Mitochondria are double-membraned organelles. The mitochondrial inner membrane has characteristic folds, called cristae, providing a large amount of surface area for chemical reactions, and it is enclosed by a permeable mitochondrial outer membrane, which completes the double membrane-bound architecture of mitochondria. Mitochondria undergo dynamic movement inside cells by fusion and fission and build large interconnected intracellular networks, a process which is called mitochondrial dynamics.
- mitochondria
- fluorescence imaging
- fluorescent chemical probes
- fluorescent nanosensors
- mitochondria-targeting peptides
1. Introduction
Mitochondria play pivotal roles in supplying cellular energy in the form of adenosine triphosphate (ATP) via oxidative phosphorylation [1]. Recent enthusiastic efforts have revealed that this ‘powerhouse of the cell’ is indeed the hub of intracellular signaling, energetics, and redox balance [2][3][4]. Considering the important role of mitochondria in calcium homeostasis, fatty acid synthesis, and biogenesis of the heme and iron-sulfur proteins, the interconnection between mitochondria and cellular signaling is essential [5][6]. Mitochondria provide energy in the form of ATP for cell survival; however, mitochondria are also actively implicated in apoptosis to promote cell death [7]. Since mitochondria are key regulators of apoptosis, they participate in developmental processes and aging [8]. Therefore, mitochondrial dysfunction is associated with aging-related phenomena including metabolic disorders, cardiomyopathies, and neurodegeneration [9]. Consequently, there is a high demand for understanding mitochondrial biology as a new frontier in health and disease.
Mitochondria are double-membraned organelles. The mitochondrial inner membrane has characteristic folds, called cristae, providing a large amount of surface area for chemical reactions, and it is enclosed by a permeable mitochondrial outer membrane, which completes the double membrane-bound architecture of mitochondria [10]. Mitochondria undergo dynamic movement inside cells by fusion and fission and build large interconnected intracellular networks, a process which is called mitochondrial dynamics [11]. It is generally believed that mitochondrial dynamics allows the cell to respond to cellular environmental changes, and results in the cell-type-specific appearance of the mitochondrial morphology. In addition, recent studies have demonstrated that mitochondrial dynamics is important for understanding multiple biological processes, and dysfunctions of mitochondrial dynamics could trigger several human diseases [8][12]. Therefore, monitoring mitochondrial morphology could provide a clue to learn many different biological processes for human diseases.
Mirroring these important roles of mitochondria in biological systems, there exists considerable interest in the development of new material for monitoring mitochondrial structure and function [4][12]. With a high sensitivity and signal-to-noise ratio, simple operation, practicality, good selectivity, and the capability for real-time detection, fluorescence has been utilized as a promising technique. From monochromatic fluorescent probes to ratiometric, multi-photon probes, and target-switchable fluorescent probes, a variety of fluorescent chemical probes has been developed [13]. Due to the high extinction coefficients, quantum yields, and modularity, the engineering of multicolored fluorescent proteins has enabled the development of various protein sensors [14]. Based on the high loading capacity and availability in multimodal imaging or sensing, there exists a continually growing attention for fluorescent nanomaterials in bioimaging [15]. The application of these fluorescent materials in mitochondrial research has disclosed a new area for monitoring mitochondrial biology in vitro and in vivo [4][16][17][18][19].
2. Fluorescent Chemical-Based Mitochondria Probes
The strategy for the development of fluorescent chemical probes for monitoring mitochondria-associated biological events is usually presented by conjugation of the mitochondria-targeting motif with an organic fluorophore using a suitable linker ( Figure 1 ). As a part of anchoring mitochondria, the lipophilic cation is mainly considered as a key motif for the accumulation of the chemical in the mitochondrial matrix [20]. A delocalized cationic character in conjugated systems such as aromatic rings is essential for the accumulation of chemicals in mitochondria [20]. Therefore, the manipulation of the lipophilic character of chemical probes can affect their ability for targeting efficiency toward mitochondria. Based on structural insights, various kinds of fluorescent probes bearing lipophilic cations in diverse fluorophores such as boron-dipyrromethene (BODIPY), Rhodamine, cyanine-based Alexa-fluor, and acedan have been developed and are already commercially available to monitor mitochondria or membrane potential, e.g., MitoTracker Green, MitoTracker Orange, MitoTracker Red, and MitoTracker Deep Red ( Figure 1 ) [19][21][22][23][24][25][26][27]. In particular, the last three dyes—MitoTracker Orange, Mitotracker Red, and MitoTracker Deep Red—are available in super-resolution imaging like stochastic optical reconstruction microscopy (STORM) [19][28].
