Graphene quantum dots (GQDs) are zero-dimensional carbon-based materials, while nanocellulose is a nanomaterial that can be derived from naturally occurring cellulose polymers or renewable biomass resources. The unique geometrical, biocompatible, and biodegradable properties of both these remarkable nanomaterials have caught the attention of the scientific community in terms of fundamental research to advanced technology. Studies have shown that the hybridisation of these novel materials not only improves existing applications but provides additional advantages as well as further improves desirable features, all of which are unattainable if GQDs and nanocellulose are used individually. Therefore, this advantageous composite material warrants remarkable applications. Potential applications for GQDs-nanocellulose composites include sensing or for analytical purposes, injectable 3D printing materials, supercapacitors, and light-emitting diodes.
1. Introduction
Over the past few years, many researchers have developed an interest in investigating carbon-based nanomaterials, such as graphene and graphene quantum dots (GQDs) [
1,
2,
3,
4]. Graphene is one layer of sp
2-hybridised carbon atoms arranged in a honeycomb lattice [
5], whereas GQDs are chopped fragments of a few graphene sheets (<10 nm lateral dimension) and <10 graphene layers which form final particles [
6]. GQDs are used in various applications owing to their higher photostability, low cytotoxicity, strong photoluminescence, dispersibility in water, fluorescence and excellent biocompatibility [
7,
8]. Furthermore, the characteristics of GQDs vary according to morphology, size, doping concentration and type [
9]. Owing to these properties and the ability to fine-tune them, GQDs are investigated for different applications in biomedicine [
10], catalyst development [
11], energy [
12] and sensing and photo electronics [
6]. Different bottom-up and top-down strategies have been used to produce GQDs, such as organic synthesis [
13], hydrothermal [
14], microwave irradiation [
15], chemical exfoliation [
16] and electrochemical exfoliation [
3,
17].
Furthermore, many researchers have also conducted extensive carbohydrate polymer research using cellulose-based nanomaterials. Cellulose is an abundantly available, naturally occurring organic polymer that is composed of repeating units of
β-glucopyranose rings that are covalently linked to one another with a
β 1-4 glycosidic bond [
18,
19]. Cellulose nanowhiskers, or nanocellulose, are cellulose particles present in the form of crystals or fibres [
20]. These particles are few micrometres in length and have a diameter <100 nm. These fibres are lightweight, biodegradable and also have a higher water-binding capacity [
21]. Cellulosic nanomaterials display a larger specific surface area; therefore, they can form many hydrogen bonds. This hydrogen bond-forming capability helps the material develop a dense and strong network [
22]. Many different kinds of nano-cellulosic materials are described in the literature, such as cellulose nanocrystals (CNCs) [
23], nanofibrillated cellulose (NFCs) [
24] and bacterial nanocellulose (BNCs) [
25]. As nanocellulose can be derived from different cellulosic sources, each displays varying properties and some are derived from natural sources. For instance, the properties of the bacterial nanocelluloses are based on different bacterial sources, whereas the properties of the cellulose nanocrystals and nanofibrillated celluloses are based on sources such as tunicin or plants [
26]. Nanocellulose has garnered significant research interest as a promising nanomaterial that can revolutionise multiple fields, such as the pharmaceutical field [
27], engineering [
28], electronics [
29] and health and environmental protection [
30].
GQD-nanocellulose is described as a hybrid material that contains nanocellulose and GQDs, which synergistically improves the properties of every individual component, such as their stability and mechanical strength [
31]. The addition of GQDs to a nanocomposite material can improve its final tensile strength, stiffness and the toughness of these GQDs–nanocellulose structures, irrespective of their GQD oxidation type and nanocellulose orientation [
32]. These structures are designed to exhibit higher conductivity, cycle stability and higher specific capacitance [
32]. Therefore, GQDs–nanocellulose composites are designed for use in many applications, such as fluorescence films, bendable and portable paper electronics, and hydrogels. The numerous -COOH and -OH functional groups are present on the surfaces of GQDs result in the formation of hydrogen bonds at the GQDs–nanocellulose interface. This can significantly affect the GQDs–nanocellulose supercells, nanocellulose lattice parameters and the morphological properties of nanocellulose, such as the dihedral angle differences in the hydroxymethyl groups, axial tilt of molecular chains and the flipping motion of terminal groups [
33]. In addition, the composites made from these renewable nanomaterials offer a greener approach than petroleum-derived composites and exhibit great potential for various technological applications [
34]. Moreover, this nanomaterial combination has recently emerged as a new class of hybrid material due to its exceptional features and notable synergistic effects [
35].
