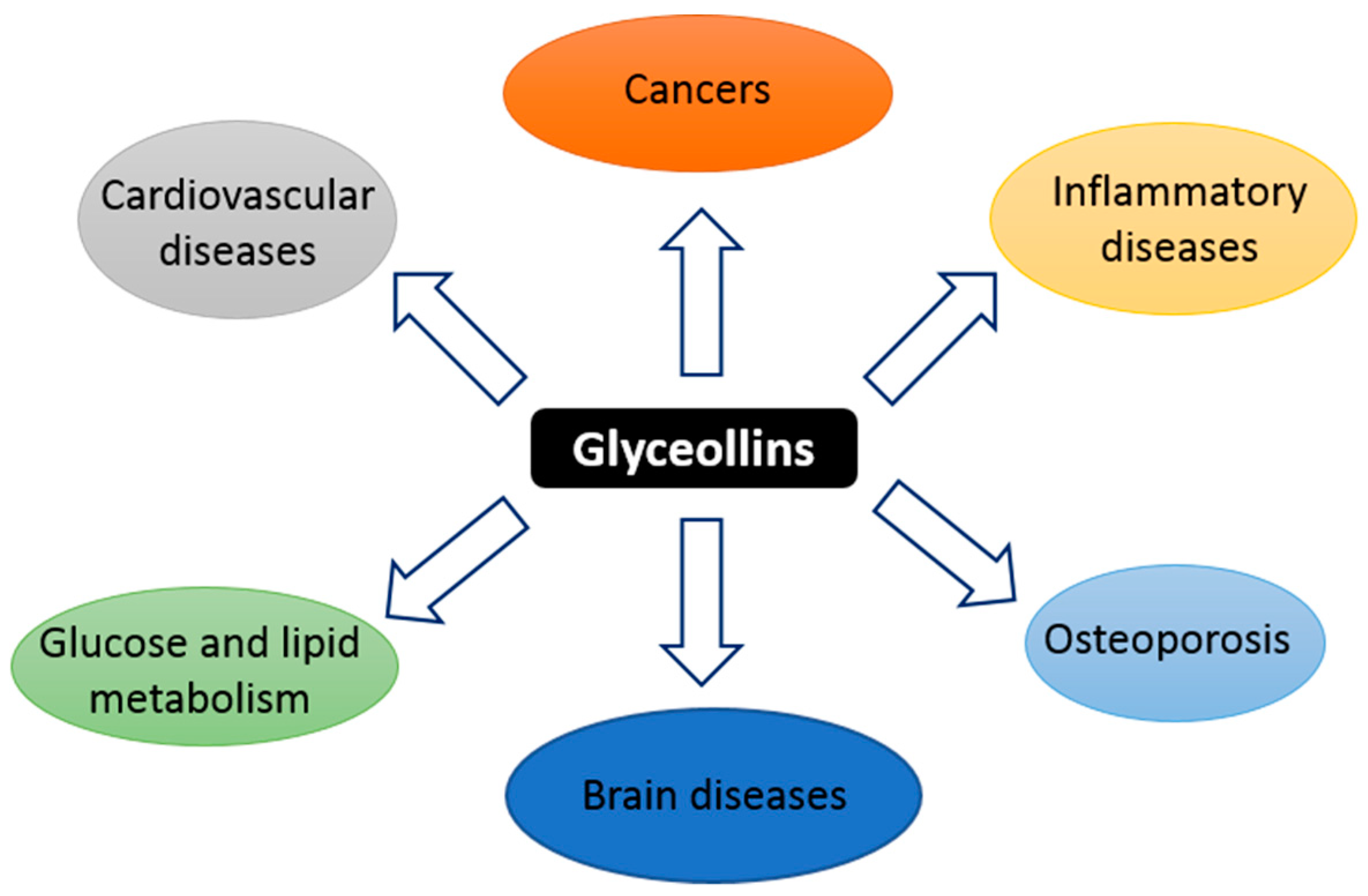
Glyceollins are a group of phytoalexins that are isolated from soybeans. They have attracted attention because they exert numerous effects on human functions and diseases, notably anticancer effects.
