Inflammatory bowel disease (IBD) is a complex inflammatory chronic and pathological condition that includes Crohn’s disease (CD) and Ulcerative colitis (UC). CD primarily affects the small and large intestine whereas the prime targets for UC are colon and rectum. Immune sentinel subsets of CD4+ T cells such as Th (T helper cells)-1, Th2, Th17, and regulatory T cells (Tregs) play a crucial role in the pathogenesis of IBD. Immunological balance between effector Th cells and Trges is essential for maintaining immune-homeostasis. Immunoregulatory Trges are characterized by the expression of transcription factor Forkhead box P3 (Foxp3), and surface marker CD25, and are functionally immunosuppressive & important for immune tolerance. Therapeutic arrangement based on Tregs is important to address the systemic inflammatory and autoimmune diseases such as IBD and rheumatoid arthritis.
- inflammatory bowels disease
- Crohn’s disease
- ulcerative colitis
- human immune system
- regulatory T cells
1. IBD Pathogenesis
The epithelial layer of the human gut consists of goblet cells, columnar cells, paneth cells, endocrine cells, M cells, tuft cells, and epithelial resident intestinal stem cells. These cells are responsible for the differentiation of gut microbiota and secretion of various mucus-containing antimicrobial peptides [1][2][3]. The intestinal barrier contains innate immune cells such as dendritic cells (DCs), neutrophils, macrophages and innate lymphoid cells (ILCs) which reside in the state of hypo-responsiveness in a healthy human gut [4][5]. The mucosal macrophages prevent the inter-conversion of Th1 and Th17 cells by producing anti-inflammatory cytokines and thus promoting the differentiation of Trges [6][7][8]. The immune cells’ balance in intestinal mucosa and luminal content is crucial for the normal functioning of the mucosal immune system since dysregulated immune effecters result in the IBD pathogenesis [9][10][11]. The impaired innate immune system is responsible for the functional abnormalities of the adaptive immune system, and interconversion of effector Th cells and Trges causing IBD pathogenesis [11][12]. Therefore, balanced gut mucosal immunity to maintain immune homeostasis and protection is inevitably critical to fighting IBD.
Activated lamina propria (intestinal mucous membrane) secrete a large number of soluble immune mediators, including pro-inflammatory cytokines viz. tumor necrosis factor (TNF), interferon-gamma (IFN-γ), Interleukin (IL)-6, IL-12, IL-21, IL-23, IL-17 and anti-inflammatory cytokines such asIL-10, transforming growth factor (TGF-β) and IL-35in local tissues [13]. The imbalance between these secreted soluble mediators especially inflammatory and anti-inflammatory cytokines secreted by immune system results in IBD pathogenesis [14][13][15][16] (Figure 1). The exogenous administration of TNF and TNF-like cytokine 1Acytokines (TLA1) regulates the balance between Th1 and Th17 cell population in the inflamed colonic tissues [17]. Further, TLA1 modulates Foxp3 expression in Tregs and its function, and murine model of colitis has seen the alleviation of colitis when treated with Tregs expressing low levels of TLA1.TLA1 may promote the maintenance of Treg suppressor function in a death domain receptor 3 (DR3) dependent manner [18]. Passive administration of anti-TLA1 antibodies prevents the development of 2,4,6-trinitrobenzene sulfonic acid (TNBS)-induced colitis in mice, partially improves dextran Sulfate sodium (DSS)-induced colitis and decreases the intestinal fibrosis in a chronic colitis model [19][20][21].
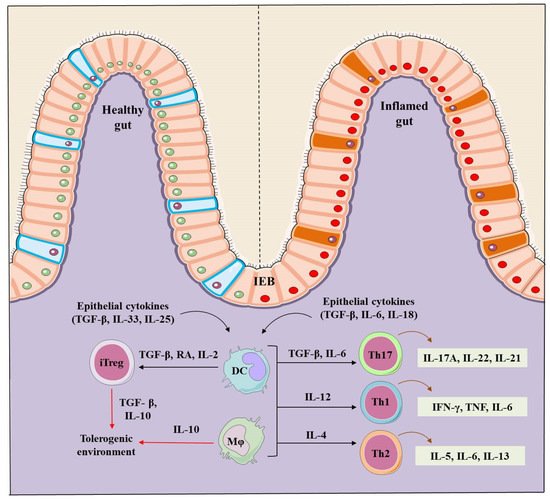
Figure 1. Regulation of the intestinal homeostasis in healthy and IBD inflamed gut. A healthy intestinal epithelial barrier (IEB) in presence of TGF-β, retinoic acid (RA) and IL-2 promote dendritic cells (DCs) and macrophages (mφ) to stimulate the generation of inducible Trges (iTregs). TGF-β and IL-10 are markers that contribute to the generation of iTregs, and establish and maintain the tolerogenic environment in a healthy gut. On the contrary, IBD induced inflammation induces intestinal epithelial barriers and secrete TGF-β, IL-6 and IL-8, stimulating DCs and mφ to produce the inflammatory Th-17 (IL-17A, IL-22, IL-21), Th-1 (IFN-γ, TNF-α, IL-6) and Th-2 type cells (IL-5, IL-6, IL-13) creating an inflammation focus and diseased intestine.
A meta-analysis has identified more than 200 genomic loci associated with IBD pathogenesis, and 68% of loci have been shared by the UC and CD disease [22]. Genome-wide association studies (GWAS) have helped the scientific community to find out genes responsible for IBD pathology and nucleotide-binding oligomerization domain 2 (NOD2) is the first gene found to be associated with IBD [23]. The associated mutations in NOD2 and polymorphisms within autophagy-related 16-like 1 (ATG16L1) genes have disseminated the pivotal role of autophagy in the pathogenicity of IBD [24][25][26]. NOD2 knockouts exhibited excessive intestinal inflammation as compared to NOD2 sufficient mice [27], and selective deletion of ATG16L1 in T cells ends with spontaneous intestinal inflammation, characterized by a decrease in Foxp3+ Tregs and aberrant expression of Th2 cells [28].
The contribution of gut homing or migration associated molecules such as α4b7 integrin, αEb7 integrin, CD62L, chemokine receptor (CCR)-4 (CCR4), CCR5, CCR7 and CCR9 in the pathogenesis of IBD is well known [29]. Defective or loss of expression of these molecules leads to the impaired Tregs trafficking to the target organs and thus inducing IBD. Loss of CCR7 and CCR4impairs the functions of Tregs in experimental colitis [30][31], and further CCR7 regulates the balance between Th1, Th17, and Trges in Crohn’s-like Murine ileitis [32]. b7 integrin deficiency impaired Tregs homing in IL-10 deficient mice and spontaneously increased IBD-induced inflammation [33]. In essence, all findings indicate the crucial role played by the number of functional Tregs since a compromised number of Tregs contribute to the pathogenesis of IBD and other associated biological impairments.
2. Regulatory T Cells (Tregs)
Tregs are heterogeneous cell populations of CD4+ T cells and possess immunosuppressive attributes. CD4+ Trges expressing high levels of IL-2 receptor α chain (CD25) and master transcription factor Foxp3 are the best-characterized populations with the immunosuppressive phenotype [34]. Foxp3 is essentially required for maintaining their immunosuppressive activity against infections, tumors, intestinal inflammation, allergy, and autoimmunity [35][36][37][38]. The absence of CD127 (IL-7 receptor α-chain) is considered as another feature of Tregs and up-regulates CD25 and Foxp3 expression upon activation. Thus a lower expression of CD127 is important alongwith elevated CD4, CD25, and Foxp3 expression to confirm the functional phenotype of immunoregulatory T cells [39][40]. Two main subsets of Trges are characterized as Foxp3 positive Trges and Foxp3 negative type 1 Treg (Tr1) cells.
