Magnesium is one of the most abundant elements on Earth and represents approximately 2.5% of its composition. Magnesium and its alloys are lightweight metallic structural materials with certain advantages, such as low density, high specific strength and high stiffness [
1,
2]. Magnesium and its alloys are considered to have great application prospects in the aerospace, transportation, electronics, biomedicine, and energy fields due to their excellent physical and chemical properties, such as low density, good damping performance, biocompatibility, recyclability, large hydrogen storage capacity, and high theoretical specific capacity [
3,
4,
5]. As the lightest structural material currently available, magnesium alloys have the potential to replace steel and aluminum in many structural applications [
6]. Thus, magnesium alloys have already found considerable applications in various fields, including the aerospace, aircraft, automotive, and electronics fields; in particular, magnesium die castings have been widely used in the automotive industry [
7]. However, despite the abovementioned advantages, magnesium and its alloys still face many difficulties in large-scale industrial applications. For instance, the poor room temperature plasticity and poor corrosion resistance of these materials still need to be addressed [
8,
9]. Currently, magnesium alloy development is focused on the production of complex structures with high efficiency and minimal environmental impact; accordingly, many new magnesium alloy preparation technologies have emerged.
Additive manufacturing (AM) is a promising new technology that can dramatically change the way components are manufactured in many different industries and greatly increase manufacturing efficiency. According to different technologies, AM can be divided into electron beam melting (EBM), direct laser forming (DLF) and selective laser melting (SLM). SLM is a technology widely used in the preparation of metal powders. Commonly used metal powders include Fe-based alloys, titanium-based alloys, Al-based alloys, Mg-based alloys, and nickel-based alloys. SLM uses a high-energy laser beam to completely melt metal powder in a protective atmosphere along a defined laser path, and this molten metal rapidly solidifies [
10]. By repeating this step and overlapping subsequent layers, a three-dimensional component is eventually formed. SLM provides a means of manufacturing geometrically complex structures, eliminating the need to build molds, which would otherwise require a considerable amount of time and money to manufacture. The cooling rate of the molten pool reaches 10
3–10
8 K/s due to the rapid movement of the laser and the molten metal pool [
11]. The characteristics of rapid solidification and layered manufacturing enable the SLM process to produce materials with a more uniform chemical composition, a more concentrated solid solution, a refined microstructure, and better mechanical properties [
12,
13]. However, due to the inherent heat treatment of the SLM process, wherein each layer is cyclically reheated by the deposition of subsequent layers, the microstructure of the material is also unique [
14]. To date, the samples produced by SLM still have some defects, such as pores and cracks, which affect the final use of the produced parts. Therefore, how to control the material density, microstructure and performance by adjusting the SLM process conditions is a research focus.
Due to several characteristics of magnesium alloys, including a low melting point, easy oxidation, and dangerous production, research on the preparation of these materials is still in its infancy worldwide. Research institutions investigating these materials include the Fraunhofer Institute for Laser Technology, Huazhong University of Science and Technology, University of Science and Technology Beijing, Central South University, Université de Technologie de Belfort-Montbéliard, Chongqing University, Hong Kong Polytechnic University, Suzhou University, and Delft University of Technology. Current research is in the small-scale trial production stage in the laboratory, and research and experimental data on the forming characteristics and mechanical properties of selective laser melted (SLMed) magnesium alloys are scarce. On the one hand, owing to the inherent physical properties of magnesium alloys, the research progress of SLMed magnesium alloys is limited. On the other hand, during the production of SLMed magnesium alloys, the generation of defects and the characteristics of microstructures affect the performance of SLMed magnesium alloys. For instance, in the Mg-Al-Zn(AZ) series of magnesium alloys, the presence of the second phase Mg
17Al
12 limits the mechanical properties—especially the elongation—of the alloy, which restricts the further development of SLMed magnesium alloys. At present, many scholars have conducted research and comprehensive reviews on the spheroidization, defects, porosity, and alloying element loss in magnesium alloys and other metals prepared by SLM [
15,
16,
17,
18,
19,
20,
21]. However, limited processable materials, immature process conditions and metallurgical defects are still problems that magnesium alloys need to face and solve in the SLM process. In the past two years, these three issues of SLMed magnesium alloys have been substantially improved by optimizing process parameters, introducing post treatment and adjusting different alloying elements. However, few review articles have been written in this regard. Therefore, it is necessary to summarize the development of SLMed magnesium alloys from the perspectives of process, element adjustment and post treatment. To improve the machinability of the material, this paper will review the research progress of the addition of alloying elements and the post-treatment to expand the processable magnesium alloy materials. Process conditions and new research progress on relative density, microstructure, mechanical properties and corrosion resistance of SLMed magnesium alloys will be reviewed. In addition, the formation mechanism of metallurgical defects, especially oxidation and cracks, will be discussed and analyzed to provide a reference for the application of SLMed magnesium alloys.