- glyceollins
- phytochemicals
- dietary compounds
- signaling pathways
- estrogen receptor
- breast cancer
1. Introduction
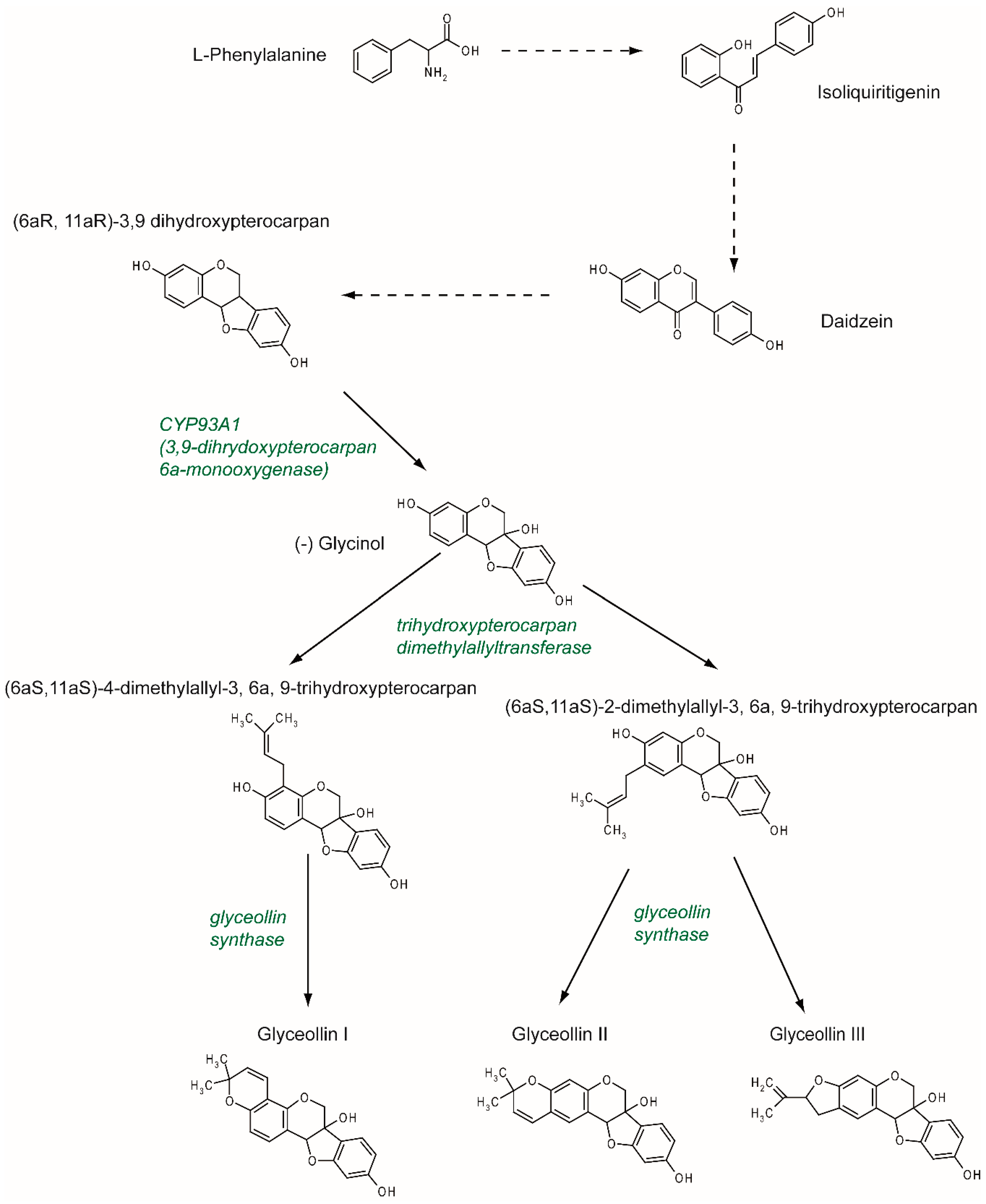
2. Synthesis and Structure
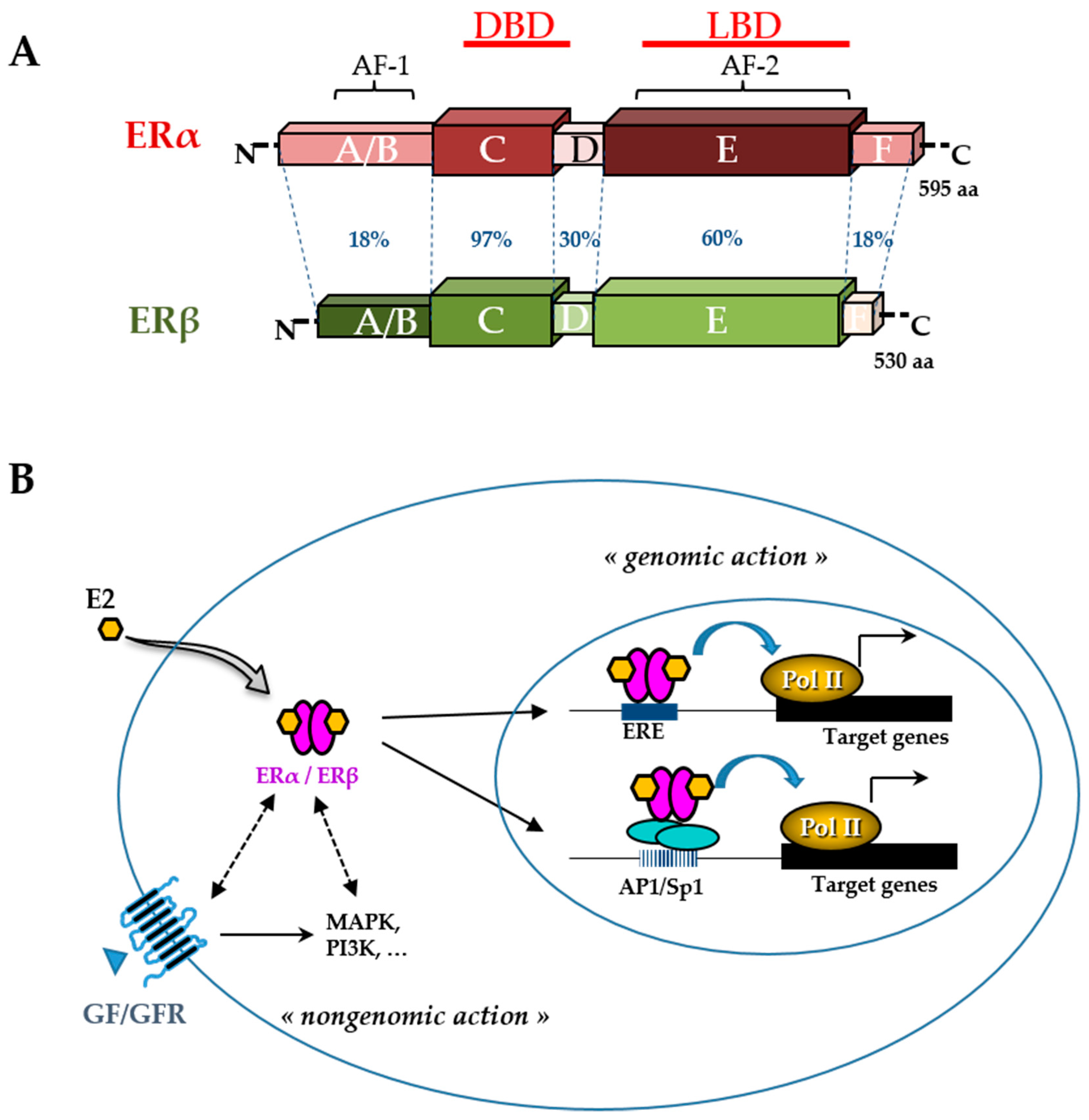
3. Metabolism and Pharmacokinetics
|
Animal Model |
Treatment |
Sample |
Time |
Method of Measure |
Major Results |
References |
|---|---|---|---|---|---|---|
|
Female monkey (Macaca fascicularis) |
Diet containing glyceollin mixture 1, 134 mg/day representing 50% of total isoflavonoids in the diet |
Plasma |
4 h and 24 h post administration (postad) |
Liquid chromatographic-photodiode array mass spectrometric analysis |
Plasma concentration of glyceollins: - 4 h: 134.2 ± 34.6 nmol/L, representing only 11.6 % of the plasma isoflavonoids level - 24 h: Undetectable ˂ 1 nmol/L |
Wood et al. [40] |
|
Male ZDSD/Pco rat |
Glyceollin mixture, gavage, 30 and 90 mg/kg |
Plasma |
20, 60, 120 and 240 minutes (min) postad |
HPLC-electrospray ionization-MS/MS |
Plama concentration of glyceollins: - 20 min: Starts to be detectable - the next 220 min: Remains stable For dose 30 mg/kg: Ranges from 81.2 to 118.4 ng/mL For dose 60 mg/kg: Ranges from 118.2 to 159.0 ng/mL - 60 min: Peak concentration |
Boué et al. 2012 [41] |
|
Glyceollin mixture, gavage, 90 mg/kg/days for 2 weeks |
Plasma, feces, and urine |
Plasma: 3 h postad Feces: Once daily for 2 weeks Urine: 24 h collection postad a single dose |
Precursor and product ion scanning using liquid chromatography coupled online with Electrospray ionization tandem mass spectrometry |
- Rapidly absorption, glyceollins undergo phase I and phase II metabolism in the small intestine and the liver - Metabolites of glyceollins were identified in the plasma, the urine, and the feces Phase I conjugation: Epoxidation, hydroxylation… Phase II conjugation: Sulfate and glucuronide conjugations… |
