Nanoparticles (NPs) have an outstanding position in pharmaceutical, biological, and medical disciplines. Polymeric NPs based on chitosan (CS) can act as excellent drug carriers because of some intrinsic beneficial properties including biocompatibility, biodegradability, non-toxicity, bioactivity, easy preparation, and targeting specificity. Drug transport and release from CS-based particulate systems depend on the extent of cross-linking, morphology, size, and density of the particulate system, as well as physicochemical properties of the drug. All these aspects have to be considered when developing new CS-based NPs as potential drug delivery systems. This review is summarizing and discussing recent advances in CS-based NPs being developed and examined for drug delivery including the following sections: (i) CS and its derivatives, basic characteristics of CS NPs, (ii) preparation procedures used for CS NPs, (iii) CS-based-nanocomposites with organic polymers and inorganic material, and (iv) implementations of CS NPs and nanocomposites in drug delivery.
- chitosan
- modified chitosan
- enhanced properties
- nanoparticles
- drug delivery
- new formulations
- targeting
- controlled release
- nanocomposites
- preparation schemes
1. Introduction
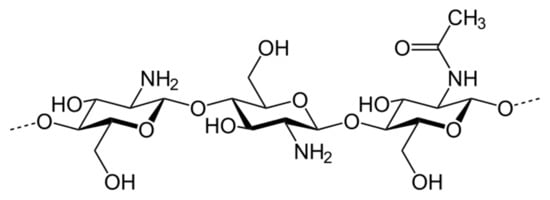
2. Enhancement of Chitosan Properties by Its Modification
| Chitosan Derivative Groups/Derivatives |
Formula |
|---|---|
| Hydrophobic derivatives | |
| Alkylated chitosan | 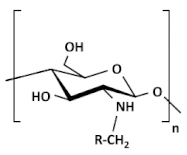 |
| Acylated chitosan | 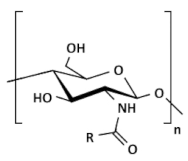 (A) N-acylated chitosan |
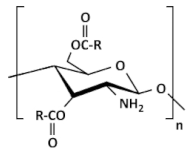 (B) O-acylated chitosan |
|
| N-phtaloylated chitosan | 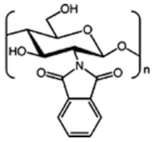 |
| Benzoylated chitosan | 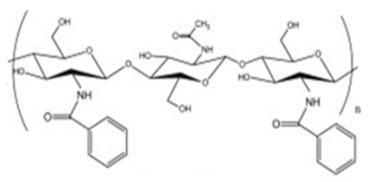 |
| Methacrylated chitosan | 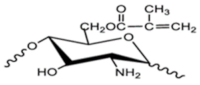 |
| Amphiphilic derivatives | |
| Cholic and deoxycholic acid-modified chitosan | 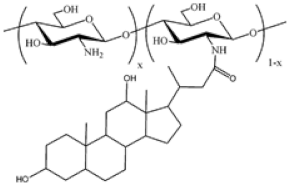 (A) Deoxycholic acid 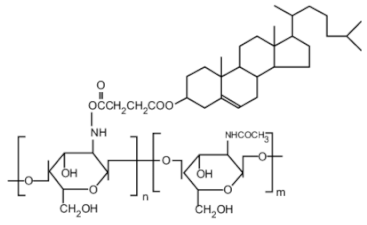 (B) Cholic acid |
| Ionic derivatives | |
| Quarternary ammonium chitosan derivatives | 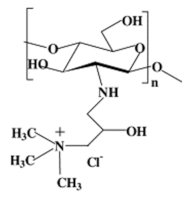 |
| Sulfated chitosan derivatives | 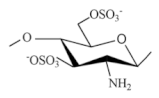 |
| Succinylated chitosan | 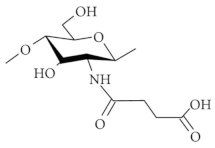 |
| Sulfonated chitosan | 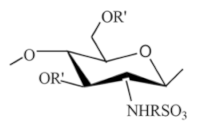 |
| Phosphorylated chitosan | 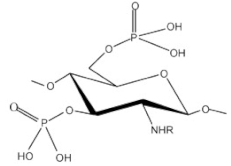 |
| Carboxyalkylated chitosan (carboxymethylchitosan) | 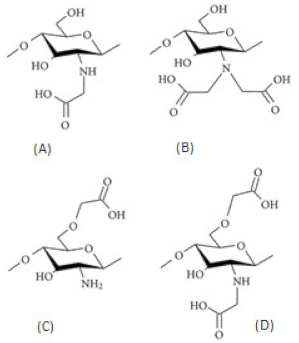 (A) N-CMC, (B) N,N-CMC, (C) O-CMC, and (D) N,O-CMC (showing the modification at the D-glucosamine unit) |
| Chitosan copolymers | |
| PEGylated chitosan | 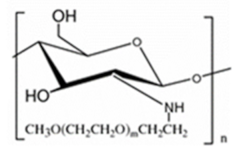 |
| PEG-methacrylated chitosan | 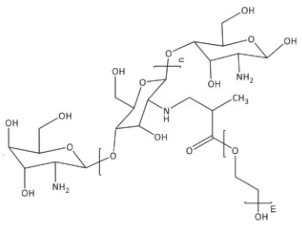 |
| Derivatives with specific substituents | |
| Sugar bound chitosan derivatives | 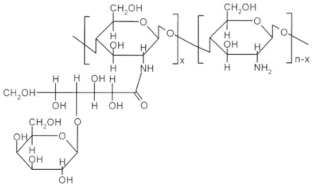 Galactosylated chitosan |
 Sialo dendrimer hybrid chitosan |
|
| Chitosan derivatives with cyclic structure |
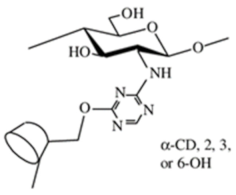 Crown ether-linked chitosan |
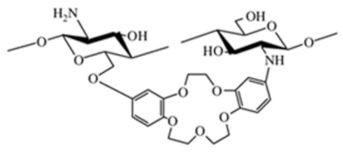 Cyclodextrin-linked chitosan |
|
| Chitosan derivatives with thiol groups | 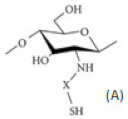 (A) Thiolated chitosan with –SH group |
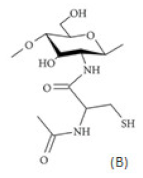 (B) Thiolated chitosan with cysteine: chitosan-N-acetyl-cysteine |
|
| Glycol chitosan |  |
| Thiosemicarbazone linked chitosan derivatives | 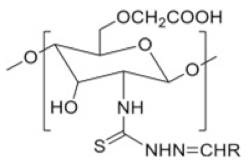 |
| Crosslinked chitosan derivatives | |
| Chitosan-glutaraldehyde crosslinked polymer | 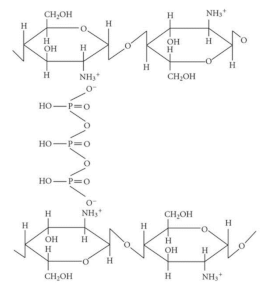 |
| Chitosan-TPP crosslinked polymer | 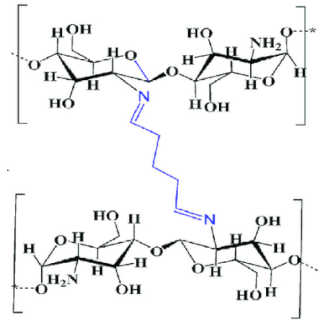 |
| Chitosan-EDTA crosslinked polymer | 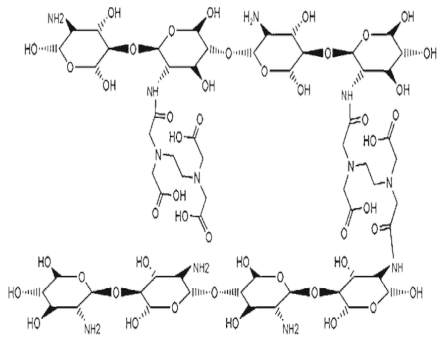 |
3. Enhancement in Drug Delivery by Chitosan Nanoparticles
3.1. Basic Aspects of (Chitosan) Nanoparticle Systems
- (i).
-
CS NSs are a matrix system, where the drug may be absorbed in the surface or encapsulated within the CS particle. As an example, Liu et al. [39] constructed the photothermal sensitive carboxymethyl CS nanospheres (CMC NSs) carrier by introducing controllable heat-sensitive groups into CMC molecules. The carrier owned high drug loading and improved the stability of coated-drug DOX. The NSs generated photothermal response through NIR irradiation to improve the drug release amount and to achieve the combined treatment effect of photodynamic therapy and chemotherapy. In vitro photothermal tests proved that the NSs had excellent light stability and photothermal conversion performance. The cytotoxicity test results showed that the NSs had no obvious toxicity, but the drug-loaded nanospheres could effectively inhibit the growth of HepG-2 cells via photo-response to release DOX and Indocyanine green for achieving photothermal-chemotherapy under NIR irradiation.
- (ii).
-
CS NCs are vesicular systems in which the drug is generally confined to a cavity consisting of an oily core covered by a CS shell. As an example, Castro et al. [65] evaluated the physicochemical and biological properties of docetaxel (DCX) loaded chitosan nanocapsules (DCX-CS NCs) functionalized with the chimeric monoclonal antibody ChiTn mAb (highly specific antigen for carcinomas) (DCX-CS/PEG-ChiTn mAb NCs) as a potential improvement treatment for cancer therapy. The NCs, formed as a polymeric shell around an oily core, allowed a 99.9% encapsulation efficiency of DCX with a monodispersity particle size in the range of 200 nm and a high positive surface charge that provided substantial stability to the nanosystems. Uptake studies and viability assay conducted in A549 human lung cancer cell line in vitro demonstrated that ChiTn mAb enhanced NPs internalization and cell viability reduction.
- (iii).
-
CS NFs can be used in various fields mainly due to the presence of -NH2 and -OH groups, along with their specific structure. Their nanofibrous structure offers enormous possibilities for chemical modifications that create new properties applicable, particularly in the biomedical field. CS NFs can be prepared by electrospinning of CS into ultrafine fibers of nano size. Owing to the large specific surface area, NFs can deliver drugs, peptides, and vaccine antigens. The release of the drug may be immediate, delayed, or modified depending on the type of interactions between the polymer and the drug. Usually, an immediate release is noticed when a composition of a water-soluble substance and a water-soluble polymer is used. The prolonged release can be achieved by integrating the drug into other nanocarriers, such as NPs, liposomes, dendrimers, then loaded into NFs or use hydrophobic polymers [18,66]. As an example, Amiri et al. [67] reported the development of a local antibiotic delivery system using chitosan/polyethylene oxide (CS/PEO) NFs for delivery of teicoplanin. Uniform and bead-less NFs were prepared via electrospinning of a CS/PEO solution containing teicoplanin. The NFs were able to release teicoplanin for up to 12 days. Antibacterial test in agar diffusion and time-kill study on Staphylococcus aureus also demonstrated that loading teicoplanin in CS/PEO NFs enhanced the antibacterial activity up to 1.5- to 2-fold. An in vivo study on a rat full-thickness wound model confirmed the safety and efficacy of applying the teicoplanin-loaded NFs and significant improvement in wound closure were observed especially with the NFs containing 4% teicoplanin.
3.2. Preparation Procedures for Chitosan Nanoparticles
3.2.1. Covalent Cross-Linking
3.2.2. Self-Assembly
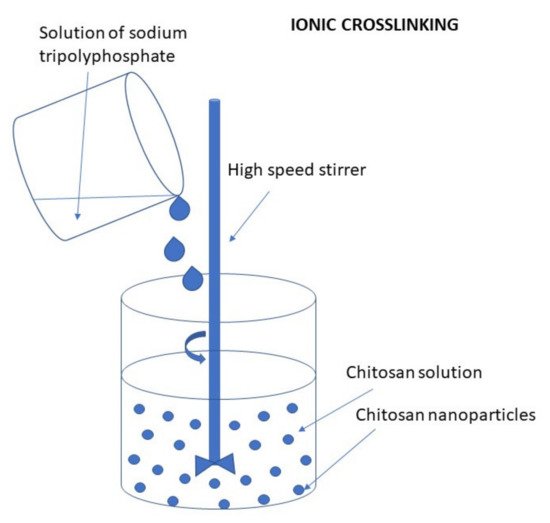
Ionic Cross-Linking (Ionic Gelation)
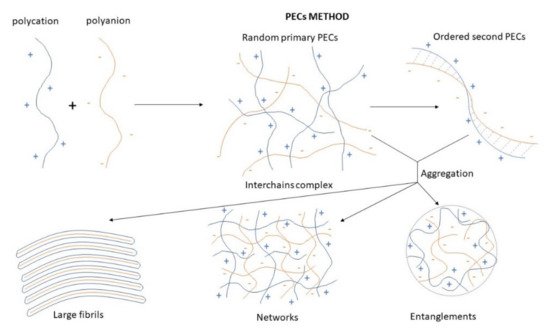
Polyelectrolyte Complex (PEC)
3.2.3. Emulsion Technique
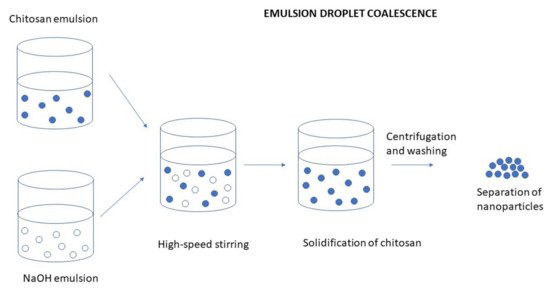
Emulsion Droplet Coalescence (Emulsion Crosslinking and Precipitation)
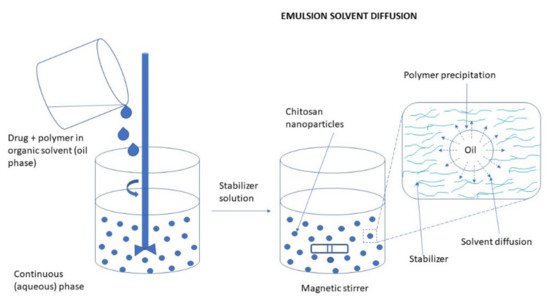
Emulsification Solvent Diffusion
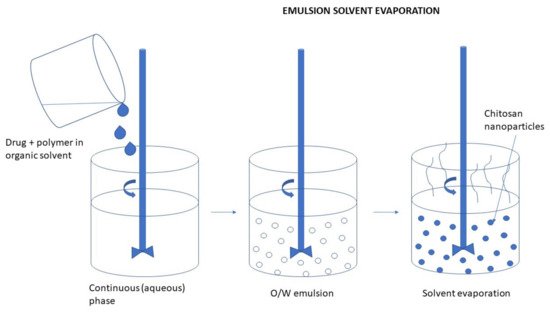
Emulsification Solvent Evaporation
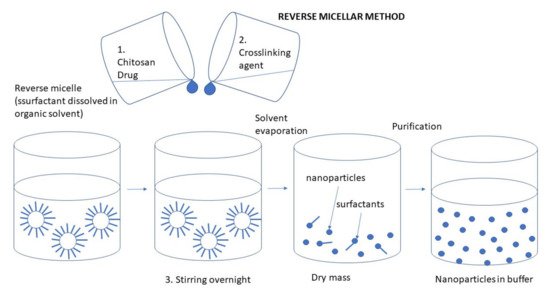
3.2.4. Reverse Micellar Method
3.2.5. Drying Methods
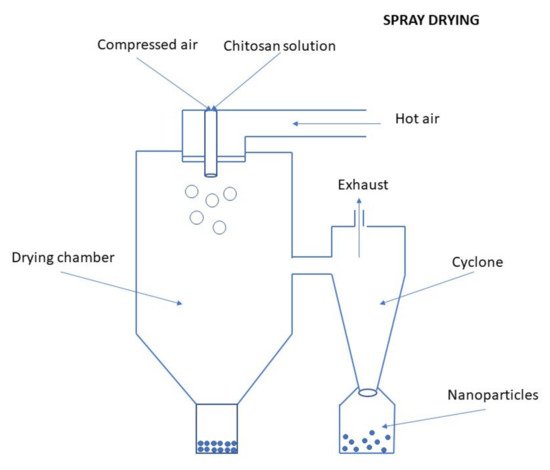
Spray Drying
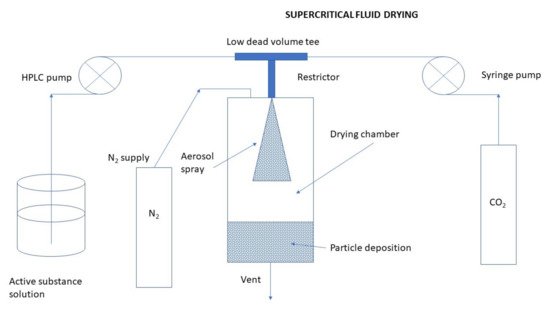
Supercritical Fluid Drying
Electrospraying Technique
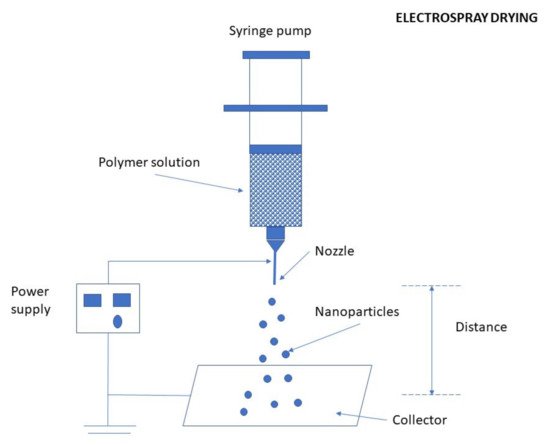
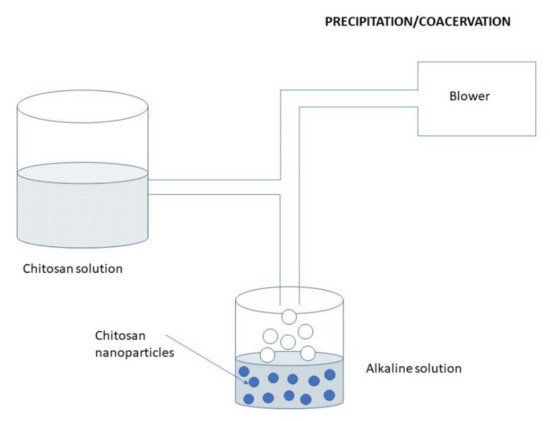
3.2.6. Precipitation/Coacervation
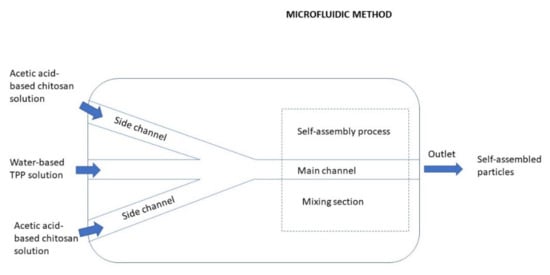
3.2.7. Microfluidic Method
3.3. Chitosan Based-Nanocomposites, Types, Their Properties, and Utilization
3.3.1. Chitosan-Polymer Nanocomposites
Synthetic Polymers
Natural Polymers
3.3.2. Chitosan-Inorganic Material Nanocomposite
Montmorillonite Clay (Mnt)
Mesoporous Silica Nanoparticles (MS NPs)
Magnetic Nanoparticles (Mag NPs)
Graphene Oxide (GO) Modified Nanocomposite
Gold Nanoparticles (Au NPs)
Zinc Oxide Nanoparticles (ZnO NPs)
Carbon Quantum Dots (CQDs)
Carbon Nanotubes (CNTs) Composite
Other Inorganic Materials Combined with CS
3.3.3. Stimuli Responsive Chitosan, Chitosan Nanoparticles, and Nanocomposites
3.4. Chitosan Nanoparticles in Drug Delivery
|
Type of CS NP |
Method of CS NP |
Formulated Drug |
In Vitro and In Vivo Tests for Biological Activity and Drug Release |
Citation |
|
Oral drug delivery of antidiabetic drugs |
||||
|
TC NPs |
Schiff-base linking with PETMP [pentaerythritol tetrakis (3-mercaptopropionate)] |
Insulin |
In vitro sustained drug release, in vitro cell viability, in vivo biodistribution, and pharmacokinetics |
[160] |
|
Snail mucin/CS NPs |
Self-assembly |
Insulin |
In vitro drug release, in vivo hypoglycemic activity in diabetic rats, and toxicity |
[161] |
|
FD/TMC NPs |
PEC method |
Insulin |
In vitro pH-dependent drug release, cytotoxicity, α-glucosidase inhibition assay |
[162] |
|
CS/Dz13Scr NPs |
Complex coacervation |
Insulin |
In vitro drug release, insulin kinetics, cytotoxicity, mucus permeation, endocytic absorption study |
[163] |
|
CS NPs |
Ionic gelation |
Polydatin |
In vitro drug release, cytotoxicity, in vivo antidiabetic activity in type 2 diabetic rats |
[164] |
|
Oral delivery of anticancer drugs |
||||
|
FD/CS NPs |
PEC method |
Methotrexate (MTX) for lung cancer therapy |
In vitro mucoadhesive study, in vitro antiproliferative assay and cellular uptake, apoptosis assay |
[165] |
|
M CS/P NPs |
Ionic gelation |
Curcumin (CUR) |
Cytotoxicity, cellular uptake |
[115] |
|
chitosan-copaiba oil-poly (isobutyl cyanoacrylate) core-shell nanocapsules |
Interfacial polymerization |
For colon cancer therapy |
In vitro mucoadhesion effect |
[166] |
|
Cys/PLA/CS NPs |
Self-assembly |
Paclitaxel (PTX) |
In vitro drug release, cytotoxicity, and cellular uptake, in vivo pharmacokinetic study, biodistribution study, antitumor efficacy |
[167] |
|
TPGS/HPMC/CS NPs |
Solvent evaporation method |
Paclitaxel (PXT) |
In vitro dissolution and swelling, Cytotoxicity, cellular uptake, transport study |
[168] |
|
Oral delivery of antihypertensive drugs |
||||
|
CS NPs |
Ionic gelation (TPP) |
Carvedilol |
In vitro drug release, in vivo pharmacokinetics on rats |
[169] |
|
Oral delivery of antioxidants |
||||
|
CS/Zein NPs |
Liquid-liquid dispersion |
Resveratrol (RVT) |
In vitro drug release, antioxidant activity, in vitro mucoadhesion study |
[170] |
|
Oral delivery of anti-inflammatory drugs |
||||
|
CS/WP-NPs |
Self-assembly |
polysaccharides from Ophiopogon japonicus (OJPs) IBD treatment |
In vitro drug release, Biocompatibility, cytotoxicity, antioxidant activity, gene expression, ex vivo mucoadhesion study |
[171] |
|
CS NPs |
Spray-drying method |
Dexketoprofen trometamol (DT) |
In vitro prolonged drug release, release kinetics, in vivo anti-inflammatory activity, HET-CAM assay |
[93] |
|
AvrA NPs-ALG/CS MPs |
Flow focusing microfluidic method |
Salmonella effector enzyme (AvrA) |
In vitro drug release, in vivo reduction of inflammation in murine dextran sulfate sodium (DSS) colitis model |
[172] |
|
Oral vaccines |
||||
|
β-CD/CS NPs |
Precipitation/coacervation method |
Ovalbumin (OVA) |
In vitro drug release, in vivo immune response in Balb/c mice |
[173] |
|
CS chloride NPs |
Ionic gelation (TPP) |
Ovalbumin (OVA) |
In vitro cell toxicity, permeability study, transepithelial electrical resistance studies, in vivo studies |
[174] |
|
ALG/CS NPs anchored with lipopolysaccharide (LPS) as an adjuvant |
Ionic gelation (TPP) |
HBsAg antigen |
In vitro drug release and mucoadhesion study, stability, cytotoxicity, in vivo immunization studies |
[175] |
|
ALG/CS coating LDHs |
Co-precipitation-hydrothermal method |
BSA |
In vitro drug release, cellular uptake, stability in biological fluids |
[176] |
|
Oral delivery of other drugs |
||||
|
Cs PLNs |
Self-assembly |
Enoxaparin |
Mucoadhesive properties, stability, in vivo anticoagulant activity in rats |
[177] |
|
CS NPS |
Double emulsification solvent evaporation method |
Salmon calcitonin (sCT) and puerarin (PR) |
In vitro drug release, stability, cellular uptake, in vivo pharmacokinetic study |
[79] |
|
SA/CS and NaCAS/CS NPs |
Ionic gelation (oxidized dextran) |
Astaxanthin (ASTX) (hepatic fibrosis treatment) |
ABTS radical scavenging assay, cytotoxicity, anti-fibrogenic activity |
[114] |
|
Soy lecithin/CS hybrid NPs |
Self-assembly |
Raloxifene hydrochloride (RLX) |
In vitro drug dissolution and release, MTT assay, intestinal drug uptake, in vivo pharmacokinetic studies, biodistribution, ex-vivo mucoadhesion studies |
[178] |
|
Ocular drug delivery |
||||
|
