The term “smart window” was coined by Granqvist in 1985. From the early 1980s, smart glazing has been a rapidly developing innovative technology that is aimed to help manage energy transfer through the building’s envelope, evading unnecessary “cooling and heating of indoor air”. The use of smart glass, which helps to regulate the amount of light (and heat) entering a building, is one of the possible ways to reduce energy consumption in buildings while maintaining an appropriate level of comfort for users. Smart glass greatly influences the building envelope performance in (i) thermal management, (ii) daylight harvesting and regulation, (iii) reduction of glare, (iv) maintenance of views, (v) power capture, and finally (vi) activating the envelope as an information display. Some technologies are currently available on the market, although—in light of the many shortcomings of the existing solutions—smart glass is the subject of ongoing “intensive research aimed at improving the technology and its widespread use”.
- smart glass
- smart window
- electrochromic
1. General Classification
2. Passive Technologies
| Type | Stimulus | Technology | Featured Systems |
|---|---|---|---|
| Passive technologies: | Heat—Thermochromic | ||
| Light—Photochromic | |||
| Heat—Phase Change Materials | |||
| Active technologies: | Gas—Gasochromic | ||
| Fluid—Optofluidic glass | |||
| Electrical current: | Microsystems | ||
| Microwrinkled Nanometric Films | |||
| Polymer dispersed liquid crystal | |||
| Suspended particle devices (SPD) | |||
| Electrochromic: | Multicolour EC | ||
| Neutral black electrochromism | |||
| Spectrally selective systems NIR/VIS | |||
| Electrochromic energy storage window | |||
| Hybrid EC/TC solutions | |||
| EC devices powered by solar cells | |||
| Nanostructures |
3. Active Technologies
3.1. Gas
3.2. Fluid
3.3. Electrical Current
3.3.1. MEMS-Based Microsystems
3.3.2. Microwrinkled Nanometric Films
3.3.3. PDLC (Polymer Dispersed Liquid Crystal)
3.3.4. SPD Windows
| No. | Team | Year | Type | ΔTsol | ΔTvis | Remarks |
|---|---|---|---|---|---|---|
| 1 | Wittwer et al. | 2004 | Active gasochromic | 71% | 72% | Switching from transparent to mirror state |
| 2 | Liang et al. | 2019 | Active gasochromic | 42% | n/a | |
| 3 | Wolfe et al. | 2018 | Optofluidic | n/a | 77% | Clear to foggy |
| 4 | Heiz et al. | 2017 | Magneto-Active Liquid | 95% | n/a | Magnetic particles in liquid |
| 5 | Hillmer et al. | 2018 | Microelectromechanical | n/a | n/a | Micromirrors. The team only measured a temperature build-up in the room. |
| 6 | Mori et al. | 2016 | Electrostatic | n/a | 17% | Micro blinds |
| 7 | Shrestha et al. | 2018 | Microwrinkled TiO2 Films | n/a | 79.2% haze | Transparent to translucent switching |
| 8 | Lampert | 1998 | PDLC | n/a | 40% haze | |
| 9 | Lampert | 2004 | PDLC | 60% | 57% haze | |
| 10 | Murray et al. | 2016 | PDLC | n/a | 25–29% haze | |
| 11 | De Filpo et al. | 2019 | PDLC | n/a | 64% haze | |
| 12 | Sol et al. | 2017 | PDLC | n/a | 61% haze | |
| 13 | Ghosh | 2017 | SPD | 46% | n/a |
3.3.5. ECDs
3.3.5.1 Switching Mechanism
Electrochromism is a reversible chemical phenomenon, where the electrochromic material changes its colour when the voltage is applied. As Kraft writes, the substances, “which change from an uncoloured oxidised state to a coloured reduced state by electrochemical reduction are called cathodic electrochromic, whereas compounds which change from an uncoloured reduced to a coloured oxidised state are called anodic electrochromic compounds” [17]. EC windows operate on the principle of the reversible electrochemical intercalation of positive ions (e.g., H+, Li+, Na+) accompanying the insertion of charge balancing electrons into the multivalent transition metal oxides (e.g., WO3, NiO, IrO, MoO3, V2O5) [27]. The basic chemical reaction featuring the most popular cathodic electrochromic compound WO3 transforming from transparent to blue is given below:
|
MxWO3 ⇄ WO3 + xe− + xM+ x ≤ 0.3 |
(4) |
while nickel oxide (NiO) can be coloured anodically to a brown colour in a reaction of
|
LiNiO ⇄ NiO + Li+ + e− |
(5) |
However, other transition metal oxides such as Co3O4, MoO3, V2O5, TiO2 also exhibit electrochromic properties [52,53]. Prussian blue (iron ferrocyanide) – originally reported by Mortimer [54] – also currently is studied as a material presenting some electrochromic behaviour [55]. Recently, polystyrene sulfonate (PEDOT:PSS) were researched as exhibiting electrochromic properties, as well as presented by Singh [51].