1 Glyceollin mixture contains glyceollin I, glyceollin II and glyceollin III.
4. Anticancer Effects
4.1. Estrogen-Dependent Effects
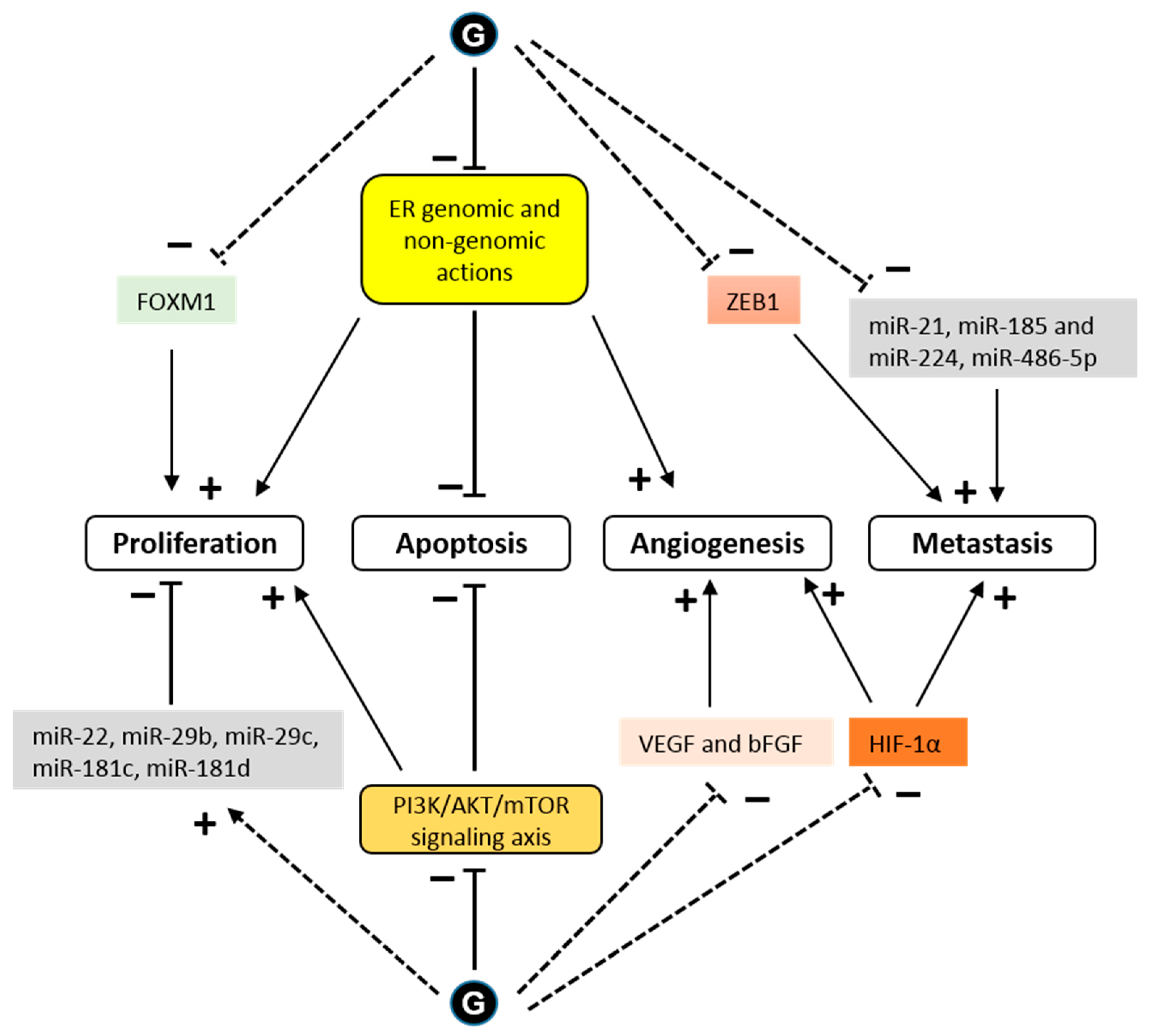
4.2. Estrogen-Independent Effect
This entry is adapted from the peer-reviewed paper 10.3390/nu11010079
References
- Tresserra-Rimbau, A.; Lamuela-Raventos, R.M.; Moreno, J.J. Polyphenols, food and pharma. Current knowledge and directions for future research. Biochem. Pharmacol. 2018, 156, 186–195.
