In this entry, we focused our attention on some case reports related to the vitamin B12 deficiency associated to anemia, neurologic disorders, and hyperhomocysteinemia. B12 deficiency reversal is simply achieved by prompt therapy, even though it is not the same for several disorders.
- vitamin B
- megaloblastic
- neurological disorders
- thrombosis
1. Introduction
During the 1940s, an intensive search for the active factor in liver extracts that prevents pernicious anemia showed that both folate and vitamin B 12 prevent megaloblastic anemia, but only vitamin B 12 can prevent neurological complications [1]. It is now known that the two vitamins act jointly in regenerating methionine from homocysteine (Hcy). Hcy accumulates if conversion to methionine is slowed because of a shortage of folate or vitamin B 12 or both, and a raised plasma Hcy suggests sub-optimal nucleic acid and amino-acid metabolism. It also has direct harmful effects, e.g., it increases the risk of cardiovascular disease through thickening the lining of blood vessels and may also increase the risk of certain cancers and dementia.
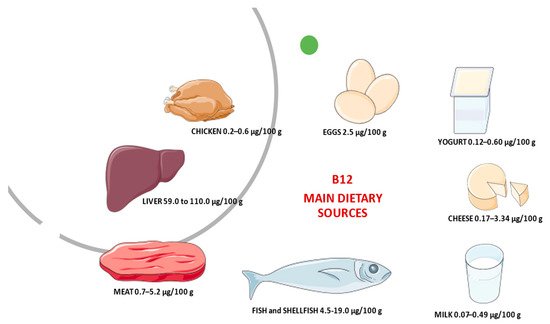
Vitamin B 12, also called cobalamin, is synthesized only by micro-organisms [2]. Dietary sources are meat (particularly liver), shellfish, some cheeses, yeast extracts, and the root nodules of legumes (peas, beans, and so on), mainly due to simultaneous bacteria presence in soil and/or their aerial surfaces. Figure 1 reports the main animal dietary B 12 source and its average content in raw/fresh amounts. Based on the absorption of labeled vitamin B 12 from some food products, such as chicken meat, rainbow trout, or eggs the bioavailability of vitamin B 12 is generally assumed to be 40% or 50% for healthy adults without alteration of gastrointestinal functioning [3]. Bioavailability also varies by type of food source. For example, dairy products have a bioavailability of vitamin B 12 three times higher than meat or fish [4]. Moreover, the bioavailability of vitamin B 12 from dairy products is considerable [5]. This aspect should take into consideration for vitamin B 12 recommendations. However, most of the information on B 12 bioavailability from foods was collected 40–50 years ago, and more recent methods to derive recommendations based on dose–response evidence are still under development [3]. Currently, to maintain a healthy hematological status and serum vitamin B 12 levels, average daily intakes of vitamin B 12 from food of 5.94 mcg for men and 3.78 mcg for women aged 20 and older have been recommended. For children aged 2–19 years old, mean daily intakes of vitamin B 12 from food range from 3.76 mcg to 4.55 mcg [6]. The original estimates of dietary folate and vitamin B 12 requirements and recommended dietary allowances (RDAs) were based on the amount needed to avoid manifest deficiency disorders (megaloblastic anemia, with sub-acute combined degeneration of the cord in the case of vitamin B 12 deficiency) and on levels observed in populations. However, these levels do not essentially represent necessary requirements.
As reported by Carmel [7], average total body stores of vitamin B 12 are 3–5 mg, mostly in the liver. After excretion from bile, it mostly is reabsorbed from the small intestine. It is first bound within the duodenum and jejunum to intrinsic factor produced by gastric parietal cells and is then absorbed in the terminal ileum [8]. Thus, when there is little or no vitamin B 12 in the diet, stores may last for up to 5–10 years before manifestations of vitamin B 12 are seen clinically. The etiology of vitamin B 12 deficiency usually includes causes related to malabsorption, such as autoimmune gastritis (pernicious anemia), celiac disease, inflammatory bowel disease, surgical gastrectomy, gastric bypass, and ileal resection. Less commonly, vitamin B 12 deficiency can occur due to nutritional habits (strict vegans, breastfed infants born to vegan mothers with decreased dietary intake of animal products), nitrous oxide abuse, Diphyllobothrium latum infection, pancreatic insufficiency, drug interference (metformin, proton pump inhibitors, drug affected purine, and pyrimidine synthesis), inherited disorders affecting intrinsic factor and other inherited disorders including methylmalonic acidemia and transcobalamin II deficiency. Moreover, alcohol abuse could represent a risk for vitamin B 12 deficiency, for its direct connection with the Hcy concentration, besides reflecting the degree of hepatocytes injury [9][10].
