Microglia play a critical role in both homeostasis and disease, displaying a wide variety in terms of density, functional markers and transcriptomic profiles along the different brain regions as well as under injury or pathological conditions, such as Alzheimer’s disease (AD). The generation of reliable models to study into a dysfunctional microglia context could provide new knowledge towards the contribution of these cells in AD. In this work, we included an overview of different microglial depletion approaches. We also reported unpublished data from our genetic microglial depletion model, Cx3cr1CreER/Csf1rflx/flx, in which we temporally controlled microglia depletion by either intraperitoneal (acute model) or oral (chronic model) tamoxifen administration. Our results reported a clear microglial repopulation, then pointing out that our model would mimic a context of microglial replacement instead of microglial dysfunction. Next, we evaluated the origin and pattern of microglial repopulation. Additionally, we also reviewed previous works assessing the effects of microglial depletion in the progression of Aβ and Tau pathologies, where controversial data are found, probably due to the heterogeneous and time-varying microglial phenotypes observed in AD. Despite that, microglial depletion represents a promising tool to assess microglial role in AD and design therapeutic strategies
- microglia
- Alzheimer’s disease
- depletion
- inflammation
1. Microglia: Micro in Size but Macro in Functions, Highly Important in Alzheimer’s Disease
Microglia, the primary immune cells of the brain, not only survey the environment for pathogens and debris, but also play other important roles in the central nervous system (CNS), providing direct sustain to neurons and supporting myelinogenesis, synaptic plasticity, and the neoformation of vessels [1,2]. These glial cells account for 10–15% of the total cells in the adult CNS in humans [3] and 5–12% in mice [4]. Microglial cells derive from myeloid progenitors of the yolk sac that at embryonic day 8.5 colonize the mouse fetal brain, and actively proliferate at early postnatal days until reaching their definitive brain density [3]. Although the number of microglial cells remains constant during lifetime in mice and humans, a rapid turnover of microglia is maintained by a balanced coupling of microglial proliferation and apoptotic death [5]. In adult life, a 28% of microglia are renewed daily, meaning the lifespan for these cells 4.2 years [6]. Spatial heterogeneity of microglia has been observed in terms of density, functional markers and transcriptomic profiles. Moreover, microglia suffer transcriptional, morphological, and functional changes during aging, injury or pathological conditions, as multiple sclerosis, Parkinson’s disease (PD), and Alzheimer’s disease (AD), among others.
In this review, we specifically focused on the role of microglia in AD. In patients, AD pathology develops along a continuum process (ATN), in which the amyloid deposition is considered the earlier event, preceding and triggering Tau pathology and neurodegeneration [7,8,9]. However, microglial role in the ATN continuum remains unsolved. Microglial activation and the loss of their homeostatic functions are considered as critical features in AD pathogenesis. Recent single-cell transcriptomic studies have identified different microglial subpopulations involved in AD [10,11,12,13,14], although the functional significance of this microglial diversity is not clearly understood. Moreover, microglial activation has been widely described in AD transgenic mice, but depending on the models, timing of pathology development and brain region, activated microglia can adopt a protective role or may acquire a cytotoxic phenotype, mediating neuronal damage. In amyloidogenic AD mouse models, a subset of activated microglial cells, named “disease-associated microglia” (DAM), cluster around amyloid plaques establishing a protective barrier [15,16]. This phenotype, characterized by the upregulation of genes involved in lysosomal, phagocytic, and lipid metabolic pathways, is ApoE- Trem2 dependent [15,16,17] and requires an oxidative metabolism [18]. Similar microglial transcriptomic profiles have also been described in several tauopathy models such as P301S and P301L mice [19,20]. However, microglial response is diverse in transgenic Tau models as, for instance, ThyTau22 mice manifest mild microglial activation, whereas P301S mice exhibit a strong microglial response [20]. Although the contribution of microglial cells to the progression and spread of pathogenic Tau species is still a matter of debate, it has recently been described that TREM2 loss of functions increases neuritic pathology and Tau spreading in amyloidogenic models [21].