- Knaze, V.; Rothwell, J.A.; Zamora-Ros, R.; Moskal, A.; Kyrø, C.; Jakszyn, P.; Skeie, G.; Weiderpass, E.; Santucci de Magistris, M.; Agnoli, C.; et al. A new food-composition database for 437 polyphenols in 19,899 raw and prepared foods used to estimate polyphenol intakes in adults from 10 European countries. Am. J. Clin. Nutr. 2018, 108, 517–524.
- Angeloni, C.; Maraldi, T.; Milenkovic, D.; Vauzour, D. Dietary Polyphenols and Their Effects on Cell Biochemistry and Pathophysiology 2014. Oxidative Med. Cell. Longev. 2015, 2015, 1–2.
- Surh, Y.-J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer 2003, 3, 768–780.
- Hennig, B.; Petriello, M.C.; Gamble, M.V.; Surh, Y.-J.; Kresty, L.A.; Frank, N.; Rangkadilok, N.; Ruchirawat, M.; Suk, W.A. The role of nutrition in influencing mechanisms involved in environmentally mediated diseases. Rev. Environ. Health 2018, 33, 87–97.
- Wongwarawipat, T.; Papageorgiou, N.; Bertsias, D.; Siasos, G.; Tousoulis, D. Olive Oil-related Anti-inflammatory Effects on Atherosclerosis: Potential Clinical Implications. Endocr. Metab. Immune Disord. Drug Targets 2017, 18.
- Zujko, M.E.; Waśkiewicz, A.; Witkowska, A.M.; Szcześniewska, D.; Zdrojewski, T.; Kozakiewicz, K.; Drygas, W. Dietary Total Antioxidant Capacity and Dietary Polyphenol Intake and Prevalence of Metabolic Syndrome in Polish Adults: A Nationwide Study. Oxidative Med. Cell. Longev. 2018, 2018, 1–10.
- Goetz, M.E.; Judd, S.E.; Safford, M.M.; Hartman, T.J.; McClellan, W.M.; Vaccarino, V. Dietary flavonoid intake and incident coronary heart disease: the REasons for Geographic and Racial Differences in Stroke (REGARDS) study. Am. J. Clin. Nutr. 2016, 104, 1236–1244.
- Liu, R.H. Health-promoting components of fruits and vegetables in the diet. Adv. Nutr. 2013, 4, 384S–392S.
- Anunciato, T.P.; da Rocha Filho, P.A. Carotenoids and polyphenols in nutricosmetics, nutraceuticals, and cosmeceuticals. J. Cosmet. Dermatol. 2012, 11, 51–54.
- Pezzuto, J.M.; Kondratyuk, T.P.; Ogas, T. Resveratrol derivatives: a patent review (2009–2012). Expert Opin. Ther. Pat. 2013, 23, 1529–1546.
- Dragan, S.; Andrica, F.; Serban, M.-C.; Timar, R. Polyphenols-rich natural products for treatment of diabetes. Curr. Med. Chem. 2015, 22, 14–22.
- Pedras, M.S.C.; Ahiahonu, P.W.K. Metabolism and detoxification of phytoalexins and analogs by phytopathogenic fungi. Phytochemistry 2005, 66, 391–411.
- Dakora, F.D.; Phillips, D.A. Diverse functions of isoflavonoids in legumes transcend anti-microbial definitions of phytoalexins. Physiol. Mol. Plant Pathol. 1996, 49, 1–20.
- Schmelz, E.A.; Huffaker, A.; Sims, J.W.; Christensen, S.A.; Lu, X.; Okada, K.; Peters, R.J. Biosynthesis, elicitation and roles of monocot terpenoid phytoalexins. Plant J. 2014, 79, 659–678.
- Wang, W.; Li, Y.; Dang, P.; Zhao, S.; Lai, D.; Zhou, L. Rice Secondary Metabolites: Structures, Roles, Biosynthesis, and Metabolic Regulation. Molecules 2018, 23, 3098.
- Park, I.S.; Kim, H.J.; Jeong, Y.-S.; Kim, W.-K.; Kim, J.-S. Differential abilities of Korean soybean varieties to biosynthesize glyceollins by biotic and abiotic elicitors. Food Sci. Biotechnol. 2017, 26, 255–261.