Currently, research suggests that there are disease implications associated with vitamin B 12 deficiency, especially in vegetarian populations [11][12][13], in pregnancy [14][15], in the elderly [16], and in developing countries [15].
2. Vitamin B12 and Anemia
The role of red blood cells (RBCs) is to transport oxygen through the body. When the number of RBCs is lower than usual body’s tissues and organs do not get enough oxygen, this blood disorder is known as anemia. Among different types of anemia, megaloblastic anemia is characterized by RBCs that are larger than normal and not able to exit the bone marrow to enter the bloodstream and deliver oxygen. However, the molecular basis of these cytomorphological aberrations remains unclear. This clinical condition indicates an altered synthesis of DNA, commonly due to a deficiency of vitamin B 12 or folic acid, both needed for the production and maturation of RBCs. In particular, the cytoplasm is excessive compared to the nucleus, causing their accumulation in the bone marrow (megaloblastosis) and macrocytosis in the peripheral blood [17]. In fact, the hallmark of megaloblastic anemia is macrocytic anemia (mean corpuscular volume, MCV > 100 fL), often associated with a reduction in the number of mature blood cells (cytopenia). A gold standard for diagnosing megaloblastic anemia is absent, so appropriate clinical and laboratory evaluation allows to establish the correct diagnosis.
Usually, adult patients come to medical attention because of symptoms related to anemia, such as fatigability, lethargy, and exertional breathlessness, pale conjunctiva, paleness and dry lips, and a disturbance of taste. We identify eight case reports all describing vitamin B 12 deficiency symptoms with etiological causes that appear to be different. A strict vegan 44-year-old woman with a history of anemia on admission presented lethargy, resulting in an inability to continue her job. Without regular medication assumption, except for the contraceptive pill. The biochemical investigations showed levels of haemoglobin (Hb) 110 g/L (reference range 115–160) and MCV 102 fL (range 80–100), and the cause of macrocytic anemia was highlighted by the slightly low levels of vitamin B 12 138 pmol/L (reference range 148–600). Test for antibodies to intrinsic factor was negative and oral contraceptive assumption could give a false lower vitamin B 12 level. Her lethargy and anemia were improved upon three months of treatment with intramuscular hydroxycobalamin 1000 μg three times a week for two weeks. Serum B 12 increases up to 400 pmol/L, Hb to 133 g/L at three months, as well as an improvement in her lethargy were observed. She was then advised to take oral cyanocobalamin supplements indefinitely as maintenance to prevent further deficiency [18].
Although glossitis rarely represents another symptom of cobalamin deficiency in addition to those related to anemia, the diagnosis of pernicious anemia with recurrent aphthous stomatitis was the clinical diagnosis of a 71-year-old woman case described by Garcia et al. [19]. Due to the vitamin B 12 malabsorption, the laboratory investigation showed a MCV of 104.1 fL and low serum vitamin B 12 (133 pg/mL). Clinical resolution was observed after two months after a treatment of 1.0 mL of hydroxocolabamin intramuscularly twice weekly over four weeks followed by 1.0 mL once weekly for four weeks. Pontes et al. [20] reported an oral manifestation of Vitamin B 12 deficiency in a 41-year-old strict vegetarian woman for 2.5 years that had not consumed milk, cheese, fish, meat, or eggs during that time. As suggested by the authors, a wide range of oral signs and symptoms may appear in anemic patients because of basic changes in the metabolism of oral epithelial cells. In fact, with regard to the clinical signs of weakness, fatigue, shortness of breath, and neurologic abnormalities, this woman had burning sensation, pale oral mucosa, glossitis with papillary atrophy, and multiple areas of painful erythema on the dorsal surface and lateral borders of the tongue and buccal mucosa were recorded. The hematologic test results showed low levels of Hb (7.2 g/dL) , MCV (fL 144), and serum cobalamin (71.8 pmol/L). After 14 days of treatment, comprising a parenteral dose of cobalamin (1000 mg/week hydroxocobalamin administered intramuscularly over 30 days) and 1 mg of folic acid daily for 30 days, the lesions had completely disappeared, as had all other symptoms. A rare presentation of dietary Vitamin B 12 deficiency anemia was reported by Pahadiya et al. [21]. A 40-year-old male presented a generalized weakness, fatigability, and hyperpigmentation with ecchymosis. His hemogram showed Hb of 9.7 g/dL, MCV 87.8 fL, and normal levels of Vitamin B 12 equal to 272 pg/ mL. Higher