Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Ophthalmology
Age-related macular degeneration (AMD) is central vision loss with aging, was the fourth main cause of blindness in 2015, and has many risk factors, such as cataract surgery, cigarette smoking, family history, hypertension, obesity, long-term smart device usage, etc. In general, AMD drug candidates from natural products are more effective at treating early and intermediate AMD.
- age-related macular degeneration (AMD)
- medication
- anti-inflammatory drugs
- natural products
1. Introduction
1.1. Definition
Age-related macular degeneration (AMD) is related to aging and is a disease that causes problems in the central region of the retina, also known as the macula [1]. AMD patients experience several physiological changes, such as the loss of central vision, drusen, retinal pigmentary changes, angiogenesis, etc., and according to angiogenesis in the retina, it can be classified as dry AMD or wet AMD, with the percentage of wet AMD cases reported to be 10–15% of the total AMD cases [2].
1.2. Health Problems, Statistics, and Risk Factors
In 2015, AMD was the fourth most common cause of blindness, and among patients with moderate to severe vision impairment, it was the third most common cause [3]. Among those aged 80 years and above, 66% have several signs of AMD [4][5], and 3% of those over 65 years old suffer visual problems caused by AMD [6]. According to Zou’s study on the global burden of AMD from 1990 to 2017, special groups can significantly suffer from AMD, such as females, the elderly, those from developing countries, and low-educated persons, etc., and from 1990 to 2017, AMD contributed to a doubling of the number of people living a low quality of life [7]. An even more serious consideration is that the number of global AMD patients has been predicted to rise from 196 million in 2020 to 288 million in 2040 [8].
1.3. Classification of Current Medication
Currently, there are several types of AMD medication, including device-based treatment (prophylactic laser or photocoagulation therapy, photodynamic therapy), anti-inflammatory drug treatment (corticosteroids, nonsteroidal anti-inflammatory drugs (NSAIDs)), anti-vascular endothelial growth factor (anti-VEGF) intravitreal injection, nutritional therapy (antioxidants, ω-3 fatty acids, lutein, zeaxanthin, long-chain polyunsaturated fatty acids, plant extracts, etc.), etc.
2. Anti-Inflammatory Drugs
Drusen is one of the hallmarks for diagnosing AMD, as they are made by the destruction of RPE cells with advancing years, and the inflammation is closely related to AMD progress, as that produces drusen [9]. It is important to suppress inflammation progress to inhibit the severity of AMD (late AMD), and as shown Figure 1, there is an anti-inflammatory pathway caused by corticosteroid drugs and nonsteroidal anti-inflammatory drugs (NSAIDs).
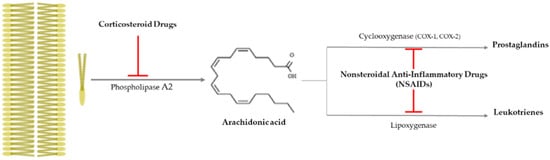
Figure 1. Anti-inflammatory pathway caused by corticosteroid drugs and nonsteroidal anti-inflammatory drugs (NSAIDs). Corticosteroid drugs inhibit the formation of arachidonic acid by causing plasma membrane breakage, which is caused by phospholipase A2. On the other hand, nonsteroidal anti-inflammatory drugs (NSAIDs) only control arachidonic acid and prostaglandins through cyclooxygenase 1 or 2 (COX-1 or -2). 

In 2008, an epidemiological study on the relation between anti-inflammatory drug use and AMD occurrence was conducted based on 614 patients (the average age and standard deviation, 72.9 ± 6.8) and 4,526 normal veterans (the average age and standard deviation, 73.2 ± 6.7) from 1997 to 2001 [10]. According to the study’s results, anti-inflammatory drug use suppressed the occurrent risk of AMD. As the macrophage-derived proinflammatory cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-1 (IL-1) especially increase when Bruch’s membrane is destroyed, and as they stimulate the increment of vascular endothelial cells, anti-inflammatory drugs can effectively suppress AMD progress [11]. In Table 1, anti-inflammatory drugs that are used in AMD treatment are summarized.
