Periodontitis (PD) is an infection-driven inflammatory disease of periodontal tissues caused by pathogenic microorganisms, which have been characterized by disruption of the tooth-supporting structures. In periodontal disease treatment, the conventional route of drug administration has many drawbacks such as poor biodistribution, low selectivity of the therapeutic effect, burst release of the drug, and damage to healthy cells. To overcome these difficulties, controlled drug delivery systems have been evolved as an alternative method to address oral infectious disease ailments. The use of drug delivery devices proves to be an excellent auxiliary method in enhancing the quality and effectiveness in periodontitis treatment, which includes inaccessible periodontal pockets. In the literature, there have been described as many various polymer-based delivery systems such as hydrogels, liposomes, micro-, and nanoparticles, which can be used in the treatment of chronic periodontal disease. The review shows the current state of knowledge regarding the applications of various polymer-based delivery systems such as hydrogels, liposomes, micro-, and nanoparticles in the treatment of chronic periodontal disease.
- Periodontitis (PD)
- Drug Delivery Systems (DDS)
- Polymeric Drug Delivery Systems
- Drug Release
- Periodontal Treatment
- Pathogenesis of Periodontitis Diseases
1. Definition
Periodontitis (PD) is an infection-driven inflammatory disease of periodontal tissues caused by pathogenic microorganisms, which have been characterized by disruption of the tooth-supporting structures.
2. Introduction
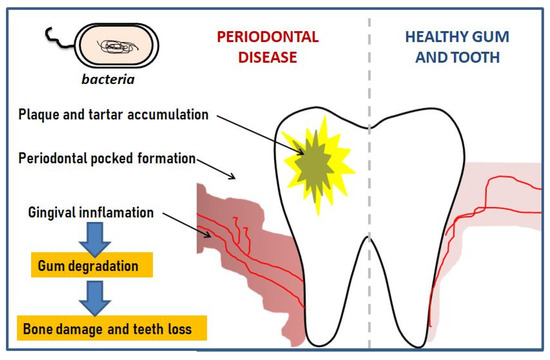
3. The Pathogenesis of Periodontitis Diseases
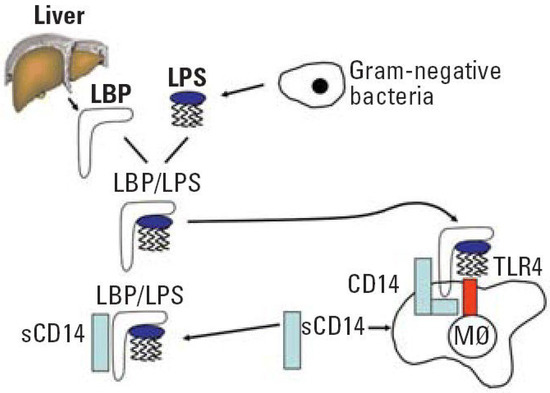
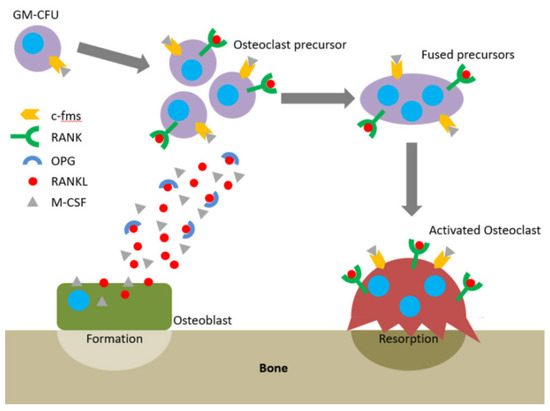
4. Conclusions and Future Perspectives
This entry is adapted from the peer-reviewed paper 10.3390/polym12071574
References
- Gayasuddin Khan; Ravi R. Patel; Sarita K. Yadav; Nagendra Kumar; Sundeep Chaurasia; Gufran Ajmal; Brahmeshwar Mishra; Brahmeshwar Mishra; Development, optimization and evaluation of tinidazole functionalized electrospun poly(ε-caprolactone) nanofiber membranes for the treatment of periodontitis. RSC Advances 2016, 6, 100214-100229, 10.1039/c6ra22072j.
- Poul Erik Petersen; Hiroshi Ogawa; Strengthening the Prevention of Periodontal Disease: The WHO Approach. Journal of Periodontology 2005, 76, 2187-2193, 10.1902/jop.2005.76.12.2187.
- Rajeshwari H R; Dinesh Dhamecha; Satveer Jagwani; Meghana Rao; Kiran Jadhav; Shabana Shaikh; Lakshmi Puzhankara; Sunil Jalalpure; H.R. Rajeshwari; Local drug delivery systems in the management of periodontitis: A scientific review.. Journal of Controlled Release 2019, 307, 393-409, 10.1016/j.jconrel.2019.06.038.
