Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Engineering, Electrical & Electronic
There are two different ways of preparing nanofluids categorized in a one-step process and a two-step process. The origin of the word “nanofluid” is connected with the work by Choi et al. back in 1995, where they researched enhancement of thermal conductivity of fluids mixed with nanoparticles, determined the direction of further research, described the theoretical study of these fluids, and set potential benefits of nanofluids. From that point, the number of scientific papers that deal with nanofluids has been exponentially increasing.
- nanofluid
1. One-Step Method
In a one-step process of preparation of nanofluids, the formation and dispersion of nanoparticles are simultaneous [1]. This method avoids the process of transportation, storage, drying, and dispersion of nanoparticles to decrease a measure of agglomeration of nanoparticles and to increase the stability of nanofluids [2][3][4]. One of the one-step preparation methods is a vapor deposition. This method was patented by Choi and Eastman in 2001 [5]. The principle consists in the formation of a thin layer of a base fluid on a vessel wall caused by a centrifugal force of a rotating disk. The material is then heated and evaporated in the vessel filled with an inert gas at a low pressure. The creation of nanofluid is finished when the raw material vapors condense by interacting with the thin film of swirling water and finally settle in the base fluid [1][5]. The second possible one-step method is the laser ablation where a highly concentrated laser beam is used for dispersion of nanoparticles from the surface of a material immersed in a base fluid. Important properties are the intensity and the wavelength of the laser beam [6][7]. There are more possible preparation methods using the one-step process such as the submerged arc method, precipitation (ion exchange) method, chemical reduction method, emulsion polymerization, sol-gel (hydrolysis) method, or microwave-assisted reaction [1][8][9]. The submerged arc nanoparticle synthesis system is an efficient one-step method to prepare nanofluids based on dielectric liquids containing copper nanoparticles [10][11].
2. Two-Step Method
In the first phase of the two-step method, nanoparticles (nanorods, nanofibers, or nanotubes) are first prepared by sol-gel method, hydrothermal synthesis, or by other techniques [1]. The sol-gel method is used for nanoparticles with high surface area and provides effective control over the texture and surface properties of nanoparticles. There are five main steps of this method starting with hydrolysis and then polycondensation, ageing, drying, and thermal decomposition [1][9][12]. According to [13][14], hydrothermal synthesis refers to the “heterogeneous reactions for synthesizing inorganic materials in aqueous media above ambient temperature and pressure” and the advantage of this method is the low energy consumption, the low-temperature processes, and the environmental impact. However, the high price of the needed autoclaves may be considered as a potential drawback [1]. The second step of the two-step method is the preparation of nanofluids using mostly ultrasonication (the bath and probe ultrasonication are of remarkably different effectivity), magnetic stirring, adjusting pH value (addition of a dispersant in Figure 3), or a combination of these processes [1]. Ultrasonication enhances performance, stability, thermophysical properties and prevents aggregation and sedimentation of nanoparticles in nanofluid, but the ideal duration of sonication is the point of research of many authors [15][16][17][18][19]. Magnetic stirring is used for dispersion of nanoparticles with a low concentration by the stirring action made by a stir bar that spins very quickly because of a rotating magnetic field created by the set of rotating magnets or electromagnets [1][20][21].
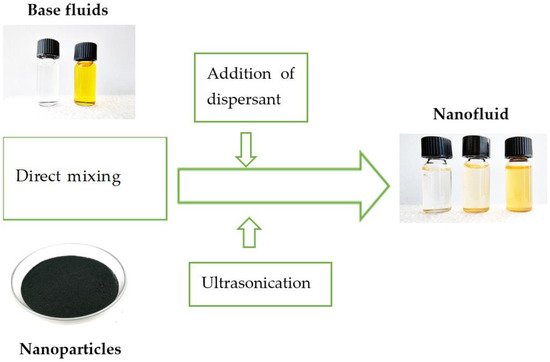
Figure 3. A simple illustration of the two-step method of preparing nanofluids. Inset photographs are from the authors archive.
Selecting a particular method, additives and timing within the applied methods are very important to make a nanofluid with excellent properties without sedimentation and agglomeration of nanoparticles. A more accurate description of preparation methods of eleven different nanoparticles is in the reference [22].