2.1. tTregs and pTre
Depending on generation, Foxp3+ Trges are further categorized as naturally occurring thymus-derived Treg (tTreg) cells and Trges developed from conventional CD4+ T cells in the periphery (pTreg). These cells possess immunosuppressive functions and maintain peripheral tolerance [41][42]. tTregs are produced by the thymus at an early stage after birth and maintain tolerance toward self-antigens [43][44]. TGF-β1 directly enhances the Foxp3 promoter and encourages the generation of tTregs [45]. Besides, exposure of naive T-cells to its cognate-antigen leads to the differentiation of pTregs under tolerogenic conditions [46][47][48], and differentiation of pTregs is facilitated by the higher concentrations of TGF-β and higher levels of Foxp3 [49][50][51]. Therefore, TGF-β1 is a key cytokine and plays a crucial role in the differentiation of both subsets of Trges. The induction of Foxp3 in peripheral naive T cells is achieved by a higher concentration of TGF-β, retinoic acid, and CD28 co-stimulation [50][52][53][54]. Both, tTreg and pTregs show similar expression levels of FoxP3, CD25, CTLA-4, GITR, ICOS, CD103, CD127 and a broad T-cell receptor (TCR) repertoire to deploy various suppressive mechanisms to control effector cells [35][39][55][56]. Foxp3+ Trges are also known to secrete IL-10, TGF-β, and IL-35 [57][58] along with granzyme A and B [59][60][61]. Furthermore, tTregs express higher levels of neuropilin-1(Nrp1), TF Ikzf2 (Helios), PD-1, and ecto nucleotidase CD73 than pTregs [62][63]. Helios and Nrp1 are considered as markers for tTregs since their greater expression is seen in tTregs as compared to pTregs [63][64][65]. Interestingly, under in-vivo conditions, pTregs could express helios [66], and a fraction of the human tTreg population did not express helios [67]. Moreover, tTregs are not differentiated based on helios and Nrp1 expression in mice [68]. Treg specific demethylated region (TSDR) is highly demthylated in tTregs, and partially demethylated along with an unstable expression of Foxp3 and CD25 in pTregs [49][69][70]. Apart from TSDR, Ig superfamily surface protein GPA33along with other Treg cell markers was recently used to identify Trges of thymus origin since this molecule is stably expressed on tTregs [71].
2.2. Type 1 T regulatory (Tr1) Cells
Tr1 cells are unique Foxp3− regulatory T cells that develop in the periphery and secrete elevated levels of immunosuppressive cytokines such as IL-10 and TGF-β [72][73]. tTreg and pTregs constitutively express Foxp3 and CD25 but these markers are expressed by Tr1 cells only in the activated state [74]. Tr1 cells are characterized by the co-expression of surface markers, CD49b and LAG-3 [75], and can be distinct due to the cytokine expression of IL-2, IL-10, IFN-γ, IL-5 and Il-17 [74] as well as granzyme B and perforin via cell death mechanism [73]. And, experimental evidences have confirmed the immunosuppressive function of Tr1 cells mediated by IL-10 [76][77].
3. Role of Tregs in IBD
Trges play a vital role in maintaining gut immune homeostasis and regulate pro-inflammatory responses elicited by the adaptive and innate immune effectors [29]. Scurfy mouse strain showing the severe autoimmune phenotype with a genetic defect in the Foxp3 gene and inhibit the Tregs development and leads to the dysregulated activation of the gut immune system [78], which mounts inflammation primarily in the gut. Further, the mutation in the human Foxp3 gene leads to a rare autoimmune dysregulation, polyendocrinopathy, enteropathy, X-linked (IPEX) syndrome along with other severe autoimmune diseases including arthritis, diabetes, allergy and IBD. This syndrome was seen due to impaired immune response mediated inflammation [79][80][81]. Furthermore Foxp3 expressing Trges are essential for maintaining the balance at the intestinal mucosal surface because intestinal inflammation gets chronic with the decreasing number of Foxp3+ Tregs [82]. DSS induced colitis in mice showings pontaneous depletion of Foxp3+ Tregs leading to an increase in the disease severity [83]. However, adoptive transfer of Tregs in Treg depleted mice (DEREG) showed a decrease in the severity and improved tissue conditions of experimental colitis [83].
Although patients with IBD showed an increased number of Tregs in the inflamed intestinal mucosa than un-inflamed mucosal part [84][85][86]. The phenotype and function of Tregs present in the inflamed mucosa or periphery of IBD patients or in experimental animals differ from those present in peripheral lymphoid organs of healthy control. Patients with IBD showed an increase in the number of peripheral Th17 cells, and a reduction in the peripheral Trges [87]. While in some cases patients with IBD showed higher expression of Foxp3 along with elevated levels of pro-inflammatory cytokines including IL-17A, IL-1β and IL-6 [87]. Moreover, the highest frequency of Foxp3+ IL-17 T-cells (Th17 and Treg intermediate cells) was seen in the inflamed mucosal tissues of patients with IBD [88][89] (Figure 2).
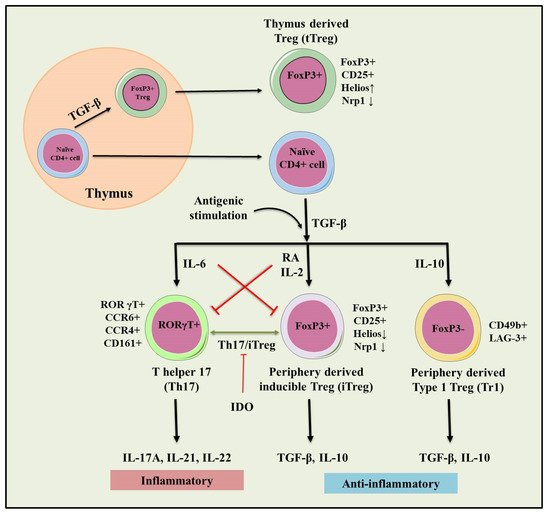
Figure 2. Role of natural and inducible regulatory T cells (iTregs) involved in the pathogenesis of IBD. Inflammation (IL-6) dependent interconversion of regulatory and effector T cell phenotype and role in dol 2, 3 dioxygenases (IDO) in the generation of inducible Tregs (iTregs). Inflammatory (IL-17A, IL-21, IL-22) and immunosuppressive (TGF-β, IL-10) conditions following antigenic stimulation were seen during the conversion of Th17 to iTreg phenotype. This interconversion plays a crucial role in maintaining tolerance towards IBD.
Foxp3+ Trges express effector T cell-specific transcription factor retinoic acid receptor-related orphan receptor gamma t (RORγt) and differentiate into Th-17 cells. These cells inhibit the immunosuppressive function of Tregs in patients with IBD [90]. Further, Tregs upregulate the expression of T-bet and express pro-inflammatory cytokine IFN-γ. Regulatory T cells are characterized as IFN-γ-expressing Th1 like Tregs and mount intestinal inflammation in patients with IBD. Th1 like Trges evoke inflammation since the IFN-γ-expressing cells accumulate at the site of inflammation in CD and UC and contribute to the IBD pathogenesis [91]. The accumulation of IFN-γ expressing Th1 like Trges is also observed in the inflamed colon in the DSS-induced colitis animal model [91]. The acquisition of pro-inflammatory behavior of Tregs in IBD most likely contributes to the uncontrolled inflammation in vivo. Furthermore, Tregs suppress the colonic inflammation by downregulating Th1 and Th17 responses in the adoptive transfer model of colitis [92][93][94], and passively transferred Trges have shown the ability to control inflammatory lesions in the experimental model of IBD [95].