2. Microstructure of SLMed Magnesium Alloy
Because SLM has the characteristics of rapid solidification, the cooling rate can reach 104–105 K∙s−1. The microstructure varied with respect to variations in the cooling rate. The microstructures in the as-cast, sub-rapidly solidified, and rapidly solidified (i.e., SLMed) samples can be compared under different cooling rates.
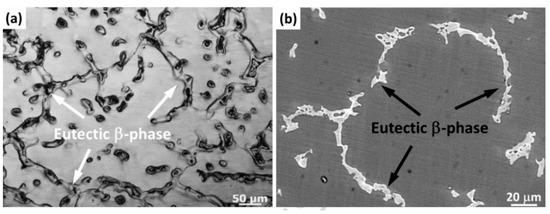
a,b are optical microscopy and SEM images of the as-cast AZ61 alloy [
163]. The as-cast alloy exhibited a typical dendritic eutectic network structure. The phase composition in the as-cast AZ61 was composed of an α-Mg matrix and a β-Mg
17Al
12 eutectic phase distributed in grain boundaries and grains. The average grain size of the as-cast AZ61 alloy was approximately 320 ± 5 μm. Under normal conditions, the grains were coarse and non-uniform. The microstructure of as-cast AZ91D is shown in a,b. As-cast AZ91 also consisted of an α-Mg solid solution and a β-Mg
17Al
12 eutectic phase. As shown in the enlarged micrograph of area A (b), some of the divorced eutectic β-Mg
17Al
12 was surrounded by lamellar eutectic. In the die-cast AZ91D ingot, β precipitates existed in the form of partially divorced eutectic structures. AZ61 and AZ91D are hypoeutectic magnesium aluminum alloys with low zinc content. In hypoeutectic Mg-Al alloys, the morphology of the eutectic phase depends on the cooling rate. A higher cooling rate resulted in a more discrete microstructure [
164]. Therefore, the inherently high cooling rate in the SLM process caused a change in β-Mg
17Al
12 between the SLMed part and the as-cast part of Mg alloy. For die-cast samples, most of the Al and Zn were concentrated in the β-Mg
17Al
12 phase [
165]. The reduction in the Al content in the α-Mg solid solution not only reduced the effect of solid solution strengthening but also deteriorated the corrosion behavior.
Sub-rapid solidification is a nonequilibrium solidification process with a solidification rate of 10
3 K/s [
69]. The sub-rapidly solidified structure changed considerably from that of the as-cast sample (c). The β-Mg
17Al
12 phase continuously distributed on the grain boundaries disappeared. The microstructure was obviously refined and was mainly composed of small equiaxed grains. The literature noted that as the thickness of the sheet decreased (i.e., the faster the solidification process), the grain size decreased [
72], and a large number of petal-like dendrites appeared in the microstructure. Owing to the high cooling rate, the dendrite structure was very small, so it was difficult to distinguish the dendrite arm spacing in the low-magnification metallographic microstructure. Under sub-rapid solidification, the grain size of the AZ61 magnesium alloy was 13.5 μm, which was much smaller than that of the as-cast AZ61 magnesium alloy.