To compare the two preparation methods, one can state that the two-step method is more economic in production of nanofluids in a larger scale and it is used most widely. However, without modifying the nanoparticle surface, the tendency to agglomerate before adding to the base liquid is considered as a major disadvantage of the two-step method. On the other hand, the one-step method cannot be effectively used to synthesize nanofluids in a large scale, but this method can yield uniformly and stably dispersed nanoparticles.
3. Stability of Nanofluids
The stability of nanofluids is of crucial importance because the sedimentation and agglomeration of nanoparticles in the base fluid cause deterioration of thermophysical properties, mainly a decrease of thermal conductivity and increase of viscosity [23][24][25]. Because of these facts, examination of stability is an important part of research and there are several methods to analyze the stability of synthesized nanofluids. The most applied methods to study the stability are a zeta potential test, a sedimentation method (photograph capturing method), ultraviolet-visible spectrophotometer and dynamic light scattering [25][26].
3.1. Zeta Potential Method
The Zeta potential (ζ-potential) method is the most used method to examine the stability of nanofluids. It can be defined as the potential difference between the stationary layer of base fluid which is attached to nanoparticles and the surface of nanoparticles [23]. It indicates the degree of repulsion between charged particles in the fluid and it can be calculated by the Helmholtz–Smoluchowski equation [27][28]:
where U is electrophoretic mobility, µ is viscosity, and ε is the dielectric constant of the base liquid. If the value of Zeta potential is over 60 mV, a nanofluid has an excellent stability, between 60 and 40 mV it has a good stability, between 40 and 30 mV the nanofluid is considered as stable, and below 30 mV it is considered as highly agglomerative [26][29][30]. Because of repulsive forces, the Zeta potential can be controlled over pH value [31][32]. A change of pH influences the surface charge on nanoparticles and modifies their interaction behavior [26]. If the pH of the nanofluid has low values, the Zeta potential will be positive. On the other hand, with higher pH values, the Zeta potential will be in negative values. The point when a pH value corresponds to zero Zeta potential is called the isoelectric point, when nanofluids are least stable, so stability rises in the positive or negative direction from that point [26]. Measurements of the Zeta potential are the most often performed by a Zeta Sizer Nano (ZSN) device [29][33][34].
ζ = μU/ε
3.2. Sedimentation Method (Photograph Capturing Method)
The sedimentation method is the simplest option of measuring the stability of nanofluids qualitatively, by observing photographs taken in different periods [29][35]. Owing to external forces (gravitation), the nanoparticles settle on the bottom of the fluid in a clear glass test tubes that can be observed by comparing photographs taken at different times [36]. In an unstable nanofluid there are three ways of sedimentation. The first one is a dispersed sedimentation where the height of a sediment rises from the bottom. The second one is a flocculated sedimentation where the sedimentation is lowered with time, and the last one is a mixed sedimentation where the behavior of both previous cases is observed at the same time [29][36][37]. The most significant impact on sedimentation has the concentration of nanoparticles and properties of the base fluid [38][39].
3.3. Ultraviolet-Visible Spectrophotometer
The ultraviolet-visible spectrophotometer is commonly used for the quantitative characterization of the colloidal stability of the dispersions [25]. One of the major advantages of this method is its suitability for all base fluids because its functioning is about the intensity of the light that becomes different because of lights scattering and absorption when passing through the fluid [26][30][40]. According to [26][40] and [41], the range of UV–visible spectrophotometer is from 200 to 900 nm wavelengths and basically, it measures various dispersions in the fluid. The stability is determined by the dispersion of nanoparticles in different time results [42].
3.4. Dynamic Light Scattering
Dynamic light scattering is a suitable method for measuring mainly spherical particles and the most significant advantage is that this method does not need drying of the dispersion (some dispersants are difficult to remove) [43][44]. A simple description of this method is that a source of monochromatic light shines on the sample and a detector collects the scattered light signals [41][45]. There is a need to know the refractive index and viscosity of a measured base liquid, and the measurement output is a signal that shows random changes due to the randomly changing relative position of the particles due to the random Brownian motion. Size as the final output is calculated by the Stokes–Einstein equation [30][31].
4. Structural Characterization of Nanofluids
The essential parameters determining the physical properties of nanofluids are the nanoparticle size distribution, morphology, crystal structure, and elemental composition. Several techniques can be used to characterize nanoparticles from these points of view. In this chapter we mention just a few techniques.
