Propolis is a natural material that honey bees (Apis mellifera) produce from various botanical sources. The therapeutic activity of propolis, including antibacterial, antifungal, and anti-inflammatory effects, have been known since antiquity. Propolis is a rich source of biologically active compounds, which affect numerous signaling pathways regulating crucial cellular processes. The results of the latest research show that propolis can inhibit proliferation, angiogenesis, and metastasis of cancer cells and stimulate apoptosis. Moreover, it may influence the tumor microenvironment and multidrug resistance of cancers.
- propolis
- propolis compounds
- cancer
- cell proliferation
- cytotoxicity
- apoptosis
- autophagy
- angiogenesis
- metastasis
- cancer therapy
1. Introduction
2. Composition of Propolis
|
Compound Name, IUPAC Name; Concentration Used |
Model |
Property |
Chemical Structure |
Reference |
|---|---|---|---|---|
|
Flavonoids, flavanones, flavones and flavonols |
||||
|
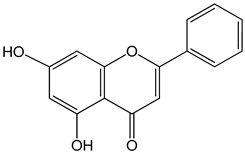
Chrysin (5,7-dihydroxy-2-phenylchromen-4-one) 50 μM 5, 25, 50, 80 µg/mL |
DU145 and PC-3 cells CAL-27 cells |
induction of apoptosis |
|
|
|
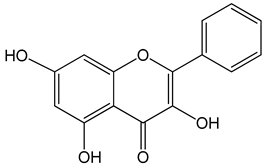
Galangin (3,5,7-trihydroxy-2-phenylchromen-4-one) 0–40 μM 0–40 μM 10, 20 and 30 mg/kg |
mice bearing B16F1 TU212, M4e, HBE, HEP-2 RTE, and HHL-5 cells BALB/c nude mice |
induction of apoptosis induction of apoptosis and inhibition of migration |
|
|
|
Genistein (5,7-dihydroxy-3-(4-hydroxyphenyl)chromen-4-one) 0–120 μM |
LNCaP cells; mouse BALB/c 3T3 and SVT2 (SV40-transformed BALB/c 3T3) fibroblasts |
inhibition of cell cycle |
|
[3] |
|
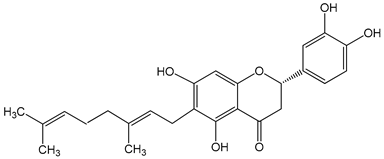
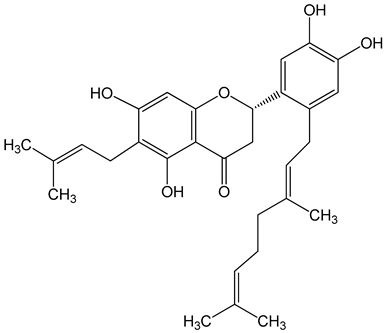
Nymphaeol A/Propolin C ((2S)-2-(3,4-dihydroxyphenyl)-6-[(2E)-3,7-dimethylocta-2,6-dienyl]-5,7-dihydroxy-2,3-dihydrochromen-4-one) 5–20 μM 2.5–20 μM |
A549 cells A549 and HCC827 cells |
anti-angiogenic activity, inhibition of proliferation inhibition of migration and invasion |
|
|
|
Nymphaeol C ((2S)-2-[2-[(2E)-3,7-dimethylocta-2,6-dienyl]-3,4-dihydroxyphenyl]-5,7-dihydroxy-6-(3-methylbut-2-enyl)-2,3-dihydrochromen-4-one) 5–20 μM |
anti-angiogenic activity, inhibition of proliferation |
|
[29] |
|
|
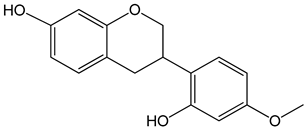
Vestitol (3-(2-hydroxy-4-methoxyphenyl)-3,4-dihydro-2H-chromen-7-ol) 0.37, 3.7, 37, and 370 μM |
HeLa cells |
cytotoxic effect |
|
[31] |
|
Aromatic acids and their derivatives |
||||
|
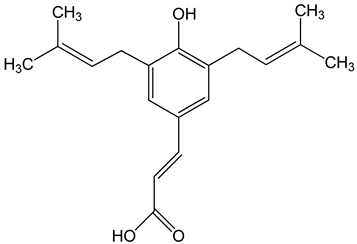
Artepillin C ((E)-3-[4-hydroxy-3,5-bis(3-methylbut-2-enyl)phenyl]prop-2-enoic acid) 250 μM 100 μg/mL 0–150 μM |
HT1080, A549, and U2OS cells BALB/c nude mice AGP-01 and HeLa cells CWR22Rv1 cells |
