Maltol, 3-hydroxy-2-methyl-4-pyrone (Figure 1A), a naturally occurring compound, can be isolated from various types of plants, such as bark and leaves of Larix deciduas, Evodiopanax innovans, Cercidiphyllum japonicum, Citharexylum spinosum, Passiflora incarnata, Panax ginseng, and different kinds of pine plants [1,2,3,4].
- maltol
- cationic surfactants
- preservative
- broad-spectrum
- personal care
- synergy
- FICI
- membrane damage.
1. Introduction
Maltol, mostly used as a flavoring molecule, also has various potential applications as a biomedical compound. Despite its extensive use in the food industry, maltol’s antimicrobial activity was evaluated only briefly, and was suggested to be insufficient on its own. Recently, we have shown that maltol can be used in conjunction with cationic surfactant species to receive higher activity against contaminant microorganisms. In this paper, we have broadened the antimicrobial efficacy studies and evidenced maltol’s mode of action against Gram-negative, Gram-positive bacteria, and fungi. In addition, to increase its efficacy, blends of maltol and two selected cationic surfactants, dodecyl-dimethyl-ammonium chloride (DDAC) and polyquaternium 80 (P-80), were appraised for their activity. Broad efficacy studies revealed synergistic interactions between maltol and both cationic surfactants against most of the tested microorganisms. Electron microscopy images were used to evaluate the microorganisms’ morphology following treatment, pinpointing the specific cell wall damage caused by each of the compounds. Our findings indicate that maltol’s effect on the microbial cell wall can be complemented by catalytic amounts of selected cationic surfactants to enhance and extend its activity. Such a solution can be used as a broad-spectrum preservative for personal care products in cosmetic applications.
Few studies demonstrated that maltol can also be produced by some actinobacteria [5,6] and a mold species [7]. Maltol is a chelating agent, which binds hard metal centers, such as Fe3+ (Ferric maltol; Figure 1B), Ga3+, Al3+, and VO2+ [8]. Due to its solubility in aqueous solution, maltol was shown to increase the absorption of several essential metals in animal and human subjects, in comparison to hydrophobic chelating molecules [9,10,11,12]. Maltol is widely used in the food industry as a flavoring agent, food additive, and a food preservative. Maltol, known for its characteristic sweet smell, is used to create a sweet aroma in fragrances, freshly baked bread, and cakes. In addition to its extensive use in the food industry, maltol was found to have broad applications, such as a biomedical compound [1,13,14,15,16] and even used as a pest control agent [17]. Maltol is a promising candidate molecule for medical purposes and an important compound in the food industry; moreover, toxicity studies have determined that maltol is non-toxic and generally recognized as safe (GRAS) [7,18].
The antimicrobial activity of maltol, on the other hand, was only briefly investigated in several previous studies [4,19,20], while its efficacy was suggested mostly as insufficient on its own [19,21]. Recent study discovered that maltol efficacy can be significantly increased by the addition of only small amounts of selected cationic surfactants [20]. In this study, the efficacy studies were widened to better understand the mode of action of maltol and its combination with two cationic surfactants, polyquaternium 80 (P-80), and didecyldimethylammonium chloride (DDAC). For this purpose, the antimicrobial mechanism of maltol and its cationic surfactant combinations were evaluated against the five pharmacopeia microorganisms, Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, Candida albicans, and Aspergillus brasiliensis, using multiple efficacy studies, morphological examination via electron microscopy imaging, and cell permeability assay.