Trges have been seen to be involved in the tissue repair mechanism of the intestine. Among the different populations of intestinal Treg i.e., Tr1 cells have been shown to mediate repair of the intestinal mucosa that co-expressed Th2 and master transcription factor GATA binding protein 3 (GATA3). Therefore, elevated expression of IL-33R, ST2, and amphiregulin (AREG), an epidermal growth factor receptor ligand was reported [96]. Furthermore, these factors are generally involved in tissue repair and are phenotypically characterized and expressed by GATA3+ Tregs [96]. Tissue repair and protection of gut is rendered by the human Tr1 Trges suppress the proliferation of T effector cells, elicit TNF and IL-1β based innate immune response and secret IL-22 to regulate the repairing of epithelium and promote barrier function [97].
4. Therapeutic Role of Tregs in IBD
Several patients with IBD showed tolerance to the current therapeutic arrangements. Therefore, the need for developing effective, safe and novel therapies for IBD is both attractive and urgent. Newer and effective immunotherapies involving anti-TNF agents (infliximab, adalimumab, and certolizumab) have shown remarkable progress toreducethe need for surgery and hospitalization of IBD patients [98][99]. However, meta-analysis conducted by Ford et al., suggested that usage of anti-TNF agents may increase the risk of getting opportunistic infections in IBD patients [100]. Other recent reports also suggest the higher chance of getting infections such as histoplasmosis, aspergillosis and cytomegalovirus infection [101] and anti-TNF monotherapy was found to be responsible for higher mycobacterial and bacterial infection; however, in a combination therapy with thiopurine will increase the risk of getting serious infections [101][102]. The REFURBISH study was reported that the risk of getting T-cell non-Hodgkin’s lymphoma in IBD patients is higher during combinational therapy with compare to anti-TNF monotherapy [103] whereas the another cohort study delineate that even the ant--TNF monotherapy is associated with lymphoma formation in small number but have the higher statistical significant. And, it put on more on risk during the combinational therapy [104]. Other than this other paradoxical side effects such as psoriasis/psoriasiform skin, development of sarcoidosis-like lesions, late occurrence of arthritis/synovitis and lupus-like syndrome (0.5 to 1% of patients) can also be developed [105][106][107][108][109]. Additionally, novel therapies including JAK inhibitor [110][111][112], anti-MAdCAM-1 [113][114][115][116][117], an anti-SMAD7 antisense oligonucleotide (mongersen) [118][119][120], S1P1 [110][121] and anti-interleukin (IL)-12/23 (ustekinumab) [122][123][124][125][126][127][128] have been under investigation for safety and other purposes.
Recently, cellular therapies have been used as potential therapeutic strategies for IBD patients [129]. The role of Tregs in the preclinical models of colitis has been well understood, and recent investigations and phase 1 clinical trials have proven the safety and efficacy of Trges. A marked difference in the number of Tregs in patients (the inflamed mucosa or peripheral blood) and experimental animal models of IBD have been observed [82][130]. Experimental model of IBD showed an increase in Tregs percentage in the inflamed ileum with a reduced immunosuppressive function and IL-10 production. The dysregulated expression between Th17 cells and Tregs was also observed in UC animal model with downregulated mRNA expression of Foxp3 and IL-10 levels in Trges [131]. In UC patients, a significant increase of IL-17 and Th17 cells and a simultaneous decrease of TGF-β and Trges in serum as compared to healthy control was seen [132]. Th-17 cells and associated cytokines IL-17 and IL-23 were found to be decreased in patients with IBD along with a decrease in the Tregs and associated cytokines IL-10 and TGF-β [133]. IL-10 is known to induce Treg mediated suppression of Th-17 cells in a STAT-3 dependent manner [134]. The improvement in the clinical and histological parameters was observed when Trges were adoptively transferred in Rag−/− or severe combined immunodeficient (SCID) mice [135]. Rapamycin-expanded Trges (Th cells cultured in presence of rapamycin) were shown to suppress colitis in SCID mice [136]. And, ovalbumin (OVA) induced Tregs from DO11.10 mice prevented colitis together with increased TGF-β and IL-10 secretion in SCID-bg mice [137]. The safety and efficacy of OVA-Treg therapy were assessed for refractory CD in an open-labeled multicenter phase I/II clinical trial. This study showed the dose-related efficacy because infusion of ova-specific Tregs treatment was well-tolerated, and 40%patients showed a reduced CD activity on 5 and 8-week post-treatment [138]. In vitro expanded CD45RA+ Tregs cells were shown to express stable Foxp3 locus, which enhanced their suppressive ability and prevented their conversion to Th17 phenotype in the SCID xenotransplant model [139]. Additionally, CD45RA− and CD45RA+ expanded Tregs expressed a high level of gut homing receptor α4β7 integrin, CD62L, and CCR7 to facilitate their intestinal homing [139]. In an active CD mucosa, CD45RA+ Tregs healed the inflammation of lamina propria and mesenteric lymph nodes [139]. Tregs isolated from the lamina propria of active IBD patients and in experimental model (DSS induced colitis) express T-bet and IFN-γ (Th-1 like Tregs) and stimulates the early stages of inflammation. Further, T-bet KO showed the development of less severe colitis with the dysregulated Th1 immune response. It suggests that T-bet expression in Tregs is required for the development of colitis [91]. In the end, Treg immunotherapy with in vitro expanded Tregs (NCT03185000) for treating the Crohn’s disease (TRIBUTE trial) is underway.
This entry is adapted from the peer-reviewed paper 10.3390/cells10081847
References
- Iftekhar, A.; Sigal, M. Defence and adaptation mechanisms of the intestinal epithelium upon infection. Int. J. Med. Microbiol. 2021, 311, 151486.
- Schroeder, B.O. Fight them or feed them: How the intestinal mucus layer manages the gut microbiota. Gastroenterol. Rep. 2019, 7, 3–12.
- Bakshani, C.R.; Morales-Garcia, A.L.; Althaus, M.; Wilcox, M.D.; Pearson, J.P.; Bythell, J.C.; Burgess, J.G. Evolutionary conservation of the antimicrobial function of mucus: A first defence against infection. npj Biofilms Microbiomes 2018, 4, 1–12.
- Lin, L.; Zhang, J. Role of intestinal microbiota and metabolites on gut homeostasis and human diseases. Bmc Immunol. 2017, 18, 1–25.
- Stagg, A.J. Intestinal dendritic cells in health and gut inflammation. Front. Immunol. 2018, 9, 2883.
- Denning, T.L.; Wang, Y.C.; Patel, S.R.; Williams, I.R.; Pulendran, B. Lamina propria macrophages and dendritic cells differentially induce regulatory and interleukin 17-producing T cell responses. Nat. Immunol. 2007, 8, 1086–1094.
- Wang, S.; Ye, Q.; Zeng, X.; Qiao, S. Functions of macrophages in the maintenance of intestinal homeostasis. J. Immunol. Res. 2019, 2019, 1512969.
- Pandiyan, P.; Bhaskaran, N.; Zou, M.; Schneider, E.; Jayaraman, S.; Huehn, J. Microbiome dependent regulation of Tregs and Th17 cells in mucosa. Front. Immunol. 2019, 10, 426.
- Yoo, J.Y.; Groer, M.; Dutra, S.V.O.; Sarkar, A.; McSkimming, D.I. Gut Microbiota and Immune System Interactions. Microorganisms 2020, 8, 1587.
- Okumura, R.; Takeda, K. Maintenance of gut homeostasis by the mucosal immune system. Proc. Jpn. Acad. Ser. B 2016, 92, 423–435.
- Yue, B.; Luo, X.; Yu, Z.; Mani, S.; Wang, Z.; Dou, W. Inflammatory bowel disease: A potential result from the collusion between gut microbiota and mucosal immune system. Microorganisms 2019, 7, 440.