GCS NPs |
Self-assembly |
Dexamethasone (DEX) |
In vitro drug release, mucoadhesive, cytotoxicity, and anti-inflammatory efficacy, in vivo study: eye irritation test and distribution test |
[179] |
|
CMC/GSH/GlySar/LDHs |
Coprecipitation–hydrothermal method |
Dexamethasone disodium phosphate (DEXP) DD to the posterior segment of the eye |
In vitro toxicity study on human conjunctival epithelial cells, cellular uptake, the in vivo precorneal retention study, the tissue distribution evaluation of rabbit’s eyes |
[148] |
|
CS/LIP |
Thin-film hydration method |
Triamcinolone acetonide (TA) Treatment of posterior eye segment diseases |
In vitro drug release, in vivo drug release |
[180] |
|
TCM/LNPs |
Emulsion solvent evaporation method |
Baicalein (BAI) |
In vitro sustained drug release, in vivo ocular irritation study, pre-corneal retention evaluation, pharmacokinetic study |
[33] |
|
CS NPs |
Ionic gelation (TPP) |
Levofloxacin (LFX) Therapy of ocular infections |
The antimicrobial study, in vitro ocular tolerance, in vivo pharmacoscintigraphic study |
[181] |
|
CS/poly(ethylene glycol) methacrylate MNPs |
Double crosslinking (ionic and covalent) in reverse emulsion |
Bevacizumab Treatment of posterior segment of the eye |
In vitro drug release kinetics, hemocompatibility, in vivo study of antiangiogenic effect (eye) |
[48] |
|
CS/PCL NPs |
Single-step emulsification method |
Dorzolamide (DRZ) Glaucoma treatment |
In vitro drug release, in vivo corneal flux experiment, corneal hydration study, ex vivo bioadhesion study, ocular tolerance study, Hen egg test-chorioallantoic membrane (HET-CAM) test |
[82] |
|
CS/gelatin gel with CUR-NPs |
- |
Latanoprost (LP) and curcumin (CUR) Glaucoma treatment |
In vitro drug release, in vitro biocompatibility, in vivo incompatibility in rabbits |
[182] |
|
Nasal drug delivery (topical) |
||||
|
CS NPs |
Ionic gelation |
Cromolyn Therapy of allergic rhinitis |
In vitro drug release, permeation, and penetration, mucoadhesion assay |
[183] |
|
DCHBC NPs |
Dialysis method |
Cetirizine (CTZ) Therapy of allergic rhinitis |
In vitro stimuli-responsive drug release, cytotoxicity, hemolysis test, protein adsorption |
[184] |
|
CS or CS maleimide NPs |
Ionic gelation (TPP) |
Japanese encephalitis-chimeric virus vaccine Nasal vaccine |
Mucoadhesive properties, antigen uptake study, in vivo study of immunization of mice |
[185] |
|
Nose to brain delivery |
||||
|
CS/HSA NPs |
Desolvation method |
Tacrine and R-flurbiprofen |
mucoadhesion properties, in vitro drug release, permeation, uptake, ex vivo diffusion experiments on rabbit nasal mucosa |
[186] |
|
PLGA NPs and PLGA/CS NPs |
Nanoprecipitation |
Ropinirole hydrochloride Antiparkinson therapy |
In vitro drug release, mucoadhesion, hemolysis assay, stability study, studies on peripheral blood mononuclear cells and RAW 264.7 macrophage cell line—cytotoxicity, cellular uptake ex vivo permeability studies |
[187] |
|
N,O-CMC NPs |
Emulsion solvent evaporation method |
Dopamine (DOPA) orTyrosine (Tyr) |
In vitro drug release, mucoadhesive properties, cytotoxicity, cellular uptake |
[188] |
|
CS NPs |
- |
Therapy of Huntington disease |
Gene silencing studies |
[189] |
|
CS NPs |
Ionic gelation (TPP) |
Zolmitriptan (ZOL) Therapy of migraine |
In vivo stability, in vivo pharmacokinetic study on Wistar rats |
[190] |
|
CS NPs |
Ionic gelation (TPP) |
Rotigotine (R) Treatment of Parkinson’s disease |
In vitro cellular uptake, cytotoxicity assay, neuroprotective activity, antioxidant activity, in vivo pharmacodynamic and pharmacokinetic study |
[191] |
|
Pulmonary (inhalation) drug delivery |
||||
|
CS NPs |
Emulsion method |
Nicotine hydrogen tartrate (NHT) Treatment of nicotine addiction |
In vitro evaluation of nose-only inhalation device, assessment of bioactivity of NHT-CS NPs via locomotor test by injection, histopathological analysis of lung tissues |
[192] |
|
CS/PLGA NPs followed by coating with chitosan |
Solvent evaporation (double-emulsion) method |
Catechin hydrate (CTH) |
In vitro drug release, ex-vivo permeation study on the nasal mucosa, cytotoxicity, in vivo comparative pulmokinetic study |
[193] |
|
CS/SLNs |
Hot ultrasonication |
Rifampicin (RIF) Tuberculosis treatment |
In vitro drug release, mucoadhesive properties, in vitro cell viability and permeability studies, stability studies |
[194] |
|
Mn-TMC NPs |
Ionic gelation (TPP) |
Etofylline (ETO) Asthma treatment |
Sustained drug release, biodegradation studies, stability, safety, and aerodynamic behavior |
[195] |
|
HA/CS NPs |
Self-assembly |
Ferulic acid (FA) Asthma treatment |
In vivo inhalation toxicity assessment |
[196] |
|
Buccal delivery |
||||
|
CS NPs |
Ionic gelation (TPP) |
Oxiplatin Anticancer therapy |
ex vivo its penetration in porcine mucosa under both passive and iontophoretic topical treatments |
[102] |
|
Cat/CS/HA NPs |
Ionic gelation |
Doxorubicin (DOX) Oral cancer treatment |
Ex vivo mucoadhesive study, in vitro drug release, cytotoxicity, cellular uptake, cancer cells death |
[103] |
|
TTEC NPs |
PEC method |
Insulin |
In vitro drug release, ex vivo permeation study on rabbit mucosa, MTT assay |
[197] |
|
CS/PEO NFs |
Electrospinning |
Sublingval delivery |
Ex vivo adhesion on porcine mucosa, swelling, compatibility |
[198] |
|
Periodontal delivery |
||||
|
CS NPs |
Ionic gelation |
Minocycline, tetracycline Periodontal disease |
Human gingival fibroblasts behavior, Cell viability and culture metabolic activity, cellular uptake, inflammatory gene expression |
[199] |
|
Core-sheath NFs: shell layer: CS core: PVA containing drug |
Coaxial electrospinning and ionic gelation (genipin) |
Tetracycline hydrochloride (TH) Periodontitis treatment |
In vitro sustained drug release, in vitro antimicrobial activity, cytotoxicity |
[200] |
|
CS/IO NPs |
- |
Chlorhexidine (CHX) Antimicrobial and antibiofilm effect against oral disease |
Determination of MIC, cytotoxicity by MTT assay |
[201] |
|
Dermal drug delivery |
||||
|
CS NPs |
Ionic gelation (TPP) |
Nicotinamide |
Clinical test, skin bioadhesion, deposition of drug in different skin layers |
[202] |
|
Poly-(ε-caprolactone)-lipid core NCs nad CS/poly-(ε-caprolactone)-lipid-core NCs |
Interfacial deposition technique |
Dutasteride Hair follicle targeting after massage procedure |
In vitro drug release, stability, in vitro skin permeation |
[203] |
|
Transdermal drug delivery |
||||
|
CS Hydroxypropyltrimonium chloride/PLGA NPs |
Antisolvent diffusion method |
Hen egg-white lysozyme (HEL) allergen immunotherapy to hair follicles using iontophoresis |
In vitro cellular uptake, ex vivo skin accumulation study, in vivo transcutaneous immunization experiment |
[204] |
|
CS NFs |
Electrospinning |
Colchicine Anti-skin cancer therapy |
Ex vivo skin permeation, deposition analysis, release kinetic and anti-melanoma efficiency against A-375 cell line |
[205] |
|
CS NPs |
Nanospray-drying technique |
5-fluorouracil (FU) |
Synergistic microwave delivery of anti-cancer |
[206] |
|
Wound healing |
||||
|
CS/PEO NFs |
Electrospinning |
Teicoplanin Local antibiotic wound healing |
In vitro drug release, antibacterial test, cytotoxicity, in vivo study on rat full-thickness wound model |
[67] |
|
PCL/CS NFs |
Electrospinning |
Curcumin (CUR) Wound dressing |
antibacterial, antioxidant properties, cell viability, and in vivo wound healing efficiency and histological assay |
[207] |
|
hydrogel membranes based on HA/PU/PVA loaded with cefepime-CS NPs |
Ionic gelation (TPP) |
Cefepime |
In vitro drug release, bacterial inhibition |
[208] |
|
CS NPs loaded hydrogel |
Ionic gelation (TPP) |
Pterocarpus marsupium heartwood extract (PM) Therapy of diabetic wounds |
In vitro drug release efficiency, in-vitro anti-microbial activity, in vivo wound healing action in streptozotocin administered diabetic rat models |
[209] |
|
Vaginal drug delivery |
||||
|
CS NPs |
Ionic gelation (TPP) |
Miconazole nitrate Therapy of vulvovaginal candidiasis |
In vivo evaluation on vulvovaginal murine model |
[210] |
|
CS NPs encapsulated in hydrophilic freeze-dried cylinders |
Ionic gelation (TPP) |
Insulin Peptide-based vaccines or delivery of microbicides |
In vitro drug release, ex vivo insulin penetration across porcine vaginal mucosa |
[211] |
|
CS and spicules NPs |
Ionic gelation (TPP) |
Calophycin A (Cal A)—seaweed-derived metabolite Therapy of vaginal candidiasis |
In vitro anti-candidal activity, in vivo on mice |
[212] |
|
Vaccine delivery |
||||
|
CS and ALG coated CS NPs |
Precipitation/coacervation method |
Hepatitis A vaccine (HAV) |
Assay of HAV-specific antibodies and their isotypes, lymphoproliferation assay, the effect of HAV formulation on the splenocytes proliferation in vaccinated mice |
[213] |
|
CS NPs |
Ionic gelation (TPP) |
Aah II toxin isolated from Androctonus australis hector (scorpion) venom |
In vitro toxin-release study, in vivo immunization trial |
[214] |
|
Gene delivery |
||||
|
MPC derived from carbonized CTS echitosan capsulated ZIF-8 |
Carbonization |
Luciferase-expressing plasmid (pGL3), and splice correction oligonucleotides (SCO) |
Cell biocompatibility, transfection efficiency, mechanism of uptake |
[215] |
|
LMW mannosylated CS NPs |
Ionic gelation |
CpG oligodeoxynucleotides |
Cytotoxicity, cellular uptake, immunostimulatory effect-cytokine release in RAW264.7 cells, efficient vector for intracellular CpG ODN delivery |
[216] |
|
TMC Cys, MABCMC, and CysMABC NPs |
Ionic gelation |
Plasmid DNA pEGFP-N1 |
In vitro DNA transfection efficiency, cytotoxicity |
[217] |
|
CS, PEI, and CMD NPs |
Self-assembly |
Anti-HIV siRNA HIV therapy |
In vitro cytotoxicity assay and siRNA delivery in two mammalian cell lines, macrophage RAW264.7, and HEK293 |
[218] |
|
TMC/DS or ALG NPs |
PEC method |
hSET1 antisense—silencing oligonucleotide Cancer therapy |
In vitro cell viability, cellular uptake, in vivo study on mice |
[219] |
|
CMC NPs labelled with FITC NPs (FITCCS/CMC) |
Self-assembly |
Anti-β-catenin siRNA Ultrasound-triggered targeted therapy of colon cancer |
In vitro drug release, cytotoxic assay, cellular uptake, therapeutic evaluation |
[220] |
|
Guanidinylated O-CMC NPs (GOCMCS) |
Self-assembly |
SiRNA delivery |
In vitro cell transfection studies with A549 cells, cellular uptake |
[221] |
|
SPION NPs encapsulated with TAT peptide/TC and TMC |
Electrostatic interaction |
siRNA Targeted anti-cancer therapy |
Cytotoxicity, cellular internalization, in vivo pharmacokinetic and biodistribution, colony formation assay, wound healing assay, Chick chorioallantoic membrane (CAM) assay |
[222] |
|
HA/PCL NPs |
Ionic gelation (TPP) |
IL6-specific siRNA and BV6 treatment of breast and colon cancer |
In vitro drug release, cellular uptake, MTT assay, apoptosis assay, Chick chorioallantoic membrane assay, wound healing assay, a clonogenic assay of tumor cells in vitro, transwell migration assay, in vivo antitumor efficacy on mice |
[223] |
|
CMD/TMC NPs |
Nanoprecipitation |
Codelivery of NIK/STAT3-specific siRNA and BV6 Cancer therapy |
Stability of NPs, in vitro drug release, cellular uptake, transfection of cells, MTT assay, Chick chorioallantoic membrane (CAM) assay, wound healing assay, colony formation assay |
[224] |
|
HA/TMC NPs |
PEC method |
IL-6- and STAT3-specific siRNAs Cancer therapy |
In vitro drug release, stability in serum, MTT cytotoxicity assay, cellular uptake, transfection efficiency, Colony formation assay Wound healing assay |
[225] |
|
CS NPs |
- |
Doxorubicin (DOX) and Bcl-2 siRNA co-delivery of therapeutics and si-RNA Cancer therapy |
In vitro drug release, in vivo tumor suppression test |
[226] |
|
Polyethyleneglycol-poly lactic acid CS (PP CS NPs) |
- |
Nerve growth factor (NGF), acteoside (ACT), and plasmid DNA (pDNA) Treatment of Parkinson’s disease |
Plasmid DNA (pDNA), nerve growth factor (NGF), acteoside (Act) |
[227] |
3.4.1. Oral Drug Delivery
Antidiabetic Drugs
Anticancer Drugs
Antihypertensive Drugs
Antioxidants
Anti-inflammatory Drugs
Oral Vaccines
Oher Drugs
3.4.2. Ocular Drug Delivery
3.4.3. Nasal Drug Delivery
Topical Nasal Delivery
Systemic Nasal Delivery
Nose to Brain Delivery
3.4.4. Pulmonary Drug Delivery
3.4.5. Buccal Drug Delivery
3.4.6. Periodontal Drug Delivery
3.4.7. Dermal and Transdermal Drug Delivery
Transdermal Drug Delivery
Dermal Delivery
3.4.8. Wound Healing
3.4.9. Vaginal Drug Delivery
Vaginal delivery is very attractive for both local and systemic administration of drugs. For the latest purpose, it shows several advantages concerning conventional oral or parenteral ways, such as the avoidance of the stomach acidic pH, the hepatic first-pass effect, or the needle-based formulations uncomfortable for the patients. The vaginal mucosa is characterized by high robustness, ease of accessibility, and rich blood supply. The effectiveness of typical vaginal formulations (creams, foams, gels, tablets, films, rings, and suppositories) can be limited by their low active residence time due to the washing-effect of the vaginal physiological fluids, small absorption area, barrier properties of the mucosa, and inadequate spreading of the formulation on vaginal surfaces. Pharmaceutical nanocarriers provide several advantages such as a high surface area and great carrier capacity, improved stability of the therapeutic agents against chemical/enzymatic degradation, enhanced bioavailability, longer drug effect in the target tissue, and drug targeting upon inclusion of specific ligands. The development of NP-based vaginal drug delivery formulations has largely been focused on biological vaccine or microbicide delivery for prevention or treatment of sexually transmitted diseases such as human immunodeficiency virus (HIV), herpes simplex virus (HSV), or human papillomavirus (HPV). The vaginal route allows a localized delivery of peptide-based vaccines/microbicides close to both the site of infection and infectible cells. An opportune vaginal drug delivery system should provide mucosal interactions that facilitate bioadhesion with mucosa increasing drug residence time at the mucosal surface, and penetration enhancement properties to allow penetration into vaginal tissue cells. In the last years, many authors have studied the mucoadhesive and penetration enhancement properties of CS in this area [11,52].