Table 1. Summary of anti-inflammatory drugs for AMD treatment.
| Classification | Application Route and Therapeutic Effect | Reference | |
|---|---|---|---|
| Corticosteroids | Dexamethasone | 1. Topical application with artemisinin
|
[12] |
2. Three combined therapies: dexamethasone, an anti-VEGF drug, and verteporfin with photodynamic therapy
|
[13] | ||
| Triamcinolone Acetonide (TA) | 1. Intravitreal injection
|
[14] | |
2. Combined intravitreal TA and bevacizumab injection
|
[15] | ||
| Spironolactone (mineralocorticoid receptor antagonist) | Oral administration
|
[16] | |
| NSAIDs | Aspirin | Topical application
|
[17][18][19] |
| Nepafenac (prodrug) | Topical application
|
[20] | |
| Diclofenac, Ketorolac | Intravitreal diclofenac and ketorolac injection
|
[21] | |
2.1. Corticosteroid Drugs
Corticosteroid drugs can perfectly inhibit the inflammation occurrence pathway, as they significantly control the arachidonic acid synthesis from plasma membrane breakage using phospholipase A2 [22] (Figure 1). Topical nanodispersion of dexamethasone and artemisinin on the eyeball effectively control CNV in AMD progress [12], and triple therapy using dexamethasone, bevacizumab, and verteporfin with photodynamic therapy is one of the anti-vascularization therapies used for AMD [13]. Triamcinolone acetonide (TA) has been widely used as a drug for macular edema and uveitis, as its effective duration is longer than that of dexamethasone [14], and combined intravitreal triamcinolone acetonide and bevacizumab injection has provided treatment to AMD patients who failed to be treated by the intravitreal injection of bevacizumab alone [15]. Spironolactone is a mineralocorticoid receptor antagonist that can be orally administered, and it is used to effectively suppress CNV in patients refractory to intravitreal anti-VEGF injections [16].
2.2. Nonsteroidal Anti-Inflammatory Drugs (NSAIDs)
Nonsteroidal anti-inflammatory drugs (NSAIDs) inhibit the synthesis of prostaglandins (PGEs) via inactivating cycloxygenase-1 (COX-1) or cycloxygenase-2 (COX-2), and they have been used as anti-inflammatory and antipyretic agents and analgesic drugs [22] (Figure 1). They are used as topical applicants for various ophthalmological purposes—for example, as an inflammation suppressor against allergic conjunctivitis and keratitis, a down-regulator for cystoid macular edema, a contractor for cataract surgery, etc. [23]. Aspirin is a famous NSAID, but although a low aspirin dose has been found to not affect AMD occurrence [17][18], a high aspirin dose might decrease AMD prevalence [19]. Nepafenac is a potent NSAID prodrug that has higher vascular permeability, longer inhibition of PGE synthesis, and better corneal penetration than others [20]. Intravitreal diclofenac and ketorolac injections have been found to effectively control inflammation in a lipopolysaccharide (LPS)-induced ocular inflammation rabbit model [21].