levels of lactate dehydrogenase (1250 IU/L) and serum bilirubin 2.43 mg/dL were observed. The authors assumed that these ecchymosed lesions might be due to chronic microhemorrhages in the superficial layer of skin because of thrombocytopenia. The ineffective erythropoiesis and the abnormal red cell membrane are the two pathophysiologic processes featuring in B 12 deficiency anemia. The resulting reduction in red cell lifespan and the associated hemolysis cause a rising in plasma bilirubin and lactic dehydrogenase (LDH). The cutaneous manifestations of Vitamin B 12 deficiency including skin hyperpigmentation was reversible after the Vitamin B 12 supplementation. Treatment with intramuscular hydroxocobalamin 1000 μg daily for five days, subsequently on alternate days for a week, and thereafter weekly for one month restored his hematological parameters to within normal limits and ecchymosis disappeared.
3. Vitamin B12 and Neurological Disorders
The neurologic manifestations of B 12 deficiency, including myelopathy, neuropathy, dementia [22][23], and rarely cerebellar ataxia and movement disorders, are difficult to diagnose. In fact, the neurological abnormalities caused by cobalamin deficiency could take place without any hematological or gastrointestinal context [24], and in the absence of anemia or an elevated mean cell volume [25]. The severity of neurological complications may be reversed only by an early treatment after onset, so a timely diagnosis is important. Despite the rapid correction of vitamin B 12 levels by prompt therapy and early clinical improvement, the recovery of polyneuropathy on nerve conduction could be slow. Deficiency of vitamin B 12, mostly in vegetarians was found to be associated with depression and adverse neurological function. Berkins [11] points out that the dietary intake of vitamin B 12 and vitamin B 6 might have an effect on brain structure. Ralapanawa et al. [26] reported a strict vegetarian 66-year-old female case with demyelinating polyneuropathy without features of anemia (Hb concentration of 12.1 g/dL, RBCs count of 4.39/mm 3, MCV of 83.3 fL), but very low serum vitamin B 12 levels (84.90 pg/mL reference range 208–963 pg/mL). To reverse neurological manifestations, after three months of therapy with intramuscular hydroxycobalamine 1000 μg for seven days, weekly for six weeks and thereafter three monthly, the patient showed clinical improvement, with repeated B 12 levels being elevated up to 308.6 pg/mL. At one- and three-year follow up for nerve conduction study previously absent, early clinical improvement was demonstrated, with a slow recovery of polyneuropathy on nerve conduction studies. Even though vitamin B 12 deficiency neuropathy is a rare debilitating disease that affects mostly the elderly, young adults with neuropathic symptoms warrant a high index of suspicion. The cause of neurological symptoms resulting from vitamin B 12 deficiency could be due to the role of methylcobalamin in myelin synthesis. The lack of cobalamin could induce the destruction of myelin sheaths or incorporation of abnormal fatty acids in myelin sheaths, thus leading to impaired neural function and/or transmission. The diagnosis of Vitamin B 12 deficiency is challenging in resource limited-settings due to limited access to diagnostic tools and unfamiliarity with the disease, owing to its rarity especially in young people. This is the case reported by Ekabe et al. [27]. A 28-year-old sub-Saharan female, presenting peripheral neuropathic symptoms, was treated with oral vitamin B 12 tablets at doses of 2 mg per day for three months. A diagnosis of vitamin B 12 deficiency related peripheral neuropathy was made based on her symptoms, ovalo-macrocytosis and hyper-segmented neutrophils on peripheral blood smear. After one month of therapy, an improvement in neurological symptoms was recorded. The authors highlighted the pivotal role of basic investigations like peripheral blood smear for the timely detection and management of vitamin B 12 associated neurological disease in resource-limited settings. A case of sub-acute combined degeneration (SCD), the most common neurological disorder, in a 33-year-old woman without anemia or macrocytosis leading was diagnosed by Maamar et al. [28] as suspected vitamin B 12 deficiency, subsequently confirmed by a low serum cobalamin. First investigations revealed Hb 12.1 g/100 mL, MCV 91 fL, other biochemical parameters were within normal limits, while magnetic resonance (MR) imaging of the spine revealed intramedullary hyperintensity in the posterior column of the cervico-dorsal spinal cord, highly suggestive of subacute combined degeneration (SCD). In fact, the patient’s vitamin B 12 serum level was low (30 pg/mL; reference range 200–700 pg/mL) while serum folate was