1.1. Graphene Quantum Dots (GQDs)
GQDs hold sp
2 hybridised carbon single-layer nanocrystals and they are highly fluorescent, regardless of whether they are in an aqueous or solid state [
36,
37]. GQDs are a distinct from the type of carbon in carbon nanodots (CNDs) and polymer dots (PDs). This is because all carbon dots possess modified chemical groups, such as oxygen groups, on the surface [
38]. However, each of them is of a different size and possesses different properties with which to perform their action. GQDs have an average lattice parameter of 0.24 nm, which corresponds to 100 in plane graphene lattice parameters [
39]. On the contrary, CNDs are divided into carbon nanoparticles without a crystal lattice and have a spherical shape [
40], while the grafted and cross-linked polymer chains of linear non-conjugated polymers form PDs [
36].
GQDs are the simplest carbon dots with connected chemical groups on the surfaces or edges [
41]. The surfaces or edges of GQDs contain triple carbene at the zigzag edges and oxygen groups at the graphene core. Additionally, the type of GQD edge plays a significant role in determining the material’s optical, electronic and magnetic properties [
36]. GQDs are produced using either the top-down or bottom-up method. Both approaches use different parameters to produce GQDs [
36]. The top-down method involves direct cutting of graphite or graphene-based materials via acid exfoliation [
16], sonochemistry [
42], solvothermal synthesis [
43], electrochemistry [
44,
45,
46,
47], or chemical oxidation [
48,
49,
50]. The advantages of these methods are an abundance of inexpensive precursor (graphite), the high graphitic nature and the formation of GQDs with high oxygen-containing functional groups, which renders good solubility and functionality. However, the drawbacks of these methods include harsh a reaction procedure, as well as the non-uniform size and thickness of the final product [
51]. Examples of the bottom-up method of GQD production from molecular precursors include cyclodehydrogenation [
52], pyrolysis [
53] and solution chemistry [
13]. Despite the difficult synthesis processes, the bottom-up approaches provide better control over the size and shape of the GQDs.
Graphene is widely used in many applications, such as electronics, solar cells and Li-ion batteries [
54], whereas GQDs have attracted tremendous interest in photoluminescence [
55], cell-imaging [
56] and drug delivery [
57,
58], among many things. GQDs have a great advantage, as their properties can be adjusted by changing their band via doping to produce amine-functionalised GQDs [
59], nitrogen-doped GQDs (N-GQDs) [
60] and sulphur–nitrogen co-doped GQDs (S, N-GQDs) [
61]. Furthermore, the photoluminescence colour of GQDs can also be changed from violet to yellow by setting the reactant concentration and temperature during the hydrothermal method [
54].
1.2. Nanocellulose
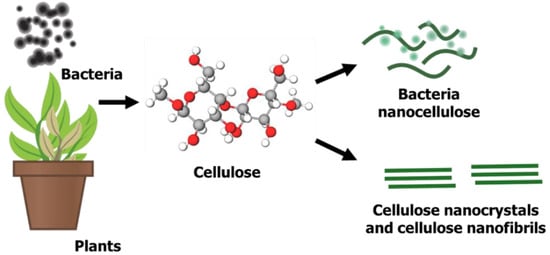
Nanocellulose, a sustainable and renewable nano-structured cellulose, has gained tremendous attention for its potential use in many applications due to its excellent surface chemistry, physical properties and remarkable biological properties [
62,
63]. Nanocellulose was first prepared in 1947 using sulfuric acid and hydrochloric acid hydrolysis from wood fibres and cotton fibres [
64]. As illustrated in
Figure 1, there are three types of nanocellulose from different precursors, bacteria nanocellulose, cellulose nanocrystals (CNCs) and cellulose nanofibrils (CNF) [
65,
66], which can be obtained from cellulose-containing precursors such as plants or bacteria. Plant cellulose is situated within a plant’s fibre walls, whereas bacteria produce exopolysaccharides to form microbial cellulose [
67]. These bacterial isolates can normally be obtained from rotten vegetables and fruits. Similar to plant-derived cellulose, some parameters or conditions, such as carbon source, nitrogen source, temperature, pH and agitation, are measured to produce a high yield of bacterial cellulose [
68,
69]. Bacterial nanocellulose is pure in nature and has a low cytotoxicity, high pore distribution and high hydrophilicity due to the presence of OH groups on its surface [
70,
71]. However, unlike bacterial cellulose, plant cellulose is impure due to the presence of lignin, hemicellulose and pectin. Apart from that, plant cellulose is slightly cytotoxic, less malleable and has small pore sizes due to less space between fibrils [
72].