3. Drug Candidates Originated from Natural Products
Lutein and zeaxanthin exist as meso-zeaxanthin (a dipolar form, dihydroxylated carotenoid) in the retina, called macular xanthophylls, [24] which are included in dark green leafy vegetables such as kale, spinach, peas, etc. and egg yolks [25], and they suppress phototoxicity-induced oxidative stress and apoptosis in the visual system [26] and also induce G2/M phase arrest in the RPE [27]. Recently, there have been many trials that have investigated AMD drug candidates from natural products, with these products especially studied for their ability to regulate the RPE’s status (Table 2). These natural product candidates can be classified into (1) anti-apoptosis inducers, (2) cell cycle arrest modulators, and (3) VEGF inactivators. Based on the apoptotic pathway, the candidates can be grouped into (1) anti-oxidative stress, (2) anti-inflammation, and (3) anti-carbonyl stress candidates. One hundred percent EtOH extract of Arctium lappa L. leaves has been found to have plentiful amounts of phenolics and flavonoids, and it effectively suppressed both oxidative stress by scavenging effects of 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)(ABTS) and apoptosis by modulating bcl-2 family and caspase cascade [28]. Genipin is a glycosidic ligand that has been found to suppress H2O2-induced oxidative stress and apoptosis via nuclear factor-erythroid 2-related factor-2 (Nrf2), which regulates the expression of heme oxygenase-1 (HO-1) and NAD(P)H: quinine oxidoreductase 1 (NQOA) [29]. Delphinidin is one of the anthocyanins that exists in several fruits and vegetables [30] and has been found to significantly inhibit the synthesis of reactive oxygen species (ROS) through modulating antioxidative enzyme activation and apoptosis via regulating the bcl-2 family and caspase cascade [31]. Glabridin (isoflavonoid) from Glycyrrhiza glabra L. root has been demonstrated to inhibit oxidative stress and apoptosis through ERK1/2 and p38MAPK inactivation [32]. Wogonin (5,7-dihydroxy-8-methoxyflavone), isolated from Scutellaria baicalensis Georgi root, has been found to control LPS-induced inflammation changes such as IL-1β, IL-6, IL-8, TNF-α, COX-2, and inducible nitric oxide synthase (iNOS) via the Toll-like receptor 4 (TLR4)/NF-κB pathway [33]. Hot water extract of Prunella vulgaris var. L has been observed to prevent oxidative stress by ROS generation, carbonyl stress by MDA production, and inflammation through Nrf2/HO-1 pathway [34]. Vaccinium uliginosum L. hot water extract has been found to suppress A2E- and blue light-induced apoptosis by controlling caspase cascade and bcl-2 pathway [35]. Baicalin, a flavonoid isolated from Scutellaria baicalensis Georgi, has been found to inhibit Aβ-induced pyroptosis through modulating miR-223/NLRP3 inflammasome pathway [36]. β-carotene, as a typical carotenoid, has anti-oxidative effects and is one of the major components in tomatoes (Lycopersicum esculentum L.) [37] and has been reported to down-regulate H2O2-induced nitrotyrosine formation and protein carbonylation [38]. Bile acid is synthesized in the liver (primary bile acid) and in the colon (secondary bile acid), and is known as bile salt when it is conjugated with taurine or glycine [39]; the major bile salts include taurocholic acid, glycocholic acid, taurochenodeoxycholic acid, and glycochenodeoxycholic acid, etc. [40], and taurocholic acid, one of the major bile salts, has been demonstrated to inhibit angiogenesis-related cell proliferation, cell migration, and tube formation [41].
Table 2. Summary of AMD drug candidates developed from natural products.