within the normal range (26 ng/mL; reference range 18–30 ng/mL). Intramuscular administration of B 12 resulted in correction of the neurological signs (paresthesis and sphincter disorders). At a seven-year follow-up, while still receiving intramuscular vitamin B 12 monthly, the patient was found to be functionally independent with no neurological deficits. Early spinal MR imaging could support the early diagnosis of SCD of the spinal cord due to Vitamin B 12 deficiency as reported in the case of a 57-year-old man by Senol et al. [29]. Following clinical and laboratory examinations, the patient was evaluated as cervical myelopathy due to Vitamin B 12 deficiency (60 pg/mL (reference range 189–883 pg/mL). The symptoms totally disappeared two months after intramuscular supplementation of vitamin B 12 (1000 µg IM daily for a week and then weekly for six weeks) and the MR imaging abnormalities significantly improved. The same diagnosis of SCD was considered and confirmed by laboratory findings in a 56-year-old man by Srikanth et al. [30]. The patient presented an acute onset of paresthesia involving both hands and feet of 15 days duration, difficulty in walking, and inability to feel the ground for the same period. Neurological examination revealed impairment of sensation of fine touch, pinprick, joint position, and vibration in both hands and feet bilaterally. All the deep tendon reflexes were exaggerated, more so in the lower limbs, with no evidence of motor weakness. Gastric endoscopy and biopsy revealed changes of atrophic gastritis and folic acid and vitamin B 12 levels in the serum were 7 micrograms and 75 picograms, respectively. Cervical MR image findings were consistent with SCD. MR imaging lesion was completely resolved treating the patient with parenteral administration of vitamin B 12 and oral folic acid. In summary, SCD is clinically characterized by predominant involvement of the dorsal columns and the lateral columns of the spinal cord, resulting in sensory deficits, paresthesia, weakness, ataxia, and gait disturbance. In some patients, MR imaging shows abnormalities of the spinal cord, indicating demyelination of the posterior column. Early diagnosis and treatment play an important role in the reversibility of neurological deficits. Delayed treatment results in irreversible disabling neurological impairment, such as spasticity and paraplegia.
The seizures rarely occur in patients with vitamin B 12 deficiency and the molecular mechanisms involving cobalamin in epileptogenesis are unknown. However, Kumar [31] reported this unusual symptom of vitamin B 12 deficiency in a 26-year-old man. A diagnosis of vitamin B 12 deficiency with multiple neuropsychiatric manifestations, namely dementia, psychosis, seizures, and myeloneuropathy, was considered. Investigations confirmed the suspicion Hb 13.2 g/dL, and MCV 114 fL. Serum B 12 assay was 26 pg/mL and folate levels were 28 ng/mL. His symptoms responded to parenteral vitamin B 12 therapy started on intramuscular vitamin B 12 injections. At 24 months follow-up, the seizures disappeared and functionality was independent. Regarding this disturbance, the author highlighted the similarities of cobalamin deficiency with multiple sclerosis and supposed that the probable impairment of cerebral neurons was due to destroyed myelin sheaths, which are more susceptible to the excitatory effects of glutamate. Mavromati and Sentissi [32] report a clinical case of delirium due to vitamin B 12 deficiency in a vegetarian female 62-year-old. Delirium could have multiple causes, so the initial diagnosis presented various difficulties. Details on neurological symptoms are reported. The laboratory and clinical examinations excluded infectious, vascular, neoplastic, metabolic, and endocrine causes. Her serum vitamin B 12 level was low (91 pmol/L) and folic acid was normal (22.2 mg/L). The patient was treated with vitamin B 12 supplementation. The vitamin B 12 level was normalized one week later (330 pmol/L). A psychiatric examination two weeks after the first evaluation revealed an important diminution of cognitive deficiency and a partial remission of the depressive symptoms (MADRS score 22, MMSE 28/30 and DRS-R-98 4; the clock test was normalized). Four weeks after the episode, there was a total remission of the depressive symptoms (MADRS score: 4) and stable mental status. The cause of the vitamin B 12 deficiency was attributed to the patient’s strict vegetarianism and this finding underlines the importance of conducting a complete laboratory test panel for delirium, including the blood levels of vitamin B 12. Table 2 summarizes the corresponding laboratory and clinical investigations of the reported cases in this section on neurological disorders and vitamin B 12 deficiency.