- Sim Yee Lim; Mali Dafydd; Jieji Ong; Launa A. Ord-McDermott; Emma Board-Davies; Kirsty Sands; David Williams; Alastair J. Sloan; Charles M. Heard; Mucoadhesive thin films for the simultaneous delivery of microbicide and anti-inflammatory drugs in the treatment of periodontal diseases. International Journal of Pharmaceutics 2020, 573, 118860, 10.1016/j.ijpharm.2019.118860.
- Deeksha Joshi; Tarun Garg; Amit K. Goyal; Goutam Rath; Advanced drug delivery approaches against periodontitis. Drug Delivery 2014, 23, 363-377, 10.3109/10717544.2014.935531.
- Zamani, F.; Jahanmard, F.; Ghasemkhah, F.; Amjad-Iranagh, S.; Bagherzadeh, R.; Amani-Tehran, M.; Latifi, M. Nanostructures for Drug Delivery; Andronescu, E., Grumezescu, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 239–270.
- Nilu Jain; Gaurav K. Jain; Shamama Javed; Zeenat Iqbal; Sushama Talegaonkar; Farhan J. Ahmad; Roop K. Khar; Recent approaches for the treatment of periodontitis. Drug Discovery Today 2008, 13, 932-943, 10.1016/j.drudis.2008.07.010.
- Rania Hamed; Ala’A Aburezeq; Ola Tarawneh; Development of hydrogels, oleogels, and bigels as local drug delivery systems for periodontitis. Drug Development and Industrial Pharmacy 2018, 44, 1488-1497, 10.1080/03639045.2018.1464021.
- Gary Greenstein; Alan Polson; The Role of Local Drug Delivery in the Management of Periodontal Diseases: A Comprehensive Review. Journal of Periodontology 1998, 69, 507-520, 10.1902/jop.1998.69.5.507.
- Aaron C. Anselmo; Samir Mitragotri; An overview of clinical and commercial impact of drug delivery systems. Journal of Controlled Release 2014, 190, 15-28, 10.1016/j.jconrel.2014.03.053.
- Sarah A. Stewart; Juan Domínguez-Robles; Ryan F. Donnelly; Eneko Larrañeta; Implantable Polymeric Drug Delivery Devices: Classification, Manufacture, Materials, and Clinical Applications.. Polymers 2018, 10, 1379, 10.3390/polym10121379.
- Bo Wang; Shuo Wang; Qi Zhang; Yixuan Deng; Xiang Li; Liangyu Peng; Xianghao Zuo; Meihua Piao; Xin Kuang; Shihou Sheng; et al. Recent advances in polymer-based drug delivery systems for local anesthetics. Acta Biomaterialia 2019, 96, 55-67, 10.1016/j.actbio.2019.05.044.
- F. De La Portilla; S. Pereira; M. Molero; F. De Marco; V. Perez-Puyana; A. Guerrero; A. Romero A; Alberto Romero; Microstructural, mechanical, and histological evaluation of modified alginate-based scaffolds. Journal of Biomedical Materials Research Part A 2016, 104, 3107-3114, 10.1002/jbm.a.35857.
- Ndidi C. Ngwuluka; Nelson A. Ochekpe; Oi Aruoma; Naturapolyceutics: The Science of Utilizing Natural Polymers for Drug Delivery. Polymers 2014, 6, 1312-1332, 10.3390/polym6051312.
- Garg, V.; Chawla, K.; Pawar, S.K; Nanotechnology controlled local drug delivery system for the treatment of periodontitisc. J. Adv. Med. Med. Res. 2018, 26, 1–17, https://doi.org/10.9734/JAMMR/2018/40828.
- Prashansa Agrawal.; Significance of Polymers in Drug Delivery System. Journal of Pharmacovigilance 2015, 3, e127, 10.4172/2329-6887.1000e127.
- Zhanhai Dong; Yining Sun; Yuhui Chen; Yan Liu; Chuhua Tang; Xiaozhong Qu; Injectable Adhesive Hydrogel through a Microcapsule Cross-Link for Periodontitis Treatment. ACS Applied Bio Materials 2019, 2, 5985-5994, 10.1021/acsabm.9b00912.
- Paster, B.J.; Boches, S.K.; Galvin, J.L.; Ericson, R.E.; Lau, C.N.; Levanos, V.A.; Sahasrabudhe, A.; Dewhirst, F.E.; Bacterial diversity in human subgingival plaque.. J. Bacteriol. 2001, 183, 3770–3783, .
- Richard P. Darveau; Periodontitis: a polymicrobial disruption of host homeostasis. Nature Reviews Microbiology 2010, 8, 481-490, 10.1038/nrmicro2337.
