Among the many modifiable factors that can increase the risk of morbidity and mortality in kidney transplant (KT) recipients, physical inactivity is often neglected. This attitude is certainly questionable since a sedentary lifestyle has been widely recognized as a “silent killer” and a major health issue in the general population, particularly in more economically developed countries. It is now well-known that physical inactivity is independently associated with a higher risk of cardiovascular disease (CVD), diabetes mellitus, obesity, osteoporosis, and breast and colonic cancer.
- physical activity
- sedentary behavior
- exercise
- sport
- cardiovascular disease
- frailty
- quality of life
- outcomes
- kidney transplant
- review
1. Introduction
Among the many modifiable factors that can increase the risk of morbidity and mortality in kidney transplant (KT) recipients, physical inactivity is often neglected. This attitude is certainly questionable since a sedentary lifestyle has been widely recognized as a “silent killer” and a major health issue in the general population, particularly in more economically developed countries [1][2][3]. It is now well-known that physical inactivity is independently associated with a higher risk of cardiovascular disease (CVD), diabetes mellitus, obesity, osteoporosis, and breast and colonic cancer [4][5]. On the other hand, there is mounting evidence that regular physical activity can effectively prevent several chronic diseases, depression, and premature death [6][7].
The World Health Organization (WHO) has defined physical activity as any bodily movement produced by skeletal muscles that requires energy expenditure. The intensity of physical activity ranges from light (slow walking) to moderate (brisk walking, jogging, stair-climbing) to vigorous (fast running, sprinting, fast cycling). In order to reduce the burden on global health, the WHO has also recently provided guidelines on physical activity and sedentary behavior for children, adolescents, adults, and older adults. The guidelines for adults (namely, subjects between 18 and 64 years) recommended a minimum of 150–300 min of moderate-intensity aerobic physical activity, 75–150 min of vigorous-intensity aerobic physical activity, or an equivalent combination of both throughout the week to achieve substantial benefits. Adults should also carry out muscle-strengthening activities at moderate or greater intensity, with the involvement of all major muscle groups, at least twice a week. For older adults, the recommendations are similar. This group should perform varied multicomponent physical activity that emphasizes functional balance and strength training at moderate or greater intensity, three or more days a week [8].
2. Physical Activity before and after Kidney Transplantation
Current acceptance criteria for KT candidates are far more liberal than in the past. Even though there is still considerable variability in recipient-selection policies among centers worldwide, it is undeniable that many conditions previously recognized as an absolute contraindication to transplantation are now considered as a marginal issue. Progressively, we have witnessed the enlistment of more and more frail patients, often at extreme ages or with multiple comorbidities. One of the many consequences of such a shift of vision is that a number of potential transplant recipients, including children, exhibit low exercise capacity and are physically inactive [9][10]. The specific burden of chronic kidney disease (CKD) should also be taken into account. Indeed, elderly subjects waiting for a KT usually show inferior physical performances and are at higher risk of disability than age-matched individuals with other non-communicable chronic diseases [11]. Additionally, it has been observed that patients on regular dialysis undergo physical activity only nine days per month, with 43.9% not exercising at all [12]. Indeed, it seems that inactivity-related morbidity increases with the duration and type of renal replacement therapy (RRT). As such, it is plausible that preemptive KT recipients may achieve better physical performances than patients requiring prolonged dialysis before transplant. On the other hand, recent studies have demonstrated that physical aerobic exercise can exert various beneficial effects on many clinical outcomes, both in patients with CKD and under dialysis [13][14].
In KT recipients, the intertwining of dialysis vintage, comorbid conditions, psychosocial, and socioeconomic factors, as well as chronic exposure to immunosuppression, can all negatively affect physical activity. Calcineurin inhibitors cyclosporine and tacrolimus may induce myopathy and muscular atrophy through inhibition of calcineurin, which is a regulator of muscle mass [15]. Mammalian targeting of rapamycin (mTOR) inhibitors sirolimus and everolimus can also cause muscular atrophy by inhibiting the Akt/mTOR pathway that regulates skeletal muscle hypertrophy [16]. Steroid administration is associated with decreased protein synthesis and increased protein catabolism and may lead to muscle atrophy [17].