Marciello et al. [211] proposed a straightforward and efficient strategy for the vaginal application and release of peptide-loaded mucoadhesive CS NPs. The CS NPs, responsible for carrying the peptide drug and allowing adhesion to the vaginal mucosal epithelium, were encapsulated in suitable hydrophilic freeze-dried cylinders. The hydrophilic freeze-dried cylinders facilitated the application and quick release of the CS NPs to the vaginal zone. Upon contact with the aqueous vaginal medium, the excipients constituting these sponge-like systems were quickly dissolved enabling the release of their content. In vitro release studies showed the ability of the sponge-like systems and the CS NPs to deliver the mucoadhesive NPs and peptides, respectively. CLSM (confocal laser scanning microscopy) micrographs proved the CS NPs ability to promote the peptide penetration inside the vaginal mucosa.
To prime adaptive immune responses from the female reproductive tract (FRT), particulate antigens must be transported to draining lymph nodes (dLNs) since no local organized lymphoid structures are being equivalent to those found in the respiratory or gastrointestinal tracts. Therefore, it is a challenge to find how to safely and effectively navigate successive barriers to transport such as crossing the epithelium and gaining access to migratory cells and lymphatic drainage that provide entry into dLNs. Park et al. [235] demonstrated that an intravaginal pretreatment with CS significantly facilitated the translocation of NPs across the multilayered vaginal epithelium to target dLNs. In addition, the CS pretreatment was found to enhance the NP associations with immunogenic antigen-presenting cells in the vaginal submucosa. These observations indicate that CS may have great potential as an adjuvant for both local and systemic protective immunity against viral infections in the FRT.
Vaginal candidiasis is a common genital tract infection caused by dimorphic fungi of the genus Candida. Sustained delivery systems prepared using biodegradable polymers, such as CS, are interesting because they may promote a slow release of the drug by controlled rates of their degradation. CS itself can reduce the microbial population. Hence, a nanoparticulate system based on CS can represent an alternative and promising therapy for the treatment of vulvovaginal candidiasis. Amaral et al. [210] fabricated polymeric NPs based on CS incorporating the common antifungal miconazole nitrate and tested them in vivo using murine vulvovaginal candidiasis (VVC). The treatment using the CS NPs with miconazole nitrate provided the same therapeutic efficacy as miconazole nitrate in a commercial cream formulation but using the antifungal content about seven-fold lower. In another work, Arumugam et al. [212] studied the killing effects of seaweed-derived metabolite Callophycin A (Cal) loaded in both chitosan (Cal-CS) and marine sponge-derived spicules (Cal-Spi) NPs. Vaginal candidiasis-induced animal model experiments confirmed that the candidicidal activity of Cal-CS NPs resulted in a significant reduction in the fungal burden of vaginal lavage. The histo-morphological alterations also evidenced that the protective role of Cal-CS NPs in the VVC model. The Cal-CS NPs could be used as an alternative strategy for the development of novel marine natural product-based topical applications.
3.4.10. Vaccine Delivery
Vaccine refers to biological products applied to prevent or control the occurrence and spread of infectious diseases. As for nature, the vaccines may be microbes or their toxins, enzymes, human or animal serum, and cells. The emergence of vaccines has made a significant contribution to the prevention and control of diseases. However, many problems are affecting the quality of the vaccines during their preparation, storage, and administration The major hurdle associated with oral mucosal immunization is enzymatic degradation of antigen at the stomach and low uptake of antigen sampling cells through the intestine. Hence, the vaccine administered through the oral route (less through the nasal route) requires the use of NPs. Nanomaterial-encapsulated vaccines have been proved effective in the delivery of antigens to immune cells. Such encapsulation can help in promoting immune responses and represents a promising vaccine transport vehicle. In oral vaccine delivery, the main target is Peyer’s patches. Due to the nanoparticle system, the vaccine is protected from enzymatic degradation while going to the mucosal tissue and taken up by the M-cells. CS is a particularly attractive choice for vaccine delivery because of its low immunogenicity, low toxicity, biocompatibility, and biodegradability. It has been widely used for mucosal but also systemic vaccine delivery and even in the preparation of DNA mucosal vaccines [11,52]. For novel, recently published oral and nasal CS NP-based vaccine delivery systems see Sections 3.4.1.6 and 3.4.3.2, respectively.
In systemic vaccine delivery, CS acts as an adjuvant. Activation of macrophages occurs after the uptake of CS [52]. CS exhibited good tolerability and excellent immune stimulation. The numerous recent hepatitis A outbreaks emphasize the need for vaccination; despite the effectiveness of the current vaccination, further development is needed to overcome its high cost plus some immune response limitations. AbdelaAllah et al. [213] evaluated the use of CS and ALG-coated CS NPs as an adjuvant/carrier for the hepatitis A (HA) vaccine against the traditional adjuvant alum (AL). Immune responses towards the HA-AL, HA-CS, and HA-ALG/CS NPs were assessed in mice. The HA-ALG/CS NPs significantly improved the immunogenicity by increasing the seroconversion rate, the hepatitis A antibodies level, and the splenocytes proliferation. Thus, the HA-ALG/CS NPs adjuvant was superior to the other classes in IFN-γ (interferon-gamma) and IL-10 (interleukin 10) development. The solution formula of the HA vaccine with CS showed comparable humoral and cellular immune responses to AL-adjuvanted suspension with a balanced Th1/Th2 immune pathway. The study demonstrated the potential of the ALG-coated CS NPs as an effective carrier for an HA vaccine.
3.4.11. Gene Delivery
Genes encoded specific proteins are essential for various physiological processes of the body and their mutations often result in disease. Gene therapy is a promising strategy to treat genetic diseases via the introduction of a foreign gene into a target cell which is then transcribed, and the genetic information is finally translated into the corresponding protein. The aim is to correct a damaged gene. For this process to be completed, the gene delivery system has to overcome several hurdles. The factors affecting transfection include targeting the delivery system to the target cell, uptake, and degradation in the endolysosomes, transport through the cell membrane, and intracellular transfer of plasmid DNA to the nucleus. Thus, DNA and RNA molecules can be destroyed by harsh acids and enzymes that are produced in the body. DNA and RNA are anionic polymers that have a good affinity with cationic polymers such as CS. CS can protect the DNA or RNA against nuclease degradation by forming a polyelectrolyte complex with negatively charged DNA or RNA. This protection can improve the transfection efficiency [11,52].
There are two main approaches for gene delivery systems, i.e., viral and nonviral ones. Considering efficiency and safety issues, polymeric vehicles would seem more convenient than other gene delivery approaches. Because of its positive charge and the small size of CS NPs (below 100 nm), endocytosis can be achieved. CS NP is therefore a promising excipient for non-viral gene delivery. In addition, chitosan–DNA-based drug complexes protect at least to some extent against degradation by DNAses in this way improving the bioavailability of DNA-based drugs delivered into the body [8]. Rahmani et al. [217] investigated in vitro DNA transfection efficiency of three CS derivatives, namely trimethyl chitosan thiolated with cysteine (CysTMC), methylated 4-N,N-dimethyl aminobenzyl N,O carboxymethyl chitosan (MABCMC), and trimethyl aminobenzyl chitosan thiolated with cysteine (CysMABC). The results showed that all the polymers could condense DNA plasmid strongly from N/P 2 and nanocomplexes had eligible sizes and zeta potentials. Moreover, the nanocomplexes had negligible cytotoxicity and CysMABC was the most effective vehicle for gene delivery in HEK-293T cells. In the two other cell lines, SKOV-3 and MCF-7, CysTMC exhibited the highest transfection efficiency.
Among several types of non-viral vectors, cell-penetrating peptides (CPPs), short peptides with 30 amino acids, are promising. CPPs show high biocompatibility and offer the potential for large-scale production. However, CPPs exhibit low transfection efficiency. Hybrid conjugation of CPPs with inorganic nanomaterials improved their efficiency and may open new venues for multifunctional treatment. Abdelhamid et al. [215] synthesized hierarchical mesoporous carbon (MPC) nanomaterials derived from the carbonized chitosan (CTS) encapsulated zeolitic imidazolate frameworks-8 (ZIF-8) and applied them for gene delivery. The MPC materials were applied as a non-viral vector for gene delivery using two oligonucleotides (ONs) called luciferase-expressing plasmid (pGL3), and splice correction oligonucleotides (SCO). The materials were biocompatible and showed insignificant toxicity. The MPC improved the transfection efficiency of cell-penetrating peptides (CPPs) (PepFect 14 (PF-14), and PF-221) by 10-fold compared to commercial vector Lipofectamine™2000 due to the synergistic effect of their action. This may be due to the positive charge of CPPs that ensure higher interactions with the negative charge of the cells.
Small interfering RNA (siRNA) is a double-stranded RNA of 20 to 25 nucleotides in length and has many important functions in biology. It primarily plays a unique role in the RNA interference events which regulates gene expression in a specific manner. siRNA-based therapies have great potential in the modulation of a large number of target mRNA molecule silencing, which ultimately decreased levels of the targeted protein. Nevertheless, there are challenges that applications of this gene silencing technology need to overcome, including rapid degradation of siRNA under physiological conditions and the difficulty of passing the cytoplasmic membrane of negatively charged siRNA. The key challenge in realizing the therapeutic potency of siRNA is the development and design of a novel degradable vector with safe and sufficient gene delivery efficacy. To improve the transfection efficiency, new safe vehicles for cancer gene delivery based on NPs have been developed. Recently some different modifications of CS have been used to produce these NPs, for example, a polyelectrolyte complex containing trimethyl chitosan (TMC) as the positive and, dextran sulfate, and alginate as the negative part [219], guanidinylated O-carboxymethyl chitosan (GOCMCS) along with poly-β-amino ester (PBAE) for siRNA delivery [221].
Yan et al. [220] fabricated the NPs of carboxymethyl chitosan (CMC) and labeled fluorescein isothiocyanate (FITC)-chitosan hydrochloride (FITCCS) as carriers for ultrasound-triggered drug delivery to treat colon cancer. The results showed the FITCCS/CMC NPs could effectively encapsulate anti-β-catenin siRNA through ionic gelation self-assembly to improve the stability of siRNA. The FITCCS/CMC NP-based pH-sensitive delivery system provided a controlled release of siRNA through responding to external stimulus (ultrasound) under favorable pH conditions. Following the transfection of anti-β-catenin siRNA for 48 h, the β-catenin protein expression of the colon cancer cells was reduced to about 40.10%, indicating the effective reduction of the protein that promotes colon cancer proliferation.
CS as a promising polysaccharide for gene/siRNA delivery requires some additional treatments to modify CS NPs. Mobarakeh et al. [218] modified CS NPs to introduce anti-HIV siRNA into two mammalian cell lines, macrophage RAW264.7, and HEK293. The CS NPs were prepared by using different concentrations of CS, polyethyleneimine (PEI), and carboxymethyl dextran (CMD) in various formulations. The results indicated that the combination of CS with both CMD and PEI significantly improved both cell viability and siRNA delivery. In the studied cell types, the NPs noticeably increased siRNA delivery efficiency with no significant cytotoxicity or apoptosis-inducing effects compared to the control cells. In addition, the NPs significantly reduced the RNA and protein expression of HIV-1 tat in both stable cells.