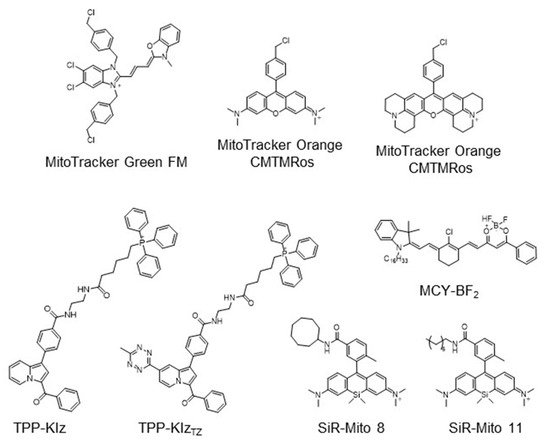
The application of fluorogenic organic dyes that induce ‘turn-on’ fluorescence by a specific event in the case of localization in mitochondria has reduced background fluorescence and obtained a high signal-to-noise ratio [23][29]. Herein, we introduce interesting examples for fluorogenic mitochondria chemical probes and systematic studies for the regulation of hydrophobicity targeting mitochondria. In addition, we focus on the development of versatile fluorescent chemical probes for monitoring mitochondrial metabolites and membrane potential.
As a mitochondria-targeting motif, TPP is the most widely used functional group in the application of chemical probes as well as other fluorescence materials [4][20][34][35]. Due to the critical impact of the fluorogenic approach, we briefly introduce recent studies in the development of chemical probes using the fluorogenic dye-bearing TPP group. Fluorogenic properties of the desired probe only induce the enhancement of fluorescent intensity when the probes are targeting mitochondria; therefore, it is considered as a key feature of fluorescent technique to enhance resolution and practicality.
During the process of energy production in mitochondria, the concentration of various ions including protons is distributed on inner and outer membranes of mitochondria, which results in mitochondrial membrane potential [13][36]. The integrity of MMP is highly related to mitochondrial functions and there is a lot of evidence that the abnormality of MMP is associated with human diseases such as Parkinson’s disease, Alzheimer’s disease, and cancer [7][9][11][13][37][38]. Therefore, fluorescent probes for detecting MMP provide a great research tool for mitochondrial biology.
3. Peptide- or Protein-Based Mitochondria Probes
Biomaterials such as peptides or proteins are major resources for the development of imaging tools for mitochondria. We focused on the recent approach for peptide- or protein-based strategies for mitochondrial visualization and their therapeutic application.
Another mitochondria-targeting peptide sequence was developed to deliver protein to mitochondria ( Figure 2 ). By in silico analysis, a novel cell-penetrating artificial mitochondria-targeting peptide (CAMP) was designed based on the structural insight between human immunodeficiency virus (HIV-1) trans-activator of transcription (TAT) peptide and the mitochondria-targeting sequence [39]. The sequence of CAMP is YGRKKRRQRRR LLRAALRK_AAL (the underscore indicates the cleavage site predicted by MitoProtII) [39]. The fusion of CAMP with EGFP or hMT1A allowed the delivery of these cargo proteins to mitochondria, then CAMP was efficiently cleaved by mitochondrial metalloprotease and released cargo proteins in cellular and in vivo mouse models [39]. These results demonstrate the capability of mitochondria-specific delivery systems using mitochondrial targeting peptides.