Even after a successful transplant, it is not uncommon that patients continue to feel as disabled as they were while on dialysis. Furthermore, in this specific group of patients, physical inactivity, defined as achieving less than 30 min of moderate-intensity exercise per week, is now recognized as a strong predictor of all-cause mortality and death with allograft function. A combination of semi-structured interviews and surveys provided two months after transplant by 88 renal allograft recipients has reported that 76% of the participants were sedentary, 13% exercised irregularly, and only 11% exercised regularly [18]. An Italian retrospective survey of KT recipients with at least a 10-year follow-up gathered information on physical activity from 6.055 patients. According to the results, 51.6% of the recipients were active whilst 48.4% were inactive. The lowest inclination to physical activity was observed in overweight and obese subjects, as well as in patients with longer dialysis vintage and older age at transplant. Noticeably, the propensity score showed that graft function was better preserved among active KT recipients [19].
Often underrated, further barriers to regular physical activity and adherence to exercise programs after transplant are the cost of fitness centers, inadequate exercise guidelines, medications side effects, feelings of total-body weakness, and the paucity of dedicated programs, as well as the need for expensive insurance coverage for participants. These issues should also be addressed to assist patients in managing their transplants [20][21]. Moreover, some patients are concerned that physical activity may interfere with the function of the allograft. In clinical practice, they often ask whether running, hiking, or gymnastics can put their transplant at risk. For all these reasons, many transplant recipients, in particular elderly subjects and children, remain sedentary. In an attempt to overcome some of these obstacles, various strategies have been proposed. For patients with end-stage kidney disease, the opportunity to offer nurse-led intra-dialytic or home-based supervised exercise programs appears particularly intriguing [22], while for transplant recipients, improved adherence to physical activity and increased walking capacity have been obtained using home-based exercise interventions with the aid of remotely-monitored wearable devices, financial incentives, or health-engagement questions [23].
3. Consequences of Physical Inactivity
Skeletal muscle is now recognized as an endocrine organ producing a panel of cytokines and peptides, also known as myokines, that can communicate with other organs, including adipose tissue [24]. Myokines production is strictly dependent on muscle contraction. As such, any muscle dysfunction may ultimately affect myokines production and potentially cause deleterious effects on multiple organs and systems. Sedentary behavior is associated with muscle wasting and visceral fat accumulation. The combination of physical inactivity and adiposity are in turn associated with elevated plasma levels of pro-inflammatory molecules, in particular interleukin-6 (IL-6) and serum c-reactive protein (CRP) [25][26]. At first glance, this observation may seem paradoxical since IL-6 and other pro-inflammatory cytokines are transiently secreted in large doses by several metabolically active tissues during exercise and muscular contraction [27]. However, it has been shown that muscle disuse may lead to IL-6 resistance with subsequently elevated levels of circulating IL-6. In this setting, the increase in plasma IL-6 concentration that accompanies obesity and physical inactivity may represent a compensatory mechanism, resulting in persistent low-grade systemic inflammation [28]. Chronically low-grade systemic inflammation has been linked to the development of many inflammation-related diseases [29].
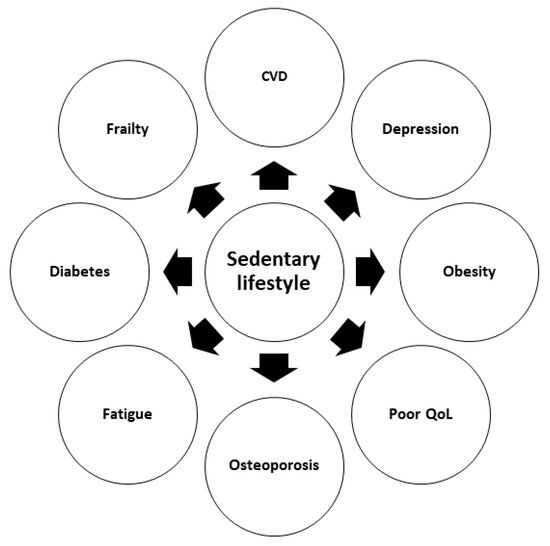
A number of retrospective studies in KT settings have suggested that low levels of physical activity independently predict post-transplant outcomes, including mortality, particularly in obese and elderly patients [30][31][32]. In another retrospective analysis on 540 KT patients, low physical activity (properly defined using validated questionnaires) was strongly associated with an increased risk of cardiovascular and all-cause mortality [33]. Similar results were obtained in an elegant prospective study evaluating long-term (8 years) outcomes of a cohort of 507 adult KT recipients assessed using the Physical Activity Scale for the Elderly at the time of transplantation. According to this scale, participants were sorted into three groups (tertiles). The overall mortality rate was about 25%, and most of the deaths were recorded in patients with a functioning allograft. Interestingly, multivariable Cox regression analysis showed a statistically significant association between the physical activity score and both recipient and transplant survival [34]. Research performed on 4034 KT recipients has reported that lower levels of physical activity are associated with a higher prevalence of obesity, diabetes mellitus, and CVD [35]. Other major consequences of poor physical activity are the loss of muscle mass and frailty, an independent risk factor for post-transplant adverse events, especially in aged patients [36][37]. As a matter of fact, in older subjects, low physical activity level combined with excessive time spent in sedentary behavior (physical activity level < 150 min per week and sedentary behavior ≥ 540 min per day) has been associated with frailty, resulting in a frailty prevalence ratio of 2.83 [38]. Physical inactivity in KT recipients is associated not only with increased mortality, but also with fatigue [39][40], depression [41], and inferior quality of life (QoL) [42][43]. Lastly, in a KT candidate, frailty may reduce the chances to be admitted to, or remain active on, the transplant waiting list [44][45][46].
