Depressive disorder in childhood and adolescence is a highly prevalent mood disorder that tends to recur throughout life. Untreated mood disorders can adversely impact a patient’s quality of life and cause socioeconomic loss. Thus, an accurate diagnosis and appropriate treatment is crucial. However, until now, diagnoses and treatments were conducted according to clinical symptoms. Objective and biological validation is lacking. This may result in a poor outcome for patients with depressive disorder. Research has been conducted to identify the biomarkers that are related to depressive disorder. Cumulative evidence has revealed that certain immunologic biomarkers including brain-derived neurotrophic factor (BDNF) and cytokines, gastrointestinal biomarkers, hormones, oxidative stress, and certain hypothalamus-pituitary axis biomarkers are associated with depressive disorder.
- pediatric
- depression
- biomarker
- BDNF
- cytokines
1. Introduction
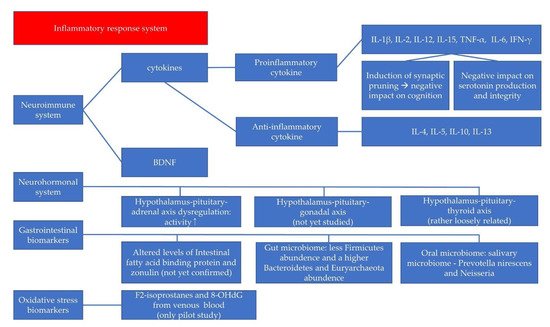
2. Neuroimmune System and Pediatric Depressive Disorder
2.1. The Cytokines in Neuroinflammation
| Study | Objective | Design | Inflammatory Markers | Findings |
|---|---|---|---|---|
| Gabbay et al., 2009 [10] | To examine the immune system in adolescents with MDD | N: 45, age 12–19 years; 13 psychotropic-free MDD Pts, 17 MDD Pts with medication, and 15 HCs | Plasma IL-6, IFN-γ, TNF-α, IL-4, and IL-1β | Significantly higher levels of plasma IFN-γ in MDD Pts and trend for IL-6 to be elevated in MDD group; Significantly increased level of IFN-γ in the unmedicated MDD group compared to HCs |
| Henje Blom et al., 2011 [11] | To investigate the effects of antidepressants on cytokines in adolescent females with anxiety disorder and/or depressive disorder | N: 102, age 14.5–18.4 years; 42 Pts (26 unmedicated Pts and 16 SSRI Pts) and 60 HCs | Plasma IL-1β, IL-2, IL-6, IL-10, TNF-α, and IFN-γ | Significantly higher values of IL-2, IL-10, and IL1-β in patient group; higher level of IL-6 in the non-medicated subgroup compared to the medicated subgroup; higher levels of IL-6 and IFN-γ were significantly related to more severe self-assessed symptoms of anxiety and depression |
| Copeland et al., 2012 [37] | To test (1) the effect of CRP levels on later depression status; (2) the effect of depression status on later CRP levels; and (3) the effect of cumulative depressive episodes on later CRP levels. | N: 1420, age 9, 11, and 13 years at intake; longitudinal study with annual assessment to age 16 and again at 19 and 21 years | CRP (dried blood spot) | CRP levels were not associated with later depression status; Cumulative depressive episodes predicted later CRP levels |
| Rengasamy et al., 2012 [39] | To examine the associations of IL-6 and TNF-α with depression severity and anhedonia severity | N: 36, age 12–18 years; 36 adolescents with depressive disorder, cross-sectional and longitudinal study | TNF-α, and IL-6 | Baseline TNFα was positively associated with baseline and follow-up SHAPS anhedonia scores, and follow-up CDRS-R |
| Amitai et al., 2016 [17] | To determine whether plasma levels of pro-inflammatory cytokines can predict response to treatment and/or are altered post fluoxetine treatment in children and adolescents. | N: 41, age 7–18 years; children and adolescents with depression and/or anxiety disorders. | Plasma IL-1β, IL-6, and TNF-α | Significantly higher levels of pro-inflammatory cytokines in SSRI-refractory than in SSRI-responsive Pts; TNF-α levels significantly reduced after 8 weeks of antidepressant treatment |
| da Silva et al., 2017 [12] | To compare serum levels of IL-6 and IL-10 between non-medicated adolescents with internalizing disorders and a comparison group of adolescents without internalizing disorders | N: 134, age 10–17 years; 76 adolescents with internalizing disorder and 58 adolescents without internalizing disorder |
Plasma IL-6, and IL-10 | Adolescents with internalizing disorders had significantly higher levels of IL-6 as compared to those without internalizing disorders |
| Pérez-Sánchez et al., 2018 [13] | To detect (1) the alterations in the cytokine profiles of adolescents during 8 weeks of treatment with fluoxetine and (2) the correlation between symptomatology and inflammatory profiles | N: 40, age 14–19 years; 22 adolescents with first episode of MDD and 18 HCs, cross-sectional and longitudinal study | Plasma IL-2, IFN-γ, IL-1β, TNF-α, IL-6, IL-15, IL-10, IL-5, IL-13, IL-1Ra, and IL-12p70 | Significantly increased levels of pro-inflammatory cytokines (IL-2, IFN-γ, IL-1β, TNF-α, IL-6, IL-12, and IL-15) and anti-inflammatory cytokines (IL-4, IL-5, and IL-13) in MDD Pts; IFN-γ, IL-1β, TNF-α, IL-6, IL-12, and IL-15 decreased only at week 4; increased IL-2 only at week 8; increased anti-inflammatory cytokines IL-4 and IL-5 at week 8 |