Although microglial activation has been reported in several brain regions of AD patients [22,23,24], it is important to point out that, in the hippocampus, the microglial response is not as strong as reported for amyloidogenic mice and several Tau models [20,25]. Apart from the individual and regional heterogeneity, this apparent discordance between transgenic models and AD patients may be associated to the aging process itself, the main risk factor for late-onset AD, and/or to the chronic pathology of AD. Mouse models bearing familial AD mutations are frequently examined at relatively young ages compared to the elderly AD patients. Nevertheless, our results and others show that microglial activation increases with age in animals models with amyloid or Tau pathologies [20,26]. Then, other comorbidities present in AD patients as vascular deficiencies, hypertension, inflammatory diseases, obesity or diabetes mellitus could be involved in this distinct microglial response. What is more, transcriptomic studies from purified microglia showed few overlaps in differentially expressed genes during aging between humans and mice, hinting that microglia may age differently in both species [27].
In the last years, microglial depletion mouse models have provided new insights into the role of these cells in physiological and pathological conditions. Here, we review different mouse models of microglial depletion, both in health and in AD, evaluating how reliable they could be as tools to study a context of microglial dysfunction or a context of microglial renewal. We recapitulate previous published data on the main microglial depletion strategies and, importantly, we also include unpublished data from our recently developed mouse model of conditional microglia depletion ( Cx3cr1 CreER /Csf1r flx/flx ). We also comment on the origin and pattern of microglial repopulation process. Additionally, we review previous works in regards to the effects of microglial depletion and repopulation in the progression of Aβ and Tau pathologies. Outcomes are diverse and sometimes contradictory, but they open new research lines regarding the mechanisms underlying microglial proliferation and migration capabilities. Selective ablation of harmful microglia within suitable time windows and their replacement by protective microglia may be a promising therapeutic strategy for AD and other neurodegenerative diseases.
2. Pharmacological and Genetic Microglial Depletion Models
Microglial viability and proliferation depend on signaling through the colony-stimulating factor1 receptor (CSF1R) [5,31,32,33] that belongs to the type III tyrosine kinase family, and is activated by two different cytokine ligands, colony stimulating factor-1 (CSF1) and interleukin-34 (IL-34) [34,35,36]. However, Csf1r is expressed on all myeloid cells [37,38], so the signaling interference through this receptor will not only affect microglial cells, but also peripheral macrophages, probably mediating an immunosuppressive effect. As it is widely known, Csf1r knock-out (KO) mice do not reach adult stage [3,39] so the suppression of this receptor should be carried out in adulthood, either through the administration of pharmacological inhibitors or through controlled genetic systems. As previously revised, different approaches give rise to variable depletion percentages, also dependent on the dose and length of treatments [40,41].
The first pharmacological approach trying to deplete microglial populations used a bisphosphonate drug, clodronate, packed in liposomes (Clo-Lip), which is rapidly taken up by phagocytic cells inducing their apoptotic death. Clo-Lip does not cross the blood–brain barrier (BBB), and consequently, needs to be administered by either intraparenchymal or intraventricular injection. Intraparenchymal Clo-Lip injection depletes between 30 and 60% of microglia 24 to 72 h after injection, but also produces astrocytic activation, releases proinflammatory cytokine and alters blood vessel integrity (reviewed in [42]). A better pharmacological strategy for microglial elimination was achieved by highly potent CSF1R tyrosine kinase inhibitors as PLX3397 and PLX647 that, after crossing the BBB, lead to microglia depletion without consequent inflammation, cytokine storm, or BBB damage, and no negative effects on mice behavior and cognition [43]. Depending on the dose and inhibitor used, different degrees of microglia depletion were reached and maintained throughout the treatment. Additional specific CSF1R inhibitors as JNJ-40346527, GW2580 and BLZ945 are available, and different studies have shown their dose-dependent effects on microglial number and phenotype [43,44]. Recently, a new and highly specific inhibitor for CSF1R, PLX5622, has been developed, improving BBB penetrance compared to PLX3397 [45]. However, and unexpectedly, the effect of these small CSF1R inhibitors is not restricted to microglia, but also affects the whole macrophage population and hematopoiesis [46]. Moreover, all these inhibitors are not specific for CSF1R, as they also inhibit three other kinases as FLT3, PDGFR, and KIT [47] and leads to broad myelosuppression, affecting macrophages, osteoclasts, and mast cells, among other cells. Additionally, it should be considered that PLX treatments may have a detrimental effect on neurons as CSF1R signaling has been demonstrated to enhance neuronal survival [48]. Actually, Shi et al. showed that PLX3397 inhibit neurite outgrowth and mildly reduce neuron number in vitro [49]. Further experimental approaches are still necessary in order to specifically deplete microglia using these small CSF1R inhibitors without affecting other cell types or tissues.