- Huang, Y.; Chen, Z. Inflammatory bowel disease related innate immunity and adaptive immunity. Am. J. Transl. Res. 2016, 8, 2490–2497.
- Friedrich, M.; Pohin, M.; Powrie, F. Cytokine networks in the pathophysiology of inflammatory bowel disease. Immunity 2019, 50, 992–1006.
- de Souza, H.S.P.; Fiocchi, C. Immunopathogenesis of IBD: Current state of the art. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 13–27.
- Neurath, M.F. Cytokines in inflammatory bowel disease. Nat. Rev. Immunol. 2014, 14, 329–342.
- Guan, Q.; Zhang, J. Recent Advances: The Imbalance of Cytokines in the Pathogenesis of Inflammatory Bowel Disease. Mediat. Inflamm. 2017, 2017, 1–8.
- Kamada, N.; Hisamatsu, T.; Honda, H.; Kobayashi, T.; Chinen, H.; Takayama, T.; Kitazume, M.; Okamoto, S.; Koganei, K.; Sugita, A.; et al. TL1A Produced by Lamina Propria Macrophages Induces Th1 and Th17 Immune Responses in Cooperation with IL-23 in Patients with Crohn’s Disease. Inflamm. Bowel Dis. 2010, 16, 568–575.
- Sidhu-Varma, M.; Shih, D.Q.; Targan, S.R. Differential Levels of Tl1a Affect the Expansion and Function of Regulatory T Cells in Modulating Murine Colitis. Inflamm. Bowel Dis. 2016, 22, 548–559.
- Takedatsu, H.; Michelsen, K.S.; Wei, B.; Landers, C.J.; Thomas, L.S.; Dhall, D.; Braun, J.; Targan, S.R. TL1A (TNFSF15) regulates the development of chronic colitis by modulating both T-helper 1 and T-helper 17 activation. Gastroenterology 2008, 135, 552–567.
- Meylan, F.; Song, Y.J.; Fuss, I.; Villarreal, S.; Kahle, E.; Malm, I.J.; Acharya, K.; Ramos, H.L.; Lo, L.; Mentink-Kane, M.M.; et al. The TNF-family cytokine TL1A drives IL-13-dependent small intestinal inflammation. Mucosal. Immunol. 2011, 4, 172–185.
- Lu, Y.; Kim, N.-M.; Jiang, Y.-W.; Zhang, H.; Zheng, D.; Zhu, F.-X.; Liang, R.; Li, B.; Xu, H.-X. Cambogin suppresses dextran sulphate sodium-induced colitis by enhancing Treg cell stability and function. Br. J. Pharm. 2018, 175, 1085–1099.
- Jostins, L.; Ripke, S.; Weersma, R.K.; Duerr, R.H.; McGovern, D.P.; Hui, K.Y.; Lee, J.C.; Schumm, L.P.; Sharma, Y.; Anderson, C.A. Host–microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature 2012, 491, 119–124.
- Shaw, M.H.; Kamada, N.; Warner, N.; Kim, Y.-G.; Nuñez, G. The ever-expanding function of NOD2: Autophagy, viral recognition, and T cell activation. Trends Immunol. 2011, 32, 73–79.
- Kuballa, P.; Huett, A.; Rioux, J.D.; Daly, M.J.; Xavier, R.J. Impaired autophagy of an intracellular pathogen induced by a Crohn’s disease associated ATG16L1 variant. PLoS ONE 2008, 3, e3391.
- Cooney, R.; Baker, J.; Brain, O.; Danis, B.; Pichulik, T.; Allan, P.; Ferguson, D.J.; Campbell, B.J.; Jewell, D.; Simmons, A. NOD2 stimulation induces autophagy in dendritic cells influencing bacterial handling and antigen presentation. Nat. Med. 2010, 16, 90–97.
- Travassos, L.H.; Carneiro, L.A.; Ramjeet, M.; Hussey, S.; Kim, Y.-G.; Magalhães, J.G.; Yuan, L.; Soares, F.; Chea, E.; Le Bourhis, L. Nod1 and Nod2 direct autophagy by recruiting ATG16L1 to the plasma membrane at the site of bacterial entry. Nat. Immunol. 2010, 11, 55–62.
- Barreau, F.; Meinzer, U.; Chareyre, F.; Berrebi, D.; Niwa-Kawakita, M.; Dussaillant, M.; Foligne, B.; Ollendorff, V.; Heyman, M.; Bonacorsi, S.; et al. CARD15/NOD2 is required for Peyer’s patches homeostasis in mice. PLoS ONE 2007, 2, e523.
- Kabat, A.; Harrison, O.; Riffelmacher, T.; Moghaddam, A.E.; Pearson, C.; Laing, A.; Abeler-Dörner, L.; Forman, S.; Grencis, R.; Sattentau, Q.; et al. The autophagy gene Atg16l1 differentially regulates Treg and TH2 cells to control intestinal inflammation. eLife 2016, 5, e12444.
- Yamada, A.; Arakaki, R.; Saito, M.; Tsunematsu, T.; Kudo, Y.; Ishimaru, N. Role of regulatory T cell in the pathogenesis of inflammatory bowel disease. World J. Gastroenterol. 2016, 22, 2195–2205.
- Schneider, M.A.; Meingassner, J.G.; Lipp, M.; Moore, H.D.; Rot, A. CCR7 is required for the in vivo function of CD4+ CD25+ regulatory T cells. J. Exp. Med. 2007, 204, 735–745.
- Yuan, Q.; Bromley, S.K.; Means, T.K.; Jones, K.J.; Hayashi, F.; Bhan, A.K.; Luster, A.D. CCR4-dependent regulatory T cell function in inflammatory bowel disease. J. Exp. Med. 2007, 204, 1327–1334.
- McNamee, E.N.; Masterson, J.C.; Veny, M.; Collins, C.B.; Jedlicka, P.; Byrne, F.R.; Ng, G.Y.; Rivera-Nieves, J. Chemokine receptor CCR7 regulates the intestinal TH1/TH17/Treg balance during Crohn’s-like murine ileitis. J. Leukoc. Biol. 2015, 97, 1011–1022.
- Sun, H.; Kuk, W.; Rivera-Nieves, J.; Lopez-Ramirez, M.A.; Eckmann, L.; Ginsberg, M.H. β7 Integrin Inhibition Can Increase Intestinal Inflammation by Impairing Homing of CD25(hi)Foxp3(+) Regulatory T Cells. Cell. Mol. Gastroenterol. Hepatol. 2020, 9, 369–385.
- Li, Z.; Li, D.; Tsun, A.; Li, B. Foxp3+ regulatory T cells and their functional regulation. Cell Mol. Immunol. 2015, 12, 558–565.
- Fontenot, J.D.; Gavin, M.A.; Rudensky, A.Y. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat. Immunol. 2003, 4, 330–336.
- Sakaguchi, S. Naturally arising Foxp3− expressing CD25+CD4+ regulatory T cells in immunological tolerance to self and non-self. Nat. Immunol. 2005, 6, 345–352.
- Pereira, L.M.S.; Gomes, S.T.M.; Ishak, R.; Vallinoto, A.C.R. Regulatory T Cell and Forkhead Box Protein 3 as Modulators of Immune Homeostasis. Front. Immunol. 2017, 8, 605.
- Hori, S.; Nomura, T.; Sakaguchi, S. Control of Regulatory T Cell Development by the Transcription FactorFoxp3. Science 2003, 299, 1057–1061.
- Liu, W.; Putnam, A.L.; Xu-Yu, Z.; Szot, G.L.; Lee, M.R.; Zhu, S.; Gottlieb, P.A.; Kapranov, P.; Gingeras, T.R.; Fazekas de St Groth, B.; et al. CD127 expression inversely correlates with Foxp3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 2006, 203, 1701–1711.
