3. Oxygen Sensing at the Epithelial Barrier in IBD
Oxygen homeostasis in healthy intestines is assured by dynamic and rapid fluctuation in cellular oxygen tension. In inflammatory bowel disease (IBD), this cellular oxygen tension is dysregulated [
40]. IBDs are chronic, immunologically mediated diseases of the gastrointestinal tract. The current understanding of IBD pathogenesis assumes that in genetically predisposed individuals, an improperly regulated interaction between the microbiota and the mucosal immune system leads to an inappropriate inflammatory response and that disease recurrence is triggered by environmental factors that remain to be fully characterized (
Figure 3) [
85]. The sensors that detect fluctuating oxygen levels appear in normal and disease states. They are an important link between metabolism and chromatin regulation; therefore, it is warranted to further address how oxygen-regulated metabolic adaptations affect the epigenome and how this impacts the function of cells [
86].
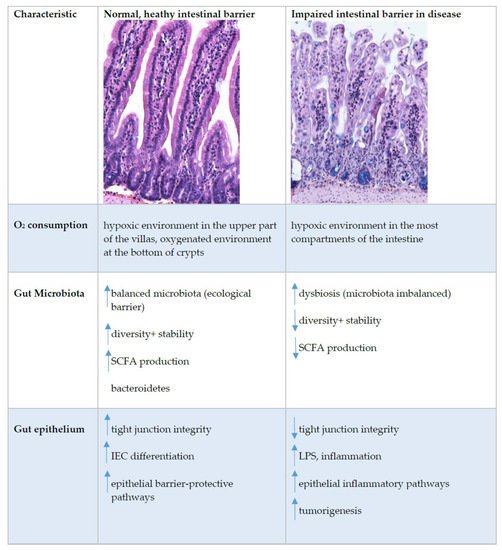
Figure 3. Intestinal epithelial barrier in health and disease. This figure shows immunohistochemistry picture of small intestine crypts–villus axis in healthy and impaired intestine in BALB/cOlaHsd mice and highlight the main characteristics where the differences at the healthy or impaired (in disease) intestinal barrier were detected. Up and down arrows indicate an increase and decrease, respectively. Abbreviations: IEC, intestinal epithelial cells; LPS, lipopolysaccharide; SCFA, short-chain fatty acids; TJ, tight junctions.
It is noteworthy that inflammation is associated with hypoxia [
87]. Infiltrating immune cells, invading pathogens, bacteria, and the increased energy demands of resident cells can force a sharp drop in available oxygen [
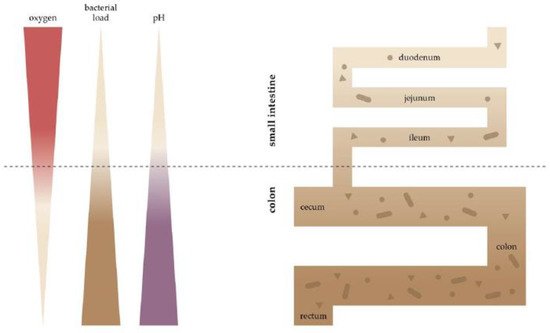
5]. In colitis, hypoxia is observed in all parts of the mucosa [
40]. These oxygen-limiting conditions impair β-oxidation, and can lead to remodelling of membrane lipids and proteins, with an increase in saturated fatty acids [
29]. In addition, with pO
2 reduction due to inflammation, IECs can release pro-inflammatory cytokines such as TNF. Furthermore, a drop in pO
2 contributes to increased epithelial cell apoptosis [
88].
The exact mechanism for the increase in hypoxia in IBD is not yet clear [
40]; it probably occurs due to several factors. Inflammation may lead to an increase in oxygen consumption by epithelial cells or an increase in vasculitis (inflammation of blood vessel walls) and thus, a decrease in oxygen availability in the inflamed areas. It is also possible that during inflammation, migrating neutrophils consume local oxygen and thus, increase hypoxia in colitis [
5,
89].