| Peters et al., 2019 [38] | 1. To compare groups with inflammation 2. To evaluate associations between inflammation and inhibitory control |
N: 70, age 12–17 years; 22 depressive adolescents with childhood trauma (DEP-T), 18 depressive adolescents (DEP), and 30 HCs | Plasma IL-1β, TNF-α, IL-6 | Significantly elevated levels of IL-6 in both DEP and DEP-T relative to HCs and significantly elevated levels of TNF-α in DEP; No group differences were detected in IL-1β; TNF-α was associated with behavior-based and observer-rated inhibitory control deficits |
| Lee et al., 2020 [14] | 1. To examine (1) the difference between inflammatory markers in MDD Pts and HCs and (2) whether these changes would be altered following antidepressant treatment 2. To investigate the relationship between cytokines’ level with the severity of depression |
N: 50, age 13–18 years; 25 medication-naïve MDD Pts and 25 HCs, cross-sectional and longitudinal study |
Plasma IL-1β, IL-2, IL-4, IL-6, IL-10, TNF-α, and IFN-γ | MDD Pts had significantly decreased level of plasm IL-2, IFN-γ, TNF-α, and IL-10 compared to healthy controls; IL-2, IFN-γ, and IL-10 showed significant increases after 12 weeks treatment compared to before treatment. IFN-γ level was negatively correlated with the CDI (r = −0.377, p < 0.01) and HDRS score (r = −0.457, p < 0.01) |
2.2. BDNF in the Neuroimmune System
| Study | Objective | Design | Findings |
|---|---|---|---|
| Pandey et al, 2010 [21] | To examine the gene expression of BDNF of pediatric depressed Pts (drug naïve or unmedicated for a period of up to 2 weeks) | N: 28, 14 MDD Pts (age 14.9 ± 1.7) and 14 HCs (age 13.0 ± 1.7) | Significantly decreased gene expression of BDNF in the lymphocytes and the protein expression in the platelets of pediatric depressed Pts. compared with HCs |
| Sasaki et al., 2011 [55] | To investigate whether serum levels of BDNF are altered in pediatric Pts with depression | N: 52, age 8–15 years; 13 male and 17 female MDD Pts; 10 male and 12 female HCs | Significantly lower levels of serum BDNF only in male MDD Pts compared to male HCs, not in female MDD Pts; Significant negative correlation between the serum BDNF levels and the duration of illness in males, but not in females |
| Pallavi et al., 2013 [56] | (1) To compare serum levels of BDNF in depression patients with healthy controls and (2) to investigate the correlation between clinical severity and serum BDNF levels | N: 148, age 13–18 years; 84 (56 males) MDD Pts, and 64 (39 males) HCs | Adolescents with depression had significantly lower levels of BDNF; BDI-II score showed a statistically significant negative correlation with BDNF in male patients, but not in female patients |
| Sun et al., 2017 [69] | To investigate (1) the correlation between serum BDNF and depression in children and (2) the change in BDNF after treatment | N: 178, age 7–16 years, 128 (55 males) MDD Pts, and 50 (25 males) HCs | Significantly lower levels of serum BDNF in MDD Pts compared to HCs; MDD Pts with comprehensive nursing showed a significant increase in BDNF expression |
| Bilgiç et al., 2020 [57] | To identify potential differences in serum BDNF levels in adolescents with MDD compared to HCs | N: 110, age 11–19 years; 70 treatment-free MDD Pts, and 40 HCs | Serum BDNF levels were significantly higher in adolescents with MDD than in HCs; No correlations between the levels of serum BDNF and the severity of depression or suicidality |
| Lee et al., 2020 [70] | To investigate whether pre-treatment BDNF levels and their early changes predict antidepressant response in MDD Pts | N: 135, age 12–17 years, 83 MDD Pts (46 responders and 37 non-responders), 52 HCs; baseline, 2 weeks, 8 weeks follow-up | No significant findings of serum BDNF between the responders, non-responders, and HCs at baseline; Early decrease in BDNF levels of responders at week 2; Early BDNF decrease predicted later SSRI response at week 8 |
3. Neurohormonal System and Pediatric Depressive Disorder
4. GI Biomarkers and Pediatric Depressive Disorder
5. Oxidative Stress Biomarkers and Pediatric Depressive Disorder
6. Summary and Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/ijms221810051
References
- Wegner, M.; Amatriain-Fernández, S.; Kaulitzky, A.; Murillo-Rodriguez, E.; Machado, S.; Budde, H. Systematic review of meta-analyses: Exercise effects on depression in children and adolescents. Front. Psychiatry 2020, 11, 81.