The solidification rate of SLM was faster than that of sub-rapid solidification. As shown in d, the SLMed microstructure was more uniform, the α-Mg grains were refined and equiaxed, and the β-Mg
17Al
12 phase precipitated at the grain boundaries. The α-Mg crystal grains and β-Mg
17Al
12 in the SLMed microstructure were more refined than those in the sub-rapidly solidified microstructures. The grain size in d was 2.46 μm (at an energy density of 208 J/mm
3). Other studies have also found the same microstructural characteristics of SLMed magnesium alloys [
26,
29,
30]. Wei et al. [
26] investigated the distribution of Mg and Al in SLMed AZ91D samples. Comparing the SLMed samples with as-cast AZ91D, the distribution of Mg and Al in the SLMed AZ91D was more uniform, and the content of Al in the matrix varied with respect to the energy density. This finding demonstrates that the chemical composition distribution was more uniform under rapid solidification, which is beneficial to reduce the segregation of the components, and the energy input has an effect on the solid solution [
166].
The rapid solidification of SLM mainly affects the generation of the second phase in the alloy. Cai et al. [
167] compared the microstructure and morphology of the second phase β-Mg
17Al
12 in the conventional as-cast part and in the part produced under rapid solidification. Compared with conventional casting, β-Mg
17Al
12 in the rapidly solidified AZ91 magnesium alloy had smaller and fewer micropores. The dispersive microporosity will act as crack initiation sites and promote crack propagation under the application of an external force, causing significant deterioration in tensile properties. These micropores were mainly formed by solidification shrinkage and dissolved gas. Liu et al. [
28] compared the microstructures under different energy densities under SLM and found that as the energy density increased, the microstructure changed. When the energy density was low, the precipitated second phase was dispersed, and the grain size was small (1.61 μm). As the energy density increased, the second phase gradually precipitated along the grain boundaries, and the grain size slightly increased. Other studies noted that the solid solution was related to the energy input. As the energy input increased, the solid solution of the elements exhibited different monotonic or nonmonotonic changes [
28,
29,
30]. The solid solution of the elements and the precipitation of the second phase were both related to the energy input and solute capture. Therefore, analysis and discussion need to be combined with the solidification path.
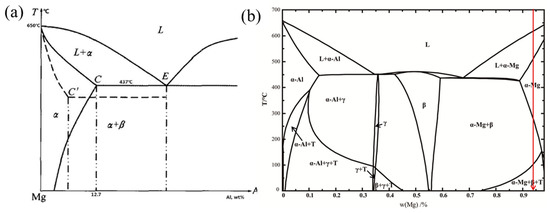
In , the left diagram is the phase diagram of the Mg-Al binary system [
168], whereas the right diagram is the phase diagram of the AZ61 Mg alloy [
28]. The addition of Zn obviously had a greater impact on the Mg-Al binary system phase diagram. Analysis of the equilibrium cooling process of AZ61 shows that the solidification path of the red line in the figure was L → L + α-Mg → α-Mg → α-Mg + β-Mg
17Al
12 → α-Mg + β-Mg
17Al
12 + T.
Taking the AZ series Mg alloy as an example, the maximum solubility of Al in Mg was 12.7 wt.% at 437 °C during equilibrium solidification, whereas the solubility of Al was only 2 wt.% at room temperature. The reaction L → α-Mg occurred first under slow cooling. The Al atoms diffused sufficiently, and the alloy composition in α-Mg gradually became uniform. As the cooling process progressed, the reaction L → α-Mg ended when the melt temperature dropped to the solid phase line. Subsequently, the second phase β-Mg17Al12 began to precipitate in the α-Mg solid solution, and the final solidified products in the equilibrium state were α-Mg solid solution and the second phase β-Mg17Al12, and there was no eutectic phase. However, SLM is a nonequilibrium process under rapid solidification, so the final phase composition of the SLMed part was slightly different from the equilibrium phase diagram. The AZ61 Mg alloy was composed of two phases of α-Mg and β-Mg17Al12 at room temperature during a relatively slow cooling process. At a faster cooling rate (higher scanning speed and lower laser power), the atoms in the primary α-Mg in the liquid phase did not have time to diffuse sufficiently. From the principle of solute redistribution, it is known that the solid solution of Al was continuously enriched at the solidification front, and the residual liquid phase between the dendrites of the α-Mg reached the eutectic composition in the late solidification period. The two phases grew independently to form a divorced eutectic structure. As a result, the temperature of the molten pool was low, and the cooling rate as fast at low energy input (138.89 J/mm3), which is equivalent to being extremely cold in the α-Mg region. Due to the lack of sufficient diffusion time, Al dissolved into the matrix. The solid solution of Al in the matrix increased. Hence, the eutectic transformation process L → α-Mg + β-Mg17Al12 was suppressed. The precipitation of β-Mg17Al12 was less prominent at low energy input. In contrast, due to the accumulation of heat and the decrease in the solidification speed, the solute capture effect was weakened when the energy input was high (156.25~208.33 J/mm3). Al diffused more fully, the content of solid solution elements decreased with increasing Ev, the eutectic transformation L → α-Mg + β-Mg17Al12 occurred, and the crystallinity of β-Mg17Al12 increased.