In summary, available data show that post-transplant lack of regular exercise may negatively impact QoL and cause severe complications. Proper evaluation of physical activity levels at the time of enlistment could represent a valuable tool for risk stratification and prediction of post-transplant patient survival.
The negative effects of sedentary behavior on KT-related outcomes are summarized in Figure 1 .
4. Physical Exercise and Sport after Transplantation
Since frailty and poor physical activity are associated with adverse outcomes after KT, physical performing tests, including the six-minute walk test and the sit-to-stand test, should be included in the evaluation of transplant candidates [47]. Furthermore, a rehabilitation program before surgery may be designed in frail candidates to prevent poor post-transplant outcomes. In a small pilot study, a pre-habilitation program consisted of weekly physical therapy sessions conducted at an outpatient clinic and at home. After two months of follow-up, enrolled subjects managed to improve their physical activity by 64% based on accelerometry. Five pre-habilitation participants received a KT during the study. Remarkably, their length of stay was shorter than in age-, sex-, and race-matched controls [48].
Fitness counseling should be offered to both patients on the transplant waiting list and KT recipients as a fundamental part of their routine medical care and follow-up programs. When promoting specific intervention strategies, healthcare professionals should always bear in mind that physical exercise may entail two major types of risk: osteo-skeletal and cardiovascular, depending on the physical activity and the underlying disease. There is a lack of formal recommendations on specific sports restrictions. The available literature is also extremely limited [49]. Even though there are examples of transplanted patients playing in professional soccer or rugby teams, it sounds reasonable to avoid full-contact impact sports that might result in injury to the area of the body where the kidney has been implanted. Prophylactic use of specifically designed padded sports clothing that protects the lower abdomen may be considered when practicing low- to moderate-risk activities. The burden of chronic allograft insufficiency and long-term immunosuppressive therapy should also be considered. Nonetheless, it seems appropriate to underline the fact that the perceived danger associated with sports and most concerns on physical activity-related safety issues after KT are often exaggerated. A systematic review of exercise training programs in KT patients has recently examined 24 studies with a cumulative population of 654 recipients and 536 controls. Included subjects were tested with an aerobic exercise program, a resistance exercise program, or a combination of both. Each intervention consisted of 20–60 min sessions, repeated 2–3 times per week for 5.5 months (median duration). The vast majority of the trials analyzed showed that all the interventions considered led to a significant increase in cardiorespiratory fitness and maximal heart rate and were associated with a substantial amelioration of muscle performance and strength. A trend towards weight loss in overweight or obese patients with stable allograft function, as well as an improvement in several aspects of QoL, were also noted. Remarkably, there was no harm in exercising or performing any physical activity [50]. These data show that regular exercise improves physical fitness and health-related QoL. It can also reduce overweight with a favorable effect on the cardiovascular system.
This entry is adapted from the peer-reviewed paper 10.3390/jpm11090927
References
- Kushi, L.H.; Fee, R.M.; Folsom, A.R.; Mink, P.J.; Anderson, K.E.; Sellers, T.A. Physical activity and mortality in postmenopausal women. JAMA 1997, 277, 1287–1292.
- Fried, L.P.; Kronmal, R.A.; Newman, A.B.; Bild, D.E.; Mittelmark, M.B.; Polak, J.F.; Robbins, J.A.; Gardin, J.M. Risk factors for 5-year mortality in older adults: The Cardiovascular Health Study. JAMA 1998, 279, 585–592.
- Andersen, L.B.; Schnohr, P.; Schroll, M.; Hein, H.O. All-cause mortality associated with physical activity during leisure time, work, sports, and cycling to work. Arch. Intern. Med. 2000, 160, 1621–1628.