- Johnson, D.; Dupuis, G.; Piche, J.; Clayborne, Z.; Colman, I. Adult mental health outcomes of adolescent depression: A systematic review. Depress. Anxiety 2018, 35, 700–716.
- Nock, M.K.; Green, J.G.; Hwang, I.; McLaughlin, K.A.; Sampson, N.A.; Zaslavsky, A.M.; Kessler, R.C. Prevalence, correlates, and treatment of lifetime suicidal behavior among adolescents: Results from the National Comorbidity Survey Replication Adolescent Supplement. JAMA Psychiatry 2013, 70, 300–310.
- Mojtabai, R.; Olfson, M.; Han, B.J.P. National trends in the prevalence and treatment of depression in adolescents and young adults. Pediatrics 2016, 138, e20161878.
- Dunlop, B.W. Prediction of treatment outcomes in major depressive disorder. Expert Rev. Clin. Pharmacol. 2015, 8, 669–672.
- Godlewska, B.R.; Browning, M.; Norbury, R.; Igoumenou, A.; Cowen, P.J.; Harmer, C.J. Predicting treatment response in depression: The role of anterior cingulate cortex. Int. J. Neuropsychopharmacol. 2018, 21, 988–996.
- Group, B.D.W.; Atkinson, A.J., Jr.; Colburn, W.A.; DeGruttola, V.G.; DeMets, D.L.; Downing, G.J.; Hoth, D.F.; Oates, J.A.; Peck, C.C.; Schooley, R.T. Biomarkers and surrogate endpoints: Preferred definitions and conceptual framework. Clin. Pharmacol. Ther. 2001, 69, 89–95.
- Jeon, S.W.; Kim, Y.K. Neuroinflammation and cytokine abnormality in major depression: Cause or consequence in that illness? World J. Psychiatry 2016, 6, 283.
- Kaufman, J.; Martin, A.; King, R.A.; Charney, D. Are child-, adolescent-, and adult-onset depression one and the same disorder? Biol. Psychiatry 2001, 49, 980–1001.
- Gabbay, V.; Klein, R.G.; Alonso, C.M.; Babb, J.S.; Nishawala, M.; De Jesus, G.; Hirsch, G.S.; Hottinger-Blanc, P.M.; Gonzalez, C.J. Immune system dysregulation in adolescent major depressive disorder. J. Affect. Disord. 2009, 115, 177–182.
- Blom, E.H.; Lekander, M.; Ingvar, M.; Åsberg, M.; Mobarrez, F.; Serlachius, E. Pro-inflammatory cytokines are elevated in adolescent females with emotional disorders not treated with SSRIs. J. Affect. Disord. 2012, 136, 716–723.
- da Silva, C.T.B.; de Abreu Costa, M.; Kapczinski, F.; de Aguiar, B.W.; Salum, G.A.; Manfro, G.G. Inflammation and internalizing disorders in adolescents. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2017, 77, 133–137.
- Pérez-Sánchez, G.; Becerril-Villanueva, E.; Arreola, R.; Martínez-Levy, G.; Hernández-Gutiérrez, M.E.; Velasco-Velásquez, M.A.; Alvarez-Herrera, S.; Cruz-Fuentes, C.; Palacios, L.; de la Peña, F. Inflammatory profiles in depressed adolescents treated with fluoxetine: An 8-week follow-up open study. Mediat. Inflamm. 2018, 2018, 1–12.
- Lee, H.; Song, M.; Lee, J.; Kim, J.-B.; Lee, M.-S. Prospective study on cytokine levels in medication-naïve adolescents with first-episode major depressive disorder. J. Affect. Disord. 2020, 266, 57–62.
- Gariup, M.; Gonzalez, A.; Lázaro, L.; Torres, F.; Serra-Pagès, C.; Morer, A. IL-8 and the innate immunity as biomarkers in acute child and adolescent psychopathology. Psychoneuroendocrinology 2015, 62, 233–242.
- Maes, M.; Scharpé, S.; Meltzer, H.Y.; Bosmans, E.; Suy, E.; Calabrese, J.; Cosyns, P. Relationships between interleukin-6 activity, acute phase proteins, and function of the hypothalamic-pituitary-adrenal axis in severe depression. Psychiatry Res. 1993, 49, 11–27.
- Amitai, M.; Taler, M.; Carmel, M.; Michaelovsky, E.; Eilat, T.; Yablonski, M.; Orpaz, N.; Chen, A.; Apter, A.; Weizman, A. The relationship between plasma cytokine levels and response to selective serotonin reuptake inhibitor treatment in children and adolescents with depression and/or anxiety disorders. J. Child. Adolesc. Psychopharmacol. 2016, 26, 727–732.