A more selective microglial depletion, with little effects on peripheral tissues, can be achieved by genetic manipulations based on the combination of cell type specific promoters coupled to suicide genes [50]. The initial approach was based on the expression of the suicide herpes simplex virus thymidine kinase ( HSV-1 TK ) transgene under the Cd11b promoter [51]. The administration of ganciclovir to Cd11b -TK mutant mice induces apoptosis of microglia, but also of CD11b+ bone marrow cells. To avoid myelotoxicity and the consequent mouse death, it is mandatory to combine this model with a bone-marrow chimera system, or alternatively administrate ganciclovir intraventricularly. Other genetic approaches to deplete myeloid population used diphtheria toxin (DT)-based models, in which myeloid promoter-driven Cre recombinase mouse lines ( Cx3cr1 Cre ) were crossed with transgenic mice harboring genes for diphtheria toxin receptor (DTR) downstream of loxP-flanked STOP sequences. In this model, the administration of DT originated the acute cell death of all myeloid cells expressing DTR [52], although reached only short-lived depletion, less than 5 days.
On the other hand, the inducible Cx3cr1 CreER line allows the targeting of microglia in a cell-type-specific and tamoxifen inducible fashion [53]. Two main Cx3cr1 CreERT2 inducible lines were created separately in which a tamoxifen-inducible Cre-recombinase is expressed under the control of the Cx3cr1 promoter: Cx3cr1 CreER/+:R26iDT-A/+ and Cx3cr1 CreER/+:R26iDTR/+ . When activated by tamoxifen, nuclear translocation of the CreER fusion protein is transient and recombination occurs only for a limited period, so only long-lived cells as microglia, but not peripheral macrophages with a shorter lifespan, will be depleted. Later, the generation of conditional knockout mice harboring a loxP-flanked exon within the Csf1r gene ( Csf1r flx/flx ) has allowed spatial and temporal control of microglia upon combination with the appropriate Cre lines [54]. Additionally, the targeting of a more specific microglia-signature gene, as Tmem119 , has allowed the generation of Tmem119 CreERT2 lines [55]. These new genetic models considerably represent an improvement in the manipulation of microglia providing a valuable tool for the functional study of these cells (reviewed in [40,41]).
3. Microglial Depletion as a Model of Microglial Replacement
Due to high mouse microglial proliferation capacity, these depletion models do not mimic a situation of microglial degeneration, as previously desired, but a context of microglial renewal. The characterization of emerging microglia’s capabilities will allow us to validate the efficacy of microglial depletion-repopulation strategies as potential therapeutic tools. This approach will be beneficial when microglia are hyperactive as well as in a context of microglial degeneration [28] or senescent [84], because in both cases microglia may contribute to neuron toxicity. As microglial chronic activation is sustained by an oxidative metabolism [18], this may compromise oxygen availability for other cellular populations. Additionally, an excessive microgliosis may induce microglial mitochondrial damage and be a major source of reactive oxidative species [85], leading to oxidative damage in neurons, to astrocyte reactivity [86] and to an exacerbation of the inflammatory cascade.
Repopulating microglia appear to fulfil functions of resident microglia and is capable of monitoring the environment and responding to acute stimuli [58,76,87]. Adult newborn microglia have been described to gradually regain steady-state maturity, transcriptionally clustering close to control microglia 2 weeks after depletion [70]. Zhan et al. (2019) also showed that the restoration of microglial homeostatic density requires NF-κB signaling as well as apoptotic egress of excessive cells [70]. In accordance, Huang et al. (2018) found no transcriptomic differences in repopulated microglia (2-month after depletion treatment) compared to resident microglia, neither in resting conditions nor after LPS challenge [68]. More recently, Gratuze et al. (2021) also described a homeostatic gene signature and equal ability to cluster around amyloid deposits in repopulated microglia in an AD mouse model [88].
