The Canadian Cancer Clinical Trials Network (3CTN) was established in 2014 to address the decline in academic cancer clinical trials (ACCT) activity. Funding was provided to cancer centres to conduct a Portfolio of ACCTs.
1. Overview
The Canadian Cancer Clinical Trials Network (3CTN) was established in 2014 to address the decline in academic cancer clinical trials (ACCT) activity. Funding was provided to cancer centres to conduct a Portfolio of ACCTs. Larger centres received core funding and were paired with smaller centres to enable support and sharing of resources. All centres were eligible for incentive-based funding for recruitment above pre-3CTN baseline. Established performance measures were collected and tracked. The overall recruitment target was 50% above pre-3CTN baseline by Year 4. An analysis was completed to identify predictive success factors and descriptive statistics were used to summarize site characteristics and outcomes. From 2014–2018, a total of 11,275 patients were recruited to 559 Portfolio trials, an overall increase of 59.6% above pre-3CTN baseline was observed in Year 4. Twenty-five (51%) adult centres met the Year 4 recruitment target and the overall recruitment target was met within three years. Three factors that correlated with sites’ achieving recruitment targets were: time period, region and number of baseline trials. 3CTN was successful in meeting its objectives and will continue to support ACCTs and member cancer centres, monitor performance over time and seek continued funding to ensure success, better trial access and outcomes for patients.
2. Framework of Canadian Cancer Clinical Trials Network
Clinical trials are essential for advancing scientific knowledge and identifying better treatment for patients. Canadians have long benefited from access to and results from practice-changing cancer clinical trials sponsored by the academic investigator community [
1,
2,
3]. However, that access is threatened by increased complexity of ethical and regulatory processes, rising costs, and limited supports available for the conduct of academic trials [
4]. Limited capacity to participate in academic sponsored trials has resulted in greater prevalence of industry sponsored clinical trials that are designed to lead to the approval of new drugs [
5]. Trials to address other important practice-changing questions are delayed or unable to be addressed.
To address the impediments to academic clinical trial activities, the Canadian Cancer Research Alliance (CCRA) 2011 report on the status of clinical trials in Canada recommended the establishment of a pan-Canadian network that would provide funding and infrastructure to support the conduct of academic clinical trials [
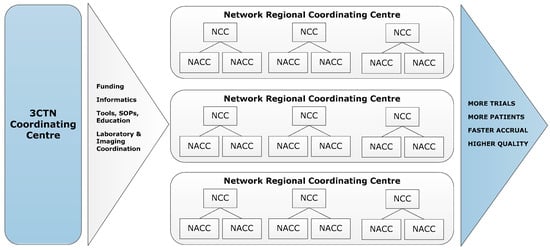
1]. In 2014, the Canadian Cancer Clinical Trials Network (3CTN; ‘the Network’) was established in collaboration with the Ontario Institute for Cancer Research (OICR), Canadian Cancer Trials Group (CCTG) and Network of Networks (N2). A total funding envelope of CAN 22 M raised was provided from 13 national and provincial charities, and public agencies. The Network has four components: (a) Coordinating Centre in Toronto serving as the administrative hub; (b) Network Regional Coordinating Centres (NRCC) that cover Canadian provinces and regions; (c) larger Network Cancer Centres (NCC); (d) smaller Network Affiliated Cancer Centres (NACC) that are linked to NCCs (
Figure 1). The Network is comprised of 66 member institutions committed to the following goals and objectives for its initial four-year strategic plan:
Figure 1. 3CTN organizational framework.
-
To improve patient access to academic clinical trials;
-
To improve site performance of academic trials;
-
To improve the trial environment for the conduct of academic clinical trials through collaboration and facilitation of important national trial initiatives;
-
To demonstrate impact of the Network and academic trials on the Canadian Health System.
The majority of adjusted funds were allocated to core staffing at Cancer Centres’ clinical trial units, to support costs of additional resources necessary for conducting academic trials [
6,
7]. Funding was provided to NRCCs for regional coordination to realize Network goals and objectives. NCCs received core funding from provincial funders to support academic cancer clinical trial (ACCT) recruitment based on population served. The smaller NACCs were paired with NCCs to form local-regional nodes to enable better support and sharing of resources and NCCs were encouraged to share a portion of their core funds with affiliated centres. In addition, both NCCs and NACCs were each eligible to receive incentive-based funding for recruitment to ACCTs above their defined, pre-3CTN baseline. Further to providing funding for trial recruitment, 3CTN provided a coordinated focus on improving ACCT trial activities, supported trial best practices, the development and implementation of patient involvement strategy for the Network and its centres, as well as the development of communication and knowledge transfer activities to meet its four-year strategic goals [
8,
9,
10]. This paper aims to describe the impact of the Network to ACCT recruitment and identify predictive success factors for adult Network sites after 3CTN’s first four-year term.