The change of microstructure (grain shape, size, second phase distribution) is related to the cooling rate. As expected, various laser parameters resulted in different solidification rates of the molten pool and thermal cycles, leading to variation in microstructures of the melted zone. In the SLM process, the combination of scanning speed and laser power is the key to controlling the cooling rate. In addition, the faster the cooling rate, the finer the microstructure; the lower the cooling rate during solidification, the longer the time available for grain coarsening. Generally speaking, as the scanning speed decreases and the laser power increases (that is, the line energy density increases), the accumulation of heat in the molten pool causes the temperature of the molten pool to rise, the cooling rate slows down, and the grain size gradually becomes coarser [
35]. The increase of the laser power or decrease of the laser scanning speed led to the coarsening of the grains in the melted zone because the higher laser power or slower laser scanning speed provided more driving force for grain boundary movement, which then promoted the growth of grains. At lower scanning speeds, the prolonged interaction time between the laser and the powder suppresses the heat dissipation in the molten pool. Therefore, due to the larger heat accumulation, the epitaxial growth kinetic conditions of the grains are enhanced [
16]. Similar results were obtained for pulsed laser samples. When the laser power was low or the scanning speed was high, the average gain size became small, since decreasing the laser power in effect acts in the same way as reducing the preheat temperature of the powder when the next pulse hits the base. The relatively high cooling rate at low laser power or high laser scanning speed restrained the growth of α-Mg grain during solidification.
As the line energy density increases (low scanning speed and high laser power), the microstructure follows the evolution of clustered finer dendrites–refined equiaxed grains–coarsened equiaxed grains [
26,
28]. The grain size of AZ61 magnesium alloy can be refined to 1.61 μm (
Ev = 138.89 J/mm
3). Then, the grains changed to an equiaxed shaped (1.79 μm) with an increase in laser energy input to 156 J/mm
3. Further increase in the laser energy input to 178 J/mm
3 and 208 J/mm
3, equiaxed grains of 2.12 μm and 2.46 μm, respectively [
28]. This is due to the decrease in cooling rate caused by changes in scanning speed and laser power and accumulation of heat in the molten pool. This same reason can explain the change of the grain size far from the center of the molten pool. The α-Mg grains inside the molten pool show equiaxed crystal morphologies. On the contrary, with the farther away from the molten pool, the α-Mg grains show a transformation from equiaxed crystal to columnar crystal and the grain size increases [
38]. The ratio G/R of the temperature gradient (G) and the solidification rate (R: the propulsion rate of the solidification interface in the normal direction) in the crystal direction of the pulsed light spot determines the microstructure morphology after solidification. Inside the molten pool, G is very high, but R tends to 0, so that the value of G/R is high, which causes the solidification structure to be equiaxed crystal. Further away from the molten pool, G decreases but R gradually increases, so that the value of G/R gradually decreases. The influence of scanning speed and laser power on the precipitation of the second phase has been described in detail above.