Induction of Hypoxia Inducible Factor (HIF) as a direct consequence of oxygen deficiency in tumor tissues is a potent stimulus of CD73 (ecto-5′nucleotidase) expression. Hypoxic environment and CD73 overexpression are associated with altered metabolism, elevated cancer cell proliferation, and tumor vascularization. Hajizadeh et al. [222] developed a delivery system for silencing CD73 and HIF-1α gene using siRNA-loaded superparamagnetic iron oxide (SPION) nanocarriers for cancer treatment. The SPIONs were encapsulated with thiolated chitosan (TC) and trimethyl chitosan (TMC) for improving their stabilization and functionalization. The produced NPs were efficiently accumulated in the tumor site, indicating their stability and targeting ability in reaching the tumor region. TAT-conjugated TMC/TC/SPIONs containing siRNAs significantly reduced the HIF-1α and CD73 expression levels in cancer cells. Moreover, siRNA-loaded NPs effectively reduced tumor growth and angiogenesis as is illustrated by CAM (cell angiogenesis) assay in Figure 33 (see Figure 33. CAM assay of HIF-1α/CD73 siRNA loaded TAT-TMC/TC/SPION nanocarriers suppressed in vivo tumor development and angiogenesis [222] in our recent paper V. Mikusova, P. Mikus, Int. J. Mol. Sci. 2021, 22, 9652). Hence, the co-silencing of CD73 and HIF-1α can be assumed as a novel anti-cancer treatment strategy with a high tumor suppression potential.
Some researchers focused on the development of delivery systems that combine silencing of inhibitor of apoptosis (IAP) genes BV6 (their increased expression is associated with cancer progression and chemoresistance) and interleukin (IL)-6, as a new promising anti-tumor treatment strategy. For this purpose, Salimifar et al. [223] prepared hyaluronate/PEGylated chitosan lactate HA/PCL) NPs to simultaneously deliver IL6-specific siRNA and BV6 to 4T1 (breast cancer) and CT26 (colon cancer) cells. They investigated the anti-tumor properties of this combined therapy both in vitro and in vivo. Such therapy synergistically increased apoptosis and decreased cell migration, proliferation, colony formation, and angiogenesis in both 4T1 and CT26 cell lines and, by that, suppressed cancer progression in tumor-bearing mice that was associated with enhanced survival time.
Another strategy is to use BV6 along with inhibition of signal transducer and activator of transcription 3 (STAT3), which is an important factor in the survival of tumor cells, and NIK as a mediator of BV6 unpredicted side effects. This combination has the potential to induce effective apoptosis in tumor cells. Nikhoo et al. [224] used carboxymethyl dextran-conjugated trimethyl chitosan (CMD/TMC) NPs loaded with NIK/STAT3-specific siRNA and BV6 to synergistically induce apoptosis in the breast, colorectal, and melanoma cancer cell lines. Their results showed that in addition to enhanced pro-apoptotic effects, this combined therapy reduced proliferation, cell migration, colony formation, and angiogenesis, along with the expression of factors including IL-10 and HIF in the tumor cells. Masjedi et al. [225] generated the active-targeted hyaluronate (HA) recoated N, N, N-trimethyl chitosan (TMC) NPs to deliver IL-6- and STAT3-specific siRNAs to the CD44-expressing cancer cells. The results showed that the synthesized NPs had high transfection efficiency, low toxicity, and controlled siRNA release. The siRNA-loaded NPs significantly inhibited the IL-6/STAT3 expression, which was associated with blockade of proliferation, colony formation, migration, and angiogenesis in the cancer cells.
A co-delivery of chemotherapeutic drugs and siRNA has gained increasing attention owing to the enhanced antitumor efficacy over the single administration. Studies in this field showed that simultaneous delivery of chemotherapeutic drugs and siRNA via a single targeted vector was more effective in treating some cancers than a sequential administration of two separate vectors with one drug in each. Yan et al. [226] developed a CS-based pH-responsive prodrug vector for the co-delivery of doxorubicin (DOX) and Bcl-2 siRNA. The accumulation of fabricated NPs in hepatoma cells was enhanced by glycyrrhetinic acid receptor-mediated endocytosis. This nanoplatform can efficiently integrate gene- and chemotherapies with a dramatically enhanced tumor inhibitory rate (88.0%) in vivo.
Gene therapy treatment strategies for Parkinson’s disease (PD) have recently come into prominence. Here, gene therapy has potential advantages to increase pre-cursor cells to synthesize dopamine and could repair or prevent degeneration of dopaminergic neurons. Gene therapy could correct a specific genetic defect by increasing, decreasing, or silencing the expression of target genes, or induce the endogenous production of a therapeutic protein. Xue et al. [227] proposed drug-loaded chitosan nanoparticles (pDNA-NGF-ActPP/CS NPs as novel candidates for the design of anti-PD drugs. They investigated the effects of chitosan polyethylene glycol-poly lactic acid (PP/CS) NPs conjugated with nerve growth factor (NGF), acteoside (Act), and plasmid DNA (pDNA) (pDNA-NGF-ActPP/CS NPs) for PD therapy using in vitro and in vivo models. Using PD cell models, they demonstrated that pDNA-NGF-ActPP/CS NPs had good neuroprotective effects. More significantly, experiments using a mouse PD model showed that pDNA-NGF-ActPP/CS NPs could ameliorate the behavioral disorders of sick mice. Immunohistochemical and Western blot (WB) analyses revealed that pDNA-NGF-ActPP/CS NPs could significantly reverse dopaminergic (DA) neuron loss in the substantia nigra and striatum of sick mice. This study opens up a novel avenue to develop anti-PD drugs.
4. Concluding Remarks
The recent review and research papers indicate CS NPs play a vital role in biomedical applications such as drug/vaccine/gene delivery, bioimaging, wound healing, tissue engineering, etc. They highlighted an outstanding position of CS as a polysaccharide able to form NPs favorable for various drug delivery purposes because of its many beneficial properties, such as mucoadhesion, controlled drug release, transfection, permeation enhancement, in situ gelation, efflux pump inhibitory properties, and stimuli-responsive properties. Many works demonstrated, via in vitro and in vivo experiments, CS NPs designed for controlled drug delivery may improve the stability of the drug and increase the efficacy of therapeutic agents. The other advantages of CS NPs DDS, presented in recent works, involved reduced therapeutical doses leading to reduced possible side effects, better bioavailability, and finally better patient compliance.
Nevertheless, the current research is still oriented towards an additional improvement of the chitosan properties. There are efforts to enhance its low solubility in physiological pH, stimuli-responsive properties, and specificity towards complex biological systems by chemical modifications of pure CS or by blending CS with other polymers or inorganic materials. In this way, new modified CS-based nanoparticles and nanocomposites possessing more or less enhanced properties were developed. Such innovative CS particulate systems provided, to a more/less extent, non-toxic, biocompatible, stable, target-specific, and biodegradable delivery devices. In addition, the systems with a proper label (e.g., metal-based nanocomposites) enabled target-specific diagnostics (due to easy dual introduction of an imaging agent together with a therapeutic agent) along with a target-specific therapy (due to a stimulus-responsive matrix). Recent newly developed native CS NPs, modified CS NPs, or CS nanocomposites were applied as potential drug carriers for many drugs and various routes of administrations. They were mainly studied for anticancer agents, proteins, vaccines, and genetic material. For example, in oral DD, new CS NPs enhanced the absorption of the drugs through the opening of tight junctions of the mucosal membrane. In ocular DD, in situ gelling properties and mucoadhesive character of CS enabled prolonging drug release. In nasal delivery, CS NPs increased the permeability of the drugs. In vaccine delivery, CS NPs enabled to formulate oral vaccines providing enhanced absorption of these hydrophilic biomolecules.
Despite apparent current progress, safety and targeting specificity are remaining among the main challenges in the future development of CS-based nanoparticulate DDSs. Therefore, systematic studies on biodistribution, in vitro and in vivo toxicity, and selectivity will further continue with newly developed CS derivatives and their NPs and nanocomposites for various administration routes.
Acknowledgments: work was supported by the projects APVV-15-0585, VEGA 1/0463/18, KEGA 027UK-4/2020, and the Toxicological and Antidoping Center at the Faculty of Pharmacy, Comenius University in Bratislava.
References
- Kumar, A.; Vimal, A.; Kumar, A. Why chitosan? From properties to perspective of mucosal drug delivery. J. Biol. Macromol. 2016, 91, 615–622, doi:10.1016/j.ijbiomac.2016.05.054.
- Shanmuganathan, R.; Edison, N.T.J.I.; Lewis Oscar, F.; Kumar, P.; Shanmugan, S.; Pugazhendhi, A. Chitosan nanopolymers: An overview of drug delivery against cancer. J. Biol. Macromol. 2019, 130, 727–736, doi:10.1016/j.ijbiomac.2019.02.060.
- Taher, F.A.; Ibrahim, S.A.; Abd El-Aziz, A.; El-Nour, M.F.A.; El-Sheikh, M.A.; El-Husseiny, N.; Mohamed, M.M. Anti-Proliferative effect of chitosan nanoparticles (extracted from crayfish Procambarus clarkii, Crustacea: Cambaridae) against MDA-MB-231 and SK-BR-3 human breast cancer cell lines. J. Biol. Macromol. 2019, 126, 478–487, doi:10.1016/j.ijbiomac.2018.12.151.
- El Rabey, H.A.; Almutairi, F.M.; Alalawy, A.I.; Al-Duais, M.A.; Sakran, M.I.; Zidan, N.S.; Tayel, A.A. Augmented control of drug-resistant Candida via fluconazole loading into fungal chitosan nanoparticles. Int. J. Biol. Macromol. 2019, 141, 511–516, doi:10.1016/j.ijbiomac.2019.09.036.
- Lim, C.; Lee, D.W.; Israelachvili, J.N.; Jho, Y.S.; Hwang, D.S. Contact time- and pH-dependent adhesion and cohesion of low molecular weight chitosan coated surfaces. Polym. 2015, 117, 887–89, doi:10.1016/j.carbpol.2014.10.033.
- Lim, C.; Hwang, D.S.; Lee, D.W. Intermolecular interactions of chitosan: Degree of acetylation and molecular weight. Polym. 2021, 259, 117782, doi:10.1016/j.carbpol.2021.117782.
- Elgadir, A.A.; Muddin, M.S.; Ferdosh, S.; Adam, A.; Chowdhury, A.J.K.; Sarker, M.Z.I. Impact of chitosan composites and chitosan nanoparticle composites on various drug delivery systems: A review. Food Drug Anal. 2015, 23, 619–629; doi:10.1016/j.jfda.2014.10.008.
- Bernkop-Schnurch, A.; Dunnhaupt, S. Chitosan-based drug delivery systems. J. Pharm. Biopharm. 2012, 81, 463–469, doi:10.1016/j.ejpb.2012.04.007.
- Saikia, C.; Gogoi, P.; Maji, T.K. Chitosan: A promising biopolymer in drug delivery applications. Mol. Genet. Med. 2015 , doi:10.4172/1747-0862.S4-006.
- Huang, G.; Liu, Y.; Chen, L. Chitosan and its derivatives as vehicles for drug delivery. Drug Deliv. 2017, 24, 108–113, doi:10.1080/10717544.2017.1399305.
- Li, J.; Cai, C.; Li, J.; Li, J.; Li, J.; Sun, T.; Wang, L.; Wu, H.; Yu, G. Chitosan-Based nanomaterials for drug delivery. 2018, 23, 2661, doi:10.3390/molecules23102661.
- Safdar, R.; Omar, A.A.; Arunagiri, A.; Regupathi, I.; Thanabalan, M. Potential of chitosan and its derivatives for controlled drug release application—A review. Drug Deliv. Sci. Technol. 2019, 49, 642–659, doi:10.1016/j.jddst.2018.10.020.
- Bakshi, P.S.; Selvakumar, D.; Kadirvelu, K.; Kumar, N.S. Chitosan as an environment friendly biomaterial—A review on recent modifications and applications. J. Biol. Macromol. 2020, 150, 1072–1083, doi:10.1016/j.ijbiomac.2019.10.113.
- Khan, F.; Pham, D.T.N.; Oloketuyi, S.F.; Manivasagan, P.; Oh, J.; Kim, Y.M. Chitosan and their derivatives: Antibiofilm drugs against pathogenic bacteria. Colloids Surf. B Biointerfaces 2020, 158, 11627, doi:10.1016/j.colsurfb.2019.110627.
- Ma, Y.; Garrido-Maestu, A.; Jeong, K.C. Application, mode of action, and in vivo activity of chitosan and its micro- and nanoparticles as microbial agents: A review. Polym. 2017, 176, 257–265, doi:10.1016/j.carbpol.2017.08.082.
- Qin, Y.; Li, P.; Gou, Z. Cationic chitosan derivatives as potential antifungals: A review of structural optimization and applications. Polym. 2020, 236, 116002, doi:10.1016/j.carbpol.2020.116002.
- Hamedi, H.; Moradi, S.; Hudson, M.; Tonelli, A.E. Chitosan based hydrogels and their applications for drug delivery in wound dressings: A review. Polym. 2018, 199, 445–460, doi:10.1016/j.carbpol.2018.06.114.
- Ali, A.; Ahmed, S. A review on chitosan and its nanocomposites in drug delivery. J. Biol. Macromol. 2018, 109, 273–286, doi:10.1016/j.ijbiomac.2017.12.078.
- Negm, N.A.; Hefni, H.H.H.; Abd-Elaal, A.A.A.; Badr, E.A.; Abou Kana, M.T.H. Advancement on modification of chitosan biopolymer and its potential applications. J. Biol. Macromol. 2020, 152, 681–702, doi:10.1016/j.ijbiomac.2020.02.196.
- Tian, Y.; Sun, Y.; Wang, X.; Kasparis, G.; Mao, S. Chapter 15—Chitosan and its derivatives-based nano-formulations in drug delivery. In Nanobiomaterials in Drug Delivery; Grumezescu, M.A, Ed.; Elsevier: Amsterdam, The Netherlands, 2016, pp.515–572.
- Mateescu, M.A.; Ispas-Szabo, P.; Assaad, E. Chitosan and its derivatives as self-assembled systems for drug delivery In Controlled Drug Delivery: The Role of Self-Assembling Multi-Task Excipients; Elsevier: Amsterdam, The Netherlands, 2015, pp. 85–125.
- Fonseca-Santos, B.; Chorilli, M. An overview of carboxymethylated derivatives of chitosan: Their use as biomaterials and drug delivery systems. Sci. Eng. C 2017, 77, 1349–1362, doi:10.1016/j.msec.2017.03.198.
- Dimassi, S.; Tabary, N.; Chai, F.; Blanchemain, N.; Martel, B. Sulfonated and sulfated chitosan derivatives for biomedical applications: A review. Polym. 2018, 202, 382–396, doi:10.1016/j.carbpol.2018.09.011.
- Tekade, M.; Maheshwari, N.; Youngren-Ortiz, S.R.; Pandey, V.; Chourasiya, Y.; Soni, V.; Deb, P.K.; Sharma, M.C. Chapter 13—Thiolated-Chitosan: A novel mucoadhesive polymer for better-targeted drug delivery. In Advances in Pharmaceutical Product Development and Research, Biomaterials and Bionanotechnology, Academic Press: Cambridge, MA, USA, 2019, pp. 459–493.
- Dang, Y.; Guan, J. Nanoparticle-based drug delivery systems for cancer therapy. Smart Mater. Med. 2020, 1, 10–19, doi:10.1016/j.smaim.2020.04.001.
- Wang, W.; Xue, C.; Mao, X. Chitosan: Structural modification, biological activity and application. J. Biol. Macromol. 2020, 164, 4532–4546, doi:10.1016/j.ijbiomac.2020.09.042.
- Chu, L.; Zhang, Y.; Feng, Z.; Yang, J.; Tian, Q.; Yao, X.; Zhao, X.; Tan, H.; Chen, Y. Synthesis and application of a series of amphipathic chitosan derivatives and the corresponding magnetic nanoparticle-embedded polymeric micelles. Polym. 2019, 223, 114966, doi:doi.org/10.1016/j.carbpol.2019.06.005.
- Burr, S.J.; Williams, P.A.; Ratcliffe, I. Synthesis of cationic alkylated chitosans and an investigation of their rheological properties and interaction with anionic surfactant. Polym. 2018, 201, 615–623, doi:10.1016/j.carbpol.2018.08.105.
- Nanda, B.; Manjappa, A.S.; Chuttani, K.; Balasinor, N.H.; Mishra, A.K.; Ramachandra Murthy, R.S. Acylated chitosan anchored paclitaxel loaded liposomes: Pharmacokinetic and biodistribution study in Ehrlich ascites tumor bearing mice. J. Biol. Macromol. 2019, 122, 367–379, doi:10.1016/j.ijbiomac.2018.10.071.
- Permadi, R.; Suk, V.R.E.; Misran, M. Synthesis and Characterization of acylated low molecular weight chitosan and acylated low molecular weight phthaloyl chitosan. Sains Malaysiana 2020, 49, 2251–2260, doi:10.17576/jsm-2020-4909-22.
- Mohamed, N.A.; Abd El-Ghany, N.A.; Abdel-Aziz, M.M. Synthesis, characterization, anti-inflammatory and anti-Helicobacter pylori activities of novel benzophenone tetracarboxylimide benzoyl thiourea cross-linked chitosan hydrogels. J. Biol. Macromol. 2021, 181, 956–965, doi:10.1016/j.ijbiomac.2021.04.095.
- Hanafy, A.F.; Abdalla, A.M.; Guda, T.K.; Gabr, K.E.; Royall, P.G.; Alqurshi, A. Ocular anti-inflammatory activity of prednisolone acetate loaded chitosan-deoxycholate self-assembled nanoparticles. Int. J. Nanomed. 2019, 14, 3679–3689, doi:10.2147/ijn.s195892.