Electrochromic devices typically consist of five thin layers that are located (sandwiched) between two panes of glass or flexible polyester foil: two external layers of transparent conductive films (usually indium tin oxide, ITO) and the counter electrode (Ni-oxide-based film), electrolyte, and electrochromic electrode in between (W-oxide-based film). The counter electrode is used for ion storage, the electrolyte for conducting ions, and the electrochromic electrode for attracting ions. When the electrical current is applied, the ions stored in the counter electrode (bleached state) migrate through the electrolyte to the electrochromic electrode, resulting in the colouration (coloured state). The mobile ions should be small. Hydrogen protons (H+) or lithium (Li+) ions are commonly used [18]. EC device can be considered as an electrical battery in which the optical absorption is related to its charge; therefore, the crucial component of an ECD is an electrolyte, which can be liquid, gel, or solid, as addressed by Cannavale et al [56]. The most popular ECD architecture is pictured in Figure 1.
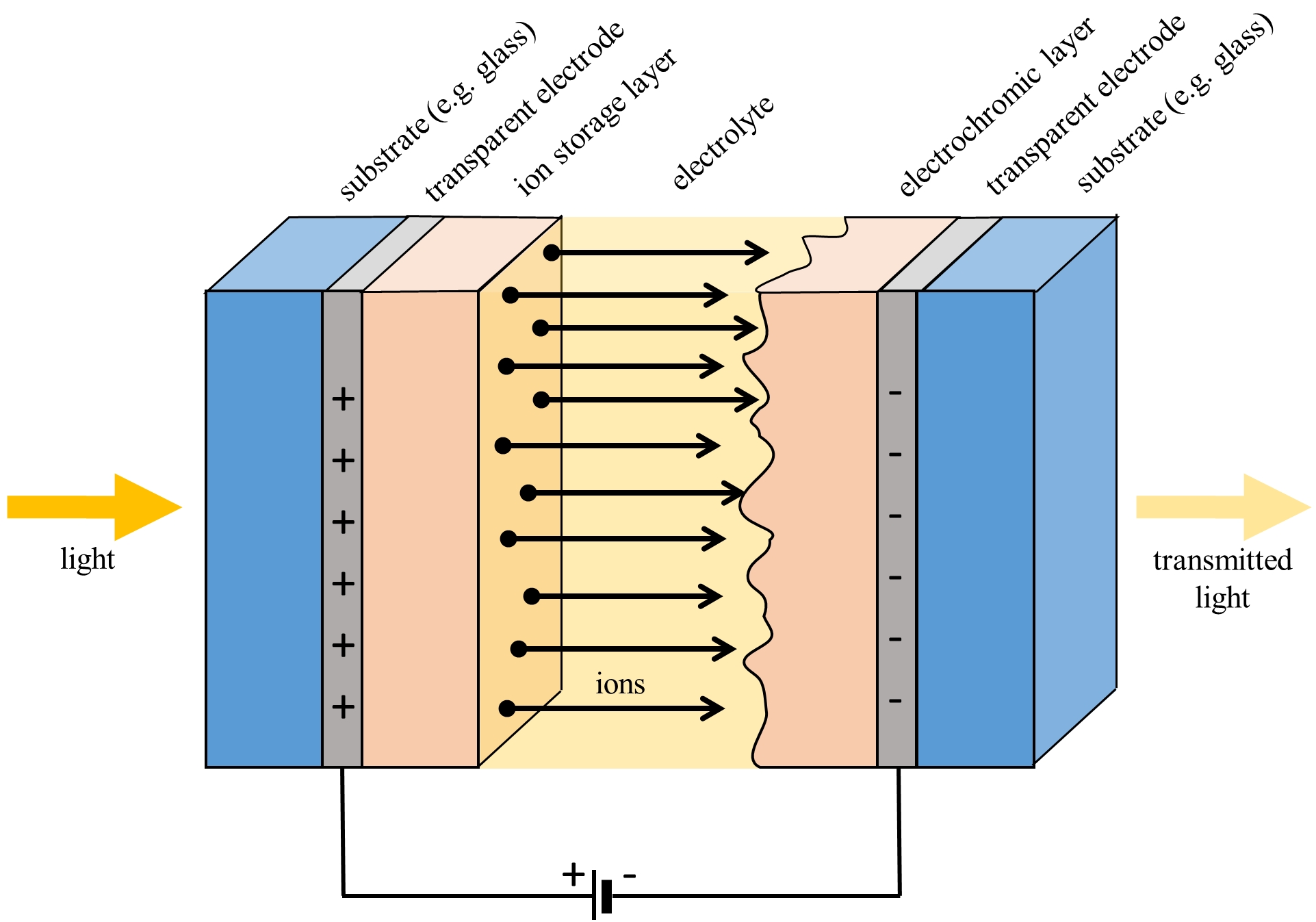
Figure 1. The most popular ECD architecture. Diagram by the author.