- Block, A.K.; Vaughan, M.M.; Schmelz, E.A.; Christensen, S.A. Biosynthesis and function of terpenoid defense compounds in maize (Zea mays). Planta 2018.
- Ejike, C.E.C.C.; Gong, M.; Udenigwe, C.C. Phytoalexins from the Poaceae: Biosynthesis, function and prospects in food preservation. Food Res. Int. 2013, 52, 167–177.
- Hidalgo, W.; Chandran, J.N.; Menezes, R.C.; Otálvaro, F.; Schneider, B. Phenylphenalenones protect banana plants from infection by Mycosphaerella fijiensis and are deactivated by metabolic conversion. Plant Cell Environ. 2016, 39, 492–513.
- Timperio, A.M.; D’Alessandro, A.; Fagioni, M.; Magro, P.; Zolla, L. Production of the phytoalexins trans-resveratrol and delta-viniferin in two economy-relevant grape cultivars upon infection with Botrytis cinerea in field conditions. Plant Physiol. Biochem. PPB 2012, 50, 65–71.
- Lee, M.R.; Kim, J.Y.; Chun, J.; Park, S.; Kim, H.J.; Kim, J.-S.; Jeong, J.-I.; Kim, J.H. Induction of glyceollins by fungal infection in varieties of Korean soybean. J. Microbiol. Biotechnol. 2010, 20, 1226–1229.
- Kim, H.J.; Lim, J.-S.; Kim, W.-K.; Kim, J.-S. Soyabean glyceollins: biological effects and relevance to human health. Proc. Nutr. Soc. 2012, 71, 166–174.
- Bamji, S.F.; Corbitt, C. Glyceollins: Soybean phytoalexins that exhibit a wide range of health-promoting effects. J. Funct. Foods 2017, 34, 98–105.
- Lecomte, S.; Chalmel, F.; Ferriere, F.; Percevault, F.; Plu, N.; Saligaut, C.; Surel, C.; Lelong, M.; Efstathiou, T.; Pakdel, F. Glyceollins trigger anti-proliferative effects through estradiol-dependent and independent pathways in breast cancer cells. Cell Commun. Signal. CCS 2017, 15, 26.
- Salvo, V.A.; Boue, S.M.; Fonseca, J.P.; Elliott, S.; Corbitt, C.; Collins-Burow, B.M.; Curiel, T.J.; Srivastav, S.K.; Shih, B.Y.; Carter-Wientjes, C.; et al. Antiestrogenic Glyceollins Suppress Human Breast and Ovarian Carcinoma Tumorigenesis. Clin. Cancer Res. 2006, 12, 7159–7164.
- Burow, M.E.; Boue, S.M.; Collins-Burow, B.M.; Melnik, L.I.; Duong, B.N.; Carter-Wientjes, C.H.; Li, S.; Wiese, T.E.; Cleveland, T.E.; McLachlan, J.A. Phytochemical Glyceollins, Isolated from Soy, Mediate Antihormonal Effects through Estrogen Receptor α and β. J. Clin. Endocrinol. Metab. 2001, 86, 1750–1758.
- Zimmermann, M.C.; Tilghman, S.L.; Boue, S.M.; Salvo, V.A.; Elliott, S.; Williams, K.Y.; Skripnikova, E.V.; Ashe, H.; Payton-Stewart, F.; Vanhoy-Rhodes, L.; et al. Glyceollin I, a Novel Antiestrogenic Phytoalexin Isolated from Activated Soy. J. Pharmacol. Exp. Ther. 2010, 332, 35–45.
- Lee, S.H.; Lee, J.; Jung, M.H.; Lee, Y.M. Glyceollins, a novel class of soy phytoalexins, inhibit angiogenesis by blocking the VEGF and bFGF signaling pathways. Mol. Nutr. Food Res. 2013, 57, 225–234.
- Lee, S.-H.; Jee, J.-G.; Bae, J.-S.; Liu, K.-H.; Lee, Y.M. A Group of Novel HIF-1α Inhibitors, Glyceollins, Blocks HIF-1α Synthesis and Decreases Its Stability via Inhibition of the PI3K/AKT/mTOR Pathway and Hsp90 Binding: GLYCEOLLINS INHIBIT HIF-1α. J. Cell. Physiol. 2015, 230, 853–862.
- Carriere, P.P.; Llopis, S.D.; Naiki, A.C.; Nguyen, G.; Phan, T.; Nguyen, M.M.; Preyan, L.C.; Yearby, L.; Pratt, J.; Burks, H.; et al. Glyceollin I Reverses Epithelial to Mesenchymal Transition in Letrozole Resistant Breast Cancer through ZEB1. Int. J. Environ. Res. Public. Health 2016, 13, 10.