| Therapeutic Mechanism |
Natural Product | Application | References | ||||
|---|---|---|---|---|---|---|---|
| Species | Applied Characteristic | Effective Compound | Model | Route | Minimum Effective Dose |
||
| Inhibition of oxidative stress and apoptosis | Arctium lappa L. leaf | 100% EtOH extract | Phenolic and flavonoid | RPE cell | Media | 30 μg/mL for 24 h | [28] |
| Mouse | I.P. | 50 mg/kg for 4 w | |||||
| Eucommia ulmoides | Genipin (glycosidic ligand) |
Genipin (glycosidic ligand) |
ARPE-19 cell | Media | 30 μM for 24 h | [29] | |
| Fruit or Vegetable | Delphinidin (anthocyanidin) |
Delphinidin (anthocyanidin) |
ARPE-19 cell | Media | 25 μg/mL for 24 h | [31] | |
| Glycyrrhiza glabra L. root | Glabridin (isoflavonoid) |
Glabridin (isoflavonoid) |
RPE cell | Media | 2 μM for 2 h | [32] | |
| Mouse | I.P. | 20 mg/kg for 1 w | |||||
| Inhibition of inflammation and apoptosis | Scutellaria baicalensis Georgi root | 5,7-dihydroxy-8-methoxyflavone (wogonin) |
5,7-dihydroxy-8-methoxyflavone (wogonin) |
ARPE-19 cell | Media | 10 μM for 24 h | [33] |
| Inhibition of oxidative stress, inflammation, and apoptosis | Prunella vulgaris var. L | Water extract | Rosmarinic Acid | ARPE-19 cell | Media | 100 μg/mL for 24 h | [34] |
| Mouse | P.O. | 100 mg/kg for 4 day | |||||
| Inhibition of apoptosis | Vaccinium uliginosum L | Water extract | Polyphenol | ARPE-19 cell | Media | 100 μg/mL for 24 h | [35] |
| Inhibition of pyroptosis | Scutellaria baicalensis Georgi | Baicalin | Baicalin | ARPE-19 cell | Media | 50 μg/mL for 72 h | [36] |
| Inhibition of carbonyl stress | Lycopersicum esculentum L. (Tomato) |
n-hexane extract | β-carotene | ARPE-19 cell | Media | 1 μM-β-carotene for 24 h |
[38] |
| Inhibition of G2/M phase arrest | Fruit or Vegetable | Lutein | Lutein | ARPE-19 cell | Media | 25 μg/mL for 24 h | [27] |
| Inhibition of VEGF activation | Bile Acid (Animal) |
Taurocholic acid | Taurocholic acid | HRPEpiC cell | Media | 100 μM for 48 h | [41] |
This entry is adapted from the peer-reviewed paper 10.3390/ijms222111837
References
- Bhutto, I.; Lutty, G. Understanding age-related macular degeneration (AMD): Relationships between the photoreceptor/retinal pigment epithelium/Bruch’s Membrane/choriocapillaris complex. Mol. Aspects Med. 2012, 33, 295–317.
- Khandhadia, S.; Cherry, J.; Lotery, A.J. Age-related macular degeneration. Adv. Exp. Med. Biol. 2012, 724, 15–36.
- Jonas, J.B.; Cheung, C.M.G.; Panda-Jonas, S. Updates on the epidemiology of age-related macular degeneration. Asia-Pac. J. Ophthalmol. 2017, 6, 493–497.
- Friedman, D.S.; O’Colmain, B.J.; Munoz, B.; Tomany, S.C.; Macarty, C.; de Jong, P.T.V.M.; Nemesure, B.; Mitchell, P.; Kempen, J.; Congdon, N. Prevalence of age-related macular degeneration in the United States. Arch. Ophthalmol. 2004, 122, 564–572.
- Bressler, N.M. Age-related macular degeneration is the leading cause of blindness. JAMA 2004, 291, 1900–1901.
- Seland, J.H.; Vingerling, J.R.; Augood, C.A.; Bentham, G.; Chakravarthy, U.; de Jong, P.T.V.M.; Rahu, M.; Soubrane, G.; Tomazzoli, L.; Topouzis, F.; et al. Visual impairment and quality of life in the older European population, the EUREYE study. Acta Ophthalmol. 2011, 89, 608–613.
- Zou, M.; Zhang, Y.; Chen, A.; Young, C.A.; Li, Y.; Zheng, D.; Jin, G. Variations and trends in global disease burden of age-related macular degeneration: 1990–2017. Acta Ophthalmol. 2021, 99, e330–e335.
- Wong, W.L.; Su, X.; Li, X.; Cheung, C.M.G.; Klein, R.; Cheng, C.Y.; Wong, T.Y. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health 2014, 2, e106–e116.
- Johnson, L.V.; Leitner, W.P.; Staples, M.K.; Anderson, D.H. Complement activation and inflammatory processes in drusen formation and age related macular degeneration. Exp. Eye Res. 2001, 73, 887–896.