3.3.5.3 Most Recent Concepts in EC Smart Windows
As is said above, EC technologies suffer from many problems that are currently being addressed by many research teams tackling the challenges of the EC windows. The concepts of the most recent solutions are Multicolour EC, Neutral black electrochromism, Spectrally selective systems NIR/VIS, Electrochromic energy storage window, Hybrid EC/TC solutions, EC devices powered by solar cells, the application of nanostructures in ECw.
For details please see: https://www.mdpi.com/2071-1050/13/17/9604/htm#B8-sustainability-13-09604
This entry is adapted from the peer-reviewed paper 10.3390/su13179604
References
- Brzezicki, M. Regulated transparency: A brief review of the existing and the most promising technologies. In Engineered Transparency 2016: Glass in Architecture and Structural Engineering; Jens, S., Bernhard, W., Eds.; Wilhelm Ernst & Sohn: Berlin, Germany, 2016; pp. 11–19.
- Nguyen, T.D.; Yeo, L.P.; Kei, T.C.; Mandler, D.; Magdassi, S.; Tok, A.I.Y. Efficient Near Infrared Modulation with High Visible Transparency Using SnO2–WO3 Nanostructure for Advanced Smart Windows. Adv. Opt. Mater. 2019, 7, 1801389.
- Jelle, B.P. Solar radiation glazing factors for window panes, glass structures and electrochromic windows in buildings—Measurement and calculation. Sol. Energy Mater. Sol. Cells 2013, 116, 291–323.
- Park, B.R.; Hong, J.; Choi, E.J.; Choi, Y.J.; Lee, C.; Moon, J.W. Improvement in Energy Performance of Building Envelope Incorporating Electrochromic Windows (ECWs). Energies 2019, 12, 1181.
- Granqvist, C.G.; Niklasson, G.A. Thermochromic Oxide-Based Thin Films and Nanoparticle Composites for Energy-Efficient Glazings. Buildings 2016, 7, 3.
- Lee, S.J.; Choi, D.S.; Kang, S.H.; Yang, W.S.; Nahm, S.; Han, S.H.; Kim, T. VO2/WO3-Based Hybrid Smart Windows with Thermochromic and Electrochromic Properties. ACS Sustain. Chem. Eng. 2019, 7, 7111–7117.
- La, T.-G.; Li, X.; Kumar, A.; Fu, Y.; Yang, S.; Chung, H.-J. Highly Flexible, Multipixelated Thermosensitive Smart Windows Made of Tough Hydrogels. ACS Appl. Mater. Interfaces 2017, 9, 33100–33106.
- Timmermans, G.; Saes, B.W.H.; Debije, M.G. Dual-responsive “smart” window and visually attractive coating based on a diarylethene photochromic dye. Appl. Opt. 2019, 58, 9823–9828.
- Li, R.; Zhou, Y.; Shao, Z.; Zhao, S.; Chang, T.; Huang, A.; Li, N.; Ji, S.; Jin, P. Enhanced Coloration/Bleaching Photochromic Performance of WO3 Based on PVP/PU Composite Matrix. ChemistrySelect 2019, 4, 9817–9821.
- Chou, H.-T.; Chen, Y.-C.; Lee, C.-Y.; Chang, H.-Y.; Tai, N.-H. Switchable transparency of dual-controlled smart glass prepared with hydrogel-containing graphene oxide for energy efficiency. Sol. Energy Mater. Sol. Cells 2017, 166, 45–51.
- Kim, D.; Yoon, J. Flexible Adaptive Solar Control Smart-films Comprising Thermo-responsive Hydrogels with Silver Naopatterned Substrates. Polym. Korea 2019, 43, 144–150.
- Wittwer, V.; Datz, M.; Ell, J.; Georg, A.; Graf, W.; Walze, G. Gasochromic windows. Sol. Energy Mater. Sol. Cells 2004, 84, 305–314.