4. Vitamin B12 Deficiency and Hyperhomocysteinemia
For example, the toxic effect of Hcy on the vascular endothelium and on the dotting cascade, as well procoagulant properties of Hcy, including the decrease of antithrombin III binding to endothelial heparan sulfate, an increase of affinity between lipoprotein(a) and fibrin, induction of tissue factor activity in endothelial cells, and inhibition of inactivation of factor V by activated protein. In all patients, clinical and biological abnormalities disappeared upon vitamin B 12 supplementation. The authors concluded that vitamin B 12 supplements can rapidly correct HHcy avoiding and preventing thrombotic events.
Tanaka et al. [33] reported a case of a 39-year-old man with inferior vena cava (IVC) thrombus. The analysis of risk factors of venous thromboembolism shown HHcy (total Hcy 83.1 µmol/L; normal range 5–15 µmol/L) due to an unbalanced diet with a deficiency of folic acid and vitamin B 12. The patient was treated with both folic acid and vitamin B 6/B 12 supplementation in association with warfarin, inducing a significant resolution of thrombus after four weeks and no evidence of recurrent IVC thrombus at six months. The authors concluded that B vitamins and folic acid therapy might be effective in patients with severe HHcy.
Ruscin et al. [34] illustrated the case of a 78-year-old nonvegetarian white woman with gastroesophageal reflux treated for long-term with histamine(2) (H(2))-receptor antagonists and a proton-pump inhibitor (PPI). During treatment, vitamin B 12 dropped from normal values (413 pg/mL) to 256 pg/mL; methylmalonic acid (MMA) and Hcy were elevated at 757 nmol/L and 27.3 micromol/L, respectively, serum folate was within the normal range (4.9 ng/mL), and serum creatinine was slightly elevated at 1.4 mg/dL. In addition, no renal dysfunction was present. After oral treatment with vitamin B 12 (1000 microg/d), MMA and Hcy concentrations decreased dramatically. The authors speculated vitamin B 12 deficiency because of cobalamin malabsorption from food intake due to drug interference, suggesting vitamin B 12 status monitoring in patients taking these medications for an extended time, particularly >4 years.
As known, elevated plasma Hcy is involved in cognitive decline, including Alzheimer’s disease, mild cognitive impairment, and dementia, especially in elderly subjects. McCaddon [35] reported seven cases of older patients (four women aged 78 years, 84 years, 77 years and 87 years, 84 years old, and two men 71 and 75 years old). They presented with cognitive impairment and/or depression, dementia, etc. Each had different vitamin B 12 status with HHcy. Treatment with N-acetylcysteine, together with B vitamin supplements, improves cognitive status in hyperhomocysteinemic patients. The authors concluded that it could be important to evaluate inadequate vitamin B 12 and folate metabolism in subjects with cognitive diseases, underlining the importance of clinical trials to evaluate the beneficial effects of a synergistic approach to cognitively impaired hyperhomocysteinaemic patients.
This entry is adapted from the peer-reviewed paper 10.3390/ijms22189694
References
- Lanska, D.J. Chapter 30 Historical aspects of the major neurological vitamin deficiency disorders: The water-soluble B vitamins. Handb. Clin. Neurol. 2009, 95, 445–476.