4.1. Transmission Electron Microscopy (TEM) and Scanning Electron Microscopy (SEM)
These methods have evolved over many years into a highly sophisticated instrument and have found different applications across many scientific disciplines, because of their excellent ability to distinguish the shape, size, and distribution of nanoparticles [46][47][48]. In [26], the methodology of transmission electron microscopy is described as: “the electrons shoot through the sample and measures how the electron beam changes as it is scattered in the sample. Scanning electron microscope images the sample surface by scanning it with electron beams in a raster scan pattern. The electrons interact with the sample atoms producing signals that contain information about the sample’s surface topography, composition and other properties”. The disadvantage of this method is that it does not capture the real situation of nanoparticles in nanofluids because there is a need for dried samples prepared in a vacuum oven [30][49].
4.2. X-ray-Based Techniques
One of the most extensively applied methods for nanoparticle characterization is X-ray diffraction (XRD) [50]. It provides information on the crystal structure, nature of the phase, crystalline grain size and lattice parameters. For this method, the nanoparticles in powder form are commonly used after drying the colloidal solution. XRD provides statistically representative, volume averaged values. For instance, this method has been applied to determine the average crystallite size of magnetite nanoparticles [51]. Another X-ray-based analytical method to determine the structure of nanoparticles in terms of averaged size or shapes is small angle X-ray scattering (SAXS) [52]. Normally, a transmission mode is used, when the X-ray beam is sent through the nanofluid sample and the average structure of all illuminated particles is measured.
4.3. Neutron Scattering Techniques
Analogously to the SAXS method, small angle neutron scattering (SANS) is often used to study the structure of nanofluids, in terms of nanoparticle size and shape distribution, but also to study assembly and alignment of nanoparticles [53]. Among the advantages of neutrons, one can highlight their larger penetration depth and an option of using contrast variation. In this way, different parts of a sample can be selectively viewed via isotopic labelling. This method has been found especially useful in structure research of magnetic nanofluids [54].
4.4. Atomic Force Microscopy
Atomic force microscopy (AFM) is a technique capable of providing three-dimensional images of surfaces. It measures the interacting forces between a fine probe and the sample. In this way, individual particles and groups of particles can be resolved and shape and size distribution of nanoparticles can be obtained [55]. AFM can scan the sample under different modes depending on the degree of proximity between the probe and the sample (contact, non-contact, and tapping mode). The tapping mode is the most common when characterizing nanoparticles [56].
Clearly, in order to get the complete picture of an unknown sample, which allows extraction of proper correlations between the structure and the improvement of the base liquid properties, one needs to employ various methods, because their results are complementary.
This entry is adapted from the peer-reviewed paper 10.3390/nano11112885
References
- Ali, H.M.; Babar, H.; Shah, T.R.; Sajid, M.U.; Qasim, M.A.; Javed, S. Preparation Techniques of TiO2 Nanofluids and Challenges: A Review. Appl. Sci. 2018, 8, 587.
- Mukherjee, S.; Paria, S. Preparation and Stability of Nanofluids-A Review. IOSR J. Mech. Civ. Eng. (IOSR-JMCE) 2013, 9, 63–69.
- Yu, W.; Xie, H. A Review on Nanofluids: Preparation, Stability Mechanisms, and Applications. J. Nanomater. 2012, 2012, 1–17.
- Li, Y.; Zhou, J.; Tung, S.; Schneider, E.; Xi, S. A Review on Development of Nanofluid Preparation and Characterization. Powder Technol. 2009, 196, 89–101.
- Choi, S.U.S.; Eastman, J.A. Enhanced Heat Transfer Using Nanofluids; Argonne National Lab. (ANL): Argonne, IL, USA, 2001.
- Huang, X.; Zhang, W. Study on Successively Preparation of Nano-TiO2 Ethanol Colloids by Pulsed Laser Ablation and Fluorescence Property. Appl. Surf. Sci. 2008, 254, 3403–3407.
- Liu, P.; Cai, W.; Wan, L.; Shi, M.; Luo, X.; Jing, W. Fabrication and Characteristics of Rutile TiO2 Nanoparticles Induced by Laser Ablation. Trans. Nonferrous Met. Soc. China 2009, 19, s743–s747.