Consequently, the grain size, the second phase precipitation, and the element solid solution can be achieved by controlling the process parameters, i.e., controlling the scanning speed and laser power (energy density). In terms of grain size, by comparing the microstructure of magnesium alloys at different cooling rates, it can be seen that faster cooling rates are beneficial for refining the microstructure. From the as-cast state to sub-rapid solidification and rapid solidification (SLM), the grain size of the magnesium alloy drops from ~320 μm to ~2 μm, which is a change of two orders of magnitude. Properly increasing the scanning speed and lowering the laser power (i.e., lowering the energy density) is beneficial for the improvement of the cooling rate and the reduction of heat accumulation in the molten pool, thereby refining the microstructure. In terms of second phase precipitation, the increase of the cooling rate is beneficial to suppress the occurrence of the phase transition L → α-Mg + β-Mg17Al12 and the precipitation of the second phase is reduced. In addition, the energy density must be controlled within a suitable range. Too high an energy input will lead to coarse grains, an increase in element segregation and a decrease in the solid solution. However, if the laser power is reduced or the scanning speed is increased blindly (too low an energy density), the powder cannot be fully melted and combined, thus causing serious pores and affecting the sample quality. Therefore, further research must be performed to more accurately control the solidification structure by controlling scanning speed or laser power and the effect of the solidification structure on the defects and mechanical properties of SLMed magnesium alloys.
4. Outlook
The development of SLM of magnesium alloys has been reviewed, and an extensive analysis of the available literature on the preparation of metals via SLM has led to the recognition of the influential process parameters and material properties. At present, the limited processable materials, immature process conditions and metallurgical defects restrict the development and application of SLMed magnesium alloys. Some efforts have been made to solve the above problems, such as adding alloy elements and applying postprocessing. However, the breakthroughs of SLMed magnesium alloys in these two areas have not been reviewed. Therefore, this article gives an overview of these three issues. In this paper, the process parameters, alloying elements, microstructure, properties, and postprocessing steps were systematically summarized. For the purpose of high efficiency, high quality, low cost, and more stable use of SLM to prepare high-performance magnesium alloys, scholars have carried out research on process parameters, alloying elements, and post-treatment steps. Although some progress has been made in terms of the relative density, mechanical properties, and corrosion resistance of magnesium alloys, the literature is still limited to unsystematic performance studies of a few types of magnesium alloys. Hence, current SLMed parts are produced under suboptimal process conditions, wherein it is difficult to control metallurgical defects.
In the future, there are still several issues that need attention in the manufacturing of magnesium alloys via SLM.
First of all, in view of the lack of processable materials, in order to expand more processable SLMed magnesium alloy materials and improve the problem of limited processable materials, we reviewed the progress of SLMed magnesium alloys in the addition of alloying elements, but there are still two problems in this area: the types of added alloying elements are limited, and the research of the addition of alloying elements needs to be in-depth. At present, most studies on SLM fabrication of magnesium alloys focus on the influence of process parameters and simple alloying elements on the microstructure and properties of binary alloys. Aside from research on Mg-Al-Zn alloys, Mg-Zn-Zr alloys, and Mg-Zn alloys, few studies have been reported on other as-cast Mg alloys and wrought Mg alloys, especially the high-strength and highly corrosion-resistant Mg alloys that are in great demand in the aerospace field (e.g., Mg-Mn alloys). We also combined the relevant research on the mature alloy element addition route in the traditional process to provide a reference for the alloy design of SLMed magnesium alloys, such as Mn and Ca, and the addition of these elements in SLMed magnesium alloys has rarely been reported. Therefore, there is a need to broaden the scope of the applicable magnesium alloys for SLM and design new materials whose compositions are suitable for the process characteristics of laser AM. In addition, post-treatment can also be applied to improve the machinability of magnesium alloys in the SLM process. We reviewed the current research progress in the postprocessing part, and the results showed that the post-processing can effectively improve the properties of SLM magnesium alloys, especially to improve the plasticity, but there are few studies at present, which is an area that needs to be further studied in the future. Traditional preparation techniques of magnesium alloys are also evaluated and related to the SLM process with a view to gaining useful insights especially with respect to postprocessing.
Second, in view of the immature process conditions, we comprehensively reviewed the new progress of different magnesium alloy materials in SLM process conditions and forming, and combined process conditions with relative density, grain size, microstructure, mechanical properties, and corrosion resistance. In the review of microstructure, the microstructure under different preparation processes (different cooling rates) was compared longitudinally, and the microstructure under different process conditions in the SLM process was also compared horizontally. In addition, it is different from the previous reviews of SLMed magnesium alloy.