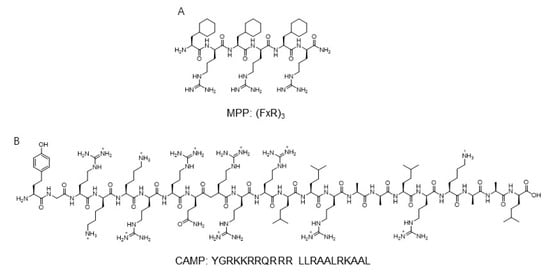
Historically, the engineered fluorescent protein has enabled real-time visualization for the protein of interest with high resolution and sensitivity. The diverse pH range of the mitochondria matrix and pH alteration induced by various perturbations such as breaking of calcium homeostasis or mitophagy have been considered as key events to study mitochondrial biology.
Due to the advantages of fast kinetics and high efficiency, bioorthogonal chemistry is widely used to label biomolecules mainly using click-chemistry without any interference in native biological processes [41]. Copper-free click chemistry such as iEDDA reactions using Tz and TCO disclosed the various applications to visualize biomolecules with fluorophore-linked Tz or TCO [42]. The incorporation of unnatural amino acids into the target protein, followed by fluorophore labeling using bioorthogonal reactions, suggested a feasible solution for monitoring mitochondrial protein [43]. By reprogramming the genetic codon, mitochondrial protein MITRAC12, which is an integral inner membrane protein, was modified by incorporation of TCO-L-lysine and subjected to iEDDA cycloaddition with a Star580-tetrazine as a fluorescent tag [44]. With the advantage of TCO-Tz bioorthogonal reactions like fast reaction kinetics, efficient reaction yield, and feasibility in physiological conditions, this approach allowed visualization of mitochondrial proteins in super-resolution imaging with a single cell level.
4. Fluorescent Nanomaterials for Mitochondria
Because of the loading capacity for various materials like drugs, fluorescent dyes, responsive units for an analyte, or mitochondria-targeting motifs at the same time, nanomaterials are considered valuable tools for theragnostic or dual-sensor systems for multiple analytes in mitochondria. Considering the regulation of complex biological processes like bioenergetic functions and redox homeostasis, the development of fluorescent tools for monitoring multiple functions that occur in mitochondria has been required in the field of mitochondria [7]. In addition, mitochondria contribute to cancer growth and survival, thereby they are regarded as a potential therapeutic target for various cancers [7][45]. Moreover, mitochondrial oxidative stress is a major pathological issue in neurodegenerative diseases such as Alzheimer’s disease [46]. Therefore, the fluorescent material for both monitoring and regulating mitochondrial function in parallel is valuable to study mitochondria-related disease. In this context, various types of nanomaterials have been developed for monitoring multiple functions of mitochondria and delivery agents to cure cancer ( Figure 3 ).
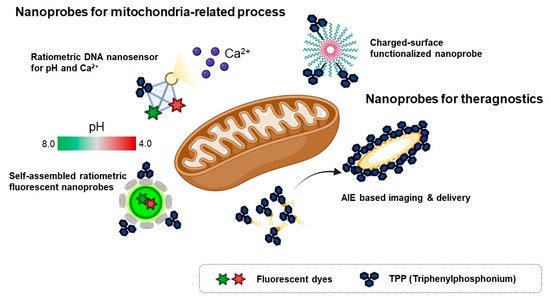
To investigate multiple physiological events in mitochondria, a ratiometric DNA nanosensor for simultaneous monitoring of both calcium and pH in mitochondria was developed [47]. A tetrahedron DNA-based nanoprobe was conjugated with four components including NIR emissive carbon dots bearing a calcium ligand (CD@Cal), pH-responsive fluorescein, TPP as a mitochondria-targeting motif, and an AF660 as a reference fluorophore for quantitative analysis [47]. Without any interference, a tetrahedron DNA nanoprobe measured calcium and pH simultaneously in vitro and quantified mitochondrial calcium concentration and pH in the neuron by real-time imaging [47]. However, self-assembled ratiometric fluorescent nanoprobes (SRFNPs) for monitoring mitochondrial pH were reported [48]. The preparation of the probe is based on a self-assembly mechanism by host-guest interaction between β-cyclodextrin polymer (β-CDP) as the host backbone and adamantine as the guest molecules [48]. Three different components, including fluorescein, rhodamine, and TPP, were introduced to the host backbone via conjugating with adamantane. Since fluorescein has pH-sensitive fluorescent intensity, the intensity ratio between fluorescein and rhodamine quantitatively reported the pH of the mitochondria in a ratiometric way. In this study, adamantane-conjugated TPP was used as a mitochondria targeting moiety [48]. SRFNP was successfully applied to monitor mitochondrial pH in the range pH 4.0–8.0 and low toxicity of SRFNP revealed the biocompatibility of the probes for live cells [48].
