Metal/carbon nanotube (CNT) composites are promising functional materials due to the various superior properties of CNTs in addition to the characteristics of metals. Electrochemical deposition can be classified into three types: (1) composite plating by electrodeposition or electroless deposition, (2) metal coating on CNT by electroless deposition, and (3) electrodeposition using CNT templates, such as CNT sheets and CNT yarns.
- metal/carbon nanotube composite
- electrochemical deposition
- electrodeposition
- electroless deposition
- composite plating
- carbon nanotube sheet
- carbon nanotube yarn
1. Introduction
2. Fabrication of Metal/CNT Composites Using Composite Plating by Electrodeposition or Electro Less Deposition
2.1. Composite Plating
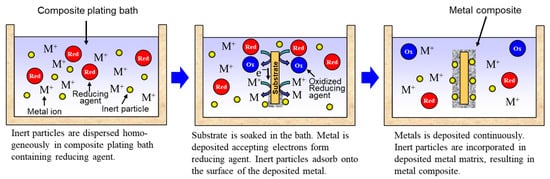
Rough schematics of composite plating by electrodeposition and electroless deposition are displayed in Figure 3 and Figure 4, respectively. In the case of electrodeposition, inert particles are dispersed homogeneously in a plating bath. When a voltage is applied, metal is electrodeposited on a cathode and the particles adsorb on the surface of the deposited metal. Then, the particles are embedded in depositing metal, resulting in a metal composite (Figure 3). In the case of CNT composite plating by electrodeposition, inert particles are dispersed homogeneously in a plating bath containing a reducing agent. When a substrate is soaked in the bath, metal is reductively deposited on the substrate accepting electrons from the reducing agent and, at the same time, the particles adsorb on the surface of the deposited metal. The particles are then embedded in depositing metal, resulting in a metal composite (Figure 4). In general, the substrate is pre-treated and catalyst particles, such as Pd particles, are fixed on the surface of the substrate before soaking into the plating bath. As far as was searched, the first article of the composite plating is on Cu/graphite composites by electrodeposition and was reported in 1928 [4]. Regarding the mechanism of the composite plating, several models have been proposed [5,6,7,8,9].
2.2. Preparation of Plating Bath for Metal/CNT Composite Plating
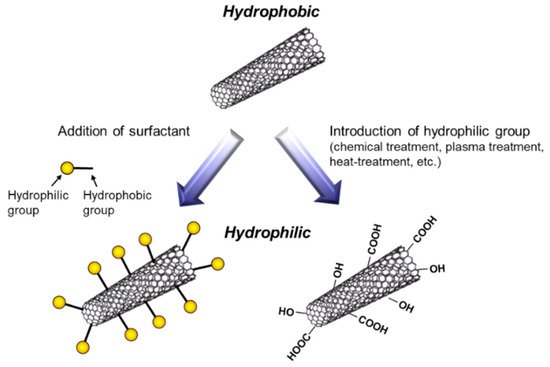
To fabricate metal/CNT composites with uniform distribution of CNTs, the preparation of plating baths with homogeneous dispersion of CNTs is important. In general, plating baths are aqueous solutions, while CNTs are hydrophobic. Therefore, hydrophilization of CNTs have been examined by the addition of surfactants or the direct introduction of hydrophilic groups on the surfaces of CNTs (Figure 5). The addition of surfactants in plating baths is a common method. Various kinds of surfactants [11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28], such as sodium dodecylbenzene sulfonate and sodium deoxycholate, have been examined for the homogeneous dispersion of CNTs in a pure water. However, effective surfactants for the dispersion in a pure water are not always effective in plating baths which contain great amounts of ions. Moreover, even if the surfactant is effective for the dispersion of CNTs in a plating bath, CNTs are not always co-deposited by electrochemical deposition. Therefore, the selection of appropriate surfactants is essential. Since the surfactants are likely incorporated in deposited metal matrix during electrochemical deposition, the concentration of surfactants should be examined. On the contrary, the direct introduction of hydrophilic groups, such as -COOH, onto the surfaces of CNTs has been examined using a chemical treatment [29], a plasma treatment [30], a heat treatment [31], and so on. These methods destroy the sp2 carbon bonding of the surfaces of CNTs. Therefore, the conditions of the treatments should be examined.