- Ma, B.; Banerjee, D. A Review of Nanofluid Synthesis. In Advances in Nanomaterials; Balasubramanian, G., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 135–176. ISBN 978-3-319-64715-9.
- Parashar, M.; Shukla, V.K.; Singh, R. Metal Oxides Nanoparticles via Sol–Gel Method: A Review on Synthesis, Characterization and Applications. J. Mater. Sci. Mater. Electron. 2020, 31, 3729–3749.
- Lo, C.-H.; Tsung, T.-T.; Chen, L.-C. Shape-Controlled Synthesis of Cu-Based Nanofluid Using Submerged Arc Nanoparticle Synthesis System (SANSS). J. Cryst. Growth 2005, 277, 636–642.
- Lo, C.-H.; Tsung, T.-T.; Chen, L.-C.; Su, C.-H.; Lin, H.-M. Fabrication of Copper Oxide Nanofluid Using Submerged Arc Nanoparticle Synthesis System (SANSS). J. Nanopart. Res. 2005, 7, 313–320.
- Isaev, A.B.; Orudjev, F.F.; Shabanov, N.S.; Magomedova, G.A.; Gasanova, F.G.; Khizrieva, I.K. Synthesis and Studies of Photocatalytic Activity of Composite Based on Nanodispersed TiO2 and SiO2. Nanotechnol. Russ. 2015, 10, 357–361.
- Huang, G.; Lu, C.-H.; Yang, H.-H. Chapter 3—Magnetic Nanomaterials for Magnetic Bioanalysis. In Novel Nanomaterials for Biomedical, Environmental and Energy Applications; Wang, X., Chen, X., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 89–109. ISBN 978-0-12-814497-8.
- Rabenau, A. The Role of Hydrothermal Synthesis in Preparative Chemistry. Angew. Chem. Int. Ed. Engl. 1985, 24, 1026–1040.
- Adio, S.A.; Sharifpur, M.; Meyer, J.P. Influence of Ultrasonication Energy on the Dispersion Consistency of Al2O3–Glycerol Nanofluid Based on Viscosity Data, and Model Development for the Required Ultrasonication Energy Density. J. Exp. Nanosci. 2016, 11, 630–649.
- Afzal, A.; Nawfal, I.; Mahbubul, I.M.; Kumbar, S.S. An Overview on the Effect of Ultrasonication Duration on Different Properties of Nanofluids. J. Anal. Calorim. 2019, 135, 393–418.
- Asadi, A.; Alarifi, I.M. Effects of Ultrasonication Time on Stability, Dynamic Viscosity, and Pumping Power Management of MWCNT-Water Nanofluid: An Experimental Study. Sci. Rep. 2020, 10, 15182.
- Mahbubul, I.M.; Saidur, R.; Amalina, M.A.; Elcioglu, E.B.; Okutucu-Ozyurt, T. Effective Ultrasonication Process for Better Colloidal Dispersion of Nanofluid. Ultrason. Sonochemistry 2015, 26, 361–369.
- Sandhya, M.; Ramasamy, D.; Sudhakar, K.; Kadirgama, K.; Harun, W.S.W. Ultrasonication an Intensifying Tool for Preparation of Stable Nanofluids and Study the Time Influence on Distinct Properties of Graphene Nanofluids—A Systematic Overview. Ultrason. Sonochemistry 2021, 73, 105479.
- Chaudhari, K.; Walke, P.; Wankhede, U.; Shelke, R. An Experimental Investigation of a Nanofluid (Al2O3 + H2O) Based Parabolic Trough Solar Collectors. Br. J. Appl. Sci. Technol. 2015, 9, 551–557.
- Naiman, I.; Devarajan, R.; Kadirgama, K. Experimental and One Dimensional Investigation on Nanocellulose and Aluminium Oxide Hybrid Nanofluid as a New Coolant for Radiator. IOP Conf. Ser. Mater. Sci. Eng. 2019, 469, 012096.
- Sidik, N.A.C.; Mohammed, H.A.; Alawi, O.A.; Samion, S. A Review on Preparation Methods and Challenges of Nanofluids. Int. Commun. Heat Mass Transf. 2014, 54, 115–125.