Third, in terms of metallurgical defects, defects such as oxidation and cracks are reviewed. Research on cracks has made progress recently, but there is lack of review of the latest research on cracks. Cracks are a serious defect that limits the application of magnesium alloys and leads to premature failure of materials. Therefore, it is necessary to review the research progress of SLMed magnesium alloy cracks. It is well known that the application of SLMed Al alloy has been limited by cracks, so the paper combines the research progress of SLMed Al alloys on cracks to provide more references for the research of SLMed magnesium alloy cracks. In terms of oxidation, inspired by the purity of molten steel, this review not only looks forward to the development of SLMed magnesium alloys from the perspective of materials and processing, but also considers the future improvement direction of SLMed magnesium alloys from a new perspective—a metallurgical perspective. In addition, this was mentioned in the previous review of SLMed magnesium alloys. In research on steel materials, the harmful effect of nonmetallic inclusions on steel properties has been widely recognized [
180,
181]. For bearing steel, the deoxidation and purification of liquid steel has always been the focus of attention. Research has found that when the total oxygen content in molten steel is reduced from 0.0026 to 0.001%, the fatigue life of bearing steel is increased by a factor of 10, and when the total oxygen content is reduced from 0.001 to 0.0004%, the fatigue life is increased by an additional factor of 10. Therefore, to improve the performance of steel materials, it is important is to minimize both the dissolved oxygen in liquid steel and the content of oxide inclusions generated during the solidification process [
182,
183]. There have been many studies on the harmful effects of high oxygen content on material properties in powder metallurgy, such as 718 alloy [
104], Al alloy [
103], and titanium alloy [
184]. Due to the highly oxyphilic nature of magnesium, the oxygen inevitably enters the magnesium alloy base metal and the magnesium alloy powder during the preparation process. However, the amount of oxygen entering the alloy system at various stages, the size and number of oxide inclusions formed in the alloy, and the mechanism of the influence of inclusions on the alloy—especially the influence of inclusions in the performance of magnesium alloys—have rarely been studied. Currently available research is limited to the understanding of the oxide film in the SLM process. It was found that the oxide film is usually located at the top of the molten pool, and the oxide film on the surface will affect the wettability, thereby weakening the interlayer bonding and resulting in the formation of defects. Improving the oxidation also means reducing the defects in the material. It was found that the use of a high-power laser could break the oxide film on the surface, but this increased the burning loss of alloying elements, and balancing the contradiction between these two aspects is a new topic. From the perspective of the preparation process of magnesium alloys, the oxygen that can enter into the alloy mainly comes from the mold cavity and the powder preparation process as well as the smelting of the magnesium base material. Future studies should focus on methods to inhibit the entry of oxygen in these processes and the mechanism of the influence of a small amount of oxygen on SLMed magnesium alloy materials.
In the future, the physical and chemical reactions in the SLMed magnesium alloy process need to be studied theoretically, especially the thermodynamics and kinetics of the formation of intermetallic compounds. For example, the thermodynamic calculation of the enthalpy of fusion of SLMed magnesium alloys with different compositions can theoretically determine the energy required for the magnesium alloy powder of a specific composition, which provides a theoretical basis for setting the laser power for actual experimental production (i.e., this approach can help ensure that the powder is melted under the optimal energy setting). In terms of dynamics research, it is necessary to establish a model of the relationship between the expansion and solidification of magnesium melt to seek the optimal energy input, control the melt temperature, and obtain the relationship between magnesium melt spreading and solidification rate to reduce the occurrence of defects, such as pores and spheroidization.
Furthermore, the influence of intermetallic compounds on SLMed magnesium alloys cannot be ignored. Intermetallic compounds have a dual effect on SLMed magnesium alloys. On the one hand, refining and dispersing intermetallic compounds can play a role in precipitation strengthening; on the other hand, intermetallic compounds can also have an adverse effect on corrosion resistance and mechanical properties. Therefore, controlling the size, shape and content of intermetallic compounds should be studied in further detail in the future. It is necessary to proceed from the theory of metallurgical physical chemistry and seek the conditions and mechanisms for the formation or decomposition of intermetallic compounds from the liquid state to the normal temperature state and finally obtain the best process parameters for controlling the intermetallic compounds in the SLM process, including an appropriate heat treatment method. According to the theoretical basis of postprocessing SLMed magnesium alloys presented above, there is still much research space in this aspect.