Figure 1. Formation of nanocellulose from cellulose-containing materials.
Nanocellulose can be prepared via mechanical or chemical methods. The most commonly used mechanical methods include high intensity ultrasonication, high-pressure homogenisation, micro-grinding and PFI milling [
62]. However, these processes require high-energy consumption [
73,
74]. Chemical methods of nanocellulose preparation, such as acid hydrolysis, enzyme hydrolysis and (2,2,6,6-tetramethylpiperidin-1-yl) oxidanyl (TEMPO) oxidation, have also been rigorously employed. Yet, the drawbacks of the chemical methods are that they are time-consuming and yield low results [
75]. Therefore, chemical and mechanical methods have been combined to overcome these issues, as well as reduce energy consumption. Acid hydrolysis is the most common type of this method used to date. The hydrolysis method reduces the sizes of nanofibres from microns to nanometres [
76,
77,
78,
79]. However, this method is not without drawbacks, such as the use of high concentrations of acid, which leads to acid waste and considerably adverse effects on the environment.
2. Preparation of Graphene Quantum Dots–Nanocellulose Composites
Over the past few years, GQDs–nanocellulose composites have been used in a variety of applications that have since generated a lot of interest in their synthesis and composite processing. Multiple studies have proven that GQDs–nanocellulose composites perform better than both GQDs and nanocellulose on their own. Different preparation techniques could impart different abilities to a composite as the preparation technique can affect the particle size, as well as the mechanical properties [
80]. However, the major issues encountered when preparing nanocellulose composites include enhancing compatibility with hydrophobic polymers, uniform dispersion within the matrix and large-scale production [
81]. Although melt-blending and in situ techniques can be employed during composite preparation, solution blending is the most common technique of GQDs–nanocellulose composite preparation. However, specific conditions, such as maintaining the temperature of the substrate, solution and solvent, are required during solution blending. Furthermore, material concentration and pH also affect the efficacy of the final composite.
Solution blending facilitates molecular mixing, which ensures that a composite is soluble in the appropriate solution. According to Ruiz-Palomero, Benítez-Martínez et al., GQDs–nanocellulose hydrogel particles can be prepared by combining carboxylated nanocellulose into sulphur–nitrogen co-doped GQDs (S, N-GQDs) [
30]. Another study found that immersing S, N-GQDs synthesized via a hydrothermal process after using thiourea as the S and N source and citric acid as the source of carbon into the nanocellulose improved the penetration of the excitation light in the hydrogel composite and maintained higher fluorescence energy [
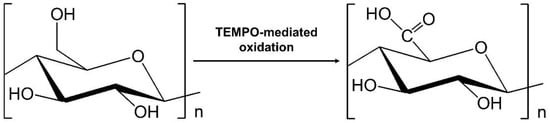
82]. Carboxylated nanocellulose can be prepared using (2,2,6,6-tetramethylpiperidine-1-oxyl) TEMPO-radical catalysed oxidation. TEMPO was found to selectively oxidise the C6 primary alcohol moiety that is present on the surface of the nanocellulose particles and formed carboxylated groups [
83], as presented in
Figure 2.
Figure 2. Oxidation of C6 hydroxyl group of cellulose in a TEMPO-system.
The regioselective conversion of the primary hydroxyl group to carboxylate weakens the adhesion between nanocellulose fibrils, as it prevents the formation of interfibrillar hydrogen bonds [
84]. The TEMPO-mediated oxidation of the cellulose slurry starts to form when the NaOCl solution is added at room temperature and the pH is maintained at 10. The temperature is maintained at 25 °C, as the contents of carboxyl groups increases as the temperature increases up to only 25 °C and subsequently decreases at 35 °C. As such, optimum temperature is critical to form complete reactions, as increases in temperature decrease carboxyl group content, leading to de-polymerisation and losses in total yield. Meanwhile, lower temperatures may cause incomplete reactions to occur, since unreacted NaOCl forms and NaOH needs to be added to the solution to neutralise the generated carboxylic acid groups and maintain a pH of 10. The amount of NaOH added is gradually increased and remains constant after a certain time, once the limiting reactant, NaOCl, is fully consumed [
82]. This technique provides a more uniform dispersion of nanocellulose in an aqueous phase. After conducting an inversion test on the composite hydrogels, Ruiz-Palomero, Benítez-Martínez et al. found that the hydrogels could be reformed several times as non-covalent interactions were involved [
30].