- Liang, R.; Liu, D.; Sun, Y.; Luo, X.; Grant, D.; Walker, G.; Wu, Y. Investigation of Mg-Y coated gasochromic smart windows for building applications. Build. Simul. 2018, 12, 99–112.
- Wolfe, D.; Goossen, K.W. Evaluation of 3D printed optofluidic smart glass prototypes. Opt. Express 2017, 26, A85–A98.
- Heiz, B.P.V.; Pan, Z.; Su, L.; Le, S.T.; Wondraczek, L. A Large-Area Smart Window with Tunable Shading and Solar-Thermal Harvesting Ability Based on Remote Switching of a Magneto-Active Liquid. Adv. Sustain. Syst. 2017, 2, 1700140.
- Lamontagne, B.; Fong, N.R.; Song, I.-H.; Ma, P.; Barrios, P.; Poitras, D. Review of microshutters for switchable glass. J. Micro/Nanolithogr. MEMS MOEMS 2019, 18, 040901.
- Hillmer, H.; Al-Qargholi, B.; Khan, M.M.; Worapattrakul, N.; Wilke, H.; Woidt, C.; Tatzel, A. Optical MEMS-based micromirror arrays for active light steering in smart windows. Jpn. J. Appl. Phys. 2018, 57, 08PA07.
- Mori, K.; Misawa, K.; Ihida, S.; Takahashi, T.; Fujita, H.; Toshiyoshi, H. A MEMS Electrostatic Roll-Up Window Shade Array for House Energy Management System. IEEE Photon. Technol. Lett. 2016, 28, 593–596.
- Hillmer, H.; Al-Qargholi, B.; Khan, M.M.; Iskhandar, M.S.Q.; Wilke, H.; Tatzel, A. Optical MEMS based micromirror arrays: Fabrication, characterization and potential applications in smart active windows. In Proceedings of the 2019 International Conference on Optical MEMS and Nanophotonics (OMN), Daejeon, Korea, 28 July–1 August 2019; pp. 188–189.
- Shrestha, M.; Asundi, A.; Lau, G.-K. Smart Window Based on Electric Unfolding of Microwrinkled TiO2 Nanometric Films. ACS Photon. 2018, 5, 3255–3262.
- Lampert, C. Smart switchable glazing for solar energy and daylight control. Sol. Energy Mater. Sol. Cells 1998, 52, 207–221.
- Lampert, C.M. Chromogenic smart materials. Mater. Today 2004, 7, 28–35.
- Murray, J.; Ma, D.; Munday, J.N. Electrically Controllable Light Trapping for Self-Powered Switchable Solar Windows. ACS Photon. 2016, 4, 1–7.
- Available online: https://www.saint-gobain-glass.com/products/priva-lite (accessed on 20 August 2021).
- De Filpo, G.; Armentano, K.; Pantuso, E.; Mashin, A.I.; Chidichimo, G.; Nicoletta, F.P. Polymer Membranes Dispersed Liquid Crystal (PMDLC): A new electro-optical device. Liq. Cryst. 2019, 46, 986–993.
- Kim, M.; Park, K.J.; Seok, S.; Ok, J.M.; Jung, H.-T.; Choe, J.; Kim, D.H. Fabrication of Microcapsules for Dye-Doped Polymer-Dispersed Liquid Crystal-Based Smart Windows. ACS Appl. Mater. Interfaces 2015, 7, 17904–17909.
- Alghamdi, H.; Almawgani, A. Smart and Efficient Energy Saving System Using PDLC Glass. In Proceedings of the 2019 Smart City Symposium Prague (SCSP), Prague, Czech Republic, 23–24 May 2019; pp. 1–5.
- Sol, J.; Timmermans, G.; Van Breugel, A.J.; Schenning, A.P.H.J.; Debije, M.G. Multistate Luminescent Solar Concentrator “Smart” Windows. Adv. Energy Mater. 2018, 8, 1702922.
- Schwartz, M. (Ed.) Smart Materials; CRC Press: Boca Raton, FL, USA, 2009.
- Ghosh, A.; Norton, B. Durability of switching behaviour after outdoor exposure for a suspended particle device switchable glazing. Sol. Energy Mater. Sol. Cells 2017, 163, 178–184.
- Deb, S.K. Reminiscences on the discovery of electrochromic phenomena in transition metal oxides. Sol. Energy Mater. Sol. Cells 1995, 39, 191–201.
- Singh, R.; Tharion, J.; Murugan, S.; Kumar, A. ITO-Free Solution-Processed Flexible Electrochromic Devices Based on PEDOT:PSS as Transparent Conducting Electrode. ACS Appl. Mater. Interfaces 2016, 9, 19427–19435.