- Watanabe, F.; Bito, T. Vitamin B12 sources and microbial interaction. Exp. Biol. Med. 2018, 243, 148–158.
- Doets, E.L.; Veld, P.H.I.’T.; Szczecińska, A.; Dhonukshe-Rutten, R.A.; Cavelaars, A.E.; Veer, P.V.’T.; Brzozowska, A.; de Groot, L. Systematic Review on Daily Vitamin B12 Losses and Bioavailability for Deriving Recommendations on Vitamin B12 Intake with the Factorial Approach. Ann. Nutr. Metab. 2013, 62, 311–322.
- Allen, L.H. Bioavailability of Vitamin B12. Int. J. Vitam. Nutr. Res. 2010, 80, 330–335.
- Vogiatzoglou, A.; Smith, D.; Nurk, E.; Berstad, P.; A Drevon, C.; Ueland, P.M.; E Vollset, S.; Tell, G.S.; Refsum, H. Dietary sources of vitamin B-12 and their association with plasma vitamin B-12 concentrations in the general population: The Hordaland Homocysteine Study. Am. J. Clin. Nutr. 2009, 89, 1078–1087.
- U.S. Department of Agriculture, Agricultural Research Service. What We Eat in America, 2017–2018. External Link Disclaimer; U.S. Department of Agriculture, Agricultural Research Service: Beltsville, MD, USA, 2020.
- Carmel, R. How I treat cobalamin (vitamin B12) deficiency. Blood 2008, 112, 2214–2221.
- Hunt, A.; Harrington, D.; Robinson, S. Vitamin B12 deficiency. BMJ 2014, 349, g5226.
- Stabler, S.P. Vitamin B12Deficiency. N. Engl. J. Med. 2013, 368, 149–160.
- Green, R.; Mitra, A.D. Megaloblastic Anemias. Med. Clin. N. Am. 2017, 101, 297–317.
- Berkins, S.; Schiöth, H.B.; Rukh, G. Depression and Vegetarians: Association between Dietary Vitamin B6, B12 and Folate In-take and Global and Subcortical Brain Volumes. Nutrients 2021, 13, 1790.
- Pawlak, R. Vitamin B12 status is a risk factor for bone fractures among vegans. Med. Hypotheses 2021, 153, 110625.
- Desmond, M.A.; Sobiecki, J.G.; Jaworski, M.; Płudowski, P.; Antoniewicz, J.; Shirley, M.K.; Eaton, S.; Książyk, J.; Cortina-Borja, M.; De Stavola, B.; et al. Growth, body composition, and cardiovascular and nutritional risk of 5- to 10-y-old children consuming vegetarian, vegan, or omnivore diets. Am. J. Clin. Nutr. 2021, 113, 1565–1577.
- Saravanan, P.; Sukumar, N.; Adaikalakoteswari, A.; Goljan, I.; Venkataraman, H.; Gopinath, A.; Bagias, C.; Yajnik, C.S.; Stallard, N.; Ghebremichael-Weldeselassie, Y.; et al. Association of maternal vitamin B12 and folate levels in early pregnancy with gestational diabetes: A prospective UK cohort study (PRiDE study). Diabetologia 2021, 64, 2170–2182.
- Behere, R.V.; Deshmukh, A.S.; Otiv, S.; Gupte, M.D.; Yajnik, C.S. Maternal Vitamin B12 Status During Pregnancy and Its Association With Outcomes of Pregnancy and Health of the Offspring: A Systematic Review and Implications for Policy in India. Front. Endocrinol. 2021, 12, 176.
- Nalder, L.; Zheng, B.; Chiandet, G.; Middleton, L.T.; de Jager, C.A. Vitamin B12 and Folate Status in Cognitively Healthy Older Adults and Associations with Cognitive Performance. J. Nutr. Heal. Aging 2021, 25, 287–294.
- Aslinia, F.; Mazza, J.J.; Yale, S.H. Megaloblastic anemia and other causes of macrocytosis. Clin. Med. Res. 2006, 4, 236–241.
- Sukumar, N.; Saravanan, P. Investigating vitamin B12 deficiency. BMJ 2019, 365, l1865.