inhibition of proliferation cytotoxic effect autophagy inhibition |
|
|
|
Baccharin ((1R,3S,4S,6R,9R,13S,15R,16S,19R,20E,22Z,26R,27S,28S)-16-hydroxy-19-[(1R)-1-hydroxyethyl]-6,15,27-trimethylspiro [2,5,11,14,18,25-hexaoxahexacyclo [2 4.2.1.03,9.04,6.09,27.013,15]nonacosa-20,22-diene-28,2′-oxirane]-12,24-dione) 0–150 μM |
CWR22Rv1 cells |
autophagy inhibition |
|
[34] |
|
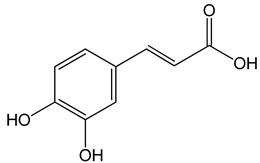
Caffeic acid ((E)-3-(3,4-dihydroxyphenyl)prop-2-enoic acid) 50 and 100 μM 65, 130, 190 µg/mL 30 μg/mL, 200 μg/mL 12.5 μM, 1 mM, 50 μM, 100 mg/kg, 20 mg/kg |
MDA-MB-231 cells CAL-27 cells Hep3, SK-Hep1, HepG2 cells |
cell cycle arrest in a dose- and time-dependent manner apoptosis activation inhibition of angiogenesis, apoptosis activation |
|
|
|
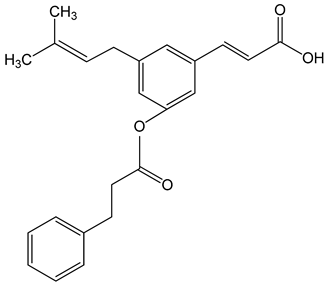
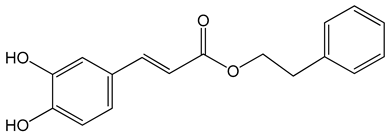
Caffeic acid phenylethyl ester (2-phenylethyl (E)-3-(3,4-dihydroxyphenyl)prop-2-enoate) 0.005–0.1 mg/mL 0.5–500 µM 10 mg/kg/day 15 mg/kg |
AGS, HCT116, HT29, YD15, HSC-4, HN22, MCF-17, MDA-MB-231, MDA-MB-468, A549, HT1080, G361, U2OS, LNCaP, PC-3, DU145, Hep2, SAS, OECM-1, TW01, TW04, SW620, H460 and PANC-1 cells Balb/c nude mice BALB/c AnM-Foxn-1 mice |
inhibition of proliferation, migration and invasion, pro-apoptotic activity anti-metastatic activity |
|
|
|
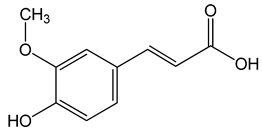
Ferulic acid ((E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enoic acid) 50, 100, 150 µg/mL |
CAL-27 cells |
apoptosis activation |
|
[26] |
|
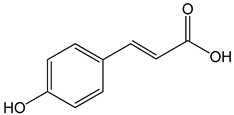
p-coumaric acid ((E)-3-(4-hydroxyphenyl)prop-2-enoic acid) 100 μg/mL 70, 140, 210 µg/mL |
AGP-01 and HeLa cells CAL-27 cells |
cytotoxic effect apoptosis activation |
|
|
|
Other |
||||
|
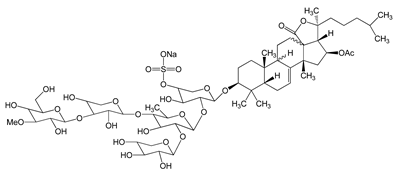
Frondoside A (sodium;[(3R,4R,5R,6S)-6-[(2S,4S,6S,12R,13R,18R)-4-acetyloxy-2,6,13,17,17-pentamethyl-6-(4-methylpentyl)-8-oxo-7-oxapentacyclo[10.8.0.02,9.05,9.013,18]icos-1(20)-en-16-yl]oxy]-5-[(2S,3R,4S,5S,6R)-5-[(2S,3R,4S,5R)-4-[(2S,3R,4S,5R,6R)-3,5-dihydroxy-6-(hydroxymethyl)-4-methoxyoxan-2-yl]oxy-3,5-dihydroxyoxan-2-yl]oxy-4-hydroxy-6-methyl-3-[(2S,3R,4S,5R)-3,4,5-trihydroxyoxan-2-yl]oxyoxan-2-yl]oxy-4-hydroxyoxan-3-yl] sulfate) 0.3–1.2 μM |
A549 cells |
anti-angiogenic activity, inhibition of proliferation |
|
[29] |
|
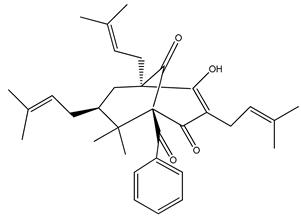
Nemorosone ((1R,5R,7S)-1-benzoyl-4-hydroxy-8,8-dimethyl-3,5,7-tris(3-methylbut-2-enyl)bicyclo[3.3.1]non-3-ene-2,9-dione) 5–50 μM |
HT-29 and THP-1 cells |
inhibition of migration and proliferation |
|
[46] |
3. The Use of Propolis and Its Components in Cancer Therapy
This entry is adapted from the peer-reviewed paper 10.3390/nu13082594