- Trzonkowski, P.; Bieniaszewska, M.; Juścińska, J.; Dobyszuk, A.; Krzystyniak, A.; Marek, N.; Myśliwska, J.; Hellmann, A. First-in-man clinical results of the treatment of patients with graft versus host disease with human ex vivo expanded CD4+ CD25+ CD127−T regulatory cells. Clin. Immunol. 2009, 133, 22–26.
- Haribhai, D.; Williams, J.B.; Jia, S.; Nickerson, D.; Schmitt, E.G.; Edwards, B.; Ziegelbauer, J.; Yassai, M.; Li, S.-H.; Relland, L.M. A requisite role for induced regulatory T cells in tolerance based on expanding antigen receptor diversity. Immunity 2011, 35, 109–122.
- Josefowicz, S.Z.; Niec, R.E.; Kim, H.Y.; Treuting, P.; Chinen, T.; Zheng, Y.; Umetsu, D.T.; Rudensky, A.Y. Extrathymically generated regulatory T cells control mucosal TH 2 inflammation. Nature 2012, 482, 395–399.
- Darrigues, J.; van Meerwijk, J.P.; Romagnoli, P. Age-dependent changes in regulatory T lymphocyte development and function: A mini-review. Gerontology 2018, 64, 28–35.
- Yang, S.; Fujikado, N.; Kolodin, D.; Benoist, C.; Mathis, D. Regulatory T cells generated early in life play a distinct role in maintaining self-tolerance. Science 2015, 348, 589–594.
- Ouyang, W.; Beckett, O.; Ma, Q.; Li, M.O. Transforming growth factor-β signaling curbs thymic negative selection promoting regulatory T cell development. Immunity 2010, 32, 642–653.
- Kretschmer, K.; Apostolou, I.; Hawiger, D.; Khazaie, K.; Nussenzweig, M.C.; von Boehmer, H. Inducing and expanding regulatory T cell populations by foreign antigen. Nat. Immunol. 2005, 6, 1219–1227.
- Knoechel, B.; Lohr, J.; Kahn, E.; Bluestone, J.A.; Abbas, A.K. Sequential development of interleukin 2–dependent effector and regulatory T cells in response to endogenous systemic antigen. J. Exp. Med. 2005, 202, 1375–1386.
- Yadav, M.; Bluestone, J.A.; Stephan, S. Peripherally induced tregs–role in immune homeostasis and autoimmunity. Front. Immunol. 2013, 4, 232.
- Zheng, Y.; Josefowicz, S.; Chaudhry, A.; Peng, X.P.; Forbush, K.; Rudensky, A.Y. Role of conserved non-coding DNA elements in the Foxp3 gene in regulatory T-cell fate. Nature 2010, 463, 808–812.
- Chen, W.; Jin, W.; Hardegen, N.; Lei, K.-j.; Li, L.; Marinos, N.; McGrady, G.; Wahl, S.M. Conversion of peripheral CD4+ CD25− naive T cells to CD4+ CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3. J. Exp. Med. 2003, 198, 1875–1886.
- Schlenner, S.M.; Weigmann, B.; Ruan, Q.; Chen, Y.; von Boehmer, H. Smad3 binding to the Foxp3 enhancer is dispensable for the development of regulatory T cells with the exception of the gut. J. Exp. Med. 2012, 209, 1529–1535.
- Tai, X.; Cowan, M.; Feigenbaum, L.; Singer, A. CD28 costimulation of developing thymocytes induces Foxp3 expression and regulatory T cell differentiation independently of interleukin 2. Nat. Immunol. 2005, 6, 152–162.
- Sun, C.-M.; Hall, J.A.; Blank, R.B.; Bouladoux, N.; Oukka, M.; Mora, J.R.; Belkaid, Y. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J. Exp. Med. 2007, 204, 1775–1785.
- Benson, M.J.; Pino-Lagos, K.; Rosemblatt, M.; Noelle, R.J. All-trans retinoic acid mediates enhanced T reg cell growth, differentiation, and gut homing in the face of high levels of co-stimulation. J. Exp. Med. 2007, 204, 1765–1774.
- Shevach, E.M.; Thornton, A.M. tTregs, pTregs, and iTregs: Similarities and differences. Immunol. Rev. 2014, 259, 88–102.
- Shevyrev, D.; Tereshchenko, V. Treg heterogeneity, function, and homeostasis. Front. Immunol. 2020, 10, 3100.
- Collison, L.W.; Workman, C.J.; Kuo, T.T.; Boyd, K.; Wang, Y.; Vignali, K.M.; Cross, R.; Sehy, D.; Blumberg, R.S.; Vignali, D.A.A. The inhibitory cytokine IL-35 contributes to regulatory T-cell function. Nature 2007, 450, 566–569.
- Collison, L.W.; Pillai, M.R.; Chaturvedi, V.; Vignali, D.A.A. Regulatory T cell suppression is potentiated by target T cells in a cell contact, IL-35- and IL-10-dependent manner. J. Immunol. 2009, 182, 6121–6128.
- Grossman, W.J.; Verbsky, J.W.; Barchet, W.; Colonna, M.; Atkinson, J.P.; Ley, T.J. Human T regulatory cells can use the perforin pathway to cause autologous target cell death. Immunity 2004, 21, 589–601.
- Gondek, D.C.; Lu, L.-F.; Quezada, S.A.; Sakaguchi, S.; Noelle, R.J. Cutting Edge: Contact-Mediated Suppression by CD4+CD25+ Regulatory Cells Involves a Granzyme B-Dependent, Perforin-Independent Mechanism. J. Immunol. 2005, 174, 1783–1786.
- Karreci, E.S.; Eskandari, S.K.; Dotiwala, F.; Routray, S.K.; Kurdi, A.T.; Assaker, J.P.; Luckyanchykov, P.; Mihali, A.B.; Maarouf, O.; Borges, T.J. Human regulatory T cells undergo self-inflicted damage via granzyme pathways upon activation. JCI Insight 2017, 2.
- Lin, X.; Chen, M.; Liu, Y.; Guo, Z.; He, X.; Brand, D.; Zheng, S.G. Advances in distinguishing natural from induced Foxp3(+) regulatory T cells. Int. J. Clin. Exp. Pathol. 2013, 6, 116–123.
- Singh, K.; Hjort, M.; Thorvaldson, L.; Sandler, S. Concomitant analysis of Helios and Neuropilin-1 as a marker to detect thymic derived regulatory T cells in naive mice. Sci. Rep. 2015, 5, 1–10.
- Delgoffe, G.M.; Woo, S.-R.; Turnis, M.E.; Gravano, D.M.; Guy, C.; Overacre, A.E.; Bettini, M.L.; Vogel, P.; Finkelstein, D.; Bonnevier, J.; et al. Stability and function of regulatory T cells is maintained by a neuropilin-1-semaphorin-4a axis. Nature 2013, 501, 252–256.
- Thornton, A.M.; Korty, P.E.; Tran, D.Q.; Wohlfert, E.A.; Murray, P.E.; Belkaid, Y.; Shevach, E.M. Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J. Immunol. 2010, 184, 3433–3441.
- Gottschalk, R.A.; Corse, E.; Allison, J.P. Expression of Helios in peripherally induced Foxp3+ regulatory T cells. J. Immunol. 2012, 188, 976–980.
- Elkord, E. Helios should not be cited as a marker of human thymus-derived Tregs. Commentary: Helios+ and Helios− cells coexist within the natural Foxp3+ T regulatory cell subset in humans. Front. Immunol. 2016, 7, 276.
- Szurek, E.; Cebula, A.; Wojciech, L.; Pietrzak, M.; Rempala, G.; Kisielow, P.; Ignatowicz, L. Differences in expression level of helios and neuropilin-1 do not distinguish thymus-derived from extrathymically-induced CD4+ Foxp3+ regulatory T cells. PLoS ONE 2015, 10, e0141161.