- Ekelund, U.; Steene-Johannessen, J.; Brown, W.J.; Fagerland, M.W.; Owen, N.; Powell, K.E.; Bauman, A.; Lee, I.M.; Lancet Physical Activity Series 2 Executive Committe; Lancet Sedentary Behaviour Working Group. Does physical activity attenuate, or even eliminate, the detrimental association of sitting time with mortality? A harmonised meta-analysis of data from more than 1 million men and women. Lancet 2016, 388, 1302–1310.
- Kyu, H.H.; Bachman, V.F.; Alexander, L.T.; Mumford, J.E.; Afshin, A.; Estep, K.; Veerman, J.L.; Delwiche, K.; Iannarone, M.L.; Moyer, M.L.; et al. Physical activity and risk of breast cancer, colon cancer, diabetes, ischemic heart disease, and ischemic stroke events: Systematic review and dose-response meta-analysis for the Global Burden of Disease Study 2013. BMJ 2016, 354, i3857.
- O’Dwyer, T.; Durcan, L.; Wilson, F. Exercise and physical activity in systemic lupus erythematosus: A systematic review with meta-analyses. Semin. Arthritis Rheum. 2017, 47, 204–215.
- Groessl, E.J.; Kaplan, R.M.; Rejeski, W.J.; Katula, J.A.; Glynn, N.W.; King, A.C.; Anton, S.D.; Walkup, M.; Lu, C.J.; Reid, K.; et al. Physical Activity and Performance Impact Long-term Quality of Life in Older Adults at Risk for Major Mobility Disability. Am. J. Prev. Med. 2019, 56, 141–146.
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.P.; Chastin, S.; Chou, R.; et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med. 2020, 54, 1451–1462.
- Wolf, M.F.; George, R.P.; Warshaw, B.; Wang, E.; Greenbaum, L.A. Physical Activity and Kidney Injury in Pediatric and Young Adult Kidney Transplant Recipients. J Pediatr. 2016, 179, 90–95.e2.
- Kutner, N.G.; Zhang, R.; Bowles, T.; Painter, P. Pretransplant physical functioning and kidney patients’ risk for posttransplantation hospitalization/death: Evidence from a national cohort. Clin. J. Am. Soc. Nephrol. 2006, 1, 837–843.
- Hartmann, E.L.; Kitzman, D.; Rocco, M.; Leng, X.; Klepin, H.; Gordon, M.; Rejeski, J.; Berry, M.; Kritchevsky, S. Physical function in older candidates for renal transplantation: An impaired population. Clin. J. Am. Soc. Nephrol. 2009, 4, 588–594.
- Manfredini, F.; Mallamaci, F.; D’Arrigo, G.; Baggetta, R.; Bolignano, D.; Torino, C.; Lamberti, N.; Bertoli, S.; Ciurlino, D.; Rocca-Rey, L.; et al. Exercise in Patients on Dialysis: A Multicenter, Randomized Clinical Trial. J. Am. Soc. Nephrol. 2017, 28, 1259–1268.
- Mallamaci, F.; Pisano, A.; Tripepi, G. Physical activity in chronic kidney disease and the EXerCise Introduction to Enhance trial. Nephrol. Dial. Transplant. 2020, 35 (Suppl. 2), ii18–ii22.
- Hargrove, N.; El Tobgy, N.; Zhou, O.; Pinder, M.; Plant, B.; Askin, N.; Bieber, L.; Collister, D.; Whitlock, R.; Tangri, N.; et al. Effect of Aerobic Exercise on Dialysis-Related Symptoms in Individuals Undergoing Maintenance Hemodialysis: A Systematic Review and Meta-Analysis of Clinical Trials. Clin. J. Am. Soc. Nephrol. 2021, 16, 560–574.
- Hudson, M.B.; Price, S.R. Calcineurin: A poorly understood regulator of muscle mass. Int. J. Biochem. Cell Biol. 2013, 45, 2173–2178.
- Bodine, S.C.; Stitt, T.N.; Gonzalez, M.; Kline, W.O.; Stover, G.L.; Bauerlein, R.; Zlotchenko, E.; Scrimgeour, A.; Lawrence, J.C.; Glass, D.J.; et al. Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat. Cell Biol. 2001, 3, 1014–1019.
- Hasselgren, P.O. Glucocorticoids and muscle catabolism. Curr. Opin. Clin. Nutr. Metab. Care 1999, 2, 201–205.