2.3. Unique Feature of Composite Plating Using CNTs as Inert Particles
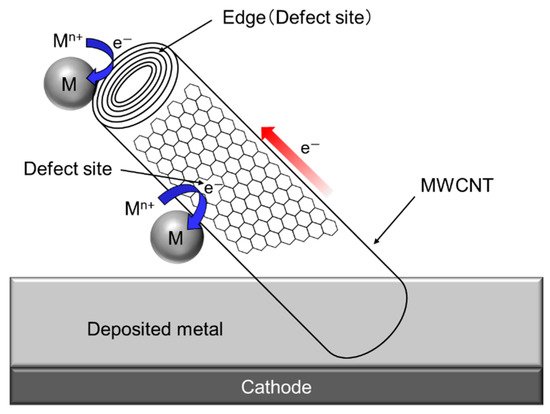
Since a single CNT, especially multi-walled CNT (MWCNT) has a fibrous shape with large aspect ratio in addition to a high electrical conductivity in the axis direction. Therefore, composite plating using CNTs as inert particles often shows a unique feature unlike other composite plating using insulation particles such as Al2O3 particles. The schematic of the unique feature is showed in Figure 7 [34]. When a part of a MWCNT is incorporated in the deposited metal matrix during electrodeposition, the metal can be electrodeposited not only on the deposited metal but also on the protruding edge (a defect site) of the MWCNT. If the defect sites exist on the sidewall of the MWCNT, the metal can also be electrodeposited on the defect sites.

2.4. Fabrication of Metal/CNT Composites Using Composite Plating by Electrodeposition
Fabrication conditions in these articles are listed in Table 1.
2.5. Fabrication of Metal/CNT Composites Using Composite Plating by Electroless Deposition
Regarding the number of published articles on metal/CNT composite plating using electroless deposition, those on the Ni-P alloy/CNT is large. In the case of electroless deposition of Ni, phosphorous compounds such as sodium hypophosphite (NaH2PO2) are usually used as the reducing agent and the P derived from the NaH2PO2 is co-deposited with Ni, resulting in Ni-P alloy deposit. Most of the purpose of the fabrication of Ni-P alloy/CNT composites is the improvement of tribological properties. Fabrication conditions in these articles are listed in Table 2.
3. Metal-Coated CNTs by Electroless Deposition
3.1. Fabrication Process
A fabrication process of metal-coated CNTs by an autocatalytic electroless deposition is schematically showed in Figure 13. Even in the case of electroless deposition, homogeneous dispersion of CNTs in the plating bath is important. The introduction of functional groups on the surface of CNTs likely effective to increase deposition sites, resulting in CNTs coated by metal films and not metal particles.
3.2. Metal-Coated CNTs
Fabrication conditions in these articles are listed in Table 3.
4. Metal/CNT Composites by Electrodeposition Using CNT Templates (Sheet, Yarn)
CNT templates, such as CNT sheets [147,148,149,150] and CNT yarns or fibers [151,152,153,154], have been developed and their various practical applications have been researched. Although a single CNT has a high electrical conductivity, electrical conductivities of those templates are far less than metals such as Cu, due to the contact resistance between each CNT of which they consist. Therefore, metallization of the CNT templates is a promising process to give them enough electrical conductivity. On the contrary, CNTs have strong anisotropy in electrical and thermal properties [155]. Therefore, the orientation of CNTs which make up the templates is also important in order to achieve the expected properties of metal/CNT composites. Fabrication conditions in these articles are listed in Table 4.
5. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/electrochem2040036