- Li, J.; Jin, X.; Yang, Y.; Zhang, L.; Liu, R.; Li, Z. Trimethyl chitosan nanoparticles for ocular baicalein delivery: Preparation, optimization, in vitro evaluation, in vivo pharmacokinetic study and molecular dynamics simulation. J. Biol. Macromol. 2020, 156, 749–761, doi:10.1016/j.ijbiomac.2020.04.115.
- Ravindran, R.; Mitra, K.; Arumugam, S.K.; Doble, M. Preparation of Curdlan sulphate—Chitosan nanoparticles as a drug carrier to target Mycobacterium smegmatis infected macrophages. Polym. 2021, 258, 117686, doi:10.1016/j.carbpol.2021.117686.
- Ravi Kumar, M.N.V.; Muzzarelli, R.A.A.; Muzzarelli, C.; Sashiwa, H.; Domb, A.J. Chitosan chemistry and pharmaceutical perspectives. Rev. 2004, 104, 6017–6084, doi:10.1021/cr030441b.
- Palacio, J.; Monsalve, Y.; Ramírez-Rodríguez, F.; López, B. Study of encapsulation of polyphenols on succinyl-chitosan nanoparticles. Drug Deliv. Sci. Technol. 2020, 57, 101610, doi:10.1016/j.jddst.2020.101610.
- Han, G.; Liu, S.; Pan, Z.; Lin, Y.; Ding, S.; Li, L.; Luo, B.; Jiao, Y.; Zhou, C. Sulfonated chitosan and phosphorylated chitosan coated polylactide T membrane by polydopamine-assisting for the growth and osteogenic differentiation of MC3T3-E1s. Polym. 2020, 229, 115517, doi:10.1016/j.carbpol.2019.115517.
- Zheng, Z.; Zhang, W.; Sun, W.; Li, X.; Duan, J.; Cui, J.; Feng, Z.; Mansour, H. Influence of the carboxymethyl chitosan anti-adhesion solution on the TGF-β1 in a postoperative peritoneal adhesion rat. Mater. Sci. Mater. Med. 2013, 24, 2549–2559, doi:10.1007/s10856-013-4981-7.
- Liu, X.; He, Z.; Chen, Y.; Zhou, C.; Wang, C.; Liu, Y.; Feng, C.; Yang, Z.; Li, P. Dual drug delivery system of photothermal-sensitive carboxymethyl chitosan nanosphere for photothermal-chemotherapy. J. Biol. Macromol. 2020, 163, 156–166, doi:10.1016/j.ijbiomac.2020.06.202.
- Wang, C.; Zhang, Z.; Chen, B.; Gu, L.; Li, Y.; Yu, S. Design and evaluation of galactosylated chitosan/graphene oxide nanoparticles as a drug delivery system. Colloid Interface Sci. 2018, 516, 332–341, doi:10.1016/j.jcis.2018.01.073.
- Sharma, A.K.; Gupta, L.; Sahu, H.; Qayum, A.; Singh, S.K.; Nakhate, K.T.; Ajazuddin; Gupta, U. Chitosan engineered PAMAM dendrimers as nanoconstructs for the enhanced anti-cancer potential and improved in vivo brain pharmacokinetics of temozolomide. Res. 2018, 35, 9, doi:10.1007/s11095-017-2324-y.
- Evangelista, T.F.S.; Andrade, G.R.S.; Nascimento, K.N.S.; dos Santos, S.B.; Costa Santos, M.F.; D’Oca, C.R.M.; Estevam, C.S.; Gimenez, I.F.; Almeida, L.E. Supramolecular polyelectrolyte complexes based on cyclodextrin-grafted chitosan and carrageenan for controlled drug release. Polym. 2020, 245, 116592, doi:10.1016/j.carbpol.2020.116592.
- Yi, Y.; Wang, Y.; Liu, H. Preparation of new crosslinked chitosan with crown ether and their adsorption for silver ion for antibacterial activities. Polym. 2003, 53, 425–430.
- Guaresti, O.; Maiz–Fernández, S.; Palomares, T.; Alonso-Varona, A.; Eceiza, A.; Pérez-Álvarez, L.; Gabilondo, N. Dual charged folate labelled chitosan nanogels with enhanced mucoadhesion capacity for targeted drug delivery. Polym. J. 2020, 134, 109847, doi:10.1016/j.eurpolymj.2020.109847.
- Adhikari, H.S.; Yadav, P.N. Anticancer activity of chitosan, chitosan derivatives, and their mechanism of action. J. Biomater. 2018, 2018, 2952085, doi:10.1155/2018/2952085.
- Qin, Y.; Xing, R.; Liu, S.; Li, K.; Meng, X.; Li, R.; Cui, J.; Li, B.; Li, P. Novel thiosemicarbazone chitosan derivatives: Preparation, characterization, and antifungal activity. Polym. 2012, 87, 2664–2670, doi:10.1016/j.carbpol.2011.11.048.
- Hsu, C.V.; Hsieh, M.H.; Xiao, M.C.; Chou, Y.H.; Wang, T.H.; Chiang, W.H. pH-Responsive polymeric micelles self-assembled from benzoic-imine-containing alkyl-modified PEGylated chitosan for delivery of amphiphilic drugs. J. Biol. Macromol. 2020, 163, 1106–1116, doi:10.1016/j.ijbiomac.2020.07.110.
- Savin, C.L.; Popa, M.; Delaite, C.; Costuleanu, M.; Costin, S.; Peptu, C.A. Chitosan grafted-poly(ethylene glycol) methacrylate nanoparticles as carrier for controlled release of bevacizumab. Sci. Eng. C 2019, 98, 843–860, doi:10.1016/j.msec.2019.01.036.
- Yu, S.; Zhang, X.; Tan, G.; Tian, L.; Liu, D.; Liu, Y.; Yang, X.; Pan, W. A novel pH-induced thermosensitive hydrogel composed of carboxymethyl chitosan and poloxamer cross-linked by glutaraldehyde for ophthalmic drug delivery. Polym. 2017, 155, 208–217, doi:10.1016/j.carbpol.2016.08.073.
- Zhuang, S.; Zhang, Q.; Wang, J. Adsorption of Co2+ and Sr2+ from aqueous solution by chitosan grafted with EDTA. Mol. Liq. 2021, 325, 115197, doi:10.1016/j.molliq.2020.115197.
- Sreekumar, S.; Goycoolea, F.M.; Moerschbacher, B.M.; Rivera-Rodriguez, G.R. Parameters influencing the size of chitosan-TPP nano- and microparticles. Rep. 2018, 8, 4695, doi:10.1038/s41598-018-23064-4.
- Garg, U.; Chauhan, S.; Nagaich, U.; Jain, N. Current advances in chitosan nanoparticles based drug delivery and targeting. Pharm. Bull. 2019, 9, 195–204, doi:10.15171/apb.2019.023.
- Agnihotri, S.A.; Mallikarjuna, N.N.; Aminabhavi, T.M. Recent advances on chitosan-based micro- and nanoparticles in drug delivery. Control. Release 2004, 100, 5–28, doi:10.1016/j.jconrel.2004.08.010.
- Shoueir, K.R.; El-Desouky, N.; Rashad, M.M.; Ahmed, M.K.; Janowska, I.; El-Kemary, M. Chitosan based-nanoparticles and nanocapsules: Overview, physicochemical features, applications of a nanofibrous scaffold, and bioprinting. J. Biol. Macromol. 2021, 167, 1176–1197, doi:10.1016/j.ijbiomac.2020.11.072.
- Zhang, A.; Meng, K.; Liu, Y.; Pan, Y.; Qu, W.; Chen, D.; Xie, S. Absorption, distribution, metabolism, and excretion of nanocarriers in vivo and their influences. Colloid Interface Sci. 2020, 284, 102261, doi:10.1016/j.cis.2020.102261.
- Cheng, X.; Tian, X.; Wu, A.; Li, J.; Tian, J.; Chong, Y.; Chai, Z.; Zhau, Y.; Chen, C.; Ge, C. Protein corona influences cellular uptake of gold nanoparticles by phagocytic and nonphagocytic cells in a size-dependent manner. ACS Appl. Mater. Interfaces 2015, 37, 20568–20575, doi:1021/acsami.5b04290.
- Malhaire, H.; Gimel, J.; Roger, E.; Benoît, J.; Lagarce, F. How to design the surface of peptide-loaded nanoparticles for efficient oral bioavailability? Drug Deliv. Rev. 2016, 106, 320–336 ,doi:10.1016/j.addr.2016.03.011.
- Lee, J.H.; Sahu, A.; Jang, C.; Tae, G. The effect of ligand density on in vivo tumor targeting of nanographene oxide. Control. Release 2015, 209, 219–228, doi:10.1016/j.jconrel.2015.04.035.
- Zhao, Z.; Ukidve, A.; Krishnan, V.; Mitragotri, S. Effect of physicochemical and surface properties on in vivo fate of drug nanocarriers. Drug Deliv. Rev. 2019, 143, 3–21, doi:10.1016/j.addr.2019.01.002.
- Sharma, G.; Valenta, D.T.; Altman, Y.; Harvey, S.; Xie, H.; Mitragotri, S.; Smith, J.W. Polymer particle shape independently influences binding and internalization by macrophages. Control. Release 2010, 147, 408–412, doi:10.1016/j.jconrel.2010.07.116.
- Banerjee, A.; Qi, J.; Gogoi, R.; Wong, J.; Mitragotri, S. Role of nanoparticle size, shape and surface chemistry in oral drug delivery. Control. Release 2016, 238, 176–185, doi:10.1016/j.jconrel.2016.07.051.
- Elci, S.G.; Jiang, Y.; Yan, B.; Kim, S.T.; Saha, K.; Moyano, D.F.; Tonga, G.Y.; Jackson, L.C.; Rotello, V.M.; Vachet, R.W. Surface Charge Controls the Suborgan Biodistributions of Gold Nanoparticles. Nano 2016, 10, 5536–5542, doi:1021/acsnano.6b02086.
- Xiao, K.; Li, Y.; Luo, J.; Lee, J.S.; Xiao, W.; Gonik, A.M.; Agarwal, R.G.; Lam, K.S. The effect of surface charge on in vivo biodistribution of PEG-oligocholic acid based micellar nanoparticles. Biomaterials 2011, 32, 3435–3446, doi:10.1016/j.biomaterials.2011.01.021.
- Wu, L.; Liu, M.; Shan, W.; Zhu, X.; Li, L.; Zhang, Z.; Huymg, Y. Bioinspired butyrate-functionalized nanovehicles for targeted oral delivery of biomacromolecular drugs. Control. Release 2017, 262, 273–283, doi:10.1016/j.jconrel.2017.07.045.
- Castro, A.; Berois, N.; Malanga, A.; Ortega, C.; Oppezzo, P.; Pristch, O.; Mombrú, A.W.; Osinaga, E.; Pardo, E. Docetaxel in chitosan-based nanocapsules conjugated with an anti-Tn antigen mouse/human chimeric antibody as a promising targeting strategy of lung tumors. J. Biol. Macromol. 2021, 182, 806–814, doi:10.1016/j.ijbiomac.2021.04.054.
- Abid, S.; Hussain, T.; Nazir, A.; Zahir, A.; Khenoussi, N. A novel double-layered polymeric nanofiber-based dressing with controlled drug delivery for pain management in burn wounds. Bull. 2019, 76, 6387–6411, doi:10.1007/s00289-019-02727-w.
- Amiri, N.; Ajami, S.; Shahroodi, A.; Jannatabadi, N.; Darban, S.A.; Bazzaz, B.S.F.; Pishavar, E.; Kalalinia, F.; Movaffagh, J. Teicoplanin-loaded chitosan-PEO nanofibers for local antibiotic delivery and wound healing. J. Biol. Macromol. 2020, 162, 645–656, doi:10.1016/j.ijbiomac.2020.06.195.
- Quinones, J.P.; Peniche, H.; Peniche, C. Chitosan based self-assembled nanoparticles in drug delivery. Polymers 2018, 235, doi:10.3390/polym10030235.
- Roy, H.; Nayak, B.S.; Nandi, S. Chitosan anchored nanoparticles in current drug development utilizing computer-aided pharmacokinetic modeling: Case studies for target specific cancer treatment and future prospective. Pharm. Des. 2020, 26, 1666–1675, doi:10.2174/1381612826666200203121241.
- Hassani, A.; Hussain, S.A.; Abdullah, N.; Kmaruddin, S. Review on micro-encapsulation with Chitosan for pharmaceutical applications. MOJ Curr. Res. Rev. 2018, 1, 77–84, doi:10.15406/mojcrr.2018.01.00013.
- Sang, Z.; Qian, J.; Han, J.; Deng, X.; Shen, J.; Li, G.; Xie, Y. Comparison of three water-soluble polyphosphate tripolyphosphate, phytic acid, and sodium hexametaphosphate as crosslinking agents in chitosan nanoparticle formulation. Polym. 2020, 230, 115577, doi:10.1016/j.carbpol.2019.115577.
- Pan, C.; Qian, J.; Zhao, C.; Yang, H.; Zhao, X.; Guo, H. Study on the relationship between crosslinking degree and properties of TPP crosslinked chitosan nanoparticles. Polym. 2020, 241, 116349, doi:10.1016/j.carbpol.2020.116349.
- Echeverri-Cuartas, C.E.; Gartner, C.; Lapitsky, Y. PEGylation and folate conjugation effects on the stability of chitosan-tripolyphosphate nanoparticles. J. Biol. Macromol. 2020, 158, 1055–1062, doi:10.1016/j.ijbiomac.2020.04.118.
- Abdelgawad, A.M.; Hudson, S.M. Chitosan nanoparticles: Polyphosphates cross-linking and protein delivery properties. J. Biol. Macromol. 2019, 136, 133–142, doi:10.1016/j.ijbiomac.2019.06.062.
- Cai, Y.; Lapitsky, Y. Biomolecular uptake effects on chitosan/tripolyphosphate micro- and nanoparticle stability. Colloids Surf. B-Biointerfaces 2020, 193, 111081, doi:10.1016/j.colsurfb.2020.111081.
- Cai, Y.; Lapitsky, Y. Pitfalls in analyzing release from chitosan/tripolyphosphate micro- and nanoparticles. J. Pharm. Biopharm. 2019, 142, 204–215, doi:10.1016/j.ejpb.2019.06.020.
- Wu, D.; Zhu, L.; Li, Y.; Zhang, X.; Xu, S.; Yang, G.; Delair, T. Chitosan-Based colloidal polyelectrolyte complexes for drug delivery: A review. Polym. 2020, 238, 116126, doi:10.1016/j.carbpol.2020.116126.
- Boudoukhani, M.; Yahoum, M.M.; Lefnaoui, S.; Moulai-Mostefa, N.; Banhobre, M. Synthesis, characterization and evaluation of deacetylated xanthan derivatives as new excipients in the formulation of chitosan-based polyelectrolytes for the sustained release of tramadol. Saudi Pharm. J. 2019, 27, 1127–1137, doi:10.1016/j.jsps.2019.09.009.
- Liu, L.; Yang, H.; Lou, Y.; Wu, J.-Y.; Miao, J.; Lu, X.-Y.; Gao, J.-Q. Enhancement of oral bioavailability of salmon calcitonin through chitosan-modified, dual drug-loaded nanoparticles. J. Pharm. 2019, 557, 170–177, doi:10.1016/j.ijpharm.2018.12.053.
- Luesakul, U.; Puthong, S.; Sansanaphongpricha, K.; Muangsin, N. Quaternizedchitosan-Coated nanoemulsions: A novel platform for improving the stability, anti-inflammatory, anti-cancer and transdermal properties of Plai extract. Polym. 2020, 230, 115625, doi:10.1016/j.carbpol.2019.115625.
- Chaudhary, S.; Kumar, S.; Kumar, V.; Sharma, R. Chitosan nanoemulsions as advanced edible coatings for fruits and vegetables: Composition, fabrication and developments in last decade. J. Biol. Macromol. 2020, 152, 154–170, doi:10.1016/j.ijbiomac.2020.02.276.
- Shahab, M.S.; Rizwanullah, M.; Alshehri, S.; Imam, S.S. Optimization to development of chitosan decorated polycaprolactone nanoparticles for improved ocular delivery of dorzolamide: In vitro, ex vivo and toxicity assessments. J. Biol. Macromol. 2020, 163, 2392–2404, doi:10.1016/j.ijbiomac.2020.09.185.
- Krishna Sailaja, A.; Amareshwar, P. Preparation of bovine serum albumin loaded chitosan nanoparticles using reverse micelle method. J. Pharm. Biol. Chem. Sci. 2011, 2, 837–846.
- Zhang, K.; Xu, Y.; Lu, L.; Shi, C.; Huang, Y.; Mao, Z.; Duan, C.; Ren, X.; Guo, Y.; Huang, C. Hydrodynamic cavitation: A feasible approach to intensify the emulsion cross-linking process for chitosan nanoparticle synthesis. Sonochem. 2021, 74, 105551, doi:10.1016/j.ultsonch.2021.105551.
- Zhang, H.; Li, X.; Kang, H. Chitosan coatings incorporated with free or nano-encapsulated Paulownia Tomentosa essential oil to improve shelf-life of ready-to-cook pork chops. LWT Food Sci. Technol. 2019, 116, 108580, doi:10.1016/j.lwt.2019.108580.
- Riegger, B.R.; Bäurer, B.; Mirzayeva, A.; Tovar, G.E.M.; Bach, M. A systematic approach of chitosan nanoparticle preparation via emulsion crosslinking as potential adsorbent in wastewater treatment. Polym. 2018, 180, 46–54, doi:10.1016/j.carbpol.2017.10.002.