- Mondal, A.C.; Fatima, M. Direct and indirect evidences of BDNF and NGF as key modulators in depression: Role of antidepressants treatment. Int. J. Neurosci. 2019, 129, 283–296.
- Yang, T.; Nie, Z.; Shu, H.; Kuang, Y.; Chen, X.; Cheng, J.; Yu, S.; Liu, H. The role of BDNF on neural plasticity in depression. Front. Cell. Neurosci. 2020, 14, 82.
- Jin, Y.; Sun, L.H.; Yang, W.; Cui, R.J.; Xu, S.B. The role of BDNF in the neuroimmune axis regulation of mood disorders. Front. Neurol. 2019, 10, 515.
- Pandey, G.N.; Dwivedi, Y.; Rizavi, H.S.; Ren, X.; Zhang, H.; Pavuluri, M.N. Brain-derived neurotrophic factor gene and protein expression in pediatric and adult depressed subjects. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2010, 34, 645–651.
- Sivri, R.Ç.; Bilgiç, A.; Kılınç, İ. Cytokine, chemokine and BDNF levels in medication-free pediatric patients with obsessive–compulsive disorder. Eur. Child. Adolesc. Psychiatry 2018, 27, 977–984.
- Ben Haim, L.; Carrillo-de Sauvage, M.-A.; Ceyzériat, K.; Escartin, C. Elusive roles for reactive astrocytes in neurodegenerative diseases. Front. Cell. Neurosci. 2015, 9, 278.
- Heneka, M.T.; Sastre, M.; Dumitrescu-Ozimek, L.; Dewachter, I.; Walter, J.; Klockgether, T.; Van Leuven, F. Focal glial activation coincides with increased BACE1 activation and precedes amyloid plaque deposition in APP transgenic mice. J. Neuroinflammation 2005, 2, 1–12.
- Maeng, S.H.; Hong, H. Inflammation as the potential basis in depression. Int. Neurourol. J. 2019, 23 (Suppl. S2), S63.
- Miller, A.H.; Haroon, E.; Raison, C.L.; Felger, J.C. Cytokine targets in the brain: Impact on neurotransmitters and neurocircuits. Depress. Anxiety 2013, 30, 297–306.
- Loftis, J.M.; Huckans, M.; Morasco, B.J. Neuroimmune mechanisms of cytokine-induced depression: Current theories and novel treatment strategies. Neurobiol. Dis. 2010, 37, 519–533.
- Maes, M.; Bosmans, E.; Meltzer, H.Y. Immunoendocrine aspects of major depression. Eur. Arch. Psychiatry Clin. Neurosci. 1995, 245, 172–178.
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Liu, H.; Sham, L.; Reim, E.K.; Lanctôt, K.L. A meta-analysis of cytokines in major depression. Biol. Psychiatry 2010, 67, 446–457.
- Belzeaux, R.; Lefebvre, M.N.; Lazzari, A.; Le Carpentier, T.; Consoloni, J.L.; Zendjidjian, X.; Abbar, M.; Courtet, P.; Naudin, J.; Boucraut, J.; et al. How to: Measuring blood cytokines in biological psychiatry using commercially available multiplex immunoassays. Psychoneuroendocrinology 2017, 75, 72–82.
- Schiepers, O.J.; Wichers, M.C.; Maes, M. Cytokines and major depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2005, 29, 201–217.
- Gabbay, V.; Klein, R.G.; Guttman, L.E.; Babb, J.S.; Alonso, C.M.; Nishawala, M.; Katz, Y.; Gaite, M.R.; Gonzalez, C.J. A preliminary study of cytokines in suicidal and nonsuicidal adolescents with major depression. J. Child. Adolesc. Psychopharmacol. 2009, 19, 423–430.
- Pallavi, P.; Sagar, R.; Mehta, M.; Sharma, S.; Subramanium, A.; Shamshi, F.; Sengupta, U.; Pandey, R.M.; Mukhopadhyay, A.K. Serum cytokines and anxiety in adolescent depression patients: Gender effect. Psychiatry Res. 2015, 229, 374–380.
- Linthorst, A.; Flachskamm, C.; Muller-Preuss, P.; Holsboer, F.; Reul, J. Effect of bacterial endotoxin and interleukin-1 beta on hippocampal serotonergic neurotransmission, behavioral activity, and free corticosterone levels: An in vivo microdialysis study. J. Neurosci. 1995, 15, 2920–2934.
- Rosenblat, J.D.; Cha, D.S.; Mansur, R.B.; McIntyre, R.S. Inflamed moods: A review of the interactions between inflammation and mood disorders. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2014, 53, 23–34.
- Gewurz, H.; Mold, C.; Siegel, J.; Fiedel, B. C-reactive protein and the acute phase response. Adv. Intern. Med. 1982, 27, 345–372.