- Burden, R.S.; Bailey, J.A. Structure of the phytoalexin from soybean. Phytochemistry 1975, 14, 1389–1390.
- Keen, N.T.; Zaki, A.I.; Sims, J.J. Biosynthesis of hydroxyphaseollin and related isoflavanoids in disease-resistant soybean hypocotyls. Phytochemistry 1972, 11, 1031–1039.
- Schopfer, C.R.; Kochs, G.; Lottspeich, F.; Ebel, J. Molecular characterization and functional expression of dihydroxypterocarpan 6a-hydroxylase, an enzyme specific for pterocarpanoid phytoalexin biosynthesis in soybean (Glycine max L.). FEBS Lett. 1998, 432, 182–186.
- Yoneyama, K.; Akashi, T.; Aoki, T. Molecular Characterization of Soybean Pterocarpan 2-Dimethylallyltransferase in Glyceollin Biosynthesis: Local Gene and Whole-Genome Duplications of Prenyltransferase Genes Led to the Structural Diversity of Soybean Prenylated Isoflavonoids. Plant Cell Physiol. 2016, 57, 2497–2509.
- Akashi, T.; Sasaki, K.; Aoki, T.; Ayabe, S.; Yazaki, K. Molecular cloning and characterization of a cDNA for pterocarpan 4-dimethylallyltransferase catalyzing the key prenylation step in the biosynthesis of glyceollin, a soybean phytoalexin. Plant Physiol. 2009, 149, 683–693.
- Sukumaran, A.; McDowell, T.; Chen, L.; Renaud, J.; Dhaubhadel, S. Isoflavonoid-specific prenyltransferase gene family in soybean: GmPT01, a pterocarpan 2-dimethylallyltransferase involved in glyceollin biosynthesis. Plant J. Cell Mol. Biol. 2018.
- Welle, R.; Grisebach, H. Induction of phytoalexin synthesis in soybean: enzymatic cyclization of prenylated pterocarpans to glyceollin isomers. Arch. Biochem. Biophys. 1988, 263, 191–198.
- Farrell, K.; Jahan, M.A.; Kovinich, N. Distinct Mechanisms of Biotic and Chemical Elicitors Enable Additive Elicitation of the Anticancer Phytoalexin Glyceollin I. Molecules 2017, 22, 1261.
- Wood, C.E.; Clarkson, T.B.; Appt, S.E.; Franke, A.A.; Boue, S.M.; Burow, M.E.; McCoy, T.; Cline, J.M. Effects of Soybean Glyceollins and Estradiol in Postmenopausal Female Monkeys. Nutr. Cancer 2006, 56, 74–81.
- Boué, S.M.; Isakova, I.A.; Burow, M.E.; Cao, H.; Bhatnagar, D.; Sarver, J.G.; Shinde, K.V.; Erhardt, P.W.; Heiman, M.L. Glyceollins, Soy Isoflavone Phytoalexins, Improve Oral Glucose Disposal by Stimulating Glucose Uptake. J. Agric. Food Chem. 2012, 60, 6376–6382.
- Quadri, S.S.; Stratford, R.E.; Boué, S.M.; Cole, R.B. Screening and Identification of Glyceollins and Their Metabolites by Electrospray Ionization Tandem Mass Spectrometry with Precursor Ion Scanning. Anal. Chem. 2013, 85, 1727–1733.
- Quadri, S.S.; Stratford, R.E.; Boué, S.M.; Cole, R.B. Identification of Glyceollin Metabolites Derived from Conjugation with Glutathione and Glucuronic Acid in Male ZDSD Rats by Online Liquid Chromatography–Electrospray Ionization Tandem Mass Spectrometry. J. Agric. Food Chem. 2014, 62, 2692–2700.
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012: Globocan 2012. Int. J. Cancer 2015, 136, E359–E386.
- Dai, X.; Xiang, L.; Li, T.; Bai, Z. Cancer Hallmarks, Biomarkers and Breast Cancer Molecular Subtypes. J. Cancer 2016, 7, 1281–1294.
- Pakdel, F. Molecular Pathways of Estrogen Receptor Action. Int. J. Mol. Sci. 2018, 19, 2591.
- Klinge, C.M. Estrogen receptor interaction with co-activators and co-repressors. Steroids 2000, 65, 227–251.