2. Results
2.1. MIC and Checkerboard Assays
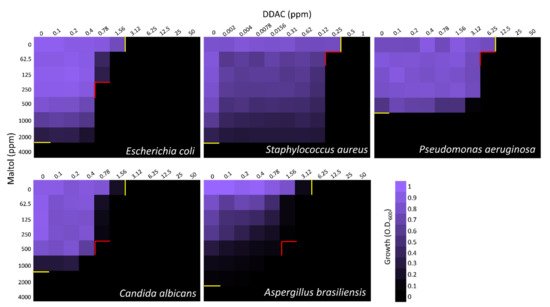
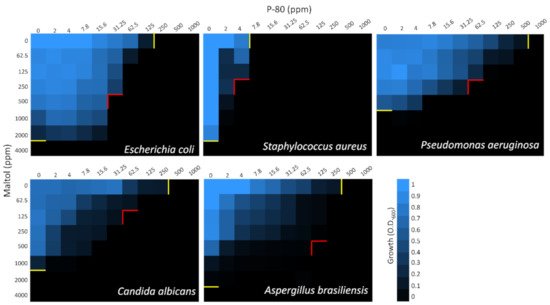
| Separate MIC100 (ppm) | Combined MIC100 (ppm) | Combined MIC100 (ppm) | |||||
|---|---|---|---|---|---|---|---|
| Maltol | DDAC | P80 | Maltol | DDAC | Maltol | P80 | |
| E. coli | 4000 | 3.12 | 250 | 250 | 0.78 | 500 | 31.25 |
| FICI = 0.31 | FICI = 0.25 | ||||||
| S. aureus | 4000 | 0.5 | 7.8 | 62.5 | 0.25 | 250 | 4 |
| FICI = 0.52 | FICI = 0.58 | ||||||
| P. aeruginosa | 1000 | 12.5 | 1000 | 62.5 | 6.25 | 250 | 62.5 |
| FICI = 0.56 | FICI = 0.31 | ||||||
| C. albicans | 2000 | 3.12 | 500 | 500 | 0.78 | 125 | 62.5 |
| FICI = 0.5 | FICI = 0.19 | ||||||
| A. brasiliensis | 4000 | 6.25 | 500 | 500 | 1.56 | 500 | 125 |
| FICI = 0.38 | FICI = 0.38 | ||||||
2.2. Potassium Leakage
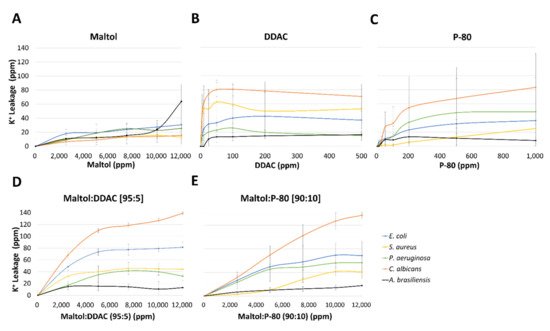
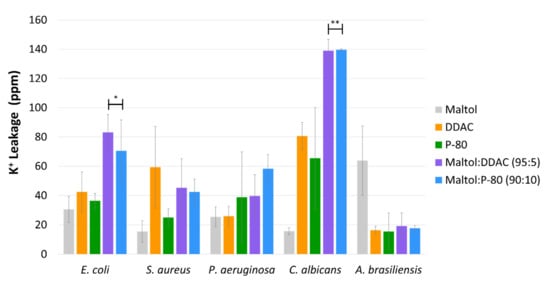
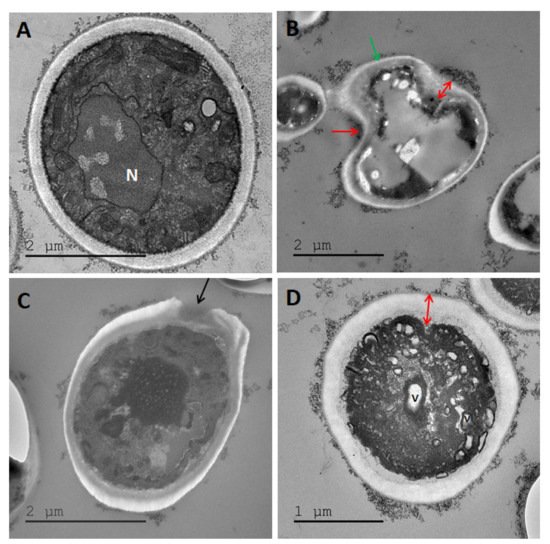
2.3. Transmission Electron Microscopy (TEM)
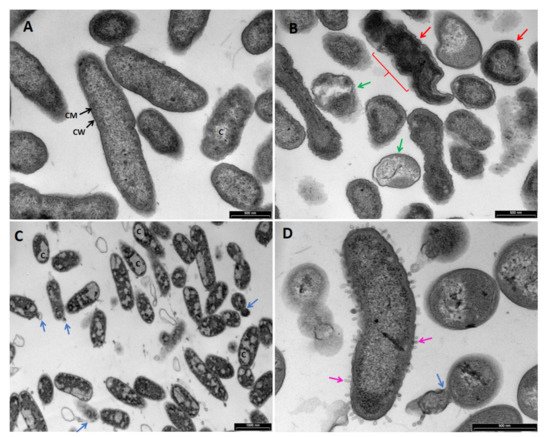
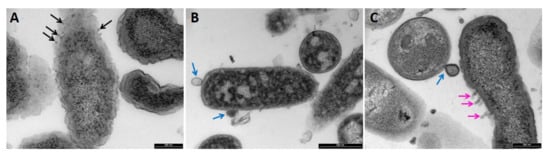
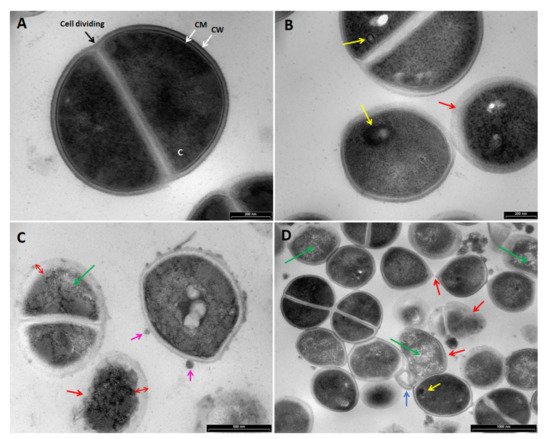