- Kumar, S.; Dilbaghi, N.; Saharan, R.; Bhanjana, G. Nanotechnology as emerging tool for enhancing solubility of poorly water-soluble drugs. BioNanoScience 2012, 4, 227–250, doi:10.1007/s12668-012-0060-7.
- Seyam, S.; Nordin, N.A.; Alfatama, M. Recent Progress of Chitosan and Chitosan Derivatives-Based Nanoparticles: Pharmaceutical Perspectives of Oral Insulin Delivery. Pharmaceuticals 2020, 13, 307, doi:10.3390/ph13100307.
- Essa, D.; Choonara, Y.E.; Kondiah, P.P.D.; Pillay, V. Comparative nanofabrication of PLGA-Chitosan-PEG systems employing microfluidics and emulsification solvent evaporation techniques. Polymers 2020, 12, 1882, doi:10.3390/polym12091882.
- Orellano, M.S.; Longo, G.S.; Porporatto, C.; Correa, N.M.; Falcone, R.D. Role of micellar interface in the synthesis of chitosan nanoparticles formulated by reverse micellar method. Colloid Surf. A Physicochem. Eng. Asp. 2020, 599, 124876, doi:10.1016/j.colsurfa.2020.124876.
- Haidar, M.K.; Demirbolat, G.M.; Timur, S.S.; Gürsoy, R.N.; Nemutlu, E.; Ulubayram, K.; Öner, L.; Eroğlu, H. Atorvastatin-Loaded nanosprayed chitosan nanoparticles for peripheral nerve injury. Bioinspired Biomimetic Nanobiomater. 2020, 9, 74–84, doi:10.1680/jbibn.19.00006.
- Lucas, J.; Ralaivao, M.; Estevinho, B.N.; Rocha, F. A new approach for the microencapsulation of curcumin by a spray drying method, in order to value food products. Powder Technol. 2020, 362, 428–435, doi:10.1016/j.powtec.2019.11.095.
- Ozturk, A.A.; Kıyan, H.T. Treatment of oxidative stress-induced pain and inflammation with dexketoprofen trometamol loaded different molecular weight chitosan nanoparticles: Formulation, characterization and anti-inflammatory activity by using in vivo HET-CAM assay. Res. 2020, 128, 103961, doi:10.1016/j.mvr.2019.103961.
- Peng, H.H.; Hong, D.X.; Guan, Y.X.; Yao, S.J.: Preparation of pH-responsive DOX-loaded chitosan nanoparticles using supercritical assisted atomization with an enhanced mixer. J. Pharm. 2019, 558, 82–90, doi:10.1016/j.ijpharm.2018.12.077.
- Peng, H.H.; Wang, Z.D.; Guan, Y.X.; Yao, S.J. Supercritical CO2 assisted preparation of chitosan-based nano-in-microparticles with potential for efficient pulmonary drug delivery. CO2 Util. 2021, 46, 101486, doi:10.1016/j.jcou.2021.101486.
- Jalvo, B.; Faraldos, M.; Bahamonde, A.; Rosal, R. Antibacterial surfaces prepared by electrospray coating of photocatalytic nanoparticles. Eng. J. 2018, 334, 1108–1118, doi:10.1016/j.cej.2017.11.057.
- Pawar, A.; Thakkar, S.; Misra, M. A Bird’s Eye view of nanoparticles prepared by electrospraying: Advancements in drug delivery field. Control. Release. 2018, 286, 179–200, doi:10.1016/j.jconrel.2018.07.036.
- Kurakula, M.; Naveen, N.R. Electrospraying: A facile technology unfolding the Chitosan based drug delivery and biomedical applications. Polym. J. 2021, 147, 110326, doi:10.1016/j.eurpolymj.2021.110326.
- Wang, Y.; Zhang, R.; Qin, W.; Dai, J.; Zhang, Q.; Lee, K.; Liu, Y. Physicochemical properties of gelatin films containing tea polyphenol-loaded chitosan nanoparticles generated by electrospray. Des. 2020, 185, 108277, doi:10.1016/j.matdes.2019.108277.
- Perera, U.M.S.P.; Rajapakse, N. Chitosan nanoparticles: Preparation, characterization, and applications. In Seafood Processing By-Products; Springer: New York, NY, USA, 2014; pp 371–387.
- Gao, Y.; Ma, Q.; Cao, J.; Wang, Y.; Yang, X.; Xu, Q.; Liang, Q.; Sun, Y. Recent advances in microfluidic-aided chitosan-based multifunctional materials for biomedical applications. J. Pharm. 2021, 600, 120465, doi:10.1016/j.ijpharm.2021.120465.
- Ma, Q.; Cao, J.; Gao, Y.; Han, S.; Liang, Y.; Zhang, T.; Wang, X.; Sun, Y. Microfluidic-mediated nano-drug delivery systems: From fundamentals to fabrication for advanced therapeutic applications. Nanoscale 2020, 12, 15512–15527, doi:10.1039/d0nr02397c.
- Zhu, C.Z.; Yao, R.Y.; Chen, Y.J.; Feng, M.R.; Ma, S.; Zhang, C.C. Self-assembly of fluorinated gradient copolymer in three-dimensional co-flow focusing microfluidic. Colloid Interface Sci. 2018, 526, 75–82, doi:10.1016/j.jcis.2018.04.076.
- Collins, D.J.; Neild, A.; de Mello, A.; Liu, A.Q.; Ai, Y. The Poisson distribution and beyond: Methods for microfluidic droplet production and single cell encapsulation. Lab Chip 2015, 15, 3439–3459, doi:10.1039/c5lc00614g.
- Gomez-Mascaraque, L.G.; Sanchez, G.; Lopez-Rubio, A. Impact of molecular weight on the formation of electrosprayed chitosan microcapsules as delivery vehicles for bioactive compounds. Polym. 2016, 150, 121–130, doi:10.1016/j.carbpol.2016.05.012.
- Lari, A.S.; Zahedi, P.; Ghourchian, H.; Khatibi, A. Microfluidic-based synthesized carboxymethyl chitosan nanoparticles containing metformin for diabetes therapy: In vitro and in vivo assessments. Polym. 2021, 261, 117889, doi:10.1016/j.carbpol.2021.117889.
- Farahani, M.; Moradikhah, F.; Shabani, I.; Soflou, R.K.; Seyedjafari, E. Microfluidic fabrication of berberine-loaded nanoparticles for cancer treatment applications. Drug Deliv. Sci. Technol. 2021, 61, 102134, doi:10.1016/j.jddst.2020.102134.
- Chisty, A.H.; Masud, R.A.; Hasan, M.M.; Khan, M.N.; Mallik, A.K.; Rahman, M.M. Chapter 3—PEGylated chitin and chitosan derivatives. In Handbook of Chitin and Chitosan; Elsevier: Amsterdam, The Netherlands, 2020; pp. 59–100.
- Malhotra, M.; Tomaro-Duchesneau, C.; Saha, S.; Prakash, S. Systemic siRNA Delivery via peptide-tagged polymeric nanoparticles, targeting PLK1 gene in a mouse xenograft model of colorectal Cancer. J. Biomater. 2013, 2013, 252531, doi:10.1155/2013/252531.
- Casey, S.L.; Wilson, L.D. Investigation of Chitosan-PVA composite films and their adsorption properties. Geosci. Environ. Prot. 2015, 3, 78–84, doi:10.4236/gep.2015.32013.
- Menazea, A.A.; Ismail, A.M.; Awwad, N.S.; Ibrahium, H.A. Physical characterization and antibacterial activity of PVA/Chitosan matrix doped by selenium nanoparticles prepared via one-pot laser ablation route. Mater. Res. Technol. 2020, 9, 9598–9606, doi:10.1016/j.jmrt.2020.06.077.
- Sohail, R.; Abbas, S.R. Evaluation of amygdalin-loaded alginate-chitosan nanoparticles as biocompatible drug delivery carriers for anticancerous efficacy. J. Biol. Macromol. 2020, 153, 36–45, doi:10.1016/j.ijbiomac.2020.02.191.
- Wang, F.; Li, J.; Tang, X.; Huang, K.; Chen, L. Polyelectrolyte three layer nanoparticles of chitosan/dextran sulfate/chitosan for dual drug delivery. Colloid Surf. B Biointerfaces 2020, 190, 110925, doi:10.1016/j.colsurfb.2020.110925.
- Hu, Q.; Hu, S.; Fleming, E.; Lee, J.Y.; Luo, Y. Chitosan-Caseinate-Dextran ternary complex nanoparticles for potential oral delivery of astaxanthin with significantly improved bioactivity. J. Biol. Macromol. 2020, 151, 747–756, doi:10.1016/j.ijbiomac.2020.02.170.
- Sabra, R.; Roberts, C.J.; Billa, N. Courier properties of modified citrus pectinate-chitosan nanoparticles in colon delivery of curcumin. Colloid Interface Sci. Commun. 2019, 32, 100192, doi:10.1016/j.colcom.2019.100192.
- Lopes, M.; Shrestha, N.; Correia, A.; Shahbazi, M.A.; Sarmento, B.; Hirvonen, J.; Veiga, F.; Seiça, R.; Ribeiro, A.; Santos, H.A. Dual chitosan/albumin-coated alginate/dextran sulfate nanoparticles for enhanced oral delivery of insulin. Control. Release 2016, 232, 29–41, doi:10.1016/j.jconrel.2016.04.012.
- Anirudhan, T.S.; Parvathy, J. Novel Thiolated Chitosan-Polyethyleneglycol blend/Montmorillonite composite formulations for the oral delivery of insulin. Carbohydr. Diet. Fibre 2018, 16, 22–29, doi:10.1016/j.bcdf.2018.02.003.
- Li, J.; Tian, S.; Tao, Q.; Zhao, Y.; Gui, R.; Yang, F.; Zang, L.; Chen, Y.; Ping, Q.; Hou, D. Montmorillonite/Chitosan nanoparticles as a novel controlled-release topical ophthalmic delivery system for the treatment of glaucoma. J. Nanomed. 2018, 13, 3975–3987, doi:10.2147/IJN.S162306.
- Luo, C.; Yang, Q.; Lin, X.; Qi, C.; Li, G. Preparation and drug release property of tanshinone IIA loaded chitosan montmorillonite microspheres. J. Biol. Macromol. 2019, 125, 721–729, doi:10.1016/j.ijbiomac.2018.12.072.
- Hou, Y.T.; Wu, K.C.E.; Lee, C.Y. Development of glycyrrhizin-conjugated, chitosan-coated, lysine-embedded mesoporous silica nanoparticles for hepatocyte-targeted liver tissue regeneration. Materialia 2020, 9, 100568, doi:10.1016/j.mtla.2019.100568.
- Chen, C.; Yao, W.; Sun, W.; Guo, T.; Lv, H.; Wang, X.; Ying, H.; Wang, Y.; Wang, P. A self-targeting and controllable drug delivery system constituting mesoporous silica nanoparticles fabricated with a multi-stimuli responsive chitosan-based thin film layer. J. Biol. Macromol. 2019, 122, 1090–1099, doi:10.1016/j.ijbiomac.2018.09.058.
- Qiu, Q.; Quan, Z.; Zhang, H.; Qin, X.; Wang, R.; Yu, J. pH-triggered sustained drug release of multilayer encapsulation system with hollow mesoporous silica nanoparticles/chitosan/polyacrylic acid. Mater. Lett. 2020, 260, 126907, doi:10.1016/j.matlet.2019.126907.
- Liao, T.; Liu, C.; Ren, J.; Chen, H.; Kuang, Y.; Jiang, B.; Chen, J.; Sun, Z.; Li, C. A chitosan/mesoporous silica nanoparticle-based anticancer drug delivery system with a “tumor-triggered targeting” property. J. Biol. Macromol. 2021, 183, 2017–2029, doi:10.1016/j.ijbiomac.2021.06.004.
- Piosik, E.; Klimczak, P.; Ziegler-Borowska, M.; Chełminiak-Dudkiewicz, A. A detailed investigation on interactions between magnetite nanoparticles functionalized with aminated chitosan and a cell model membrane. Sci. Eng. C 2020, 109, 110616, doi:10.1016/j.msec.2019.110616.
- Salmanian, G.; Hassanzadeh-Tabrizi, S.A.; Koupaei, N. Magnetic chitosan nanocomposites for simultaneous hyperthermia and drug delivery applications: A review. J. Biol. Macromol. 2021, 184, 618–635; doi:10.1016/j.ijbiomac.2021.06.108.
- Araujo, H.C.; Gomes da Silva, A.C.; Paião, L.I.; Magario, M.K.W.; Frasnelli, F.C.T.; Oliveira, S.H.P.; Pessan, J.P.; Monteiro, D.R. Antimicrobial, antibiofilm and cytotoxic effects of a colloidal nanocarrier composed by chitosan-coated iron oxide nanoparticles loaded with chlorhexidine. Dent. 2020, 101, 103453, DOI:10.1016/j.jdent.2020.103453.
- Khmara, I.; Molcan, M.; Antosova, A.; Bednarikova, Z.; Zavisova, V.; Kubovcikova, K.; Jurikova, A.; Girman, V.; Baranovicova, E.; Koneracka, M.; et al. Bioactive properties of chitosan stabilized magnetic nanoparticles—Focus on hyperthermic and anti-amyloid activities. Magn. Magn. Mater. 2020, 513, 167056, doi:10.1016/j.jmmm.2020.167056.
- Bandeira, A.C.; de Oliveira Matos, A.; Evangelista, B.S.; da Silva, S.M.; Nagib, P.R.A.; de Moraes Crespo, A.; Amaral, A.C. Is it possible to track intracellular chitosan nanoparticles using magnetic nanoparticles as contrast agent? Med. Chem. 2019, 27, 2637–2643, doi:10.1016/j.bmc.2019.04.011.
- Anirudhan, T.S.; Sekhar, V.C.; Athira, V.S. Graphene oxide based functionalized chitosan polyelectrolyte nanocomposite for targeted and pH responsive drug delivery. J. Biol. Macromol. 2020, 150, 468–479, doi:10.1016/j.ijbiomac.2020.02.053.
- Rebekah, A.; Sivaselvam, S.; Viswanathan, C.; Prabhu, D.; Gautam, R.; Ponpandian, N. Magnetic nanoparticle-decorated graphene oxide-chitosan composite as an efficient nanocarrier for protein delivery. Colloid Surf. A Physicochem. Eng. Asp. 2021, 610, 125913, doi:10.1016/j.colsurfa.2020.125913.
- Baktash, M.S.; Zarrabi, A.; Avazverdi, E.; Reis, N.M. Development and optimization of a new hybrid chitosan-grafted graphene oxide/magnetic nanoparticle system for theranostic applications. Mol. Liq. 2021, 322, 114515, doi:10.1016/j.molliq.2020.114515.
- Khademi, Z.; Lavaee, P.; Ramezani, M.; Alibolandi, M.; Abnous, K.; Taghdisi, S.M. Co-Delivery of doxorubicin and aptamer against Forkhead box M1 using chitosan-gold nanoparticles coated with nucleolin aptamer for synergistic treatment of cancer cells. Polym. 2020, 248, 116735, doi:10.1016/j.carbpol.2020.116735.
- Salem, D.S.; Hegazy, S.F.; Obayya, S.S.A. Nanogold-loaded chitosan nanocomposites for pH/light-responsive drug release and synergistic chemo-photothermal cancer therapy. Colloid Interface Sci. Commun. 2021, 41, 100361, doi:10.1016/j.colcom.2021.100361.
- Saravanakumar, K.; Mariadoss, A.V.A.; Sathiyaseelan, A.; Wang, M.H. Synthesis and characterization of nano-chitosan capped gold nanoparticles with multifunctional bioactive properties. J. Biol. Macromol. 2020, 165, 747–757, doi:10.1016/j.ijbiomac.2020.09.177.
- López-Pérez, G.; Prado-Gotor, R.; Fuentes-Rojas, J.A.; Martin-Valero, M.J. Understanding gold nanoparticles interactions with chitosan: Crosslinking agents as novel strategy for direct covalent immobilization of biomolecules on metallic surfaces. Mol. Liq. 2020, 302, 112381, doi:10.1016/j.molliq.2019.112381.
- Wu, H.; Zhang, J. Chitosan-based zinc oxide nanoparticle for enhanced anticancer effect in cervical cancer: A physicochemical and biological perspective. Saudi Pharm. J. 2018, 26, 205–210, doi:10.1016/j.jsps.2017.12.010.
- Singh, T.A.; Das, J.; Sil, P.C. Zinc oxide nanoparticles: A comprehensive review on its synthesis, anticancer and drug delivery applications as well as health risks. Colloid Interface Sci. 2020, 286, 102317, doi:10.1016/j.cis.2020.102317.
- George, D.; Maheswari, P.U.; Begum, K.M.M.S. Cysteine conjugated chitosan based green nanohybrid hydrogel embedded with zinc oxide nanoparticles towards enhanced therapeutic potential of naringenin. Funct. Polym. 2020, 148, 104480, doi:10.1016/j.reactfunctpolym.2020.104480.
- Ghaffari, S.B.; Sarrafzadeh, M.H.; Salami, M.; Khorramizadeh, M.R. A pH-sensitive delivery system based on N-succinyl chitosan-ZnO nanoparticles for improving antibacterial and anticancer activities of curcumin. J. Biol. Macromol. 2020, 151, 428–440, doi:10.1016/j.ijbiomac.2020.02.141.
- Kandra, R.; Bajpai, S. Synthesis, mechanical properties of fluorescent carbon dots loaded nanocomposites chitosan film for wound healing and drug delivery. J. Chem. 2020, 13, 4882–4894, doi:10.1016/j.arabjc.2019.12.010.