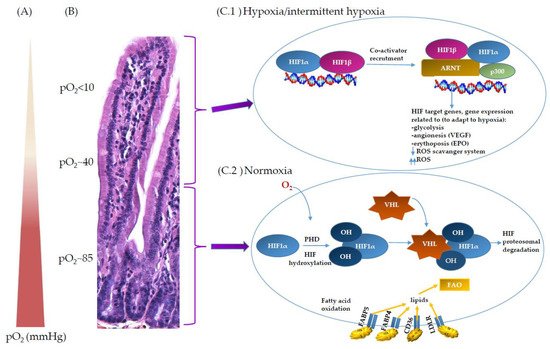
In the inflamed mucosa, HIF-1α increases barrier protection genes, enhances innate immune responses, and activates an antimicrobial response by increasing β-defensins, antimicrobial peptides implicated in the resistance of the surface of epithelia to microbial colonization. β-defensin expression requires low-oxygen conditions and HIF-1α activation [
90]. All of these activities of HIF-1α in colitis result in a protective response. On the contrary to HIF-1α, HIF-2α are essential for the maintenance of an epithelial inflammatory response and, when chronically activated, can also increase the pro-inflammatory response, intestinal injury, and cancer [
40]. However, more work is needed to fully understand the dynamic regulation of HIF-1α and HIF-2α in inflamed mucosa.
IBD is classified into two broad groups: ulcerative colitis (UC) and Crohn’s disease (CD). In UC patients, inflammation is localised in the colonic region. In CD patients, inflammatory lesions are seen throughout the gastrointestinal tract, with some extra ulcerated mucosa and granulomas [
91]. Both HIF-1α and HIF-2α expression are increased in the intestinal epithelium of UC and CD patients and in a mouse model of colitis [
87].
The composition of the microbiota is frequently associated with IBD [
92]. In IBD, there is an overall decrease in microbial diversity, including decreased abundance and even loss of major phyla of obligated anaerobe bacteria such as
Firmicutes and
Bacteroidetes, which, in healthy individuals, represent 90% of all microbes present [
56,
84,
93,
94]. This suggests the presence of elevated oxygen levels. Following this, the abundance of facultative anaerobic bacteria, such as Enterobacteriaceae, is markedly elevated in individuals with IBD. The abundance of facultative anaerobic bacteria in the colon is correlated with the distribution of oxygen emanating from the host tissue [
56,
84,
95]. Whether oxygen pressure alterations are causative or a result of the dysbiosis found in IBD remains to be determined.
A homeostatic role for SCFA in the distal gut during inflammation is important in the progression of inflammatory diseases. It was reported that a reduction in SCFA is associated with IBD [
63,
92]. In addition, the epithelial transporter—the cell-specific butyrate transporter (encoded by SLC16A1)—is downregulated in the inflamed colonic mucosa of patients with IBD. The reduction in butyrate and subsequent inhibition of β-oxidation may be particularly detrimental in the context of intestinal inflammation [
96,
97]. Administration of exogenous butyrate promotes resistance to experimental colitis [
98]. Butyrate can also protect mice from
Clostridium difficile-induced colitis through a HIF-1-dependent mechanism [
37]. SCFAs are currently the most studied bacterial metabolites that, from studies, can be identified as beneficial to colon homeostasis. IBD is correlated with disruption of commensal bacteria and the levels of expression of genes in commensal bacteria [
99]. Although patients with IBD exhibit decreased overall diversity of the microbiome with alternation in microbiota, it is still not clear if these changes initiate disease pathogenesis or are secondary to inflammation [
100]. The microbiota of patients with active CD is 50% less diverse and the microbiota of patients with UC is 30% less diverse [
101]
Increased hypoxia, a lack of metabolites from the microbiota and a dysfunctional microbiota are the main characteristics of IBD. These characteristics of IBD underpin the disease course and contribute to extra-intestinal symptoms. Although of diagnostic value, which of the alterations are causative and which offer viable entry points for therapy beyond reducing symptoms remain to be investigated.