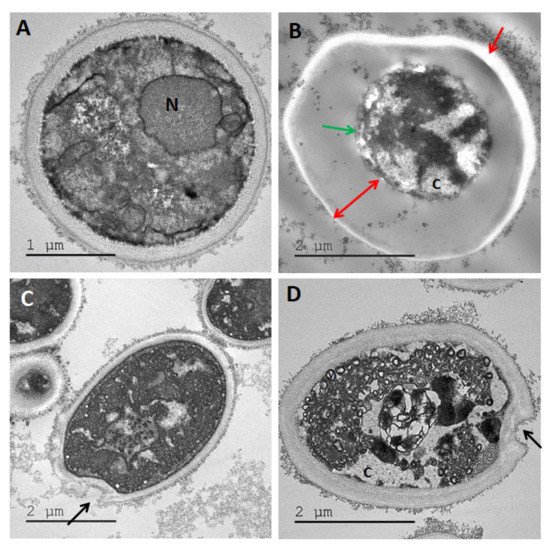

5. Conclusions
1. Han, Y.; Xu, Q.; Hu, J.N.; Han, X.Y.; Li, W.; Zhao, L.C. Maltol, a food flavoring agent, attenuates acute alcohol-induced oxidative
damage in mice. Nutrients 2015, 7, 682–696, doi:10.3390/nu7010682.
2. Tiefel, P.; Berger, R.G. Seasonal variation of the concentrations of maltol and maltol glucoside in leaves of Cercidiphyllum
japonicum. J. Sci. Food Agric. 1993, 63, 59–61, doi:10.1002/jsfa.2740630110.
3. Mar, A.; Pripdeevech, P. Chemical composition and antibacterial activity of essential oil and extracts of Citharexylum spinosum
flowers from Thailand. Nat. Prod. Commun. 2014, 9, 707–710, doi:10.1177/1934578x1400900532.
4. Hayata, Y. Studies on Passiflora incarnata dry extract. I. Isolation of maltol and pharmacological action of maltol and ethyl maltol.
Chem. Pharm. Bull. 1974, 2091.
5. Kornsakulkarn, J.; Saepua, S.; Supothina, S.; Chanthaket, R.; Thongpanchang, C. Sporaridin and sporazepin from actinomycete
Streptosporangium sp. BCC 24625. Phytochem. Lett. 2014, 10, 149–151, doi:10.1016/j.phytol.2014.08.021.