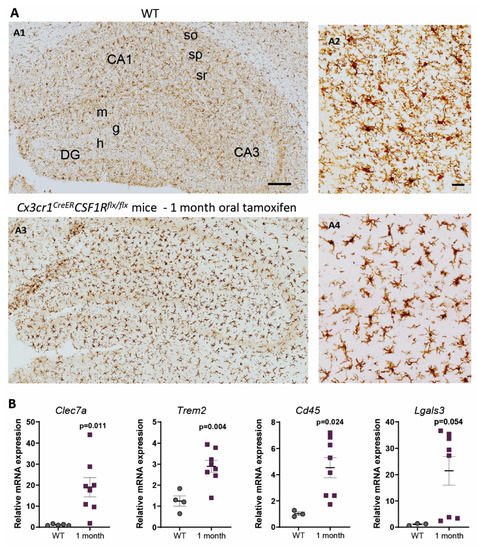
In contrast, under 1-month continuous tamoxifen treatment in our Cx3cr1 CreER /Csf1r flx/flx model, we observed that emerging microglia displayed an active morphology, with a thickening of cell body and shortening and thickening of microglial processes ( Figure 3 A). Furthermore, the increased expression of Clec7a , Cd45 , Trem2 and Lgals3 corroborated microglial activation after 1-month treatment ( Figure 3 B). We should take into account that, unlike previously mentioned publications, we maintained the depletion inductor, so a continuous microglial depletion and repopulation was taking place. An active phenotype is typical of phagocytic microglia, which could be eliminating cellular debris of dead microglia [32]. In this sense, an upregulation of scavenger-associated proteins, such as Cd36 [68], and of the phagocytic marker Cd68 [49] have been reported in repopulating microglia. However, given the small remaining microglial population at some points (for instance, at the end of our acute treatment ( Figure 1 A,B)), the CNS must count with an additional mechanism to eliminate all cellular debris, among which phagocytic activity of astrocytes has been proposed [69,89]. Konishi et al. (2020) demonstrated that, after specific microglial depletion (using Siglech dtr mice), astrocytes, rather than CNS-associated macrophages or circulating monocytes, clear microglial debris [69].
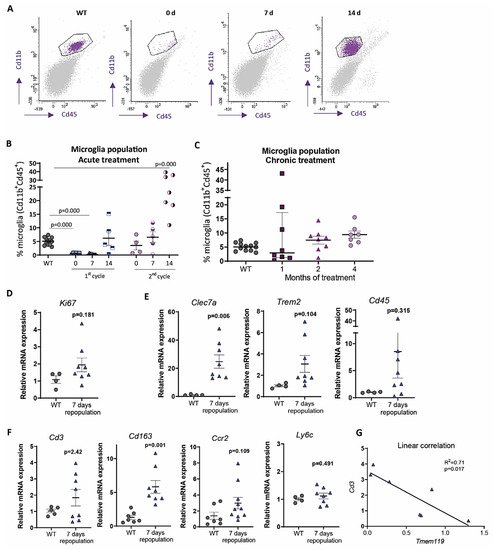
Finally, when drawing conclusion from chronic depletion models, we must be aware that changes observed are difficult to be unequivocally addressed to one factor. They may be due to: (i) a reduction in total microglial levels, (ii) a reduction of previous, sometimes burnt out, microglia, or (iii) the presence of emerging microglia whose characteristics are still not thoroughly described. Same challenges will occur with other state-of-the-art microglial manipulation techniques, such as chimeric mice. These models have been proposed as emerging tools to substitute exhausted microglia and/or to characterize human microglial response in AD pathology. Under this approach, mouse microglia is depleted to be replaced with human iPS-derived microglia in immunosuppressed mice [90]. After this manipulation, alterations in the progression of AD pathologies could be addressed to: (i) a reduction in the number of mouse microglia, (ii) the effect of human microglia, (iii) the immunosuppression of mice, (iv) the interaction between mouse and human microglia. Additionally, taking into account the stablished interaction between microglia and astrocytes [91], in depletion as well as in chimeric mice, modifications in the pathologies may also be caused by changes occurred in other glial cells. In essence, although they are promising tools, we should be cautious when drawing conclusions from these models.