- Gordon, E.J.; Prohaska, T.R.; Gallant, M.P.; Sehgal, A.R.; Strogatz, D.; Conti, D.; Siminoff, L.A. Prevalence and determinants of physical activity and fluid intake in kidney transplant recipients. Clin. Transplant. 2010, 24, E69–E81.
- Masiero, L.; Puoti, F.; Bellis, L.; Lombardini, L.; Totti, V.; Angelini, M.L.; Spazzoli, A.; Nanni Costa, A.; Cardillo, M.; Sella, G.; et al. Physical activity and renal function in the Italian kidney transplant population. Ren. Fail. 2020, 42, 1192–1204.
- Gordon, E.J.; Prohaska, T.R.; Gallant, M.; Siminoff, L.A. Self-care strategies and barriers among kidney transplant recipients: A qualitative study. Chronic Illn. 2009, 5, 75–91.
- Zelle, D.M.; Klaassen, G.; van Adrichem, E.; Bakker, S.J.; Corpeleijn, E.; Navis, G. Physical inactivity: A risk factor and target for intervention in renal care. Nat. Rev. Nephrol. 2017, 13, 152–168.
- Perez-Dominguez, B.; Casaña-Granell, J.; Garcia-Maset, R.; Garcia-Testal, A.; Melendez-Oliva, E.; Segura-Orti, E. Effects of exercise programs on physical function and activity levels in patients undergoing hemodialysis. A randomized controlled trial. Eur. J. Phys. Rehabil. Med. 2021. Epub ahead of print.
- Serper, M.; Barankay, I.; Chadha, S.; Shults, J.; Jones, L.S.; Olthoff, K.M.; Reese, P.P. A randomized, controlled, behavioral intervention to promote walking after abdominal organ transplantation: Results from the LIFT study. Transpl. Int. 2020, 33, 632–643.
- Gomarasca, M.; Banfi, G.; Lombardi, G. Myokines: The endocrine coupling of skeletal muscle and bone. Adv. Clin. Chem. 2020, 94, 155–218.
- Pedersen, B.K.; Febbraio, M.A. Muscles, exercise and obesity: Skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 2012, 8, 457–465.
- Burini, R.C.; Anderson, E.; Durstine, J.L.; Carson, J.A. Inflammation, physical activity, and chronic disease: An evolutionary perspective. Sports Med. Health Sci. 2020, 2, 1–6.
- Pedersen, B.K.; Steensberg, A.; Schjerling, P. Muscle-derived interleukin-6: Possible biological effects. J. Physiol. 2001, 536, 329–337.
- Pedersen, B.K.; Steensberg, A.; Fischer, C.; Keller, C.; Keller, P.; Plomgaard, P.; Febbraio, M.; Saltin, B. Searching for the exercise factor: Is IL-6 a candidate? J. Muscle Res. Cell Motil. 2003, 24, 113–119.
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832.
- Katzmarzyk, P.T.; Janssen, I.; Ardern, C.I. Physical inactivity, excess adiposity and premature mortality. Obes. Rev. 2003, 4, 257–290.
- Hu, F.B.; Willett, W.C.; Li, T.; Stampfer, M.J.; Colditz, G.A.; Manson, J.E. Adiposity as compared with physical activity in predicting mortality among women. N. Engl. J. Med. 2004, 351, 2694–2703.
- Yango, A.F.; Gohh, R.Y.; Monaco, A.P.; Reinert, S.E.; Gautam, A.; Dworkin, L.D.; Morrissey, P.E. Excess risk of renal allograft loss and early mortality among elderly recipients is associated with poor exercise capacity. Clin. Nephrol. 2006, 65, 401–407.
- Zelle, D.M.; Corpeleijn, E.; Stolk, R.P.; de Greef, M.H.; Gans, R.O.; van der Heide, J.J.; Navis, G.; Bakker, S.J. Low physical activity and risk of cardiovascular and all-cause mortality in renal transplant recipients. Clin. J. Am. Soc. Nephrol. 2011, 6, 898–905.
- Rosas, S.E.; Reese, P.P.; Huan, Y.; Doria, C.; Cochetti, P.T.; Doyle, A. Pretransplant physical activity predicts all-cause mortality in kidney transplant recipients. Am. J. Nephrol. 2012, 35, 17–23.
- Kang, A.W.; Garber, C.E.; Eaton, C.B.; Risica, P.M.; Bostom, A.G. Physical Activity and Cardiovascular Risk among Kidney Transplant Patients. Med. Sci. Sports Exerc. 2019, 51, 1154–1161.