- Copeland, W.E.; Shanahan, L.; Worthman, C.; Angold, A.; Costello, E.J. Cumulative depression episodes predict later C-reactive protein levels: A prospective analysis. Biol. Psychiatry 2012, 71, 15–21.
- Peters, A.T.; Ren, X.; Bessette, K.L.; Goldstein, B.I.; West, A.E.; Langenecker, S.A.; Pandey, G.N. Interplay between pro-inflammatory cytokines, childhood trauma, and executive function in depressed adolescents. J. Psychiatr. Res. 2019, 114, 1–10.
- Rengasamy, M.; Marsland, A.; McClain, L.; Kovats, T.; Walko, T.; Pan, L.; Price, R.B. Longitudinal relationships of cytokines, depression and anhedonia in depressed adolescents. Brain Behav. Immun. 2021, 91, 74–80.
- Kim, J.-W.; Szigethy, E.M.; Melhem, N.M.; Saghafi, E.M.; Brent, D.A. Inflammatory markers and the pathogenesis of pediatric depression and suicide: A systematic review of the literature. J. Clin. Psychiatry 2014, 75, 1242–1253.
- Colasanto, M.; Madigan, S.; Korczak, D.J. Depression and inflammation among children and adolescents: A meta-analysis. J. Affect. Disord. 2020, 1, 940–948.
- Gill, H.; El-Halabi, S.; Majeed, A.; Gill, B.; Lui, L.M.; Mansur, R.B.; Lipsitz, O.; Rodrigues, N.B.; Phan, L.; Chen-Li, D. The association between adverse childhood experiences and inflammation in patients with major depressive disorder: A systematic review. J. Affect. Disord. 2020, 272, 1–7.
- Slopen, N.; Kubzansky, L.D.; Koenen, K.C. Internalizing and externalizing behaviors predict elevated inflammatory markers in childhood. Psychoneuroendocrinology 2013, 38, 2854–2862.
- Chi, S.; Lee, M.-S. Personalized Medicine Using Neuroimmunological Biomarkers in Depressive Disorders. J. Pers. Med. 2021, 11, 114.
- Dantzer, R. Cytokine-induced sickness behaviour: A neuroimmune response to activation of innate immunity. Eur. J. Pharmacol. 2004, 500, 399–411.
- Brymer, K.J.; Romay-Tallon, R.; Allen, J.; Caruncho, H.J.; Kalynchuk, L.E. Exploring the potential antidepressant mechanisms of TNFα antagonists. Front. Neurosci. 2019, 13, 98.
- Grosse, L.; Ambrée, O.; Jörgens, S.; Jawahar, M.C.; Singhal, G.; Stacey, D.; Arolt, V.; Baune, B.T. Cytokine levels in major depression are related to childhood trauma but not to recent stressors. Psychoneuroendocrinology 2016, 73, 24–31.
- Moreira, F.P.; Wiener, C.D.; Jansen, K.; Portela, L.V.; Lara, D.R.; de Mattos Souza, L.D.; da Silva, R.A.; Oses, J.P. Childhood trauma and increased peripheral cytokines in young adults with major depressive: Population-based study. J. Neuroimmunol. 2018, 319, 112–116.
- D’Acunto, G.; Nageye, F.; Zhang, J.; Masi, G.; Cortese, S. Inflammatory cytokines in children and adolescents with depressive disorders: A systematic review and meta-analysis. J. Child. Adolesc. Psychopharmacol. 2019, 29, 362–369.
- Chaiton, M.; O’Loughlin, J.; Karp, I.; Lambert, M. Depressive symptoms and C-reactive protein are not associated in a population-based sample of adolescents. Int. J. Behav. Med. 2010, 17, 216–222.
- Miller, G.E.; Cole, S.W. Clustering of depression and inflammation in adolescents previously exposed to childhood adversity. Biol. Psychiatry 2012, 72, 34–40.
- Tabatabaeizadeh, S.-A.; Abdizadeh, M.F.; Meshkat, Z.; Khodashenas, E.; Darroudi, S.; Fazeli, M.; Ferns, G.A.; Avan, A.; Ghayour-Mobarhan, M. There is an association between serum high-sensitivity C-reactive protein (hs-CRP) concentrations and depression score in adolescent girls. Psychoneuroendocrinology 2018, 88, 102–104.
- De Punder, K.; Entringer, S.; Heim, C.; Deuter, C.E.; Otte, C.; Wingenfeld, K.; Kuehl, L.K. Inflammatory measures in depressed patients with and without a history of adverse childhood experiences. Front. Psychiatry 2018, 9, 610.
- Kraus, T.; Haack, M.; Schuld, A.; Hinze-Selch, D.; Koethe, D.; Pollmächer, T.J.P. Body weight, the tumor necrosis factor system, and leptin production during treatment with mirtazapine or venlafaxine. Pharmacopsychiatry 2002, 35, 220–225.