6. Zarrini, J. Introducing acidophilic and acid tolerant actinobacteria as new sources of antimicrobial agents against Helicobacter
pylori. Arch. Razi Inst. 2021, 76, 1–23, doi:10.22092/ARI.2019.128039.1401.
7. Rajamanikyam, M.; Gade, S.; Vadlapudi, V.; Parvathaneni, S.P.; Koude, D.; Kumar Dommati, A.; Kumar Tiwari, A.; Misra, S.;
Sripadi, P.; Amanchy, R.; et al. Biophysical and biochemical characterization of active secondary metabolites from Aspergillus
allahabadii. Process Biochem. 2017, 56, 45–56, doi:10.1016/j.procbio.2017.02.010.
8. Kontoghiorghe, C.N.; Kolnagou, A.; Kontoghiorghes, G.J. Phytochelators intended for clinical use in iron overload, other
diseases of iron imbalance and free radical pathology. Molecules 2015, 20, 20841–20872, doi:10.3390/molecules201119725.
9. Kelsey, S.M.; Hider, R.C.; Bloor, J.R.; Blake, D.R.; Gutteridge, C.N.; Newland, A.C. Absorption of Low and Therapeutic Doses
of Ferric Maltol, a Novel Ferric Iron Compound, in Iron Deficient Subjects Using a Single Dose Iron Absorption Test. J. Clin.
Pharm. Ther. 1991, 16, 117–122, doi:10.1111/j.1365-2710.1991.tb00292.x.
10. Gasche, C.; Ahmad, T.; Tulassay, Z.; Baumgart, D.C.; Bokemeyer, B.; Büning, C.; Howaldt, S.; Stallmach, A. Ferric maltol is
effective in correcting iron deficiency anemia in patients with inflammatory bowel disease: Results from a phase-3 clinical trial
program. Inflamm. Bowel Dis. 2015, 21, 579–588, doi:10.1097/MIB.0000000000000314.
11. Kaneko, N.; Yasui, H.; Takada, J.; Suzuki, K.; Sakurai, H. Orally administrated aluminum-maltolate complex enhances oxidative
stress in the organs of mice. J. Inorg. Biochem. 2004, 98, 2022–2031, doi:10.1016/j.jinorgbio.2004.09.008.
12. Bernstein, L.R.; Tanner, T.; Godfrey, C.; Noll, B. Chemistry and pharmacokinetics of gallium maltolate, a compound with high
oral gallium bioavailability. Met. Based. Drugs 2000, 7, 33–47, doi:10.1155/MBD.2000.33.
Liver Fibrosis through TGF-β1-mediated Activation of PI3K/Akt Signaling Pathway. J. Agric. Food Chem. 2019, 67, 1392–
1401, doi:10.1021/acs.jafc.8b05943.
14. Li, W.; Su, X.M.; Han, Y.; Xu, Q.; Zhang, J.; Wang, Z.; Wang, Y.P. Maltol, a Maillard reaction product, exerts anti-tumor efficacy
in H22 tumor-bearing mice via improving immune function and inducing apoptosis. RSC Adv. 2015, 5, 101850–101859,
doi:10.1039/c5ra17960b.
15. Yasumoto, E.; Nakano, K.; Nakayachi, T.; Morshed, S.R.M.; Hashimoto, K.; Kikuchi, H.; Nishikawa, H.; Kawase, M.; Sakagami,
H. Cytotoxic Activity of Deferiprone, Maltol and Related Hydroxyketones against Human Tumor Cell Lines. Anticancer Res.
2004, 24, 755–762.
16. Jo, B.G.; Park, N.J.; Kim, S.N.; Jegal, J.; Choi, S.; Lee, S.W.; Yi, L.W.; Lee, S.R.; Kim, K.H.; Yang, M.H. Isolation of maltol
derivatives from Stellera chamaejasme and the anti-atopic properties of maltol on skin lesions in DNCB-stimulated mice. RSC
Adv. 2019, 9, 2125–2132, doi:10.1039/C8RA09743G.
17. Williams, M.; Eveleigh, E.; Forbes, G.; Lamb, R.; Roscoe, L.; Silk, P. Evidence of a direct chemical plant defense role for maltol
against spruce budworm. Entomol. Exp. Appl. 2019, 167, 755–762, doi:10.1111/eea.12822.