A theragnostic approach for tumor targeting, imaging, and drug delivery with mitochondria-targeting nanoparticles was reported [49]. In this study, the author employed the AIE approach to display playing multiple roles in selective drug delivery, anticancer activity, and mitochondria targeting, showing potential ability for theragnostic tools in cancer therapy [49]. A self-assembled nanoparticle with cyanostilbene and a long alkyl chain was designed for targeting mitochondria by incorporation of a TPP moiety and revealed the accumulation in mitochondria by AIE phenomena [49]. Moreover, encapsulation of doxorubicin was successfully delivered to mitochondria and induced anti-cancer activity in both cell-based and in vivo systems [49]. Furthermore, photothermal properties of hydrophilic nanoparticles are expected to be a theragnostic approach based on photothermal therapy and photothermal/photoacoustic imaging [51]. Wang et al. , reported Mito-BDP5 nanoparticles based on a purely organic BODIPY core with a modification by TPP and an ethylene glycol chain which successfully visualized mitochondria in HeLa cells and tumors in mice by photothermal and photoacoustic imaging with good bioavailability and enhanced permeability and retention effects [51]. TPP-based mitochondria-targeting graphene oxide nanocomposites loaded with indocyanin green TPP-PPG@ICG suggested a new class of fluorescence imaging-guided phototherapy [52]. Due to the preferential accumulation of TPP-PPG@ICG in tumors and availability in NIR light sources, this nanocomposite proved a therapeutic potential with enhanced photothermal efficacy and suppressed ATP production, which led to overcoming drug resistance [52]. Besides cancer therapy, mitochondria-targeting nanoparticles can be used as theragnostic tools for neurodegenerative disease. Ceria nanoparticles are known for their antioxidant activity by scavenging reactive oxygen species [46]. Mitochondria-targeting TPP-conjugated ceria nanoparticles were localized to mitochondria and suppressed neuronal death by regulating reactive oxygen species of damaged mitochondria in an Alzheimer’s disease mouse model [46].
To improve photostability and reduce photobleaching, CdSe/ZnS-based quantum dots and iron-oxide-based nanoprobes, which were coated with polyacrylate and covalently linked with TPP, were reported as functionalized nanoprobes for imaging mitochondria [50]. It was confirmed that inorganic nanoparticle quantum dot-TPPs with low surface charge and high colloidal stability enhanced mitochondrial targeting efficiency with low nonspecific binding and bypass endosomal trafficking [50].
This entry is adapted from the peer-reviewed paper 10.3390/ma14154180
References
- Jonckheere, A.I.; Smeitink, J.A.; Rodenburg, R.J. Mitochondrial ATP synthase: Architecture, function and pathology. J. Inherit. Metab. Dis. 2012, 35, 211–225.
- Tan, J.X.; Finkel, T. Mitochondria as intracellular signaling platforms in health and disease. J. Cell Biol. 2020, 219, e202002179.
- Handy, D.E.; Loscalzo, J. Redox regulation of mitochondrial function. Antioxid. Redox Signal. 2012, 16, 1323–1367.
- Wisnovsky, S.; Lei, E.K.; Jean, S.R.; Kelley, S.O. Mitochondrial Chemical Biology: New Probes Elucidate the Secrets of the Powerhouse of the Cell. Cell Chem. Biol. 2016, 23, 917–927.
- Vamecq, J.; Dessein, A.F.; Fontaine, M.; Briand, G.; Porchet, N.; Latruffe, N.; Andreolotti, P.; Cherkaoui-Malki, M. Mitochondrial dysfunction and lipid homeostasis. Curr. Drug Metab. 2012, 13, 1388–1400.