- Sasaki, T.; Niitsu, T.; Hashimoto, T.; Kanahara, N.; Shiina, A.; Hasegawa, T.; Kimura, H.; Ishikawa, M.; Tone, J.; Yamauchi, A. Decreased levels of serum brain-derived neurotrophic factor in male pediatric patients with depression. Open Clin. Chem. J. 2011, 4, 28–33.
- Pallavi, P.; Sagar, R.; Mehta, M.; Sharma, S.; Subramanium, A.; Shamshi, F.; Sengupta, U.; Qadri, R.; Pandey, R.M.; Mukhopadhyay, A.K. Serum neurotrophic factors in adolescent depression: Gender difference and correlation with clinical severity. J. Affect. Disord. 2013, 150, 415–423.
- Bilgiç, A.; Çelikkol Sadıç, Ç.; Kılınç, İ.; Akça, Ö.F. Exploring the association between depression, suicidality and serum neurotrophin levels in adolescents. Int. J. Psychiatry Clin. Pract. 2020, 24, 143–150.
- Carniel, B.P.; da Rocha, N.S. Brain-derived neurotrophic factor (BDNF) and inflammatory markers: Perspectives for the management of depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2020, 108, 110151.
- Martinowich, K.; Lu, B. Interaction between BDNF and serotonin: Role in mood disorders. Neuropsychopharmacology 2008, 33, 73–83.
- Kowiański, P.; Lietzau, G.; Czuba, E.; Waśkow, M.; Steliga, A.; Moryś, J. BDNF: A key factor with multipotent impact on brain signaling and synaptic plasticity. Cell. Mol. Neurobiol. 2018, 38, 579–593.
- Groves, J. Is it time to reassess the BDNF hypothesis of depression? Mol. Psychiatry 2007, 12, 1079–1088.
- Hashimoto, K. Brain-derived neurotrophic factor as a biomarker for mood disorders: An historical overview and future directions. Psychiatry Clin. Neurosci. 2010, 64, 341–357.
- Molendijk, M.; Spinhoven, P.; Polak, M.; Bus, B.; Penninx, B.; Elzinga, B. Serum BDNF concentrations as peripheral manifestations of depression: Evidence from a systematic review and meta-analyses on 179 associations (N = 9484). Mol. Psychiatry 2014, 19, 791–800.
- Matrisciano, F.; Bonaccorso, S.; Ricciardi, A.; Scaccianoce, S.; Panaccione, I.; Wang, L.; Ruberto, A.; Tatarelli, R.; Nicoletti, F.; Girardi, P. Changes in BDNF serum levels in patients with major depression disorder (MDD) after 6 months treatment with sertraline, escitalopram, or venlafaxine. J. Psychiatr. Res. 2009, 43, 247–254.
- Luan, S.; Zhou, B.; Wu, Q.; Wan, H.; Li, H. Brain-derived neurotrophic factor blood levels after electroconvulsive therapy in patients with major depressive disorder: A systematic review and meta-analysis. Asian J. Psychiatry 2020, 51, 101983.
- Zhao, X.; Li, Y.; Tian, Q.; Zhu, B.; Zhao, Z. Repetitive transcranial magnetic stimulation increases serum brain-derived neurotrophic factor and decreases interleukin-1β and tumor necrosis factor-α in elderly patients with refractory depression. J. Int. Med. Res. 2019, 47, 1848–1855.
- Kim, Y.-K.; Lee, H.-P.; Won, S.-D.; Park, E.-Y.; Lee, H.-Y.; Lee, B.-H.; Lee, S.-W.; Yoon, D.; Han, C.; Kim, D.-J. Low plasma BDNF is associated with suicidal behavior in major depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2007, 31, 78–85.
- Khan, M.S.; Wu, G.W.; Reus, V.I.; Hough, C.M.; Lindqvist, D.; Westrin, Å.; Nier, B.M.; Wolkowitz, O.M.; Mellon, S.H. Low serum brain-derived neurotrophic factor is associated with suicidal ideation in major depressive disorder. Psychiatry Res. 2019, 273, 108–113.
- Sun, Q.; Cui, C.; Fu, Y.; Ma, S.; Li, H. Nursing interventions in depressed children with low serum levels of BDNF. Exp. Ther. Med. 2017, 14, 2947–2952.
- Lee, J.; Lee, K.H.; Kim, S.H.; Han, J.Y.; Hong, S.-B.; Cho, S.-C.; Kim, J.-W.; Brent, D. Early changes of serum BDNF and SSRI response in adolescents with major depressive disorder. J. Affect. Disord. 2020, 265, 325–332.
- Carbone, D.L.; Handa, R.J. Sex and stress hormone influences on the expression and activity of brain-derived neurotrophic factor. Neuroscience 2013, 239, 295–303.
- Cubeddu, A.; Bucci, F.; Giannini, A.; Russo, M.; Daino, D.; Russo, N.; Merlini, S.; Pluchino, N.; Valentino, V.; Casarosa, E. Brain-derived neurotrophic factor plasma variation during the different phases of the menstrual cycle in women with premenstrual syndrome. Psychoneuroendocrinology 2011, 36, 523–530.