- Swanson, M.W.; McGwin, G., Jr. Anti-inflammatory drug use and age-related macular degeneration. Optom. Vis. Sci. 2008, 85, 947–950.
- Apte, R.S. Regulation of angiogenesis by macrophages. Adv. Exp. Med. Biol. 2010, 664, 15–19.
- Ponnusamy, C.; Sugumaran, A.; Krishnaswami, V.; Kandasamy, R.; Natesan, S. Design and development of artemisinin and dexamethasone loaded topical nanodispersion for the effective treatment of age-related macular degeneration. IET Nanobiotechnol. 2019, 13, 868–874.
- Ehmann, D.; Garcia, R. Triple therapy for neovascular age-related macular degeneration: Verteporfin PDT, bevacizumab, and dexamethasone. Retina 2007, 27, 133–140.
- Francis, B.A.; Chang, E.L.; Haik, B.G. Particle size and drug interactions of injectable corticosteroids used in ophthalmic practice. Ophthalmology 1996, 103, 1884–1888.
- Jonas, J.B.; Libondi, T.; Golubkina, L.; Spandau, U.H.; Schlichtenbrede, F.; Rensch, F. Combined intravitreal bevacizumab and triamcinolone in exudative age-related macular degeneration. Acta Ophthalmol. 2010, 88, 630–634.
- Zhao, M.; Mantel, I.; Gelize, E.; Li, X.; Xie, X.; Arboleda, A.; Seminel, M.; Levy-Boukris, R.; Derngoghossian, M.; Prunotto, A.; et al. Mineralocorticoid receptor antagonism limits experimental choroidal neovascularization and structural changes associated with neovascular age-related Macular degeneration. Nat. Commun. 2019, 10, 369.
- Christen, W.G.; Glynn, R.J.; Chew, E.Y.; Burning, J.E. Low-dose aspirin and medical record-confirmed age-related macular degeneration in a randomized trial of women. Ophthalmology 2009, 116, 2386–2392.
- Ye, J.; Xu, Y.F.; He, J.J.; Lou, L.X. Association between aspirin use and age-related macular degeneration: A meta-analysis. Investig. Ophthalmol. Vis. Sci. 2014, 55, 2687–2696.
- McGeer, P.L.; Sibley, J. Sparing of age-related macular degeneration in rheumatoid arthritis. Neurobiol. Aging. 2005, 26, 1199–1203.
- Gamache, D.A.; Graff, G.; Brady, M.T.; Spellman, J.M.; Yanni, J.M. Nepafenac, a unique nonsteroidal prodrug with potential utility in the treatment of trauma-induced ocular inflammation: I. Assessment of anti-inflammatory efficacy. Inflammation 2000, 24, 357–370.
- Baranano, D.E.; Kim, S.J.; Edelhauser, H.F.; Durairaj, C.; Kompella, U.B.; Handa, J.T. Efficacy and pharmacokinetics of intravitreal non-steroidal anti-inflammatory drugs for intraocular inflammation. Br. J. Ophthalmol. 2009, 93, 1387–1390.
- Hitner, H.; Nagle, B. Chapter 25. Respiratory Pharmacology, Treatment of Asthma and COPD. In Pharmacology: An Introduction, 7th ed.; McGraw Hill Education Korea, Ltd. and Medical Science: Seoul, Korea, 2019; pp. 385–402.
- Kim, S.J.; Flach, A.J.; Jampol, L.M. Nonsteroidal anti-inflammatory drugs in ophthalmology. Surv. Ophthalmol. 2010, 55, 108–133.
- Subczynski, W.K.; Wisniewska, A.; Widomska, J. Location of macular xanthophylls in the most vulnerable regions of photoreceptor outer-segment membranes. Arch. Biochem. Biophys. 2010, 504, 61–66.
- Perry, A.; Rasmussen, H.; Johnson, E.J. Xanthophyll (lutein, zeaxanthin) content of fruits, vegetables and corn and egg products. J. Food. Comp. Anal. 2009, 22, 9–15.