- Stehling, O.; Lill, R. The role of mitochondria in cellular iron-sulfur protein biogenesis: Mechanisms, connected processes, and diseases. Cold Spring Harb. Perspect. Biol. 2013, 5, a011312.
- Burke, P.J. Mitochondria, Bioenergetics and Apoptosis in Cancer. Trends Cancer 2017, 3, 857–870.
- Duchen, M.R. Mitochondria in health and disease: Perspectives on a new mitochondrial biology. Mol. Asp. Med. 2004, 25, 365–451.
- Norat, P.; Soldozy, S.; Sokolowski, J.D.; Gorick, C.M.; Kumar, J.S.; Chae, Y.; Yagmurlu, K.; Prada, F.; Walker, M.; Levitt, M.R.; et al. Mitochondrial dysfunction in neurological disorders: Exploring mitochondrial transplantation. NPJ Regen. Med. 2020, 5, 22.
- Frey, T.G.; Mannella, C.A. The internal structure of mitochondria. Trends Biochem. Sci. 2000, 25, 319–324.
- Chen, H.; Chan, D.C. Mitochondrial dynamics—Fusion, fission, movement, and mitophagy—In neurodegenerative diseases. Hum. Mol. Genet. 2009, 18, R169–R176.
- Westermann, B. Mitochondrial fusion and fission in cell life and death. Nat. Rev. Mol. Cell Biol. 2010, 11, 872–884.
- Li, X.; Zhao, Y.; Yin, J.; Lin, W. Organic fluorescent probes for detecting mitochondrial membrane potential. Coord. Chem. Rev. 2020, 420, 213419.
- Hoffman, R.M. Advantages of multi-color fluorescent proteins for whole-body and in vivo cellular imaging. J. Biomed. Opt. 2005, 10, 41202.
- Wolfbeis, O.S. An overview of nanoparticles commonly used in fluorescent bioimaging. Chem. Soc. Rev. 2015, 44, 4743–4768.
- Mitra, K.; Lippincott-Schwartz, J. Analysis of mitochondrial dynamics and functions using imaging approaches. Curr. Protoc. Cell Biol. 2010, 46, 4.25.1–4.25.21.
- Sun, N.; Malide, D.; Liu, J.; Rovira, I.I.; Combs, C.A.; Finkel, T. A fluorescence-based imaging method to measure in vitro and in vivo mitophagy using mt-Keima. Nat. Protoc. 2017, 12, 1576–1587.
- Jakobs, S. High resolution imaging of live mitochondria. Biochim. Biophys. Acta 2006, 1763, 561–575.
- Samanta, S.; He, Y.; Sharma, A.; Kim, J.; Pan, W.; Yang, Z.; Li, J.; Yan, W.; Liu, L.; Qu, J.; et al. Fluorescent Probes for Nanoscopic Imaging of Mitochondria. Chem 2019, 5, 1697–1726.
- Murphy, M.P. Targeting lipophilic cations to mitochondria. Biochim. Biophys. Acta 2008, 1777, 1028–1031.
- Lincoln, R.; Greene, L.E.; Zhang, W.; Louisia, S.; Cosa, G. Mitochondria Alkylation and Cellular Trafficking Mapped with a Lipophilic BODIPY-Acrolein Fluorogenic Probe. J. Am. Chem Soc. 2017, 139, 16273–16281.
- Petrat, F.; Pindiur, S.; Kirsch, M.; de Groot, H. “Mitochondrial” photochemical drugs do not release toxic amounts of 1O(2) within the mitochondrial matrix space. Arch. Biochem. Biophys. 2003, 412, 207–215.
- Li, H.; Xin, C.; Zhang, G.; Han, X.; Qin, W.; Zhang, C.-W.; Yu, C.; Jing, S.; Li, L.; Huang, W. A mitochondria-targeted two-photon fluorogenic probe for the dual-imaging of viscosity and H2O2 levels in Parkinson’s disease models. J. Mater. Chem. B 2019, 7, 4243–4251.