- Knorr, U.; Søndergaard, M.H.G.; Koefoed, P.; Jørgensen, A.; Faurholt-Jepsen, M.; Vinberg, M.; Kessing, L.V. Increased blood BDNF in healthy individuals with a family history of depression. Psychiatry Res. 2017, 256, 176–179.
- Bus, B.; Molendijk, M.; Tendolkar, I.; Penninx, B.; Prickaerts, J.; Elzinga, B.; Voshaar, R.O. Chronic depression is associated with a pronounced decrease in serum brain-derived neurotrophic factor over time. Mol. Psychiatry 2015, 20, 602–608.
- Pariante, C.M.; Lightman, S.L. The HPA axis in major depression: Classical theories and new developments. Trends Neurosci. 2008, 31, 464–468.
- Lopez-Duran, N.L.; Kovacs, M.; George, C.J. Hypothalamic-pituitary-adrenal axis dysregulation in depressed children and adolescents: A meta-analysis. Psychoneuroendocrinology 2009, 34, 1272–1283.
- Gjerstad, J.K.; Lightman, S.L.; Spiga, F. Role of glucocorticoid negative feedback in the regulation of HPA axis pulsatility. Stress 2018, 21, 403–416.
- Heim, C.; Newport, D.J.; Mletzko, T.; Miller, A.H.; Hemeroff, C.B. The link between childhood trauma and depression: Insights from HPA axis studies in humans. Psychoneuroendocrinology 2008, 33, 693–710.
- Vreeburg, S.A.; Hartman, C.A.; Hoogendijk, W.J.; van Dyck, R.; Zitman, F.G.; Ormel, J.; Penninx, B.W. Parental history of depression or anxiety and the cortisol awakening response. Br. J. Psychiatry 2010, 197, 180–185.
- Rao, U.; Hammen, C.L.; Poland, R.E. Risk markers for depression in adolescents: Sleep and HPA measures. Neuropsychopharmacology 2009, 34, 1936–1945.
- Daria, S.; Proma, M.A.; Shahriar, M.; Islam, S.M.A.; Bhuiyan, M.A.; Islam, M.R. Serum interferon-gamma level is associated with drug-naive major depressive disorder. SAGE Open Med. 2020, 8, 2050312120974169.
- Koo, J.W.; Russo, S.J.; Ferguson, D.; Nestler, E.J.; Duman, R.S. Nuclear factor-kappaB is a critical mediator of stress-impaired neurogenesis and depressive behavior. Proc. Natl. Acad. Sci. USA 2010, 107, 2669–2674.
- Fischer, S.; Ehlert, U.; Amiel Castro, R. Hormones of the hypothalamic-pituitary-gonadal (HPG) axis in male depressive disorders—A systematic review and meta-analysis. Front. Neuroendocr. 2019, 55, 100792.
- Dwyer, A.A.; Phan-Hug, F.; Hauschild, M.; Elowe-Gruau, E.; Pitteloud, N. Transition in endocrinology: Hypogonadism in adolescence. Eur. J. Endocrinol. 2015, 173, R15–R24.
- Dorn, L.D.; Burgess, E.S.; Dichek, H.L.; Putnam, F.W.; Chrousos, G.P.; Gold, P.W. Thyroid hormone concentrations in depressed and nondepressed adolescents: Group differences and behavioral relations. J. Am. Acad. Child. Adolesc. Psychiatry 1996, 35, 299–306.
- Dorn, L.D.; Dahl, R.E.; Birmaher, B.; Williamson, D.E.; Kaufman, J.; Frisch, L.; Perel, J.M.; Ryan, N.D. Baseline thyroid hormones in depressed and non-depressed pre- and early-pubertal boys and girls. J. Psychiatr. Res. 1997, 31, 555–567.
- Hirtz, R.; Libuda, L.; Hinney, A.; Focker, M.; Buhlmeier, J.; Antel, J.; Holterhus, P.M.; Kulle, A.; Kiewert, C.; Hebebrand, J.; et al. Lack of Evidence for a Relationship Between the Hypothalamus-Pituitary-Adrenal and the Hypothalamus-Pituitary-Thyroid Axis in Adolescent Depression. Front. Endocrinol. 2021, 12, 662243.
- Yarandi, S.S.; Peterson, D.A.; Treisman, G.J.; Moran, T.H.; Pasricha, P.J. Modulatory effects of gut microbiota on the central nervous system: How gut could play a role in neuropsychiatric health and diseases. J. Neurogastroenterol. Motil. 2016, 22, 201.
- Rao, M.; Gershon, M.D. The bowel and beyond: The enteric nervous system in neurological disorders. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 517.
- Ohlsson, L.; Gustafsson, A.; Lavant, E.; Suneson, K.; Brundin, L.; Westrin, Å.; Ljunggren, L.; Lindqvist, D. Leaky gut biomarkers in depression and suicidal behavior. Acta Psychiatr. Scand. 2019, 139, 185–193.