18. Regulation (EC) No 1334/2008 of the European Parliament and of the Council of 16 December 2008 on flavourings and certain
food ingredients with flavouring properties for use in and on foods and amending Council Regulation (EEC) No 1601/91,
Regulations (EC) No 2232/96 and (EC) No 110/2008 and Directive 2000/13/EC (Text with EEA relevance). Off. j. Eur. Union. 2018,
2016, 48–119.
19. Schved, F.; Pierson, M.D.; Juven, B.J. Sensitization of Escherichia coli to nisin by maltol and ethyl maltol. Lett. Appl. Microbiol.
1996, 22, 189–191, doi:10.1111/j.1472-765X.1996.tb01139.x.
20. Salama, P.; Gliksberg, A. The use of catalytic amounts of selected cationic surfactants in the design of new synergistic
preservative solutions. Cosmetics 2021, 8, doi:10.3390/cosmetics8020054.
21. Jay, J.M.; Rivers, G.M. Antimicrobial Activity of Some Food Flavoring Compounds. J. Food Saf. 1984, 6, 129–139,
doi:10.1111/j.1745-4565.1984.tb00609.x.
22. Odds, F.C. Synergy, antagonism, and what the chequerboard puts between them. J. Antimicrob. Chemother. 2003, 52, 1,
doi:10.1093/jac/dkg301.
23. Ziklo, N.; Tzafrir, I.; Shulkin, R.; Salama, P. Salicylate UV-filters in sunscreen formulations compromise the preservative system
efficacy against Pseudomonas aeruginosa and Burkholderia cepacia. Cosmetics 2020, 7, doi:10.3390/COSMETICS7030063.
24. Falk, N.A. Surfactants as Antimicrobials: A Brief Overview of Microbial Interfacial Chemistry and Surfactant Antimicrobial
Activity. J. Surfactants Deterg. 2019, 22, 1119–1127, doi:10.1002/jsde.12293.
25. Dolezal, R.; Soukup, O.; Malinak, D.; Savedra, R.M.L.; Marek, J.; Dolezalova, M.; Pasdiorova, M.; Salajkova, S.; Korabecny, J.;
Honegr, J.; et al. Towards understanding the mechanism of action of antibacterial N-alkyl-3-hydroxypyridinium salts: Biological
activities, molecular modeling and QSAR studies. Eur. J. Med. Chem. 2016, 121, 699–711, doi:10.1016/j.ejmech.2016.05.058.
26. Gilbert, P.; Moore, L.E. Cationic antiseptics: Diversity of action under a common epithet. J. Appl. Microbiol. 2005, 99, 703–715,
doi:10.1111/j.1365-2672.2005.02664.x.
27. Yoshimatsu, T.; Hiyama, K.I. Mechanism of the action of didecyldimethylammonium chloride (DDAC) against Escherichia coli
and morphological changes of the cells. Biocontrol Sci. 2007, 12, 93–99, doi:10.4265/bio.12.93.
28. Saito, H.; Sakakibara, Y.; Sakata, A.; Kurashige, R.; Murakami, D.; Kageshima, H.; Saito, A.; Miyazaki, Y. Antibacterial activity
of lysozyme-chitosan oligosaccharide conjugates (LYZOX) against Pseudomonas aeruginosa, Acinetobacter baumannii and
Methicillin-resistant Staphylococcus aureus. PLoS One 2019, 14, 1–26, doi:10.1371/journal.pone.0217504.
29. Atay, H.Y. Antibacterial Activity of Chitosan-Based Systems; S., J., S., J., Eds.; Springer Nature Singapore Pte Ltd, The Gateway,
Singapore, 2019; ISBN 9789811502637.
30. Clifton, L.A.; Skoda, M.W.A.; Le Brun, A.P.; Ciesielski, F.; Kuzmenko, I.; Holt, S.A.; Lakey, J.H. Effect of divalent cation removal
on the structure of gram-negative bacterial outer membrane models. Langmuir 2015, 31, 404–412, doi:10.1021/la504407v.
This entry is adapted from the peer-reviewed paper 10.3390/cosmetics8030086