- Koushan, K.; Rusovici, R.; Li, W.; Ferguson, L.R.; Chalam, K.V. The role of lutein in eye-related disease. Nutrients 2013, 5, 1823–1839.
- Liu, H.; Liu, W.; Zhou, X.; Long, C.; Kuang, X.; Hu, J.; Tang, Y.; Liu, L.; He, J.; Huang, Z.; et al. Protective effect of lutein on ARPE-19 cells upon H2O2-induced G2/M arrest. Mol. Med. Rep. 2017, 16, 2069–2074.
- Kim, D.H.; Choi, Y.R.; Shim, J.; Choi, Y.S.; Kim, Y.T.; Kim, M.K.; Kim, M.J. Suppressive effect of Arctium lappa L. leaves on retinal damage against A2E-induced ARPE-19 cells and mice. Molecules 2020, 25, 1737.
- Zhao, H.; Wang, R.; Ye, M.; Zhang, L. Genipin protects against H2O2-induced oxidative damage in retinal pigment epithelial cells by promoting Nrf2 signaling. Int. J. Mol. Med. 2019, 43, 936–944.
- Kong, J.M.; Chia, L.S.; Goh, N.K.; Chia, T.F.; Brouillard, R. Analysis and biological activities of anthocyanins. Phytochemistry 2003, 64, 923–933.
- Ni, T.; Yang, W.; Xing, Y. Protective effects of delphinidin against H2O2-induced oxidative injuries in human retinal pigment epithelial cells. Biosci. Rep. 2019, 39, BSR20190689.
- Aung, K.H.; Liu, H.; Ke, Z.; Jiang, S.; Huang, J. Glabridin attenuates the retinal degeneration induced by sodium iodate in vitro and in vivo. Front. Pharmacol. 2020, 11, 566699.
- Chen, C.; Guo, D.; Lu, G. Wogonin protects human retinal pigment epithelium cells from LPS-induced barrier dysfunction and inflammatory responses by regulating the TLR4/NF-κB signaling pathway. Mol. Med. Rep. 2017, 15, 2289–2295.
- Kim, J.; Cho, K.; Choung, S.Y. Protective effect of Prunella vulgaris var. L extract against blue light induced damages in ARPE-19 cells and mouse retina. Free Radic. Biol. Med. 2020, 152, 622–631.
- Yoon, S.M.; Lee, B.L.; Guo, Y.R.; Choung, S.Y. Preventive effect of Vaccinium uliginosum L. extract and its fractions on age-related macular degeneration and its action mechanisms. Arch. Pharm. Res. 2016, 39, 21–32.
- Sun, H.J.; Jin, X.M.; Xu, J.; Xiao, Q. Baicalin alleviates age-related macular degeneration via miR-223/NLRP3-regulated pyroptosis. Pharmacology 2020, 105, 28–38.
- Marti, R.; Rosello, S.; Cebolla-Cornejo, J. Tomato as a source of carotenoids and polyphenols targeted to cancer prevention. Cancer 2016, 8, 58.
- Chichili, G.R.; Nohr, D.; Frank, J.; Flaccus, A.; Fraser, P.D.; Enfissi, E.M.A.; Biesalski, H.K. Protective effects of tomato extract with elevated β-carotene levels on oxidative stress in ARPE-19 cells. Br. J. Nutr. 2006, 96, 643–649.
- Chiang, J.Y.L. Bile acids: Regulation of synthesis. J. Lipid Res. 2009, 50, 1955–1966.
- Hofmann, A.F. The continuing importance of Bile Acids in liver and intestinal disease. Arch. Intern. Med. 1999, 159, 2647–2658.
- Warden, C.; Barnett, J.M.; Brantley, M.A., Jr. Taurocholic acid inhibits features of age-related macular degeneration in vitro. Exp. Eye Res. 2020, 193, 107974.
This entry is offline, you can click here to edit this entry!