- Asbjornsdottir, B.; Snorradottir, H.; Andresdottir, E.; Fasano, A.; Lauth, B.; Gudmundsson, L.S.; Gottfredsson, M.; Halldorsson, T.I.; Birgisdottir, B.E. Zonulin-dependent intestinal permeability in children diagnosed with mental disorders: A systematic review and meta-analysis. Nutrients 2020, 12, 1982.
- Calarge, C.A.; Devaraj, S.; Shulman, R.J. Gut permeability and depressive symptom severity in unmedicated adolescents. J. Affect. Disord. 2019, 246, 586–594.
- Simkin, D.R. Microbiome and mental health, specifically as it relates to adolescents. Curr. Psychiatry Rep. 2019, 21, 1–12.
- Furet, J.-P.; Firmesse, O.; Gourmelon, M.; Bridonneau, C.; Tap, J.; Mondot, S.; Doré, J.; Corthier, G. Comparative assessment of human and farm animal faecal microbiota using real-time quantitative PCR. FEMS Microbiol. Ecol. 2009, 68, 351–362.
- Kuo, P.-H.; Chung, Y.-C.E. Moody microbiome: Challenges and chances. J. Formos. Med. Assoc. 2019, 118, S42–S54.
- Jiang, H.Y.; Pan, L.Y.; Zhang, X.; Zhang, Z.; Zhou, Y.Y.; Ruan, B. Altered gut bacterial–fungal interkingdom networks in patients with current depressive episode. Brain Behav. 2020, 10, e01677.
- Michels, N.; Van de Wiele, T.; Fouhy, F.; O’Mahony, S.; Clarke, G.; Keane, J. Gut microbiome patterns depending on children’s psychosocial stress: Reports versus biomarkers. Brain Behav. Immun. 2019, 80, 751–762.
- Thapa, S.; Sheu, J.C.; Venkatachalam, A.; Runge, J.K.; Luna, R.A.; Calarge, C.A. Gut microbiome in adolescent depression. J. Affect. Disord. 2021, 292, 500–507.
- Wingfield, B.; Lapsley, C.; McDowell, A.; Miliotis, G.; McLafferty, M.; O’Neill, S.M.; Coleman, S.; McGinnity, T.M.; Bjourson, A.J.; Murray, E.K. Variations in the oral microbiome are associated with depression in young adults. Sci. Rep. 2021, 11, 1–9.
- Simpson, C.A.; Adler, C.; du Plessis, M.R.; Landau, E.R.; Dashper, S.G.; Reynolds, E.C.; Schwartz, O.S.; Simmons, J.G. Oral microbiome composition, but not diversity, is associated with adolescent anxiety and depression symptoms. Physiol. Behav. 2020, 226, 113126.
- Black, C.N.; Bot, M.; Scheffer, P.G.; Cuijpers, P.; Penninx, B.W. Is depression associated with increased oxidative stress? A systematic review and meta-analysis. Psychoneuroendocrinology 2015, 51, 164–175.
- Lindqvist, D.; Dhabhar, F.S.; James, S.J.; Hough, C.M.; Jain, F.A.; Bersani, F.S.; Reus, V.I.; Verhoeven, J.E.; Epel, E.S.; Mahan, L. Oxidative stress, inflammation and treatment response in major depression. Psychoneuroendocrinology 2017, 76, 197–205.
- Horn, S.R.; Leve, L.D.; Levitt, P.; Fisher, P.A. Childhood adversity, mental health, and oxidative stress: A pilot study. PLoS ONE 2019, 14, e0215085.
- Guney, E.; Ceylan, M.F.; Tektas, A.; Alisik, M.; Ergin, M.; Goker, Z.; Dinc, G.S.; Ozturk, O.; Korkmaz, A.; Eker, S. Oxidative stress in children and adolescents with anxiety disorders. J. Affect. Disord. 2014, 156, 62–66.
- Tobore, T.O. On the potential harmful effects of E-Cigarettes (EC) on the developing brain: The relationship between vaping-induced oxidative stress and adolescent/young adults social maladjustment. J. Adolesc. 2019, 76, 202–209.
- Moradi-Kor, N.; Dadkhah, M.; Ghanbari, A.; Rashidipour, H.; Bandegi, A.R.; Barati, M.; Kokhaei, P.; Rashidy-Pour, A. Protective Effects of Spirulina platensis, Voluntary Exercise and Environmental Interventions Against Adolescent Stress-Induced Anxiety and Depressive-Like Symptoms, Oxidative Stress and Alterations of BDNF and 5HT-3 Receptors of the Prefrontal Cortex in Female Rats. Neuropsychiatr. Dis. Treat. 2020, 16, 1777.
- Ting, E.Y.; Yang, A.C.; Tsai, S.J. Role of Interleukin-6 in Depressive Disorder. Int. J. Mol. Sci. 2020, 21, 2194.