- Safe, S.; Kim, K. Nuclear Receptor-Mediated Transactivation through Interaction with Sp Proteins. In Progress in Nucleic Acid Research and Molecular Biology; Elsevier: Amsterdam, The Netherlands, 2004; Volume 77, pp. 1–36. ISBN 978-0-12-540077-0.
- Mehta, R.G.; Hawthorne, M.; Mehta, R.R.; Torres, K.E.O.; Peng, X.; McCormick, D.L.; Kopelovich, L. Differential Roles of ERα and ERβ in Normal and Neoplastic Development in the Mouse Mammary Gland. PLoS ONE 2014, 9, e113175.
- Payton-Stewart, F.; Khupse, R.S.; Boué, S.M.; Elliott, S.; Zimmermann, M.C.; Skripnikova, E.V.; Ashe, H.; Tilghman, S.L.; Beckman, B.S.; Cleveland, T.E.; et al. Glyceollin I enantiomers distinctly regulate ER-mediated gene expression. Steroids 2010, 75, 870–878.
- Boué, S.M.; Tilghman, S.L.; Elliott, S.; Zimmerman, M.C.; Williams, K.Y.; Payton-Stewart, F.; Miraflor, A.P.; Howell, M.H.; Shih, B.Y.; Carter-Wientjes, C.H.; et al. Identification of the Potent Phytoestrogen Glycinol in Elicited Soybean (Glycine max). Endocrinology 2009, 150, 2446–2453.
- Bratton, M.R.; Martin, E.C.; Elliott, S.; Rhodes, L.V.; Collins-Burow, B.M.; McLachlan, J.A.; Wiese, T.E.; Boue, S.M.; Burow, M.E. Glyceollin, a novel regulator of mTOR/p70S6 in estrogen receptor positive breast cancer. J. Steroid Biochem. Mol. Biol. 2015, 150, 17–23.
- Fingar, D.C.; Blenis, J. Target of rapamycin (TOR): an integrator of nutrient and growth factor signals and coordinator of cell growth and cell cycle progression. Oncogene 2004, 23, 3151–3171.
- Brown, K. Breast cancer chemoprevention: risk-benefit effects of the antioestrogen tamoxifen. Expert Opin. Drug Saf. 2002, 1, 253–267.
- Payton-Stewart, F.; Schoene, N.W.; Kim, Y.S.; Burow, M.E.; Cleveland, T.E.; Boue, S.M.; Wang, T.T.Y. Molecular effects of soy phytoalexin glyceollins in human prostate cancer cells LNCaP. Mol. Carcinog. 2009, 48, 862–871.
- Rhodes, L.V.; Tilghman, S.L.; Boue, S.M.; Wang, S.; Khalili, H.; Muir, S.E.; Bratton, M.R.; Zhang, Q.; Wang, G.; Burow, M.E.; et al. Glyceollins as novel targeted therapeutic for the treatment of triple-negative breast cancer. Oncol. Lett. 2012, 3, 163–171.
- Guarino, M.; Rubino, B.; Ballabio, G. The role of epithelial-mesenchymal transition in cancer pathology. Pathology 2007, 39, 305–318.
- Schmalhofer, O.; Brabletz, S.; Brabletz, T. E-cadherin, beta-catenin, and ZEB1 in malignant progression of cancer. Cancer Metastasis Rev. 2009, 28, 151–166.
- Carmeliet, P. Angiogenesis in health and disease. Nat. Med. 2003, 9, 653–660.
- Kaelin, W.G.; Ratcliffe, P.J. Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol. Cell 2008, 30, 393–402.
- Isaacs, J.S.; Jung, Y.-J.; Mimnaugh, E.G.; Martinez, A.; Cuttitta, F.; Neckers, L.M. Hsp90 regulates a von Hippel Lindau-independent hypoxia-inducible factor-1 alpha-degradative pathway. J. Biol. Chem. 2002, 277, 29936–29944.
- Semenza, G.L. Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene 2010, 29, 625–634.
- Ke, Q.; Costa, M. Hypoxia-inducible factor-1 (HIF-1). Mol. Pharmacol. 2006, 70, 1469–1480.
- Kim, H.J.; Jung, C.L.; Jeong, Y.S.; Kim, J.-S. Soybean-derived glyceollins induce apoptosis through ROS generation. Food Funct. 2014, 5, 688–695.
- Kim, B.R.; Seo, J.-Y.; Sung, M.-K.; Park, J.H.Y.; Suh, H.-J.; Liu, K.H.; Kim, J.-S. Suppression of 7,12-dimethylbenz(a)anthracene-induced mammary tumorigenesis by glyceollins. Mol. Nutr. Food Res. 2015, 59, 907–917.