- Saha, P.C.; Chatterjee, T.; Pattanayak, R.; Das, R.S.; Mukherjee, A.; Bhattacharyya, M.; Guha, S. Targeting and Imaging of Mitochondria Using Near-Infrared Cyanine Dye and Its Application to Multicolor Imaging. ACS Omega 2019, 4, 14579–14588.
- Levi, S.; Corsi, B.; Bosisio, M.; Invernizzi, R.; Volz, A.; Sanford, D.; Arosio, P.; Drysdale, J. A human mitochondrial ferritin encoded by an intronless gene. J. Biol. Chem. 2001, 276, 24437–24440.
- Samudio, I.; Konopleva, M.; Hail, N., Jr.; Shi, Y.X.; McQueen, T.; Hsu, T.; Evans, R.; Honda, T.; Gribble, G.W.; Sporn, M.; et al. 2-Cyano-3,12-dioxooleana-1,9-dien-28-imidazolide (CDDO-Im) directly targets mitochondrial glutathione to induce apoptosis in pancreatic cancer. J. Biol. Chem. 2005, 280, 36273–36282.
- Zhou, R.; Yazdi, A.S.; Menu, P.; Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature 2011, 469, 221–225.
- Bates, M.; Jones, S.A.; Zhuang, X. Preparation of photoswitchable labeled antibodies for STORM imaging. Cold Spring Harb. Protoc. 2013, 2013, 540–541.
- Choi, S.K.; Rho, J.; Yoon, S.E.; Seok, J.H.; Kim, H.; Min, J.; Yoon, W.; Lee, S.; Yun, H.; Kwon, O.P.; et al. Full Color Tunable Aggregation-Induced Emission Luminogen for Bioimaging Based on an Indolizine Molecular Framework. Bioconjug Chem. 2020, 31, 2522–2532.
- Choi, S.-K.; Lee, Y.; Yoon, S.E.; Choi, H.; Kim, J.; Kim, J.H.; Lee, S.; Kim, W.; Kim, E. A tetrazine-fused aggregation induced emission luminogen for bioorthogonal fluorogenic bioprobe. Sens. Actuators B Chem. 2021, 340, 129966.
- Sung, J.; Rho, J.G.; Jeon, G.G.; Chu, Y.; Min, J.S.; Lee, S.; Kim, J.H.; Kim, W.; Kim, E. A New Infrared Probe Targeting Mitochondria via Regulation of Molecular Hydrophobicity. Bioconjug Chem. 2019, 30, 210–217.
- Chu, Y.; Shin, M.C.; Sung, J.; Park, J.; Kim, E.; Lee, S. Development of Theragnostic Tool Using NIR Fluorescence Probe Targeting Mitochondria in Glioma Cells. Bioconjug Chem. 2019, 30, 1642–1648.
- Xiao, H.; Li, P.; Zhang, W.; Tang, B. An ultrasensitive near-infrared ratiometric fluorescent probe for imaging mitochondrial polarity in live cells and in vivo. Chem. Sci. 2016, 7, 1588–1593.
- Ma, C.; Xia, F.; Kelley, S.O. Mitochondrial Targeting of Probes and Therapeutics to the Powerhouse of the Cell. Bioconjug Chem. 2020, 31, 2650–2667.
- Zielonka, J.; Joseph, J.; Sikora, A.; Hardy, M.; Ouari, O.; Vasquez-Vivar, J.; Cheng, G.; Lopez, M.; Kalyanaraman, B. Mitochondria-Targeted Triphenylphosphonium-Based Compounds: Syntheses, Mechanisms of Action, and Therapeutic and Diagnostic Applications. Chem. Rev. 2017, 117, 10043–10120.
- Perry, S.W.; Norman, J.P.; Barbieri, J.; Brown, E.B.; Gelbard, H.A. Mitochondrial membrane potential probes and the proton gradient: A practical usage guide. Biotechniques 2011, 50, 98–115.
- Lin, M.T.; Beal, M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 2006, 443, 787–795.
- Keeney, P.M.; Xie, J.; Capaldi, R.A.; Bennett, J.P., Jr. Parkinson’s disease brain mitochondrial complex I has oxidatively damaged subunits and is functionally impaired and misassembled. J. Neurosci. 2006, 26, 5256–5264.