- Lee, S.; Park, K.; Kim, J.; Min, H.; Seong, R.H. Foxp3 expression in induced regulatory T cells is stabilized by C/EBP in inflammatory environments. Embo Rep. 2018, 19, e45995.
- Polansky, J.K.; Kretschmer, K.; Freyer, J.; Floess, S.; Garbe, A.; Baron, U.; Olek, S.; Hamann, A.; von Boehmer, H.; Huehn, J. DNA methylation controls Foxp3 gene expression. Eur. J. Immunol. 2008, 38, 1654–1663.
- Opstelten, R.; de Kivit, S.; Slot, M.C.; van den Biggelaar, M.; Iwaszkiewicz-Grześ, D.; Gliwiński, M.; Scott, A.M.; Blom, B.; Trzonkowski, P.; Borst, J. GPA33: A marker to identify stable human regulatory T cells. J. Immunol. 2020, 204, 3139–3148.
- Levings, M.K.; Gregori, S.; Tresoldi, E.; Cazzaniga, S.; Bonini, C.; Roncarolo, M.G. Differentiation of Tr1 cells by immature dendritic cells requires IL-10 but not CD25+CD4+ Tr cells. Blood 2005, 105, 1162–1169.
- Groux, H.; O’Garra, A.; Bigler, M.; Rouleau, M.; Antonenko, S.; De Vries, J.E.; Roncarolo, M.G. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature 1997, 389, 737–742.
- Zeng, H.; Zhang, R.; Jin, B.; Chen, L. Type 1 regulatory T cells: A new mechanism of peripheral immune tolerance. Cell Mol. Immunol. 2015, 12, 566–571.
- Gagliani, N.; Magnani, C.F.; Huber, S.; Gianolini, M.E.; Pala, M.; Licona-Limon, P.; Guo, B.; De’Broski, R.H.; Bulfone, A.; Trentini, F. Coexpression of CD49b and LAG-3 identifies human and mouse T regulatory type 1 cells. Nat. Med. 2013, 19, 739.
- Barrat, F.J.; Cua, D.J.; Boonstra, A.; Richards, D.F.; Crain, C.; Savelkoul, H.F.; de Waal-Malefyt, R.; Coffman, R.L.; Hawrylowicz, C.M.; O’Garra, A. In vitro generation of interleukin 10-producing regulatory CD4(+) T cells is induced by immunosuppressive drugs and inhibited by T helper type 1 (Th1)- and Th2-inducing cytokines. J. Exp. Med. 2002, 195, 603–616.
- Levings, M.K.; Sangregorio, R.; Galbiati, F.; Squadrone, S.; de Waal Malefyt, R.; Roncarolo, M.-G. IFN-α and IL-10 induce the differentiation of human type 1 T regulatory cells. J. Immunol. 2001, 166, 5530–5539.
- Wildin, R.S.; Ramsdell, F.; Peake, J.; Faravelli, F.; Casanova, J.L.; Buist, N.; Levy-Lahad, E.; Mazzella, M.; Goulet, O.; Perroni, L.; et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat. Genet. 2001, 27, 18–20.
- Bacchetta, R.; Barzaghi, F.; Roncarolo, M.-G. From IPEX syndrome to FOXP3 mutation: A lesson on immune dysregulation. Ann. N. Y. Acad. Sci. 2018, 1417, 5–22.
- Bennett, C.L.; Christie, J.; Ramsdell, F.; Brunkow, M.E.; Ferguson, P.J.; Whitesell, L.; Kelly, T.E.; Saulsbury, F.T.; Chance, P.F.; Ochs, H.D. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat. Genet. 2001, 27, 20–21.
- Boehm, F.; Martin, M.; Kesselring, R.; Schiechl, G.; Geissler, E.K.; Schlitt, H.-J.; Fichtner-Feigl, S. Deletion of Foxp3+ regulatory T cells in genetically targeted mice supports development of intestinal inflammation. BMC Gastroenterol. 2012, 12, 97.
- Maul, J.; Loddenkemper, C.; Mundt, P.; Berg, E.; Giese, T.; Stallmach, A.; Zeitz, M.; Duchmann, R. Peripheral and intestinal regulatory CD4+ CD25(high) T cells in inflammatory bowel disease. Gastroenterology 2005, 128, 1868–1878.
- Boschetti, G.; Kanjarawi, R.; Bardel, E.; Collardeau-Frachon, S.; Duclaux-Loras, R.; Moro-Sibilot, L.; Almeras, T.; Flourié, B.; Nancey, S.; Kaiserlian, D. Gut Inflammation in Mice Triggers Proliferation and Function of Mucosal Foxp3+ Regulatory T Cells but Impairs Their Conversion from CD4+ T Cells. J. Crohns. Colitis. 2016, 11, 105–117.
- Holmén, N.; Lundgren, A.; Lundin, S.; Bergin, A.-M.; Rudin, A.; Sjövall, H.; Ohman, L. Functional CD4+CD25high regulatory T cells are enriched in the colonic mucosa of patients with active ulcerative colitis and increase with disease activity. Inflamm. Bowel Dis. 2006, 12, 447–456.
- Hovhannisyan, Z.; Treatman, J.; Littman, D.R.; Mayer, L. Characterization of interleukin-17-producing regulatory T cells in inflamed intestinal mucosa from patients with inflammatory bowel diseases. Gastroenterology 2011, 140, 957–965.
- Wang, Y.; Liu, X.P.; Zhao, Z.B.; Chen, J.H.; Yu, C.G. Expression of CD4+ forkhead box P3 (FOXP3)+ regulatory T cells in inflammatory bowel disease. J. Dig. Dis 2011, 12, 286–294.
- Eastaff-Leung, N.; Mabarrack, N.; Barbour, A.; Cummins, A.; Barry, S. Foxp3+ Regulatory T Cells, Th17 Effector Cells, and Cytokine Environment in Inflammatory Bowel Disease. J. Clin. Immunol. 2009, 30, 80–89.
- Li, J.; Ueno, A.; Iacucci, M.; Fort Gasia, M.; Jijon, H.B.; Panaccione, R.; Kaplan, G.G.; Beck, P.L.; Luider, J.; Barkema, H.W.; et al. Crossover Subsets of CD4+ T Lymphocytes in the Intestinal Lamina Propria of Patients with Crohn’s Disease and Ulcerative Colitis. Dig. Dis. Sci. 2017, 62, 2357–2368.
- Ueno, A.; Jijon, H.; Chan, R.; Ford, K.; Hirota, C.; Kaplan, G.G.; Beck, P.L.; Iacucci, M.; Fort Gasia, M.; Barkema, H.W.; et al. Increased prevalence of circulating novel IL-17 secreting Foxp3 expressing CD4+ T cells and defective suppressive function of circulating Foxp3+ regulatory cells support plasticity between Th17 and regulatory T cells in inflammatory bowel disease patients. Inflamm. Bowel Dis. 2013, 19, 2522–2534.
- Yang, B.H.; Hagemann, S.; Mamareli, P.; Lauer, U.; Hoffmann, U.; Beckstette, M.; Föhse, L.; Prinz, I.; Pezoldt, J.; Suerbaum, S.; et al. Foxp3+ T cells expressing RORγt represent a stable regulatory T-cell effector lineage with enhanced suppressive capacity during intestinal inflammation. Mucosal Immunol. 2016, 9, 444–457.
- Di Giovangiulio, M.; Rizzo, A.; Franzè, E.; Caprioli, F.; Facciotti, F.; Onali, S.; Favale, A.; Stolfi, C.; Fehling, H.-J.; Monteleone, G.; et al. Tbet Expression in Regulatory T Cells Is Required to Initiate Th1-Mediated Colitis. Front. Immunol. 2019, 10.