- Purnima S. Kumar; Ann L. Griffen; Melvin L. Moeschberger; Eugene J. Leys; Identification of Candidate Periodontal Pathogens and Beneficial Species by Quantitative 16S Clonal Analysis. Journal of Clinical Microbiology 2005, 43, 3944-3955, 10.1128/jcm.43.8.3944-3955.2005.
- Purnima S. Kumar; Eugene J. Leys; Jennifer M. Bryk; Francisco J. Martinez; Melvin L. Moeschberger; Ann L. Griffen; Changes in Periodontal Health Status Are Associated with Bacterial Community Shifts as Assessed by Quantitative 16S Cloning and Sequencing. Journal of Clinical Microbiology 2006, 44, 3665-3673, 10.1128/jcm.00317-06.
- Lu, Q.; Jin, L.; Darveau, R.P.; Samaranayake, L.P.; Expression of human β-defensins-1 and -2 peptides in unresolved chronic periodontitis. J. Periodontal Res. 2004, 39, 221–227, .
- Hui Zeng; Huixia Wu; Valerie Sloane; Rheinallt M. Jones; Yimin Yu; Patricia Lin; Andrew T. Gewirtz; Andrew S. Neish; Flagellin/TLR5 responses in epithelia reveal intertwined activation of inflammatory and apoptotic pathways. American Journal of Physiology-Gastrointestinal and Liver Physiology 2006, 290, G96-G108, 10.1152/ajpgi.00273.2005.
- Mehmet A. Eskan; George Hajishengallis; Denis F. Kinane; Differential Activation of Human Gingival Epithelial Cells and Monocytes by Porphyromonas gingivalis Fimbriae. Infection and Immunity 2006, 75, 892-898, 10.1128/iai.01604-06.
- Yong-Chen Lu; Wen-Chen Yeh; Pamela S. Ohashi; LPS/TLR4 signal transduction pathway. Cytokine 2008, 42, 145-151, 10.1016/j.cyto.2008.01.006.
- Je-Kyung Ryu; Soo Jin Kim; Sang-Hyun Rah; Ji In Kang; Hi Eun Jung; Dongsun Lee; Heung Kyu Lee; Jie-Oh Lee; Beom Seok Park; Tae-Young Yoon; et al. Reconstruction of LPS Transfer Cascade Reveals Structural Determinants within LBP, CD14, and TLR4-MD2 for Efficient LPS Recognition and Transfer. Immunity 2017, 46, 38-50, 10.1016/j.immuni.2016.11.007.
- Steven L. Raymond; David C. Holden; Juan C. Mira; Julie A. Stortz; Tyler J. Loftus; Alicia M. Mohr; Lyle L. Moldawer; Frederick A. Moore; Shawn D. Larson; Philip A. Efron; et al. Microbial recognition and danger signals in sepsis and trauma. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2017, 1863, 2564-2573, 10.1016/j.bbadis.2017.01.013.
- Karin Nylander; Bo Danielsen; Ole Fejerskov; Erik Dabelsteen; Expression of the Endothelial Leukocyte Adhesion Molecule-1 (ELAM-1) on Endothelial Cells in Experimental Gingivitis in Humans. Journal of Periodontology 1993, 64, 355-357, 10.1902/jop.1993.64.5.355.
- M. S. Tonetti; Martin A. Imboden; Niklaus P Lang; Neutrophil Migration Into the Gingival Sulcus Is Associated With Transepithelial Gradients of Interleukin-8 and ICAM-1. Journal of Periodontology 1998, 69, 1139-1147, 10.1902/jop.1998.69.10.1139.
- Darryl C. Zeldin; Peyton Eggleston; Martin D. Chapman; Giovanni Piedimonte; Harard Renz; David Peden; How Exposures to Biologics Influence the Induction and Incidence of Asthma. Environmental Health Perspectives 2006, 114, 620-626, 10.1289/ehp.8379.
- Lei Ren; Lijian Jin; Wai Keung Leung; Local expression of lipopolysaccharide-binding protein in human gingival tissues. Journal of Periodontal Research 2004, 39, 242-248, 10.1111/j.1600-0765.2004.00732.x.
- Elia Burns; Gilad Bachrach; Lior Shapira; Gabriel Nussbaum; Cutting Edge: TLR2 is required for the innate response to Porphyromonas gingivalis: activation leads to bacterial persistence and TLR2 deficiency attenuates induced alveolar bone resorption.. The Journal of Immunology 2006, 177, 8296-8300, 10.4049/jimmunol.177.12.8296.
- Dana T. Graves; D. Cochran; The Contribution of Interleukin-1 and Tumor Necrosis Factor to Periodontal Tissue Destruction. Journal of Periodontology 2003, 74, 391-401, 10.1902/jop.2003.74.3.391.