- Che Sidik, N.A.; Mahmud Jamil, M.; Aziz Japar, W.M.A.; Muhammad Adamu, I. A Review on Preparation Methods, Stability and Applications of Hybrid Nanofluids. Renew. Sustain. Energy Rev. 2017, 80, 1112–1122.
- Rafiq, M.; Shafique, M.; Azam, A.; Ateeq, M. Transformer Oil-Based Nanofluid: The Application of Nanomaterials on Thermal, Electrical and Physicochemical Properties of Liquid Insulation—A Review. Ain. Shams Eng. J. 2021, 12, 555–576.
- Sajid, M.U.; Ali, H.M. Thermal Conductivity of Hybrid Nanofluids: A Critical Review. Int. J. Heat Mass Transf. 2018, 126, 211–234.
- Setia, H.; Gupta, R.; Wanchoo, R.K. Stability of Nanofluids. MSF 2013, 757, 139–149.
- Hwang, Y.; Lee, J.-K.; Lee, J.-K.; Jeong, Y.-M.; Cheong, S.; Ahn, Y.-C.; Kim, S.H. Production and Dispersion Stability of Nanoparticles in Nanofluids. Powder Technol. 2008, 186, 145–153.
- Munson, B.R.; Young, D.F.; Okiishi, T.H. Fundamentals of Fluid Mechanics, 3rd ed.; John Wiley & Sons: New Jersey, NJ, USA, 1998; Chapter 3.
- Ali, N.; Teixeira, J.A.; Addali, A. A Review on Nanofluids: Fabrication, Stability, and Thermophysical Properties. J. Nanomater. 2018, 2018, 6978130.
- Ghadimi, A.; Saidur, R.; Metselaar, H.S.C. A Review of Nanofluid Stability Properties and Characterization in Stationary Conditions. Int. J. Heat Mass Transf. 2011, 54, 4051–4068.
- Wang, X.-J.; Li, X.-F.; Wang, N.; Wen, X.-Y.; Long, Q. Influence of SDBS on Stability of Al2O3 Nano-Suspensions. In Nanophotonics, Nanostructure, and Nanometrology II, Proceedings of the Photonics Asia 2007, Beijing, China, 12–14 November 2007; Zhu, X., Chou, S.Y., Arakawa, Y., Eds.; SPIE: Beijing, China, 2008; Volume 6831, p. 683113.
- Jiang, L.; Gao, L.; Sun, J. Production of Aqueous Colloidal Dispersions of Carbon Nanotubes. J. Colloid Interface Sci. 2003, 260, 89–94.
- Mondragon, R.; Julia, J.E.; Barba, A.; Jarque, J.C. Characterization of Silica–Water Nanofluids Dispersed with an Ultrasound Probe: A Study of Their Physical Properties and Stability. Powder Technol. 2012, 224, 138–146.
- Chang, H.; Jwo, C.S.; Fan, P.S.; Pai, S.H. Process Optimization and Material Properties for Nanofluid Manufacturing. Int. J. Adv Manuf. Technol. 2007, 34, 300–306.
- Wei, X.; Zhu, H.; Kong, T.; Wang, L. Synthesis and Thermal Conductivity of Cu2O Nanofluids. Int. J. Heat Mass Transf. 2009, 52, 4371–4374.
- Ali, N.; Teixeira, J.A.; Addali, A. Aluminium Nanofluids Stability: A Comparison between the Conventional Two-Step Fabrication Approach and the Controlled Sonication Bath Temperature Method. J. Nanomater. 2019, 2019, e3930572.
- Ilyas, S.U.; Pendyala, R.; Marneni, N. Settling Characteristics of Alumina Nanoparticles in Ethanol-Water Mixtures. AMM 2013, 372, 143–148.
- Ilyas, S.U.; Pendyala, R.; Marneni, N. Stability and Agglomeration of Alumina Nanoparticles in Ethanol-Water Mixtures. Procedia Eng. 2016, 148, 290–297.
- Ilyas, S.U.; Pendyala, R.; Marneni, N. Preparation, Sedimentation, and Agglomeration of Nanofluids. Chem. Eng. Technol. 2014, 37, 2011–2021.
- Lee, K.; Hwang, Y.; Cheong, S.; Kwon, L.; Kim, S.; Lee, J. Performance Evaluation of Nano-Lubricants of Fullerene Nanoparticles in Refrigeration Mineral Oil. Curr. Appl. Phys. 2009, 9, e128–e131.