- Lin, C.; Sun, K.; Zhang, C.; Tan, T.; Xu, M.; Liu, Y.; Xu, C.; Wang, Y.; Li, L.; Whittaker, A. Carbon dots embedded metal organic framework @ chitosan core-shell nanoparticles for vitro dual mode imaging and pH-responsive drug delivery. Microporous Mesoporous Mater. 2020, 293, 109775, doi:10.1016/j.micromeso.2019.109775.
- Sheng, Y.; Dai, W.; Gao, J.; Li, H.; Tan, W.; Wang, J.; Deng, L.; Kong, Y. pH-sensitive drug delivery based on chitosan wrapped graphene quantum dots with enhanced fluorescent stability. Sci. Eng. C 2020, 112, 110888, doi:10.1016/j.msec.2020.110888.
- Dong, X.; Wei, C.; Liang, J.; Liu, T.; Kong, D.; Lv, F. Thermosensitive hydrogel loaded with chitosan-carbon nanotubes for near infrared light triggered drug delivery. Colloid Surf. B Biointerfaces 2017, 154, 253–262, doi:10.1016/j.colsurfb.2017.03.036.
- Singh, R.P.; Sonali, G.S.; Singh, S.; Bharti, S.; Pandey, B.L.; Koch, B.; Muthu, M.S. Chitosan-Folate decorated carbon nanotubes for site specific lung cancer delivery. Sci. Eng. C 2017, 77, 446–458, doi:10.1016/j.msec.2017.03.225.
- Ghaffar, I.; Imran, M.; Perveen, S.; Kanwal, T.; Saifullah, S.; Bertino, M.F.; Ehrhardt, C.J.; Yadavalli, V.K.; Shah, M.R. Synthesis of chitosan coated metal organic frameworks (MOFs) for increasing vancomycin bactericidal potentials against resistant S. aureus Mater. Sci. Eng. C 2019, 105, 110111, doi:10.1016/j.msec.2019.110111.
- Li, L.; Han, S.; Zhao, S.; Li, X.; Liuab, B.; Liu, Y. Chitosan modified metal–organic frameworks as a promising carrier for oral drug delivery. RSC Adv. 2020, 10, 45130–45138, doi:10.1039/D0RA08459J.
- Xu, T.; Zhang, J.; Chi, H.; Cao, F. Multifunctional properties of organic-inorganic hybrid nanocomposites based on chitosan derivatives and layered double hydroxides for ocular drug delivery. Acta Biomaterialia 2016, 36, 152–163, doi:10.1016/j.actbio.2016.02.041.
- Wang, Y.; Zhou, L.; Fang, L.; Cao, F. Multifunctional carboxymethyl chitosan derivatives-layered double hydroxide hybrid nanocomposites for efficient drug delivery to the posterior segment of the eye. Acta Biomaterialia 2020, 104, 104–114, doi:10.1016/j.actbio.2020.01.008.
- Elanchezhiyan, S.S.; Meenakshi, S. Synthesis and characterization of chitosan/Mg-Al layered double hydroxide composite for the removal of oil particles from oil-in-water emulsion. J. Biol. Macromol. 2017, 104, 1586–1595, doi:10.1016/j.ijbiomac.2017.01.095.
- Sabourian, P.; Tavakolian, M.; Yazdani, H.; Frounchi, M.; van de Ven, T.G.M.; Maysinger, D.; Kakkar, A. Stimuli-responsive chitosan as an advantageous platform for efficient delivery of bioactive agents. J. Control. Release 2020, 317, 216–231, doi:10.1016/j.jconrel.2019.11.029.
- Constantin, M.; Bucatariu, S.M.; Doroftei, F.; Fundueanu, G. Smart composite materials based on chitosan microspheres embedded in thermosensitive hydrogel for controlled delivery of drugs. Polym. 2017, 157, 493–502, doi:10.1016/j.carbpol.2016.10.022.
- Liu, H.; Liu, J.; Xie, X.; Li, X. Development of photo-magnetic drug delivery system by facile-designed dual stimuli-responsive modifed biopolymeric chitosan capped nano-vesicle to improve efficiency in the anesthetic effect and its biological investigations. Photochem. Photobiol. B Biol. 2020, 202, 111716, doi:10.1016/j.jphotobiol.2019.111716.
- Daund, V.; Chalke, S.; Sherje, A.P.; Kale, P.P. ROS responsive mesoporous silica nanoparticles for smart drug delivery: A review. Drug Deliv. Sci. Technol. 2021, 64, 102599, doi:10.1016/j.jddst.2021.102599.
- Jiao, J.; Li, X.; Zhang, S.; Liu, J.; Di, D.; Zhang, Y.; Zhao, Q.; Wang, S. Redox and pH dual-responsive PEG and chitosan-conjugated hollow mesoporous silica for controlled drug release. Sci. Eng. C 2016, 67, 26–33, doi:10.1016/j.msec.2016.04.091.
- Lin, J.T.; Liu, Z.K.; Zhu, Q.L.; Rong, X.H. Liang, C.L.; Wang, J.; Ma, D.; Sun, J.; Wan, G.H. Redox-Responsive nanocarriers for drug and gene co-delivery based on chitosan derivatives modified mesoporous silica nanoparticles. Colloid Surf. B Biointerfaces 2017, 155, 41–50, doi:10.1016/j.colsurfb.2017.04.002.
- Rastegari, B.; Karbalaei-Heidari, H.R.; Zeinali, S.; Sheardown, H. The enzyme-sensitive release of prodigiosin grafted β-cyclodextrin and chitosan magnetic nanoparticles as an anticancer drug delivery system: Synthesis, characterization and cytotoxicity studies. Colloid Surf. Biointerfaces 2017, 158, 589–601, doi:10.1016/j.colsurfb.2017.07.044.
- Baghbani, F.; Chegeni, M.; Moztarzadeh, F.; Hadian-Ghazvini, S.; Raz, M. Novel ultrasound-responsive chitosan/perfluorohexane nanodroplets for image-guided smart delivery of an anticancer agent: Curcumin. Sci. Eng. C 2017, 74, 186-193, DOI:10.1016/j.msec.2016.11.107.
- Mathew, S.A.; Prakash, P.A.; Jaabir, M.S.M.; Dhanavel, S.; Manikandan, R.; Stephen, A. Dopamine-Conjugated CuS/chitosan nanocomposite for targeted photothermal drug delivery: In vitro cytotoxicity study to establish bio-compatibility. Drug Deliv. Sci. Technol 2021, 61, 102193, doi:10.1016/j.jddst.2020.102193.
- Bhatta, A.; Krishnamoorthy, G.; Marimuthu, N.; Dihingia, A.; Manna, P.; Biswal, H.T.; Das, M.; Krishnamoorthy, G. Chlorin e6 decorated doxorubicin encapsulated chitosan nanoparticles for photo-controlled cancer drug delivery. J. Biol. Macromol. 2019, 136, 951–961, doi:10.1016/j.ijbiomac.2019.06.127.
- Sudhakar, S.; Chandran, S.V.; Selvamurugan, N.; Nazeer, R.A. Biodistribution and pharmacokinetics of thiolated chitosan nanoparticles for oral delivery of insulin in vivo. J. Biol. Macromol. 2020, 150, 281–288, doi:10.1016/j.ijbiomac.2020.02.079.
- Mumuni, M.A.; Kenechukwu, F.C.; Ofokansi, K.C.; Attama, A.A.; Díaz Díaz, D. Insulin-loaded mucoadhesive nanoparticles based on mucin-chitosan complexes for oral delivery and diabetes treatment. Polym. 2020, 229, 115506, doi:10.1016/j.carbpol.2019.115506.
- Tsai, L.C.; Chen, C.H.; Lin, C.W.; Ho, Y.C.; Mi, F.L. Development of multifunctional nanoparticles self-assembled from trimethyl chitosan and fucoidan for enhanced oral delivery of insulin. J. Biol. Macromol. 2019, 126, 141–150, doi:10.1016/j.ijbiomac.2018.12.182.
- Wong, C.Y.; Al-Salami, H.; Dass, C.R. Formulation and characterisation of insulin-loaded chitosan nanoparticles capable of inducing glucose uptake in skeletal muscle cells in vitro. J. Drug Deliv. Sci. Technol. 2020, 57, 101738, doi:10.1016/j.jddst.2020.101738.
- Abdel-Moneim, A.; El-Shahawy, A.; Yousef, A.I.; Abd El-Twab, S.M.; Elden, Z.E.; Taha, M. Novel polydatin-loaded chitosan nanoparticles for safe and efficient type 2 diabetes therapy: In silico, in vitro and in vivo approaches. J. Biol. Macromol. 2020, 154, 1496–1504, doi:10.1016/j.ijbiomac.2019.11.031.
- Coutinho, A.J.; Costa Lima, S.A.; Afonso, C.M.M.; Reis, S. Mucoadhesive and pH responsive fucoidan-chitosan nanoparticles for the oral delivery of methotrexate. J. Biol. Macromol. 2020, 158, 180–188, doi:10.1016/j.ijbiomac.2020.04.233.
- Xavier, F.H., Jr.; Gueutin, C.; Chacun, H.; Vauthier, C.; Egito, E.S.T. Mucoadhesive paclitaxel-loaded chitosan-poly (isobutyl cyanoacrylate) core-shell nanocapsules containing copaiba oil designed for oral drug delivery. Drug Deliv. Sci. Technol. 2019, 53, 101194, doi:10.1016/j.jddst.2019.101194.
- Du, X.; Yin, S. Xu, L.; Ma, J.; Yu, H.; Wang, G.; Li, J. Polylysine and cysteine functionalized chitosan nanoparticle as an efficient platform for oral delivery of paclitaxel. Polym. 2020, 229, 115484, doi:10.1016/j.carbpol.2019.115484.
- Tran, P.H.L.; Wang, T.; Yang, C.; Tran, T.T.D.; Duan, W. Development of conjugate-by-conjugate structured nanoparticles for oral delivery of docetaxel. Sci. Eng. C 2020, 107, 110346, doi:mat10.1016/j.msec.2019.110346.
- Sharma, M.; Sharma, R.; Jain, D.K.; Saraf, A. Enhancement of oral bioavailability of poorly water soluble carvedilol by chitosan nanoparticles: Optimization and pharmacokinetic study. J. Biol. Macromol. 2019, 135, 246–260, doi:10.1016/j.ijbiomac.2019.05.162.
- Pauluk, D.; Padilha, A.K.; Khalil, N.M.; Mainardes, R.M. Chitosan-Coated zein nanoparticles for oral delivery of resveratrol: Formation, characterization, stability, mucoadhesive properties and antioxidant. Food Hydrocoll. 2019, 94, 411–417, doi:10.1016/j.foodhyd.2019.03.042.
- Lin, C.; Kuo, T.C.; Lin, J.C.; Ho, Y.C.; Mi, F.L. Delivery of polysaccharides from Ophiopogon japonicus (OJPs) using OJPs/chitosan/whey protein co-assembled nanoparticles to treat defective intestinal epithelial tight junction barrier. J. Biol. Macromol. 2020, 160, 558–570, doi:10.1016/j.ijbiomac.2020.05.151.
- Ling, K.; Wu, H.; Neish, A.S.; Champion, J.A. Alginate/Chitosan microparticles for gastric passage and intestinal release of therapeutic protein nanoparticles. Control. Release 2019, 29, 174–186, doi:10.1016/j.jconrel.2018.12.017.
- Muye, H.; Chen, Z.; Huibing, H.; Yu, J.; Yanzuo, C.; Kaiyan, L.; Feng. G. Cyclodextrin/chitosan nanoparticles for oral ovalbumin delivery: Preparation, characterization and intestinal mucosal immunity in mice. Asian J. Pharm. Sci. 2019, 14, 193–203, doi:10.1016/j.ajps.2018.04.001.
- Cole, H.; Bryan, D.; Lancaster, L.; Mawas, F.; Vllasaliu, D. Chitosan nanoparticle antigen uptake in epithelial monolayers can predict T mucosal but not systemic in vivo immune response by oral delivery. Polym. 2018, 190, 248–254, doi:10.1016/j.carbpol.2018.02.084.
- Saraf, S.; Jain, S.; Sahoo, R.N.; Mallick, S. Lipopolysaccharide derived alginate coated Hepatitis B antigen loaded chitosan nanoparticles for oral mucosal immunization. J. Biol. Macromol. 2020, 154, 466–476, doi:10.1016/j.ijbiomac.2020.03.124.
- Yu, X.; Wen, T.; Cao, P.; Shan, L.; Li, L. Alginate-chitosan coated layered double hydroxide nanocomposites for enhanced oral vaccine delivery. Colloid Interface Sci. 2019, 556, 258–265, doi:10.1016/j.jcis.2019.08.027.
- Dong, W.; Wang, X.; Liu, C.; Zhang, X.; Zhang, X.; Chen, X.; Kou, Y.; Mao, S. Chitosan based polymer-lipid hybrid nanoparticles for oral delivery of enoxaparin. J. Pharm. 2018, 547, 499–505, doi:10.1016/j.ijpharm.2018.05.076.
- Murthy, A.; Ravi, P.R.; Kathuria, H.; Vats, R. Self-Assembled lecithin-chitosan nanoparticles improve the oral bioavailability and alter the pharmacokinetics of raloxifene. J. Pharm. 2020, 588, 119731, doi:10.1016/j.ijpharm.2020.119731.
- Yu, A.; Shi, H.; Liu, H.; Bao, Z.; Dai, M.; Lin, D.; Lin, D.; Xu, X.; Li, X.; Wang, Y. Mucoadhesive dexamethasone-glycol chitosan nanoparticles for ophthalmic drug delivery. J. Pharm. 2020, 575, 118943, doi:10.1016/j.ijpharm.2019.118943.
- Khalil, M.; Hashmi, U.; Riaz, R.; Abbas, S.R. Chitosan coated liposomes (CCL) containing triamcinolone acetonide for sustained delivery: A potential topical treatment for posterior segment Diseases. J. Biol. Macromol. 2020, 143, 483–491, doi:10.1016/j.ijbiomac.2019.10.256.
- Ameeduzzafar; Imam, S.S.; Bukhari, S.N.A.; Ahmad, J.; Ali, A. Formulation and optimization of levofloxacin loaded chitosan nanoparticle for ocular delivery: In-vitro characterization, ocular tolerance and antibacterial activity. J. Biol. Macromol. 2018, 108, 650–659, doi:10.1016/j.ijbiomac.2017.11.170.
- Cheng, Y.H.; Ko, Y.C.; Chang, Y.F.; Huang, S.H.; Liu, C.J. Thermosensitive chitosan-gelatin-based hydrogel containing curcuminloaded nanoparticles and latanoprost as a dual-drug delivery system for glaucoma treatment. Eye Res. 2019, 179, 179–187, doi:10.1016/j.exer.2018.11.017.
- Abruzzo, A.; Cerchiara, T.; Bigucci, F.; Zuccheri, G.; Cavallari, C.; Saladini, B.; Luppi, B. Cromolyn-Crosslinked chitosan nanoparticles for the treatment of allergic Rhinitis. Eur. J. Pharm. Sci. 2019, 131, 136–145, doi:10.1016/j.ejps.2019.02.015.
- Sun, M.; Yu, X.; Wang, T.; Bi, S.; Liu, Y.; Chen, X. Nasal adaptive chitosan-based nano-vehicles for anti-allergic drug delivery. J. Biol. Macromol. 2019, 135, 1182–1192, doi:10.1016/j.ijbiomac.2019.05.188.
- Dumkliang, E.; Pamornpathomkul, B.; Patrojanasophon, P.; Ngawhirunpat, T.; Rojanarata, T.; Yoksan, S.; Opanasopit, P. Feasibility of chitosan-based nanoparticles approach for intranasal immunisation of live attenuated Japanese encephalitis vaccine. J. Biol. Macromol. 2021, 183, 1096–1105, doi:10.1016/j.ijbiomac.2021.05.050.
- Piazzini, V.; Landucci, E.; D’Ambrosio, M.; Fasiolo, L.T.; Cinci, L.; Colombo, G.; Pellegrini-Giampietro, D.E.; Bilia, A.R.; Luceri, C.; Bergonzi, M.C. Chitosan coated human serum albumin nanoparticles: A promising strategy for nose-to-brain drug delivery. J. Biol. Macromol. 2019, 129, 267–280, doi:10.1016/j.ijbiomac.2019.02.005.
- Chatzitaki, A.T.; Karavasili, S.J.C.; Andreadis, D.; Fatouros, D.G.; Borges, O. Chitosan-Coated PLGA nanoparticles for the nasal delivery of ropinirole hydrochloride: In vitro and ex vivo evaluation of efficacy and safety. J. Pharm. 2020, 589, 119776, doi:10.1016/j.ijpharm.2020.119776.
- Cassano, R.; Trapani, A.; Di Gioia, M.L.; Mandracchia, D.; Pellitteri, R.; Tripodo, G.; Trombino, S.; Di Gioia, S.; Conese, M. Synthesis and characterization of novel chitosan-dopamine or chitosantyrosine conjugates for potential nose-to-brain delivery. J. Pharm. 2020, 589, 119829; doi:10.1016/j.ijpharm.2020.119829.
- Sava, V.; Fihurka, O.; Khvorova, A.; Sanchez-Ramos, J. Enriched chitosan nanoparticles loaded with siRNA are effective in lowering Huntington’s disease gene expression following intranasal administration. Nanotechnol. Biol. Med. 2020, 24, 102119, doi:10.1016/j.nano.2019.102119.