3. 2014-2018 Outcomes
Since the formation of 3CTN as a pan-Canadian cancer network, there has been an increase in academic cancer clinical trial recruitment. From 2014 to 2018, a total of 11,275 patients were enrolled in Portfolio trials, with 3907 patients recruited in the last fiscal year, which was an increase of 59.6% above pre-3CTN baseline yearly totals, exceeding the recruitment target for the period. This was accomplished despite challenges due to declining numbers of Portfolio Phase III and other large study population randomized clinical trials during the same period.
There were three factors that correlated with successful recruitment: time period, region and number of baseline trials. Centres with lower numbers of baseline trials had greater increases in accrual, whereas centres with larger numbers of trials had less of an increase. 3CTN support may have had less of an impact on those centres that had larger numbers of existing trial staff and infrastructure compared to smaller centres. However, the number of trials and trial personnel could not continue to expand capacity limits. Further investigation into this ceiling effect is required.
The lack of association between funding and achieving accrual targets was surprising. It is possible that sites may have secured new funding for their trial units outside of 3CTN. In addition, it is possible that the limited envelope of core and incentive funding was not sufficient to independently improve recruitment to ACCTs. The CAN 22 M total budget was substantial and unprecedented, but less than the CAN 36 M original budget developed based on adaption of the UK model.
Improved communication, best practices implementation and other 3CTN activities to improve trial performance may have also contributed to recruitment success across the Network. Improved connections and collaborative problem solving among Network centres are among advancements reported by sites and observed during Network-wide meetings. Smaller centres with more limited resources to conduct trials were linked to supports from larger centres to increase clinical trial activity. Some NCCs allocated core funding to NACCs to hire staff, while others provided educational opportunities, shared resources and coordinated activities, such as research ethics board submissions. These changes allowed smaller centres to increase their accrual by a larger percentage compared to larger centres, although total numbers were greater for larger centres with established clinical trial programs and patient catchment areas. Whether to prioritize support for smaller centres to increase their trial activity and accrual, which may allow patients more access to clinical trials, or larger centres which can accrue more patients in absolute terms, is an ongoing debate.
3CTN provided Network sites with access to clinical research professionals to share best practices, tools, and templates. In addition, 3CTN worked with stakeholders to develop and implement initiatives to address challenges in the clinical trial environment. These initiatives included supporting the roll out of clinical trials management systems, development of a recruitment best practices repository and a clinical trial education and awareness campaign. Although it will require more time to quantify the impact, these ACCT-focused improvements are expected to contribute to a more efficient and high-quality research environment for all clinical trials.
There was regional variation in attaining accrual targets. Most regions saw increased clinical trial activity above pre-3CTN baseline; however, some were unable to meet recruitment targets, while others did not sustain baseline trial activity. Twelve sites were delayed in joining the Network and may benefit from additional r time to improve trial recruitment activities. Certain regions had smaller populations and trial opportunities and capacities to participate in more trials. Core funding amounts were variable for each centre, determined by the size of the population served and availability of funds. Access to 3CTN core funding varied across provinces, partly due to available funding from the provincial funder, and partly due to the funding model based on patient population. For example, Network cancer centres located in Quebec did not receive core funding directly; instead, centralized roles were created to support sites. A more targeted approach to improving and sustaining capacity for trial conduct at centres is needed.
There are several limitations to our analysis. Unfortunately, pre-3CTN baseline recruitment data from 2011–2013 were not collected on a trial-by-trial basis, and this has limited the ability to carry out more in-depth analysis of changes in individual trials and trial portfolios over time. The reliance on trial information from trial registries and other public sources to review and supplement data provided by member cancer centres limits understanding of trial complexity. Although 3CTN membership reflects the majority of sites conducting cancer trials in Canada, the total number of sites is relatively small, and the evaluation timeframe is relatively short, resulting in somewhat limited statistical sample size and power. Furthermore, the focus was on academic multicentre trials, which may not represent the broader conduct of clinical trials, which would include single centre studies and industry-sponsored trials.
The UK experience has shown that when adequately funded, a coordinated and managed approach to clinical research is highly successful and can be sustained in the long term [
13]. 3CTN is the only Canadian network that provides direct support to clinical trial units to conduct ACCTs, and met its key objectives to increase trial recruitment and performance, and achieved greater integration across clinical trial centres through its activities. Although 3CTN met its overall objectives and its initial success is encouraging, support for ACCT conduct needs to be sustained. Insufficient support for clinical trial units remains a challenge and the resulting focus on industry sponsored studies continues to be concerning [
14]. 3CTN has developed the structure, platforms and linkages needed to deliver on its objectives and with time will adapt and evolve into a more sustainable network. 3CTN will continue to support ACCT and Network sites, monitor performance over time, assess for factors that contribute to successful ACCT and seek continued funding to ensure the success of the Network, resulting in better trial access and outcomes for patients.
This entry is adapted from the peer-reviewed paper 10.3390/curroncol28040248