- Ogino, H.; Nakamura, K.; Ihara, E.; Akiho, H.; Takayanagi, R. CD4+CD25+ Regulatory T Cells Suppress Th17-Responses in an Experimental Colitis Model. Dig. Dis. Sci. 2011, 56, 376–386.
- Uhlig, H.H.; Coombes, J.; Mottet, C.; Izcue, A.; Thompson, C.; Fanger, A.; Tannapfel, A.; Fontenot, J.D.; Ramsdell, F.; Powrie, F. Characterization of Foxp3+ CD4+CD25+ and IL-10-secreting CD4+CD25+ T cells during cure of colitis. J. Immunol. 2006, 177, 5852–5860.
- Harrison, O.J.; Srinivasan, N.; Pott, J.; Schiering, C.; Krausgruber, T.; Ilott, N.E.; Maloy, K.J. Epithelial-derived IL-18 regulates Th17 cell differentiation and Foxp3+ Treg cell function in the intestine. Mucosal Immunol. 2015, 8, 1226–1236.
- Martin, B.; Banz, A.; Bienvenu, B.; Cordier, C.; Dautigny, N.; Bécourt, C.; Lucas, B. Suppression of CD4+ T lymphocyte effector functions by CD4+ CD25+ cells in vivo. J. Immunol. 2004, 172, 3391–3398.
- Monticelli, L.A.; Osborne, L.C.; Noti, M.; Tran, S.V.; Zaiss, D.M.; Artis, D. IL-33 promotes an innate immune pathway of intestinal tissue protection dependent on amphiregulin–EGFR interactions. Proc. Natl. Acad. Sci. USA 2015, 112, 10762–10767.
- Cook, L.; Stahl, M.; Han, X.; Nazli, A.; MacDonald, K.N.; Wong, M.Q.; Tsai, K.; Dizzell, S.; Jacobson, K.; Bressler, B. Suppressive and gut-reparative functions of human type 1 T regulatory cells. Gastroenterology 2019, 157, 1584–1598.
- Ananthakrishnan, A.N.; Cagan, A.; Cai, T.; Gainer, V.S.; Shaw, S.Y.; Savova, G.; Churchill, S.; Karlson, E.W.; Kohane, I.; Liao, K.P. Comparative effectiveness of infliximab and adalimumab in Crohn’s disease and ulcerative colitis. Inflamm. Bowel Dis. 2016, 22, 880–885.
- Vulliemoz, M.; Brand, S.; Juillerat, P.; Mottet, C.; Ben-Horin, S.; Michetti, P. TNF-Alpha Blockers in Inflammatory Bowel Diseases: Practical Recommendations and a User’s Guide: An Update. Digestion 2020, 101, 20–30.
- Ford, A.C.; Peyrin-Biroulet, L. Opportunistic Infections with Anti-Tumor Necrosis Factor-α Therapy in Inflammatory Bowel Disease: Meta-Analysis of Randomized Controlled Trials. Am. J. Gastroenterol. 2013, 108, 1268–1276.
- Fellermann, K. Adverse events of tumor necrosis factor inhibitors. Dig. Dis. 2013, 31, 374–378.
- Kirchgesner, J.; Lemaitre, M.; Carrat, F.; Zureik, M.; Carbonnel, F.; Dray-Spira, R. Risk of serious and opportunistic infections associated with treatment of inflammatory bowel diseases. Gastroenterology 2018, 155, 337–346.e310.
- Deepak, P.; Sifuentes, H.; Sherid, M.; Stobaugh, D.; Sadozai, Y.; Ehrenpreis, E.D. T-cell non-Hodgkin’s lymphomas reported to the FDA AERS with tumor necrosis factor-alpha (TNF-α) inhibitors: Results of the REFURBISH study. Off. J. Am. Coll. Gastroenterol. ACG 2013, 108, 99–105.
- Lemaitre, M.; Kirchgesner, J.; Rudnichi, A.; Carrat, F.; Zureik, M.; Carbonnel, F.; Dray-Spira, R. Association between use of thiopurines or tumor necrosis factor antagonists alone or in combination and risk of lymphoma in patients with inflammatory bowel disease. JAMA 2017, 318, 1679–1686.
- Eickstaedt, J.B.; Killpack, L.; Tung, J.; Davis, D.; Hand, J.L.; Tollefson, M.M. Psoriasis and psoriasiform eruptions in pediatric patients with inflammatory bowel disease treated with anti–tumor necrosis factor alpha agents. Pediatric Dermatol. 2017, 34, 253–260.
- Decock, A.; Van Assche, G.; Vermeire, S.; Wuyts, W.; Ferrante, M. Sarcoidosis-like lesions: Another paradoxical reaction to anti-TNF therapy? J. Crohns. Colitis 2017, 11, 378–383.
- Alivernini, S.; Pugliese, D.; Tolusso, B.; Bui, L.; Petricca, L.; Guidi, L.; Mirone, L.; Rapaccini, G.L.; Federico, F.; Ferraccioli, G. Paradoxical arthritis occurring during anti-TNF in patients with inflammatory bowel disease: Histological and immunological features of a complex synovitis. Rmd Open 2018, 4, e000667.
- Perše, M.; Unkovič, A. The role of TNF in the pathogenesis of inflammatory bowel disease. In Biological Therapy for Inflammatory Bowel Disease; IntechOpen: London, UK, 2019.
- Shovman, O.; Tamar, S.; Amital, H.; Watad, A.; Shoenfeld, Y. Diverse patterns of anti-TNF-α-induced lupus: Case series and review of the literature. Clin. Rheumatol. 2018, 37, 563–568.
- Sandborn, W.J.; Feagan, B.G.; Wolf, D.C.; D’Haens, G.; Vermeire, S.; Hanauer, S.B.; Ghosh, S.; Smith, H.; Cravets, M.; Frohna, P.A. Ozanimod induction and maintenance treatment for ulcerative colitis. N. Engl. J. Med. 2016, 374, 1754–1762.
- Sandborn, W.J.; Sands, B.E.; Danese, S.; D’Haens, G.R.; Vermeire, S.; Schreiber, S.; Feagan, B.G.; Reinisch, W.; Friedman, G.; Woodworth, D.A. Efficacy and safety of oral tofacitinib as maintenance therapy in patients with moderate to severe ulcerative colitis: Results from a phase 3 randomised controlled trial. Gastroenterology 2017, 152, S199.
- D’Haens, G.R.; Panaccione, R.; Higgins, P.; Colombel, J.-F.; Feagan, B.G.; Moscariello, M.; Chan, G.; Healey, P.J.; Niezychowski, W.; Wang, W. 856 Efficacy and safety of oral tofacitinib for maintenance therapy in patients with moderate to severe Crohn’s disease: Results of a phase 2B randomized placebo-controlled trial. Gastroenterology 2016, 150, S183.
- Villablanca, E.J.; Cassani, B.; Von Andrian, U.H.; Mora, J.R. Blocking lymphocyte localization to the gastrointestinal mucosa as a therapeutic strategy for inflammatory bowel diseases. Gastroenterology 2011, 140, 1776–1784.e1775.
- Feagan, B.G.; Rutgeerts, P.; Sands, B.E.; Hanauer, S.; Colombel, J.-F.; Sandborn, W.J.; Van Assche, G.; Axler, J.; Kim, H.-J.; Danese, S. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N. Engl. J. Med. 2013, 369, 699–710.