- Garcia, B.G.; Cardoso, M.F.P.; Faria, O.; Gomez, R.S.; Mesquita, R.A. A Case Report of Pernicious Anemia and Recurrent Aph-thous Stomatitis. J. Contemp. Dent. Pract. 2009, 10, 83–89.
- Pontes, H.A.; Neto, N.C.; Ferreira, K.B.; Fonseca, F.P.; Vallinoto, G.M.; Pontes, F.S.; Pinto, D.S. Oral manifestations of vitamin B12 deficiency: A case report. J. Can. Dent. Assoc. 2009, 75, 533–537.
- Pahadiya, H.R.; Lakhotia, M.; Choudhary, S.; Prajapati, G.R.; Pradhan, S. Reversible ecchymosis and hyperpigmented. lesions: A rare presentation of dietary Vitamin B12 deficiency. J. Fam. Med. Prim. Care 2016, 5, 485–487.
- Hemmer, B.; Glocker, F.X.; Schumacher, M.; Deuschl, G.; Lücking, C.H. Subacute combined degeneration: Clinical, electro-physiological, and magnetic resonance imaging finding. J. Neurol. Neurosurg. Psychiatry. 1998, 65, 822–827.
- Maamar, M.; Tazi-Mezalek, Z.; Harmouche, H.; Ammouri, W.; Zahlane, M.; Adnaoui, M.; Aouni, M.; Mohattane, A.; Maaouni, A. Neurological manifestations of vitamin B12 deficiency: A retrospective study of 26 cases. Rev. Med. Interne 2006, 27, 442–447.
- Green, R. Vitamin B12 deficiency from the perspective of a practicing hematologist. Blood 2017, 129, 2603–2611.
- Lindenbaum, J.; Healton, E.B.; Savage, D.G.; Brust, J.C.; Garrett, T.J.; Podell, E.R.; Marcello, P.D.; Stabile, S.; Allen, R.H. Neuro-psychiatric disorders caused by cobalamin deficiency in the absence of anemia or macrocytosis. N. Engl. J. Med. 1988, 318, 1720–1728.
- Ralapanawa, D.M.P.U.K.; Jayawickreme, K.P.; Ekanayake, E.M.M.; Jayalath, W.A.T.A. B12 deficiency with neurological manifestations in the absence of anaemia. BMC Res. Notes 2015, 8, 458.
- Ekabe, C.J.; Kehbila, J.; Abanda, M.H.; Kadia, B.M.; Sama, C.-B.; Monekosso, G.L. Vitamin B12 deficiency neuropathy; a rare diagnosis in young adults: A case report. BMC Res. Notes 2017, 10, 72.
- Maamar, M.; Mezalek, Z.T.; Harmouche, H.; Adnaoui, M.; Aouni, M.; Maaouni, A. Contribution of spinal MRI for unsuspected cobalamin deficiency in isolated sub-acute combined degeneration. Eur. J. Intern. Med. 2008, 19, 143–145.
- Senol, M.G.; Sonmez, G.; Ozdag, F.; Saracoglu, M. Reversible myelopathy with vitamin B12 deficiency. Singap. Med. J. 2008, 49, e330–e332.
- Srikanth, S.G.; Jayakumar, P.N.; Vasudev, M.K.; Taly, A.B.; Chandrashekar, H.S. MRI in subacute combined degeneration of spinal cord: A case report and review of literature. Neurol. India 2002, 50, 310.
- Kumar, S. Recurrent seizures: An unusual manifestation of vitamin B12 deficiency. Neurol. India 2004, 52, 122–123.
- Mavrommati, K.; Sentissi, O. Delirium as a result of vitamin B12 deficiency in a vegetarian female patient. Eur. J. Clin. Nutr. 2013, 67, 996–997.
- Tanaka, M.; Taniguchi, T.; Saito, N.; Kimura, T. Inferior vena cava thrombus due to hyperhomocysteinemia. J. Cardiol. Cases 2018, 18, 168–170.
- Ruscin, J.M.; Page, R.L.; Valuck, R.J. Vitamin B12 Deficiency Associated with Histamine2-Receptor Antagonists and a Proton-Pump Inhibitor. Ann. Pharmacother. 2002, 36, 812–816.
- McCaddon, A. Homocysteine and cognitive impairment; a case series in a General Practice setting. Nutr. J. 2006, 5, 6.