- Kang, Y.C.; Son, M.; Kang, S.; Im, S.; Piao, Y.; Lim, K.S.; Song, M.Y.; Park, K.S.; Kim, Y.H.; Pak, Y.K. Cell-penetrating artificial mitochondria-targeting peptide-conjugated metallothionein 1A alleviates mitochondrial damage in Parkinson’s disease models. Exp. Mol. Med. 2018, 50, 1–13.
- Horton, K.L.; Stewart, K.M.; Fonseca, S.B.; Guo, Q.; Kelley, S.O. Mitochondria-penetrating peptides. Chem. Biol. 2008, 15, 375–382.
- Prescher, J.A.; Bertozzi, C.R. Chemistry in living systems. Nat. Chem. Biol. 2005, 1, 13–21.
- Oliveira, B.L.; Guo, Z.; Bernardes, G.J.L. Inverse electron demand Diels-Alder reactions in chemical biology. Chem. Soc. Rev. 2017, 46, 4895–4950.
- Kim, E.; Koo, H. Biomedical applications of copper-free click chemistry: In vitro, in vivo, and ex vivo. Chem. Sci. 2019, 10, 7835–7851.
- Saal, K.A.; Richter, F.; Rehling, P.; Rizzoli, S.O. Combined Use of Unnatural Amino Acids Enables Dual-Color Super-Resolution Imaging of Proteins via Click Chemistry. ACS Nano 2018, 12, 12247–12254.
- van Loo, G.; Saelens, X.; van Gurp, M.; MacFarlane, M.; Martin, S.J.; Vandenabeele, P. The role of mitochondrial factors in apoptosis: A Russian roulette with more than one bullet. Cell Death Differ. 2002, 9, 1031–1042.
- Kwon, H.J.; Cha, M.Y.; Kim, D.; Kim, D.K.; Soh, M.; Shin, K.; Hyeon, T.; Mook-Jung, I. Mitochondria-Targeting Ceria Nanoparticles as Antioxidants for Alzheimer’s Disease. ACS Nano 2016, 10, 2860–2870.
- Liu, Z.; Pei, H.; Zhang, L.; Tian, Y. Mitochondria-Targeted DNA Nanoprobe for Real-Time Imaging and Simultaneous Quantification of Ca(2+) and pH in Neurons. ACS Nano 2018, 12, 12357–12368.
- Feng, Z.; Ma, Y.; Li, B.; He, L.; Wang, Q.; Huang, J.; Liu, J.; Yang, X.; Wang, K. Mitochondria targeted self-assembled ratiometric fluorescent nanoprobes for pH imaging in living cells. Anal. Methods 2019, 11, 2097–2104.
- Kim, K.Y.; Jin, H.; Park, J.; Jung, S.H.; Lee, J.H.; Park, H.; Kim, S.K.; Bae, J.; Jung, J.H. Mitochondria-targeting self-assembled nanoparticles derived from triphenylphosphonium-conjugated cyanostilbene enable site-specific imaging and anticancer drug delivery. Nano Res. 2017, 11, 1082–1098.
- Chakraborty, A.; Jana, N.R. Design and Synthesis of Triphenylphosphonium Functionalized Nanoparticle Probe for Mitochondria Targeting and Imaging. J. Phys. Chem. C 2015, 119, 2888–2895.
- Wang, J.-L.; Zhang, L.; Zhao, M.-J.; Zhang, T.; Liu, Y.; Jiang, F.-L. Mitochondria-Targeted BODIPY Nanoparticles for Enhanced Photothermal and Photoacoustic Imaging In Vivo. ACS Appl. Bio Mater. 2021, 4, 1760–1770.
- Zeng, W.N.; Yu, Q.P.; Wang, D.; Liu, J.L.; Yang, Q.J.; Zhou, Z.K.; Zeng, Y.P. Mitochondria-targeting graphene oxide nanocomposites for fluorescence imaging-guided synergistic phototherapy of drug-resistant osteosarcoma. J. Nanobiotechnol. 2021, 19, 79.