- Boyle, W.J.; Simonet, W.S.; Lacey, D.L; Osteoclast differentiation and activation. Boyle Ocl. Rev. 2003, 423, 337–342, .
- Robert Owen; Gwendolen C. Reilly; In vitro Models of Bone Remodelling and Associated Disorders. Frontiers in Bioengineering and Biotechnology 2018, 6, 134, 10.3389/fbioe.2018.00134.
- Cochran, D.L.; Inflammation and bone loss in periodontal disease. J. Periodontol. 2008, 79, 1569–1576, 10.3389/fbioe.2018.00134.
- Tania Crotti; Malcolm D. Smith; Robert Hirsch; Steven Soukoulis; Helen Weedon; Maria Capone; Michael J. Ahern; D.R. Haynes; Receptor activator NF κB ligand (RANKL) and osteoprotegerin (OPG) protein expression in periodontitis. Journal of Periodontal Research 2003, 38, 380-387, 10.1034/j.1600-0765.2003.00615.x.
- Nawarat Wara-Aswapati; Rudee Surarit; Anek Chayasadom; Jason A. Boch; W Pitiphat; RANKL Upregulation Associated With Periodontitis andPorphyromonas gingivalis. Journal of Periodontology 2007, 78, 1062-1069, 10.1902/jop.2007.060398.
- R Assuma; T Oates; D Cochran; S Amar; Dana T. Graves; IL-1 and TNF antagonists inhibit the inflammatory response and bone loss in experimental periodontitis.. The Journal of Immunology 1998, 160, 403–409, .
- Pamela J. Baker; Mark Dixon; R. Todd Evans; Lisa Dufour; Ellis Johnson; Derry C. Roopenian; CD4+ T Cells and the Proinflammatory Cytokines Gamma Interferon and Interleukin-6 Contribute to Alveolar Bone Loss in Mice. Infection and Immunity 1999, 67, 2804-2809, .
- Cavalla Franco; Hernández-Ríos Patricia; Timo Sorsa; Cláudia Cristina Biguetti; Hernández Marcela; Matrix Metalloproteinases as Regulators of Periodontal Inflammation. International Journal of Molecular Sciences 2017, 18, 440, 10.3390/ijms18020440.
- Päivi Mäntylä; M. Stenman; D. Kinane; T. Salo; K. Suomalainen; S. Tikanoja; T. Sorsa; Monitoring periodontal disease status in smokers and nonsmokers using a gingival crevicular fluid matrix metalloproteinase-8-specific chair-side test. Journal of Periodontal Research 2006, 41, 503-512, 10.1111/j.1600-0765.2006.00897.x.
- Hernández Ríos, M.; Sorsa, T.; Obregón, F.; Tervahartiala, T.; Valenzuela, M.A.; Pozo, P.; Dutzan, N.; Lesaffre, E.; Molas, M.; Gamonal, J.; et al. Proteolytic roles of matrix metalloproteinase (MMP)-13 during progression of chronic periodontitis: Initial evidence for MMP-13/MMP-9 activation cascade. J. Clin. Periodontol. 2009, 36, 1011–1017, .
- Rinkee Mohanty; Swati Joshi Asopa; M. Derick Joseph; Bhupender Singh; Jagadish Prasad Rajguru; K. Saidath; Uma Sharma; Red complex: Polymicrobial conglomerate in oral flora: A review.. Journal of Family Medicine and Primary Care 2019, 8, 3480-3486, 10.4103/jfmpc.jfmpc_759_19.
- Camille Zenobia; George Hajishengallis; Porphyromonas gingivalis virulence factors involved in subversion of leukocytes and microbial dysbiosis. Virulence 2015, 6, 236-243, 10.1080/21505594.2014.999567.
- Coats, S.R.; Jones, J.W.; Do, C.T.; Braham, P.H.; Bainbridge, B.W.; To, T.T.; Goodlett, D.R.; Ernst, R.K.; Darveau, R.P.; Human Toll-like receptor 4 responses to P. gingivalis are regulated by lipid A 1-and 4’-phosphatase activities. Cell. Microbiol. 2009, 11, 1587–1599, .
- George Hajishengallis; Min Wang; Shuang Liang; Martha Triantafilou; Kathy Triantafilou; Pathogen induction of CXCR4/TLR2 cross-talk impairs host defense function. Proceedings of the National Academy of Sciences 2008, 105, 13532-13537, 10.1073/pnas.0803852105.
- Mariko Naito; Eiko Sakai; Yixin Shi; Hiroshi Ideguchi; Mikio Shoji; Naoya Ohara; Kenji Yamamoto; Koji Nakayama; Porphyromonas gingivalis-induced platelet aggregation in plasma depends on Hgp44 adhesin but not Rgp proteinase. Molecular Microbiology 2006, 59, 152-167, 10.1111/j.1365-2958.2005.04942.x.