The same researchers also reported a similar GQDs–nanocellulose hydrogel preparation procedure for sensing 2,4,5-trichlorophenol [
31]. In short, the carboxylated nanocellulose was mixed with an aqueous solvent that contained S, N-GQDs, where the nanocellulose acted as a gelator. The sample was mixed using a vortex and sonication, then centrifuged for 0.5 min at 1300 rpm and heated for 20 s in a vial. The transparent hydrogel was observed following cooling to room temperature after the last centrifugation stage. An inversion test was used to assess gel formation. Due to the interacting surface hydroxyl and carboxyl groups, the carboxylated nanocellulose was found to be an effective gelator. This caused a significant self-association that resulted in nano-fibre entanglement due to hydrogen bonding. Therefore, nanocellulose is a suitable gel matrix for hosting GQDs. The study also found that 10
wt% of carboxylated nanocellulose provided the most stable hydrogel when mixed with the S, N-GQDs. This successful gel formation and stability confirmed the compatibility and suitability of the nanocellulose in hosting the GQDs and that it was potentially capable of detecting various analytes. This was due to its exceptional optical features which were accentuated by the network formation combined with the photoluminescence behaviour of the GQDs, which rendered them suitable for analytical purposes.
Apart from nanocellulose and GQDs concentration, the pH of the solvent containing GQDs and the doping characteristics of the GQDs play a significant role in the successful formation of a GQDs–nanocellulose hydrogel composite. The integrity of a composite structure can be weak in some cases due to the pH of the media or material concentration. For instance, Ruiz-Palomero, Soriano et al. [
31] reported that only a solution of dialysed GQDs that had a neutral pH could support gel formation. Moreover, pure undoped GQDs produced weak gels, while N-doped GQDs did not form hydrogels even after purification. However, the S-GQDs produced low photoluminescence albeit gels. Doping GQDs with a combination of S and N heteroatoms (S, N-GQDs) produced the most stable gels with strong fluorescence features. On the other hand, the concentration of GQDs needed to be optimised as a very high concentration may de-stabilise the hydrogen bonding network via additional π–π stacking interactions. The study revealed that an 8 mg mL
−1 concentration of S, N-GQDs and 10
wt% carboxylated nanocellulose produced the best and strongest hydrogel with the highest photoluminescent features.
Tetsuka et al. [
85] was one of the earliest attempts at producing a GQDs–nanocellulose composite via solvent blending by producing a transparent clay film comprising of amino-functionalised GQDs (af-GQDs) and cellulose nanofibrils (CNF) as a colour converting material for blue light-emitting diode (LED) [
85]. Treating the heavy oxidised graphene sheets (OGSs) with a mild amino-hydrothermal method produced uniform-sized af-GQDs with a tuneable photoluminescence. The amino groups were found to alter the electronic structures and shift the HOMO levels to a higher energy with a maximal photoluminescence at the long wavelength [
85]. The af-GQDs–CNF composite was prepared by mixing the CNF suspension with aqueous solution containing af-GQDs. A clay suspension was prepared by using a high shear mixer to disperse clay in a solution containing 15
wt% of the af-GQDs–CNF. The mixture was then degassed and centrifuged to remove any flocculated clay impurities before it was poured on a glass mould. It was then heated at 60 °C overnight and detached from the glass mould to produce af-GQDs–CNF clay film. Unlike Ruiz-Palomero, Soriano et al. [
31], Tetsuka et al. [
85] introduced the incorporation of clay in the composite matrix via electrostatic interactions between the clay and af-GQDs–CNF to form a flexible and transparent film. The resultant film exhibited bright colourful photoluminescence and is a promising future light emitting diode application.
Khabibullin et al. [
86] produced injectable fluorescent hydrogels composed of GQDs and cellulose nanocrystals (CNCs). Unlike Ruiz-Palomero, Soriano, et al. [
31] and Tetsuka et al. [
85], who used the hydrothermal method, Khabibullin et al. [
86] employed the Hummers method to prepare the GQDs. The CNCs were functionalised with amino groups before being subjected to the composite hydrogel preparation. The hydrogel was prepared by dispersing the powdered GQDs in aqueous solution containing CNCs. The sample was mixed via vortex, then left to rest. The time of hydrogel formation varied depending on the concentration of GQDs and CNCs. The sample with 50 mg/mL of CNCs and 7 mg/mL of GQDs yielded a strong hydrogel formation (gelation within 30 min), while the sample with lower concentrations of CNCs and GQDs yielded a weak hydrogel formation (gelation within four hours) and the sample with <20 mg/mL CNCs and <5 mg/mL GQDs yielded no hydrogel formation. Similar to the neutral pH that facilitated hydrogel formation in Ruiz-Palomero, Soriano, et al.’s [
31] study, the pH used for the preparation of hydrogel was maintained at seven. As the produced GQDs–CNCs composite possessed a shear thinning behaviour, it was a suitable injectable material for 3D printing with additional fluorescence features.
