, T.-H.; Kazmierczak, J.; Tyliszczak, T.; Guyot, F.; Brown, G.E., Jr. Nanoscale Detection of Organic Signatures in Carbonate Microbialites. Proc. Natl. Acad. Sci. USA 2006, 103, 9440–9445.
- Madix, R.J.; Solomon, J.L.; Stöhr, J. The Orientation of the Carbonate Anion on Ag(110). Surf. Sci. 1988, 197, L253–L259.