- Ferreira, L.M.B.; Kiill, C.P.; Pedreiro, L.N.; Santos, A.M.; Gremião, M.P.D. Chapter 5—Supramolecular design of hydrophobic and hydrophilic polymeric nanoparticles. In Design and Development of New Nanocarriers; Grumezescu, A.M., Ed.; William Andrew Publishing: Norwich, NY, USA, 2018; pp. 181–221. ISBN 978-0-12-813627-0.
- Mansour, D.A.; Atiya, E.G. Application of UV/Vis Spectroscopy to Assess the Stability of Oil-Based Nanofluids. In Proceedings of the 2016 IEEE Conference on Electrical Insulation and Dielectric Phenomena (CEIDP), Toronto, ON, Canada, 16–19 October 2016; pp. 671–674.
- Amaro-Gahete, J.; Benítez, A.; Otero, R.; Esquivel, D.; Jiménez-Sanchidrián, C.; Morales, J.; Caballero, Á.; Romero-Salguero, F. A Comparative Study of Particle Size Distribution of Graphene Nanosheets Synthesized by an Ultrasound-Assisted Method. Nanomaterials 2019, 9, 152.
- Badaire, S.; Poulin, P.; Maugey, M.; Zakri, C. In Situ Measurements of Nanotube Dimensions in Suspensions by Depolarized Dynamic Light Scattering. Langmuir 2004, 20, 10367–10370.
- Schärtl, W. Light Scattering from Polymer Solutions and Nanoparticle Dispersions; Springer Laboratory; Springer: Berlin/Heidelberg, Germany, 2007; ISBN 978-3-540-71950-2.
- Smith, D.J. Chapter 1 Characterization of Nanomaterials Using Transmission Electron Microscopy. In Nanocharacterisation: Edition 2; The Royal Society of Chemistry: Cambridge, UK, 2015; pp. 1–29.
- Vladár, A.E.; Hodoroaba, V.-D. Chapter 2.1.1—Characterization of nanoparticles by scanning electron microscopy. In Characterization of Nanoparticles; Hodoroaba, V.-D., Unger, W.E.S., Shard, A.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 7–27. ISBN 978-0-12-814182-3.
- Majeed, N.; Abdulmajeed, B.; Yaseen, A. The Influence of the Preparation and Stability of Nanofluids for Heat Transfer. J. Eng. 2019, 25, 45–54.
- Liu, M.S.; Lin, M.C.C.; Tsai, C.Y.; Wang, C.-C. Enhancement of Thermal Conductivity with Cu for Nanofluids Using Chemical Reduction Method. Int. J. Heat Mass Transf. 2006, 49, 3028–3033.
- Mourdikoudis, S.; Pallares, R.; Thanh, N. Characterization Techniques for Nanoparticles: Comparison and Complementarity upon Studying Nanoparticle Properties. Nanoscale 2018, 10, 12871–12934.
- Upadhyay, S.; Parekh, K.; Pandey, B. Influence of Crystallite Size on the Magnetic Properties of Fe3O4 Nanoparticles. J. Alloys Compd. 2016, 678, 478–485.
- Li, T.; Senesi, A.; Lee, B. Small Angle X-ray Scattering for Nanoparticle Research. Chem. Rev. 2016, 116, 11128–11180.
- Hollamby, M. Practical Applications of Small-Angle Neutron Scattering. Phys. Chem. Chem. Phys. 2013, 15, 10566–10579.
- Avdeev, M.V.; Aksenov, V.L. Small-Angle Neutron Scattering in Structure Research of Magnetic Fluids. Physics-Uspekhi 2010, 53, 971.
- Rao, A.; Schoenenberger, M.; Gnecco, E.; Glatzel, T.; Meyer, E.; Brändlin, D.; Scandella, L. Characterization of Nanoparticles Using Atomic Force Microscopy. J. Phys. Conf. Ser. 2007, 61, 971–976.
- Mechler, A.; Kopniczky, J.; Kokavecz, J.; Hoel, A.; Granqvist, C.; Heszler, P. Anomalies in Nanostructure Size Measurements by AFM. Phys. Rev. B 2005, 72, 125407.
This entry is offline, you can click here to edit this entry!