4. Do Microglia Refresh or Poison AD Progression?
In order to further characterize microglial role in AD, microglial depletion models are being combined with AD mouse models bearing either Aβ and/or Tau pathologies ( Table 1 ). Currently, controversial data are found and new studies are needed to clarify if microglia is beneficial or detrimental to AD pathology.
| Pathology | Depletion Model | Outcomes | References |
|---|---|---|---|
| Aβ | PLX5622 in 5xFAD mice, from 4- to 5-month-old. | 50% microglia depletion. Reduction of microgliosis and plaque burden, enhancement of neuritic dystrophies. | [62] |
| Aβ | PLX3397 in 5xFAD mice, from 9- to 10-month-old. | Around 50% microglia depletion. Decrease in Aβ deposition and rescue of dopaminergic signaling. | [65] |
| Aβ | PLX5622 in APP/PS1 mice, from 12- to 13-month-old. | Diminution of leukotriene biosynthesis and the neuronal 5-lipoxygenase. | [92] |
| Aβ | PLX5622 in 5xFAD mice from 1.5 to 4- or 7-month-olds. | 97% microglia depletion. Reduction of plaque deposition, but increase of cerebral amyloid angiopathy formation. | [45] |
| Aβ | PLX3397 in 5xFAD mice from 2- to 5-month-old. | 70–80% microglia depletion. Reduction of intraneuronal amyloid, neuritic plaque deposition and improvement in cognitive functions (fear conditioning tests). | [93] |
| Aβ | Diphtheria toxin in 15 months-old Cx3cr1CreER/+:R26DTR/+/APPxPS1 mice, for 1–2 weeks. | 90% depletion. No changes in the number of Aβ plaques, but an increase in size. | [94] |
| Aβ | GWS2580 in APP/PS1 mice from 6- to 9-month-old. | 30% reduction of microglia. No changes in the number of Aβ plaques. Improved performance in memory and behavioral tasks. | [43] |
| Aβ | PLX3397 in 5xFAD from 10- to 11-month-old. | 90% microglia depletion. No alterations in β-amyloid levels or plaque load, but rescue of dendritic spine loss and improvements in contextual memory. | [56] |
| Aβ and Tau | PLX5562 in 3xTg mice for 3 months. | 30% microglia depletion. No changes in total or phosphorylated Tau. Improvements in cognition. | [64] |
| Aβ and Tau | PLX3397 from 5.5- to 7-month-old in 5xFAD/PS19 Tau -injected mice. | 81% microglia depletion. Higher reduction in non-plaque-associated microglia. No changes in Aβ pathology, reduction in Tau pathology and neurodegeneration. | [71] |
| Aβ and Tau | PLX3397 from 6- to 9-month-old in 5xFAD mice injected with AD-Tau. | Improved cognitive and neuronal deficits. Enhancement of Tau seeding and spreading around plaques. | [88] |
| Tau | Cx3cr1CreER/R26DTA/hTAU mice, treated with tamoxifen for 2–3 months at different ages. | 60% microglia depletion. No changes in soluble oligomeric, phosphorylated or total aggregated Tau levels. | [66] |
| Tau | PLX3397 in P301S APOE E4 mice from 6- to 9-month-old. | Total microglia depletion. Protection from brain volume loss and neurodegeneration. Reduction of Tau pathology progression. | [49] |
| Tau | PLX3397 in rTg4510 mice, from 12- to 15-month-old. | 30% microglia depletion. No changes in Tau burden, cortical atrophy, blood vessels or glial activation. | [63] |
| Tau | (a) Clodronate liposomes and PLX3397 in AAV-GFP/Tau injected C57BL/6 mice. (b) PLX3397 in PS19 mice. In both cases, from 3.5- to 4.5-month-old. | 70–80% (a) and 90% (b) microglia depletion. Reduction of phospho-Tau. | [95] |
Relevant results in AD mouse models of microglia depletion. Mouse models used and main outcomes are shown.
Studies of microglial depletion in other CNS pathologies showed similarly controversial results. Rice et al. (2017) and Acharya et al. (2016) described significant improvements subsequent to microglial depletion in neuronal damage models [59,117]. In the same line, Li et al. (2017) showed neuroprotection following microglial depletion in a model of intracerebral hemorrhage [118]. However, other publications reported an increase in neuroinflammation and brain damage after microglial depletion in ischemia models [119,120].
Therefore, novel approaches to increase microglial renewal may be a promising tool to assess microglial role in the ATN continuum and to design potential therapies for AD.
This entry is adapted from the peer-reviewed paper 10.3390/ijms22189734