Alizadehgiashi et al. [
8] also employed a solution mixing procedure for the preparation of GQDs–nanocellulose composites. Similar to Tetsuka et al. [
85], Alizadehgiashi et al. [
8] utilised amino-functionalised GQDs (af-GQDs) that had been prepared using the hydrothermal method, except that an aldehyde-modified CNCs was used for the composite hydrogel formation instead. The composite hydrogel was prepared by mixing various concentrations of aldehyde-functionalised CNCs (from 10 to 60 mg/mL) and af-GQDs (from 2.5 to 60 mg/mL) suspensions in various volumetric ratios, which may change the structure of the composite from lamellar to nanofibrillar, as well as enabling the controlling of the permeability of the hydrogel. A thick lamellar structure (large pores) was observed at a lower CNC concentration ratio, while a nanofibrillar structure (small pores) was observed at a high CNC concentration ratio (low af-GQDs content). The transition of the structure can be attributed to the number of crosslinking points available (higher CNC content imparts fewer crosslinking points), which, in turn, determines the thickness of the wall and size of the pores. In terms of permeability, the hydrogel composite with lower CNC content (lamellar structure) had higher permeability than the composite with higher CNC content (nanofibrillar structure) due to the larger pore size [
87].
On the other hand, Rosddi et al. [
88] produced a GQDs–nanocellulose composite by dissolving carboxylated GQDs in an aqueous solution containing cationically modified CNCs. A thin film of the composite was then formed using the spin coating technique and the composite showed potential in analytical applications. The same researchers later reported the modification of surface plasmon resonance gold film with carboxylated GQDs–CNCs to enhance the detection sensitivity of glucose [
89]. Similar to Ruiz-Palomero, Benítez-Martínez, et al. [
30] and Ruiz-Palomero, Soriano, et al. [
31], Mahmoud et al. [
90] prepared GQDs–nanocellulose using S, N-GQDs, except that the nanocellulose used was not modified with the carboxyl group and no hydrogel formation was reported. The GQDs–nanocellulose was prepared by mixing of equal concentrations of S, N-GQDs and nanocellulose (3 mg/mL each). The composite was then sonicated and drop-casted onto glassy carbon electrode (GCE) to fabricate a modified GCE sensor. More recently, Xiong et al. [
91] prepared a flexible GQDs–nanocellulose film via a combination of electrolysis and liquid dispersion for sensor and supercapacitor applications.
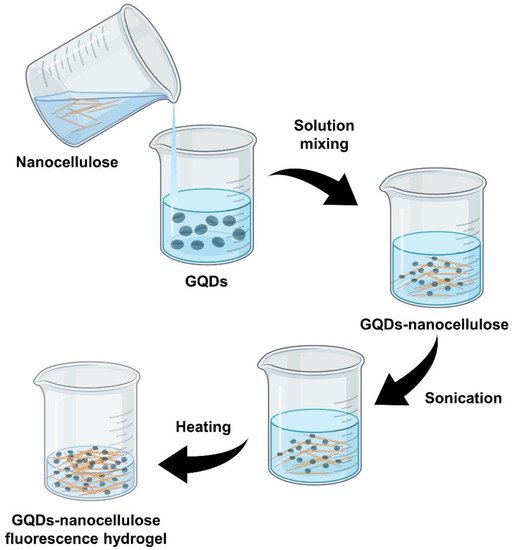
The average particle size of GQDs–nanocellulose composites can be controlled by altering the reaction conditions. The reactive chemical groups and its distinctive morphology make nanocellulose a good biological template for GQDs–nanocellulose synthesis. Solution mixing is the most common method of preparing nanocomposite films by far. This method is suitable for obtaining good dispersion of nanocellulose with a polymer solution due to the good dispersion of these nanoparticles in water. Figure 3 presents a schematic illustration of GQDs–nanocellulose hydrogel formation.
Figure 3. Preparation of GQDs–nanocellulose fluorescence hydrogel via solution mixing.
This entry is adapted from the peer-reviewed paper 10.3390/molecules26206158