- Khezri, F.A.N.Z.; Lakshmi, C.S.R.; Bukka, R.; Nidhi, M.; Nargund, S.L. Pharmacokinetic study and brain tissue analysis of Zolmitriptan loaded chitosan nanoparticles in rats by LC-MS method. J. Biol. Macromol. 2020, 142, 52–62, doi:10.1016/j.ijbiomac.2019.08.236.
- Bhattamisra, S.K.; Shak, A.T.; Xi, L.W.; Safian, N.H.; Choudhury, H.; Lim, W.M.; Shahzad, N.; Alhakamy, N.A.; Anwer, K.; Radhakrishnan, A.K.; et al. Nose to brain delivery of rotigotine loaded chitosan nanoparticles in human SH-SY5Y neuroblastoma cells and animal model of Parkinson’s disease. J. Pharm. 2020, 579, 119148, doi:10.1016/j.ijpharm.2020.119148.
- Wang, H.; Holgate, J.; Bartlett, S.; Islam, N. Assessment of nicotine release from nicotine-loaded chitosan nanoparticles dry powder inhaler formulations via locomotor activity of C57BL/6 mice. J. Pharm. Biopharm. 2020, 154, 175–185, doi:10.1016/j.ejpb.2020.07.011.
- Ahmad, N.; Ahmad, R.; Alrasheed, R.A.; Almatar, H.M.A.; Al-Ramadan, A.S.; Buheazah, T.M.; AlHomoud, H.S.; Al-Nasif, H.A.; Alam, A. A Chitosan-PLGA based catechin hydrate nanoparticles used in targeting of lungs and cancer treatment. Saudi J. Biol. Sci. 2020, 27, 2344–2357, doi:10.3390/pr9040621.
- Vieira, A.C.C.; Chaves, L.L.; Pinheiro, M.; Costa Lima, S.; Rolim Neto, P.J.; Ferreira, D.; Sarmento, B.; Reis, S. Lipid nanoparticles coated with chitosan using a one-step association method to target rifampicin to alveolar macrophages. Polym. 2021, 25, 116978, doi:10.1016/j.carbpol.2020.116978.
- Pardeshi, C.V.; Agnihotri, V.V.; Patil, K.Y.; Pardeshi, S.R.; Surana, S.J. Mannose-Anchored N,N,N-trimethyl chitosan nanoparticles for pulmonary administration of etofylline. J. Biol. Macromol. 2020, 165, 445–459, doi:10.1016/j.ijbiomac.2020.09.163.
- Dhayanandamoorthy, Y.; Antoniraj, M.G.; Kandregula, C.A.B.; Kandasamy, R. Aerosolized hyaluronic acid decorated, ferulic acid loaded chitosan nanoparticle: A promising asthma control strategy. J. Pharm. 2020, 591, 119958, doi:10.1016/j.ijpharm.2020.119958.
- Rahbarian, M.; Mortazavian, E.; Dorkoosh, F.A.; Tehrani, M.R. Preparation, evaluation and optimization of nanoparticles composed of thiolated triethyl chitosan: A potential approach for buccal delivery of Insulin. Drug Deliv. Sci. Technol. 2018, 44, 254–263, doi:10.1016/j.jddst.2017.12.016.
- Stie, M.B.; Gätke, J.R.; Wan, F.; Chronakis, J.S.; Jacobsen, J.; Nielsen, H.M. Swelling of mucoadhesive electrospun chitosan/polyethylene oxide nanofibers facilitates adhesion to the sublingual mucosa. Polym. 2020, 24, 116428, doi:10.1016/j.carbpol.2020.116428.
- Martin, V.; Ribeiro, I.A.C.; Alves, M.M.; Gonçalves, L.; Almeida, A.J.; Grenho, L.; Fernandes, M.H.; Santos, C.F.; Gomes, P.S.; Bettencourt, A.F. Understanding intracellular trafficking and anti-inflammatory effects of minocycline chitosan-nanoparticles in human gingival fibroblasts for periodontal disease treatment. J. Pharm. 2019, 572, 118821, doi:10.1016/j.ijpharm.2019.118821.
- Dos Santos, D.M.; Chagas, P.A.M.; Leite, I.S.; Inada, N.M.; de Annunzio, S.R.; Fontana, C.R.; Campana-Filho, S.P.; Correa, D.S. Core-sheath nanostructured chitosan-based nonwovens as a potential drug delivery system for periodontitis treatment. J. Biol. Macromol. 2020, 142, 521–534, doi:10.1016/j.ijbiomac.2019.09.124.
- Araujo, H.C.; da Silva, A.C.G.; Paiao, L.I.; Magario, M.K.W.; Frasnelli, S.C.T.; Oliveira, S.H.P.; Pessan, J.P.; Monteiro, D.R. Antimicrobial, antibiofilm and cytotoxic effects of a colloidal nanocarrier composed by chitosan-coated iron oxide nanoparticles loaded with chlorhexidine. Dent. 2020, 101, 103453, doi:10.1016/j.jdent.2020.103453.
- Abd-Allah, H.; Abdel-Aziz, R.T.A.; Nasr, M. Chitosan nanoparticles making their way to clinical practice: A feasibility study on their topical use for acne treatment. J. Biol. Macromol. 2020, 156, 262–270, doi:10.1016/j.ijbiomac.2020.04.040.
- Ushirobira, C.Y.; Afiune, L.A.F.; Pereira, M.N.; Cunha-Filho, M.; Gelfuso, G.M.; Gratieri, T. Dutasteride nanocapsules for hair follicle targeting: Effect of chitosan-coating and physical stimulus. J. Biol. Macromol. 2020, 151, 56–61, doi:10.1016/j.ijbiomac.2020.02.143.
- Takeuchi, I.; Suzuki, T.; Makino, K. Iontophoretic transdermal delivery using chitosan-coated PLGA nanoparticles for transcutaneous immunization. Colloid Surf. A Physicochem. Eng. Asp. 2020, 608, 125607, doi:10.1016/j.colsurfa.2020.125607.
- Morad, H.; Jahanshahi, M.; Akbari, J.; Saeedi, M.; Gill, P.; Enayatifard, R. Novel topical and transdermal delivery of colchicine with chitosan based biocomposite nanofiberous system; formulation, optimization, characterization, ex vivo skin deposition/permeation, and anti-melanoma evaluation. Chem. Phys. 2021, 263, 124381, doi:10.1016/j.matchemphys.2021.124381.
- Nawaz, A.; Wong, T.W. Microwave as skin permeation enhancer for transdermal drug delivery of chitosan-5-fluorouracil nanoparticles. Polym. 2017, 157, 906–919, doi:10.1016/j.carbpol.2016.09.080.
- Fahimirad, S.; Abtahi, H.; Satei, P.; Ghaznavi-Rad, E.; Moslehi, M.; Ganji, A. Wound healing performance of PCL/Chitosan based electrospun nanofiber electrosprayed with curcumin loaded chitosan nanoparticles. Polym. 2021, 259, 117640, doi:10.1016/j.carbpol.2021.117640.
- Shafique, M.; Sohail, M.; Minhas, M.U.; Khaliq, T.; Kousar, M.; Khan, S.; Hussain, Z.; Mahmood, A.; Abbasi, M.; Aziz, H.C.; et al. Bio-Functional hydrogel membranes loaded with chitosan nanoparticles for accelerated wound healing. J. Biol. Macromol. 2021, 170, 207–221, doi:10.1016/j.ijbiomac.2020.12.157.
- Manne, A.A.; Arigela, B.; Giduturi, A.K.; Komaravolu, R.K.; Mangamuri, U.; Poda, S. Pterocarpus marsupium Roxburgh heartwood extract/chitosan nanoparticles loaded hydrogel as an innovative wound healing agent in the diabetic rat model. Today Commun. 2021, 26, 101916, doi:10.1016/j.mtcomm.2020.101916.
- Amaral, A.C.; Saavedra, P.H.V.; Souza, A.C.O.; de Melo, M.T.; Tedesco, A.C.; Morais, P.C.; Felipe, M.S.S., Bocca, A.L. Miconazole loaded chitosan-based nanoparticles for local treatment of vulvovaginal candidiasis fungal infections. Colloids Surf. B Biointerfaces 2019, 174, 409–415, doi:10.1016/j.colsurfb.2018.11.048.
- Marciello, M.; Rossi, S.; Caramella, C.; Remunán-López, C. Freeze-Dried cylinders carrying chitosan nanoparticles for vaginal peptide delivery. Polym. 2017, 170, 43–51, doi:10.1016/j.carbpol.2017.04.051.
- Arumugam, G.; Rajendran, R. Callophycin A loaded chitosan and spicules based nanocomposites as an alternative strategy to overcome vaginal candidiasis. J. Biol. Macromol. 2020, 161, 656–665, doi:10.1016/j.ijbiomac.2020.06.119.
- Abdel Allah, N.H.; Gaber, Y.; Rashed, M.E.; Azmy, A.F.; Abou-Taleb, H.A.; Abdel Ghani, S. Alginate-Coated chitosan nanoparticles act as effective adjuvant for hepatitis A vaccine in mice. J. Biol. Macromol. 2020, 152, 904–912, doi:10.1016/j.ijbiomac.2020.02.287.
- Rebbouh, F.; Martin-Eauclaire, M.F.; Laraba-Djebari, F. Chitosan nanoparticles as a delivery platform for neurotoxin II from Androctonus australis hector scorpion venom: Assessment of toxicity and immunogenicity. Acta Tropica 2020, 205, 105353, doi:10.1016/j.actatropica.2020.105353.
- Abdelhamid, H.N.; Dowaidar, M.; Langel, U. Carbonized chitosan encapsulated hierarchical porous zeolitic imidazolate frameworks nanoparticles for gene delivery. Microporous Mesoporous Mater. 2020, 302, 110200, doi:10.1016/j.micromeso.2020.110200.
- Babii, O.; Wang, Z.; Liu, G.; Martinez, E.C.; van den Hurk, S.D.L.; Chen, L. Low molecular weight chitosan nanoparticles for CpG oligodeoxynucleotides delivery: Impact of molecular weight, degree of deacetylation, and mannosylation on intracellular uptake and cytokine induction. J. Biol. Macromol. 2020, 159, 46–56, doi:10.1016/j.ijbiomac.2020.05.048.
- Rahmani, S.; Hakimi, S.; Esmaeily, A.; Samadi, F.Y.; Mortazavian, E.; Nazari, M.; Mohammadi, Z.; Tehrani, N.R.; Tehrani, M.R. Novel chitosan based nanoparticles as gene delivery systems to cancerous and noncancerous cells. J. Pharm. 2019, 560, 306–314, doi:10.1016/j.ijpharm.2019.02.016.
- Mobarakeh, V.I.; Modarressi, M.H.; Rahimi, P.; Bolhassani, A.; Arefian, E.; Atyabi, F.; Vahabpour, R. Optimization of chitosan nanoparticles as an anti-HIV siRNA delivery vehicle. J. Biol. Macromol. 2019, 129, 305–315, doi:10.1016/j.ijbiomac.2019.02.036.
- Baghaei, M.; Tekie, F.S.M.; Khoshayand, M.R.; Varshochian, R.; Hajiramezanali, M.; Kachousangi, M.J.; Dinarvand, R.; Atyabiet, F. Optimization of chitosan-based polyelectrolyte nanoparticles for gene delivery, using design of experiment: In vitro and in vivo study, Sci. Eng. C 2021, 118, 111036, doi:10.1016/j.msec.2020.11103.
- Yan, L.; Gao, S.; Shui, S.; Liu, S.; Qu, H.; Liu, C.; Zheng, L. Small interfering RNA-loaded chitosan hydrochloride/carboxymethyl chitosan nanoparticles for ultrasound-triggered release to hamper colorectal cancer growth in vitro. J. Biol. Macromol. 2020, 162, 1303–1310, doi:10.1016/j.ijbiomac.2020.06.246.
- Tang, Y.; Liu, Y.; Xie, Y.; Chen, J.; Dou, Y. Apoptosis of A549 cells by small interfering RNA targeting survivin delivery using poly-β-amino ester/guanidinylated O-carboxymethyl chitosan nanoparticles. Asian J. Pharm. Sci. 2020, 15, 121–128, doi:10.1016/j.ajps.2018.09.009.
- Hajizadeh, F.; Ardebili, S.M.; Moornani, M.B.; Masjedi, A.; Atyabi, F.; Kiani, M.; Namdar, A.; Karpisheh, V.; Izadi, S.; Baradaran, B.; Azizi, G.; Ghalamfarsa, G.; Sabz, G.; Yousefi, M.; Jadidi-Niaragh, F. Silencing of HIF-1α/CD73 axis by siRNA-loaded TAT-chitosan-spion nanoparticles robustly blocks cancer cell progression. Eur. J. Pharmacol. 2020, 882, 173235, doi:10.1016/j.ejphar.2020.173235.
- Salimifard, S.; Kiani, F.K.; Eshaghi, F.S.; Izadi, S.; Shahdadnejad, K.; Masjedi, A.; Heydari, M.; Ahmadi, A.; Hojjat-Farsangi, M.; Hassannia, H.; et al. Codelivery of BV6 and anti-IL6 siRNA by hyaluronate-conjugated PEGchitosan-lactate nanoparticles inhibits tumor progression. Life Sci. 2020, 260, 118423, doi:1016/j.lfs.2020.118423.
- Nikkhoo, A.; Rostami, N.; Farhadi, S.; Esmaily, M.; Ardebili, S.M.; Atyabi, F.; Baghaei, M.; Haghnavaz, N.; Yousefi, M.; Aliparasti, M.R.; et al. Codelivery of STAT3 siRNA and BV6 by carboxymethyl dextran trimethyl chitosan nanoparticles suppresses cancer cell progression. J. Pharm. 2020, 581, 119236, doi:10.1016/j.ijpharm.2020.119236.
- Masjedi, A.; Ahmadi, A.; Atyabi, F.; Farhadi, S.; Irandoust, M.; Khazaei-Poul, Y.; Chaleshtari, M.G.; Fathabad, M.E.; Baghaei, M.; Haghnavaz, N.; et al. Silencing of IL-6 and STAT3 by siRNA loaded hyaluronate-N,N,N-trimethyl chitosan nanoparticles potently reduces cancer cell progression. J. Biol. Macromol. 2020, 149, 487–500, doi:10.1016/j.ijbiomac.2020.01.273.
- Yan, T.; Zhu, S.; Hui, W.; He, J.; Liu, Z.; Cheng, J. Chitosan based pH-responsive polymeric prodrug vector for enhanced tumor targeted co-delivery of doxorubicin and siRNA. Polym. 2020, 250, 116781, doi:10.1016/j.carbpol.2020.116781.
- Xue, Y.; Wang, N.; Zeng, Z.; Huang, J.; Xiang, Z.; Guan, J.Q. Neuroprotective effect of chitosan nanoparticle gene delivery system grafted with acteoside (ACT) in Parkinson’s disease models. Mater. Sci. Technol. 2020, 43, 197–207, doi:10.1016/j.jmst.2019.10.013.
- Lang, X.; Wang, T.; Sun, M.; Chen, X.; Liu, Y. Advances and applications of chitosan-based nanomaterials as oral delivery carriers. J. Biol. Macromol. 2020, 154, 433–445, doi:10.1016/j.ijbiomac.2020.03.148.
- Alhajj, N.; Zakaria, Z.; Naharudin, I.; Ahsan, F.; Li, W.; Wong, T.W. Critical physicochemical attributes of chitosan nanoparticles admixed lactose-PEG 3000 microparticles in pulmonary inhalation. Asian J. Pharm. Sci. 2020, 15, 374–384, doi:10.1016/j.ajps.2019.02.001.
- Macedo, A.S.; Castro, P.M.; Roque, L.; Thome, N.G.; Reis, C.P.; Pintado, M.E.; Fonte, P. Novel and revisited approaches in nanoparticle systems for buccal drug delivery. Control. Release 2020, 320, 125–141, doi:10.1016/j.jconrel.2020.01.006.
- Matos, B.N.; Pereira, M.N.; Bravo, M.O.; Cunha-Filho, M.; Saldanha-Araújo, F.; Gratieri, T.; Gelfuso, G. Chitosan nanoparticles loading oxaliplatin as a mucoadhesive topical treatment of oral tumors: Iontophoresis further enhances drug delivery ex vivo. J. Biol. Macromol. 2020, 154, 1265–1275, doi:10.1016/j.ijbiomac.2019.11.001.
- Pornpitchanarong, C.; Rojanarata, T.; Opanasopit, P.; Ngawhirunpat, T.; Patrojanasophon, P. Catechol-modified chitosan/hyaluronic acid nanoparticles as a new avenue for local delivery of doxorubicin to oral cancer cells. Colloids Surf. BBiointerfaces 2020, 196, 111279, doi:10.1016/j.colsurfb.2020.111279.
- Sah, A.K.; Dewangan, M.; Suresh, P.K. Potential of chitosan-based carrier for periodontal drug delivery. Colloids Surf. B Biointerfaces 2019, 178, 185–198, doi:10.1016/j.colsurfb.2019.02.044.
- Nair, S.S. Chitosan-based transdermal drug delivery systems to overcome skin barrier functions. Drug Deliv. Ther. 2019, 9, 266–270, doi:10.22270/jddt.v9i1.2180.
- Park, J.; Ramanathan, R.; Pham, L.; Kim, B.S.; Woodrow, A. Chitosan enhances nanoparticle delivery from the reproductive tract to target draining lymphoid organs. Nanotechnol. Biol. Med. 2017, 13, 2015–2025, doi:10.1016/j.nano.2017.04.012.
This entry is adapted from the peer-reviewed paper 10.3390/ijms22179652