- Harhay, M.N.; Rao, M.K.; Woodside, K.J.; Johansen, K.L.; Lentine, K.L.; Tullius, S.G.; Parsons, R.F.; Alhamad, T.; Berger, J.; Cheng, X.S.; et al. An overview of frailty in kidney transplantation: Measurement, management and future considerations. Nephrol. Dial. Transplant. 2020, 35, 1099–1112.
- McAdams-DeMarco, M.A.; Ying, H.; Olorundare, I.; King, E.A.; Haugen, C.; Buta, B.; Gross, A.L.; Kalyani, R.; Desai, N.M.; Dagher, N.N.; et al. Individual Frailty Components and Mortality in Kidney Transplant Recipients. Transplantation 2017, 101, 2126–2132.
- Da Silva, V.D.; Tribess, S.; Meneguci, J.; Sasaki, J.E.; Garcia-Meneguci, C.A.; Carneiro, J.A.O.; Virtuoso, J.S., Jr. Association between frailty and the combination of physical activity level and sedentary behavior in older adults. BMC Public Health 2019, 19, 709.
- Bossola, M.; Pepe, G.; Vulpio, C. Fatigue in kidney transplant recipients. Clin. Transplant. 2016, 30, 1387–1393.
- Goedendorp, M.M.; Hoitsma, A.J.; Bloot, L.; Bleijenberg, G.; Knoop, H. Severe fatigue after kidney transplantation: A highly prevalent, disabling and multifactorial symptom. Transpl. Int. 2013, 26, 1007–1015.
- Zelle, D.M.; Dorland, H.F.; Rosmalen, J.G.; Corpeleijn, E.; Gans, R.O.; Homan van der Heide, J.J.; van Son, W.J.; Navis, G.; Bakker, S.J. Impact of depression on long-term outcome after renal transplantation: A prospective cohort study. Transplantation 2012, 94, 1033–1040.
- Takahashi, A.; Hu, S.L.; Bostom, A. Physical Activity in Kidney Transplant Recipients: A Review. Am. J. Kidney Dis. 2018, 72, 433–443.
- Macdonald, J.H.; Kirkman, D.; Jibani, M. Kidney transplantation: A systematic review of interventional and observational studies of physical activity on intermediate outcomes. Adv. Chronic Kidney Dis. 2009, 16, 482–500.
- Vleut, R.; Abramowicz, D.; Hellemans, R. Frailty: A new comorbidity in kidney transplant candidates? Nephrol. Dial. Transplant. 2020, 35, 1085–1087.
- Ponticelli, C.; Podestà, M.A.; Graziani, G. Renal transplantation in elderly patients. How to select the candidates to the waiting list? Transplant. Rev. 2014, 28, 188–192.
- Tennankore, K.K.; Gunaratnam, L.; Suri, R.S.; Yohanna, S.; Walsh, M.; Tangri, N.; Prasad, B.; Gogan, N.; Rockwood, K.; Doucette, S.; et al. Frailty and the Kidney Transplant Wait List: Protocol for a Multicenter Prospective Study. Can. J. Kidney Health Dis. 2020, 7, 2054358120957430.
- Cheng, X.S.; Myers, J.; Han, J.; Stedman, M.R.; Watford, D.J.; Lee, J.; Discipulo, K.V.; Chan, K.N.; Chertow, G.M.; Tan, J.C. Physical Performance Testing in Kidney Transplant Candidates at the Top of the Waitlist. Am. J. Kidney Dis. 2020, 76, 815–825.
- McAdams-DeMarco, M.A.; Ying, H.; Van Pilsum Rasmussen, S.; Schrack, J.; Haugen, C.E.; Chu, N.M.; González Fernández, M.; Desai, N.; Walston, J.D.; Segev, D.L. Prehabilitation prior to kidney transplantation: Results from a pilot study. Clin. Transplant. 2019, 33, e13450.
- Totti, V.; Zancanaro, M.; Trerotola, M.; Nanni Costa, A.; Antonetti, T.; Anedda, A.; Roi, G.S. Quality of life and energy expenditure in transplant recipient football players. Transplant. Proc. 2013, 45, 2758–2760.
- Calella, P.; Hernández-Sánchez, S.; Garofalo, C.; Ruiz, J.R.; Carrero, J.J.; Bellizzi, V. Exercise training in kidney transplant recipients: A systematic review. J. Nephrol. 2019, 32, 567–579.