- Sands, B.E.; Feagan, B.G.; Rutgeerts, P.; Colombel, J.-F.; Sandborn, W.J.; Sy, R.; D’Haens, G.; Ben-Horin, S.; Xu, J.; Rosario, M. Effects of vedolizumab induction therapy for patients with Crohn’s disease in whom tumor necrosis factor antagonist treatment failed. Gastroenterology 2014, 147, 618–627.e613.
- Colombel, J.-F.; Sands, B.E.; Rutgeerts, P.; Sandborn, W.; Danese, S.; D’Haens, G.; Panaccione, R.; Loftus, E.V.; Sankoh, S.; Fox, I. The safety of vedolizumab for ulcerative colitis and Crohn’s disease. Gut 2017, 66, 839–851.
- Loftus Jr, E.V.; Colombel, J.-F.; Feagan, B.G.; Vermeire, S.; Sandborn, W.J.; Sands, B.E.; Danese, S.; D’Haens, G.R.; Kaser, A.; Panaccione, R. Long-term efficacy of vedolizumab for ulcerative colitis. J. Crohns. Colitis 2017, 11, 400–411.
- Monteleone, G.; Neurath, M.F.; Ardizzone, S.; Di Sabatino, A.; Fantini, M.C.; Castiglione, F.; Scribano, M.L.; Armuzzi, A.; Caprioli, F.; Sturniolo, G.C.; et al. Mongersen, an oral SMAD7 antisense oligonucleotide, and Crohn’s disease. N. Engl. J. Med. 2015, 372, 1104–1113.
- Marafini, I.; Monteleone, I.; Dinallo, V.; Di Fusco, D.; De Simone, V.; Laudisi, F.; Fantini, M.C.; Di Sabatino, A.; Pallone, F.; Monteleone, G. CCL20 is negatively regulated by TGF-β1 in intestinal epithelial cells and reduced in Crohn’s disease patients with a successful response to Mongersen, a Smad7 antisense oligonucleotide. J. Crohns. Colitis 2017, 11, 603–609.
- Feagan, B.G.; Sands, B.E.; Rossiter, G.; Li, X.; Usiskin, K.; Zhan, X.; Colombel, J.-F. Effects of mongersen (GED-0301) on endoscopic and clinical outcomes in patients with active Crohn’s disease. Gastroenterology 2018, 154, 61–64.e66.
- Danese, S.; Furfaro, F.; Vetrano, S. Targeting S1P in inflammatory bowel disease: New avenues for modulating intestinal leukocyte migration. J. Crohns. Colitis 2018, 12, S678–S686.
- Leonardi, C.L.; Kimball, A.B.; Papp, K.A.; Yeilding, N.; Guzzo, C.; Wang, Y.; Li, S.; Dooley, L.T.; Gordon, K.B.; Investigators, P.S. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 76-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 1). Lancet 2008, 371, 1665–1674.
- Sandborn, W.J.; Feagan, B.G.; Fedorak, R.N.; Scherl, E.; Fleisher, M.R.; Katz, S.; Johanns, J.; Blank, M.; Rutgeerts, P.; Ustekinumab Crohn’s Disease Study Group. A randomized trial of Ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with moderate-to-severe Crohn’s disease. Gastroenterology 2008, 135, 1130–1141.
- Sandborn, W.J.; Gasink, C.; Gao, L.-L.; Blank, M.A.; Johanns, J.; Guzzo, C.; Sands, B.E.; Hanauer, S.B.; Targan, S.; Rutgeerts, P. Ustekinumab induction and maintenance therapy in refractory Crohn’s disease. N. Engl. J. Med. 2012, 367, 1519–1528.
- Hibi, T.; Imai, Y.; Murata, Y.; Matsushima, N.; Zheng, R.; Gasink, C. Efficacy and safety of ustekinumab in Japanese patients with moderately to severely active Crohn’s disease: A subpopulation analysis of phase 3 induction and maintenance studies. Intest Res. 2017, 15, 475.
- Nigam, G.B.; Limdi, J.K. An update on the role of anti-IL-12/IL23 agents in the management of inflammatory bowel disease. Br. Med. Bull. 2021, 138, 29–40.
- Jefremow, A.; Neurath, M.F. All are Equal, Some are More Equal: Targeting IL 12 and 23 in IBD—A Clinical Perspective. Immunotargets Ther. 2020, 9, 289.
- Kashani, A.; Schwartz, D.A. The Expanding Role of Anti–IL-12 and/or Anti–IL-23 Antibodies in the Treatment of Inflammatory Bowel Disease. Gastroenterol. Hepatol. 2019, 15, 255.
- Misselwitz, B.; Juillerat, P.; Sulz, M.C.; Siegmund, B.; Brand, S. Emerging treatment options in inflammatory Bowel disease: Janus Kinases, stem cells, and more. Digestion 2020, 101, 69–82.
- Sun, X.; He, S.; Lv, C.; Sun, X.; Wang, J.; Zheng, W.; Wang, D. Analysis of murine and human Treg subsets in inflammatory bowel disease. Mol. Med. Rep. 2017, 16, 2893–2898.
- Ma, Y.-H.; Zhang, J.; Chen, X.; Xie, Y.-F.; Pang, Y.-H.; Liu, X.-J. Increased CD4 + CD45RA - Foxp3 low cells alter the balance between Treg and Th17 cells in colitis mice. World J. Gastroenterol. 2016, 22, 9356.
- Gong, Y.; Lin, Y.; Zhao, N.; He, X.; Lu, A.; Wei, W.; Jiang, M. The Th17/Treg Immune Imbalance in Ulcerative Colitis Disease in a Chinese Han Population. Mediat. Inflamm. 2016, 2016, 7089137.
- Geng, X.; Xue, J. Expression of Treg/Th17 cells as well as related cytokines in patients with inflammatory bowel disease. Pak. J. Med. Sci. 2016, 32, 1164–1168.
- Chaudhry, A.; Samstein, R.M.; Treuting, P.; Liang, Y.; Pils, M.C.; Heinrich, J.-M.; Jack, R.S.; Wunderlich, F.T.; Brüning, J.C.; Müller, W.; et al. Interleukin-10 signaling in regulatory T cells is required for suppression of Th17 cell-mediated inflammation. Immunity 2011, 34, 566–578.
- Song-Zhao, G.X.; Maloy, K.J. Experimental mouse models of T cell-dependent inflammatory bowel disease. Methods Mol. Biol. 2014, 1193, 199–211.
- Ogino, H.; Nakamura, K.; Iwasa, T.; Ihara, E.; Akiho, H.; Motomura, Y.; Akahoshi, K.; Igarashi, H.; Kato, M.; Kotoh, K.; et al. Regulatory T cells expanded by rapamycin in vitro suppress colitis in an experimental mouse model. J. Gastroenterol. 2012, 47, 366–376.
- Zhou, P.; Borojevic, R.; Streutker, C.; Snider, D.; Liang, H.; Croitoru, K. Expression of dual TCR on DO11.10 T cells allows for ovalbumin-induced oral tolerance to prevent T cell-mediated colitis directed against unrelated enteric bacterial antigens. J. Immunol. 2004, 172, 1515–1523.
- Desreumaux, P.; Foussat, A.; Allez, M.; Beaugerie, L.; Hébuterne, X.; Bouhnik, Y.; Nachury, M.; Brun, V.; Bastian, H.; Belmonte, N.; et al. Safety and efficacy of antigen-specific regulatory T-cell therapy for patients with refractory Crohn’s disease. Gastroenterology 2012, 143, 1207–1217.e1202.
- Canavan, J.B.; Scottà, C.; Vossenkämper, A.; Goldberg, R.; Elder, M.J.; Shoval, I.; Marks, E.; Stolarczyk, E.; Lo, J.W.; Powell, N.; et al. Developing in vitro expanded CD45RA+ regulatory T cells as an adoptive cell therapy for Crohn’s disease. Gut 2016, 65, 584–594.
